Abstract
Mesenchymal stem cells sheets have been verified as a promising non-scaffold strategy for bone regeneration. Alveolar bone marrow mesenchymal stem cells, derived from neural crest, have the character of easily obtained and strong multi-differential potential. However, the bone regenerative features of alveolar bone marrow mesenchymal stem cells sheets in the craniofacial region remain unclear. The purpose of the present study was to compare the osteogenic differentiation and bone defect repairment characteristics of bone marrow mesenchymal stem cells sheets derived from alveolar bone (alveolar bone marrow mesenchymal stem cells) and iliac bone (Lon-bone marrow mesenchymal stem cells)
Introduction
Craniofacial bone defect is a common disease, which would seriously affect the health of patients and brings difficulties for medical treatment. Currently, autogenous bone, autografts and allografts substitutes are the standard treatments for bone tissue reconstruction, but with many drawbacks. The autogenous bone practical clinical application is restricted by both donor site morbidity and the limited availability of bone volume. 1 Besides, commercial bone grafts substitutes have poor efficiency in large bone defect reconstruction with the limited osteogenic and osteoinductive ability. 2 In recent decades, stem cell-based tissue engineering has emerged as a therapeutic approach to repair of bone defects. 3 The stem cell-based tissue engineering depends on biomaterials and stem cells. Although substantial advances have been made, there are many challenges in craniofacial bone tissue engineering, such as complex physiological structures, suitable biomaterials and superior osteoinductive stem cells. 4
Recently, cell sheet engineering has been developed as a unique, scaffold-free method of tissue regeneration,
5
with the advantage of maintenance of extracellular matrix (ECM) and cell-cell junctions, without the impairment induce by biomaterial and its degradation.
6
Since the mesenchymal stem cells (MSCs) sheets were first used for the regeneration of osteogenic tissue in 2006,
7
more and more studies have been made to investigate the function of MSC sheet in craniofacial bone regeneration. By being used alone or in conjunction with exogenous biomaterials, MSCs sheets have been verified effective in bone regeneration.
8
Bone marrow MSCs (BMSCs), with the character of high proliferation and osteogenic capacity, are the predominant MSCs used in preclinical and clinical studies of cell sheet based craniofacial tissue regeneration.
9
However, several disadvantages of BMSCs have been found in the application, such as the invasive and painful harvest procedure, impairment of self-renewal and differentiation capacity due to donor age,
10
and weak stemness maintenance during expansion
Generally, the BMSCs are majorly derived from iliac crest bone marrow. They were also discovered in alveolar bone, which are named alveolar bone MSCs (Al-BMSCs), have the potential of osteogenic, chondrogenic or adipogenic differentiation,
12
and can effectively regenerate bone tissue
The purpose of this study was to compare the histology character, osteogenic differentiation, and specific gene expression of BMSCs sheets derived from alveolar bone (Al-BMSCs) and iliac bone (Lon-BMSCs)
Materials and methods
Rabbit
10 adult female New Zealand white rabbits weighing between 3.0 and 3.5 kg were used in this study. The animal study was approved by the Animal Care and Use Committee of Beijing Stomatological Hospital, Capital Medical University (Ethical code: KQYY-201812-002). The rabbits were purchased from Beijing Fang Yuanyuan Laboratory Animal (Beijing, China) and maintained in a specific pathogen-free animal facility and kept under conventional conditions with free access to water and food.
BMSCs derived from iliac bone and alveolar bone cultures
BMSCs derived from alveolar bone (Al-BMSCs) were cultured from bone marrow complex during the preparation of the implant hole according to the previous study. 17 Briefly, the bone marrow complex (about 0.1–0.2 mL) were obtained from dental implant treatment of five healthy male patients (30–50 years old), with their consent and approval from the Ethics Committee of China Rehabilitation Research Center (No. 2018-094-1). The bone marrow complex was put into the centrifugal tubes with medium and were grown in alpha-MEM (Invitrogen, Carlsbad, CA, USA), supplemented with 15% fetal bovine serum (FBS; Invitrogen, Carlsbad, CA, USA), 2 mmol/L glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin (Invitrogen, Carlsbad, CA, USA) in a humidified incubator under 5% CO2 at 37°C. The culture medium was changed every 3 days.
The human BMSCs extracted from iliac bone marrow (Lon-BMSCs) were obtained from the Department of Experimental Hematology, Beijing Institute of Radiation Medicine. The Lon-BMSCs and Al-BMSCs were expanded by detachment with 0.5% trypsin- ethylenediaminetetraacetic acid (EDTA) solution when the respective cultures reached 80% confluence. All the cells at passages 3–5 were used in subsequent experiments.
Stem cell sheet induction and histological observation
To induce the cell sheets, Lon-BMSCs and Al-BMSCs at third passages were sub-cultured in 10 cm dishes with 2 × 105cells/well, and cultured in complete medium containing 20 µg/mL vitamin C. 18 About 10 days later, the cells on the edge of the dishes wrapped, indicating that cell sheets had formed. After detaching the cell sheets were fixed with 4% formalin and then embedded in paraffin. Sections (8-µm) were prepared, deparaffinized, and stained with hematoxylin-eosin (HE).
Osteogenesis potential detection
To evaluate the osteogenic differentiation potential, Lon-BMSCs and Al-BMSCs or Lon-BMSCs and Al-BMSCs sheets were incubated in the osteogenic medium (Invitrogen). The medium was changed every 2 days. On day 5 after induction, cells or cell sheets were fixed with 4% paraformaldehyde for alkaline phosphatase (ALP) staining following the manufacturer’s protocol (Sigma-Aldrich). On day 14 after induced for osteogenic induction, cells or cell sheets were fixed with 70% ethanol, and stained with solution contained 2% Alizarin Red (Sigma-Aldrich). ALP and Alizarin Red staining were measured by using the Image-Pro Plus 6.0 program (Media Cybernetics, Rockville, MD, USA).
Real-time PCR for assessing gene expression
Cells or cell sheets were treated with Trizol (Invitrogen). Total mRNA was extracted by RNAprep pure Cell Kit (TIANGEN, Beijing, China). Then cDNA was synthesized with FastQuant RT Kit (TIANGEN, Beijing, China). We obtained GAPDH primer, forward 5’- CGGACCAATACGACCAAATCCG -3,’ reverse 5’- AGCCACATCGCTCAGACACC -3’; OPN primer, forward 5’- ATGATGGCCGAGGTGATAGT-3,’ reverse 5’- ACCATTCAACTCCTCGCTTT-3’; OCN primer, forward 5’- AGCAAAGGTGCAGCCTTTGT-3,’ reverse 5’- GCGCCTGGGTCTCTTCACT-3’; Runx2 primer, forward 5’- TCTTAGAACAAATTCTGCCCTTT-3,’ reverse 5’- TGCTTTGGTCTTGAAATCACA-3’; BSP primer, forward 5’- CAGGCCACGATATTATCTTTACA-3,’ reverse 5’- CTCCTCTTCTTCCTCCTCCTC-3,’ from Primer 3. Real-time polymerase chain reaction (PCR) reactions were performed with the SuperReal PreMix Plus SYBR Green PCR kit (TIANGEN, Beijing, China).
Cell sheets transplantation in Rabbit Calvarial Bone Defect model
Ten rabbits were anesthetized via intramuscular injection with 7.5 mg/kg tiletamine-zolazepam (Zoletil 50, Virbac, Carros Cedex, France) and 2 mg/kg xylazine hydrochloride (Sumianxin II, Jilin Huamu, Changchun, China). The rabbit calvarial bone defect was created according to the previous study. 19 After midline skin incision, muscle dissection, and periosteal elevation, the calvarial bone was carefully exposed. Two symmetrical round defects, each 5 mm in diameter and 2 mm in depth, were prepared in the calvaria using a trephine bur under the copious irrigation of sterile saline. Two experimental modalities were randomly allocated to the 20 defects, as follows: (1) Lon-BMSCs sheet and (2) Al-BMSCs sheet. Cell sheets were collected and immediately implanted into the bone defects. After incision closed, the animals received 30 mg/kg penicillin by intramuscular injection to prevent infection. The animals had free access to food and water and were monitored daily for any complications or abnormal behaviors during the healing period.
Sequential fluorescent labeling
According to the time of execution, the experimental animals were divided into two groups (n = 5). In the 4-week group, the animals were intramuscularly injected with 90 mg/kg Xylenol orange (Sigma, USA), 20 mg/kg calcein (Sigma, USA), and 30 mg/kg alizarin red S (Sigma, USA), respectively, at 1, 2, and 3 weeks after the operation. In the 8-week group, the time point was 2, 4, and 6 weeks.
Micro-computed tomography measurements
After 4 and 8 weeks of healing, the rabbits were sacrificed. The skull bones were harvested and fixed in 10% neutral buffered formalin, the samples were processed and scanned with a micro-CT (Siemens Inveon, Siemens, Germany) running at a voltage of 80 kV, an electric current of 500 mA, exposure time of 2000 ms and a pixel resolution of 15 µm with a 0.5 mm aluminum filter. Three-dimensional (3D) image models were reconstructed using Inveon Research Workplace 4.2 (Siemens, Germany). A 5 mm diameter circular region was placed in the center of the initial defect area and was defined as the region of interest (ROI). The optimal threshold for discriminating between the bone and grafting materials was determined as 615. Finally, the bone volume density (BV/TV %) within the ROI, was determined and expressed as mean ± standard deviation (M ± SD).
Histology analysis
For hard tissue slices examination, samples were dehydrated with ethanol of ascending concentrations, embedded in polymethylmethacrylate (PMMA) and cut into sections using a microtome (LEICA SP1600, Germany). The fluorescent labeling was observed using the Stereo Microscope and Laser Scanning Confocal Microscope (OLYMPUSFV1000, Japan). The area of three fluorescent stained bone was quantified by Image-Pro Plus 6.0 software. For HE staining, the samples were decalcified in 10% EDTA (pH 7.0) for 21 days, stained with HE (Sigma, St. Louis, MO) following standard protocols, and observed using a standard light microscope Leica DM 4000 microscope (Leica, Germany).
Immunofluorescence analysis
For fluorescent immunohistochemistry (IHC) of fibronectin and integrin β1, deparaffinized slides were subjected to antigen retrieval in boiled sodium citrate buffer solution (pH 6.0) for 10 min, then blocked for 20 min in blocking buffer (5% bovine serum albumin and 0.01% Triton X-100 in phosphate buffer solution (PBS; Sigma). After blocking, samples were incubated with rabbit polyclonal anti-fibronectin (Abcam, ab2413, 1:300) and rabbit polyclonal anti-integrin β1 (Abcam, ab183666, 1:300) overnight at 4°C. Alexa Fluor 594 goat anti-rabbit IgG (Life Technologies, USA, 1:500) were used to detect primary antibodies, and then counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Invitrogen). Immunofluorescent images were captured using a Leica TCS SP5 confocal microscope (Leica). The expression intensity was measured using the Image-Pro Plus 6.0 program (Media Cybernetics, Rockville, MD, USA).
Bioinformatics analysis gene expression profile of Lon-BMSCs and Al-BMSCs
Gene expression dataset GSE58474 was downloaded from the Gene Expression Omnibus(GEO) database, which compared the gene expression between human Al-BMSCs and Lon-BMSCs. More gene expression data were derived from S Yu et al., 20 which compared the gene expression between human periodontal ligament mesenchymal stem cells (PDLSCs) and umbilical cord MSCs (UCMSCs). Those genes with an adjusted P-value < 0.05 and the absolute value of fold-change (FC) > 2 were analyzed. Then differentially expressed genes were subjected to Gene Ontology (GO) and The Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis to explore the gene function of individual genomic products based on defined features and understand the biological meaning underlying particular gene lists. In this study, both GO and KEGG analyses of differentially expressed genes were performed with a criterion false discovery rate (FDR) < 0.05.
Statistical analysis
All statistical calculations were performed. All values are described as the mean ± standard deviation (M ± SD). Statistics were calculated by using SPSS 13.0 statistical software. Student t-test was used to determine statistical significance. P ⩽ 0.05 was considered statistically significant.
Results
Higher osteogenic differentiation of Al-BMSCs compared to Lon-BMSCs
AL-BMSCs and Lon-BMSCs were derived from 5 individuals, the stem cells were identified by flow cytometric analysis and osteogenesis differentiation (Supplemental Figure 1). Both the ALP staining and Alizarin Red staining showed Al-BMSCs have higher osteogenic differentiation compared to Lon-BMSCs (Figure 1(a), (b)). The results of RT-PCR showed that the expression of BSP, OPN, OCN, Runx2 were higher expressed in Al-BMSCs than Lon-BMSCs at 0 day, 7 days and 14 days of osteogenic induction (Figure 1(c-f)), at 28 days, the expression of OPN and OCN were also up-regulated in Al-BMSCs than Lon-BMSCs (Supplemental Figure 2).
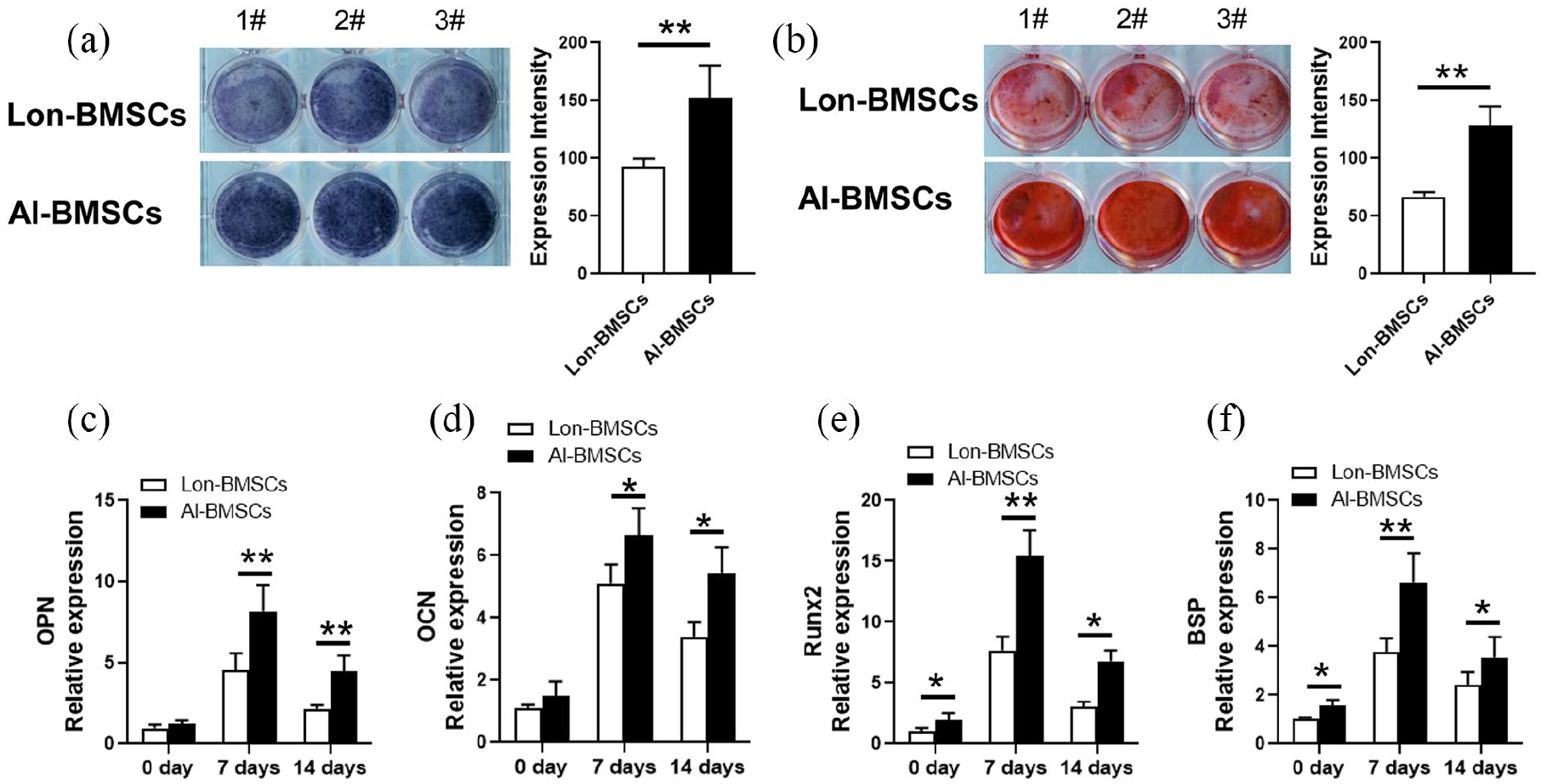
Comparison of osteogenic differentiation capacities of Lon-BMSCs and Al-BMSCs. (a) ALP (alkaline phosphatase) staining and qualitative measurement of Lon-BMSCs and Al-BMSCs following 7 days of osteogenic induction. (b) Alizarin red staining and qualitative measurement of Lon-BMSCs and Al-BMSCs following 14 days of osteogenic induction. (c, d, e, and f) Real-time PCR showed the expression of OPN, OCN, Runx2 and BSP were up-regulated in Al-BMSCs compared to Lon-BMSCs. GAPDH was used as an internal control. Student’s t-test was used to determine statistical significance. Values are mean ± SD, Error bars represent the s.d, (n = 5), *P ⩽ 0.05, **P ⩽ 0.01.
Histological analyses of Lon-BMSCs and Al-BMSCs sheets
Cell sheets were formed after 10 days of culture (Figure 2(a), (b)). Histological analysis showed that both sheets consisted of 2–3 layers of cells, which were closely connected (Figure 2(c, d)). The immunofluorescence staining indicated that there was no significant difference in the expression of fibronectin and integrin β1 between Lon-BMSCs and Al-BMSCs sheets (Figure 2(e-h), Supplemental Figure 3A).

Histological analyses of Lon-BMSCs and Al-BMSCs sheets. (a, b): The gross morphology of Lon-BMSCs and Al-BMSCs sheet showed no difference, with Vit C induced for 10 days. (c, d): The Hematoxylin and eosin (HE) staining of Lon-BMSCs and Al-BMSCs sheet all sheets consisted of 2-3 layers of cells. (n = 5; scale bar: 100 μm). Immunofluorescence analysis showed (e, f) fibronectin and (g, h) integrin β1 were highly expressed in the Lon-BMSCs and Al-BMSCs sheets, with no significant difference between them. Sections were counterstained with DAPI (blue). (n = 5, scale bar: 100 μm).
Higher osteogenic differentiation of Al-BMSCs sheet than Lon-BMSCs sheet
Al-BMSCs and Lon-BMSCs derived from 5 individuals were induced to cell sheet. The result of ALP staining and Alizarin Red staining showed Al-BMSCs sheets have higher osteogenic differentiation compared to Lon-BMSCs sheet (Figure 3(a, b), Supplemental Figure 3B, C). The results of RT-PCR showed that the expression of BSP, OPN, OCN, Runx2 were higher expressed in Al-BMSCs sheet than Lon-BMSCs sheet at 0 day and 7 days osteogenic induction (Figure 3(c-f)). The results showed Al-BMSCs sheets have higher osteogenic differentiation than Lon-BMSCs sheets, although the difference was decreased at 14 days of osteogenic induction.

Comparison of osteogenic differentiation capacities of Lon-BMSCs sheet and Al-BMSCs sheet. (a) ALP staining and qualitative measurement showed Al-BMSCs and Lon-BMSCs sheets following 7 days of osteogenic induction. (b) Alizarin red staining and qualitative measurement of Al-BMSCs and Lon-BMSCs sheets following 14 days of osteogenic induction. (c, d, e) Real-time PCR showed the expression of OPN, OCN, Runx2 and BSP were up-regulated in Al-BMSCs sheet compared to Lon-BMSCs sheet. GAPDH was used as an internal control. Student’s t-test was used to determine statistical significance. Values are mean ± SD, Error bars represent the s.d, (n = 5), *P ⩽ 0.05, **P ⩽ 0.01.
Bone regeneration characteristics of Lon-BMSCs and Al-BMSCs sheets in rabbit calvarial defects
In the calvarial defect rabbit model, it cannot be repaired by self-healing (Supplemental Figure 3D, E). The skull bones were harvested at 4 and 8 weeks. Regeneration of the calvarial defects was evaluated using micro-CT. The 3D reconstruction of the images showed that the bone defect area was decreased when transplanted with Lon-BMSCs and Al-BMSCs sheet at 4 weeks. More newly formed bones were witnessed in Al-BMSCs sheet transplanted group than Lon-BMSCs sheet group (Figure 4(a)). The BV/TV (bone volume/tissue volume) ratios (Figure 4(b)), and trabecula thickness (Figure 4(d)) in the defect areas of the Al-BMSCs sheet group were significantly higher than Lon-BMSCs sheet group (P < 0.05), while the BSA/BV (bone surface/bone volume) ratios (Figure 4(c)) in the defect areas of the Al-BMSCs sheet group were significantly lower than Lon-BMSCs sheet group (P < 0.05). HE staining and sequential fluorescent labeling showed much more new bone formation was formed and diminishes the diameter of the defect in AL-BMSCs sheet group than Lon-BMSCs sheet group (Figure 4(e, f)). Larger regenerated tissues were found in AL-BMSCs sheet group than Lon-BMSCs sheet group (P < 0.05) (Figure 4(g)). T-bet (Th1 marker), CCR7 (type I macrophage marker) and CD206 (type II macrophage marker) were analyzed by IHC analysis. At 4 weeks the count of Th1, M1 and M2 were witnessed in blank control, Lon-BMSCs sheet and AL-BMSCs sheet group, without any difference (Supplemental Figure 4).

Analyses of bone defect repairment by Lon-BMSCs and Al-BMSCs sheets in rabbit calvarial defect at 4 weeks. (a) 3D-reconstruction of Micro-CT images of defect regions in Lon-BMSCs and Al-BMSCs sheet at 8 weeks. (b, c, d) Qualitative measurement of BV/TV, BSA/BV and thickness values between Lon-BMSCs and Al-BMSCs sheets groups (*P < 0.05, **P < 0.01). (e) HE stained images of bone defect regions at 8 weeks in Lon-BMSCs and Al-BMSCs sheets groups. (f) Sequential fluorescent labeling of bone formation and mineralization. Bone formation and mineralization in the defect area of the rabbit at 8 weeks. Red, green and yellow represent labeling by Alizarin Red S, Calcein and Xylenol orange. (g) New bone area percentages in the defect region were assessed by histomorphometric analyses. (Scale bars = 0.5 mm) (*P < 0.05, **P < 0.01).
At 8 weeks, much great bone formation was found both in Al-BMSCs and Lon-BMSCs sheet groups, but the data showed the similar tendency of 4 weeks, more newly formed bones were witnessed in Al-BMSCs sheet group compared to Lon-BMSCs sheet group (Figure 5).

Analyses of bone defect repairment by Lon-BMSCs and Al-BMSCs sheets in rabbit calvarial defect at 8 weeks. (a) 3D-reconstruction of Micro-CT images of defect regions in Lon-BMSCs and Al-BMSCs sheet at 8 weeks. (b, c, d) Qualitative measurement of BV/TV, BSA/BV and thickness values between Lon-BMSCs and Al-BMSCs sheets groups (*P < 0.05, **P < 0.01). (e) HE stained images of bone defect regions at 8 weeks in Lon-BMSCs and Al-BMSCs sheets groups. (f) Sequential fluorescent labeling of bone formation and mineralization. Bone formation and mineralization in the defect area of the rabbit at 8 weeks. Red, green and yellow represent labeling by Alizarin Red S, Calcein and Xylenol orange. (g) New bone area percentages in the defect region were assessed by histomorphometric analyses. (Scale bars = 0.5 mm) (*P < 0.05, **P < 0.01).
Identification of key genes and pathways specific in Al-BMSCs using an integrated bioinformatics analysis
Al-BMSCs and dental MSCs (PDLSCs, dental pulp stem cells (DPSCs), SCAPs, etc.) are derived from the neural crest, and they have similar characteristics. Though integrated bioinformatics analysis with other neural crest derived MSCs, it would reveal the key genes and pathways in neural crest derived MSCs compared to other tissue derived MSCs. The published gene expression datasets between human Al-BMSCs and Lon-BMSCs, and the gene expression between human PDLSCs and Umbilical Cord MSC (UCMSCs) were used. The results showed a total 173 significantly changed genes were identified (94 up-regulated and 79 down-regulated) in Al-BMSCs compared to Lon-BMSCs (Figure 6(a)). While total 1059 significantly changed genes were identified (588 up-regulated and 471 down-regulated) in PDLSCs compared to UCMSCs (Figure 6(b)). Total 71 genes overlapped across the two datasets, suggesting that these genes may be conserved in neural crest derived MSCs. Among these 71 genes, 41 were up-regulated while 29 were down-regulated (Table). GO and KEGG analyses were performed on the 71 genes. The GO analysis revealed that most of the proteins encoded by these differentially expressed genes were ECM proteins located in the extracellular space (Figure 6(c)). The molecular functions (MF) enriched in this dataset were primarily associated with platelet-derived growth-factor binding and ECM structural constitution, while the enriched biological processes (BP) were primarily those associated with ECM organization and cell adhesion. The KEGG analysis revealed that the primary enriched signaling pathways were associated with ECM-receptor interaction, protein digestion and absorption, focal adhesion, and the PI3 K-Akt signaling pathway (Figure 6(d)). Interestingly, the Homeobox genes like

Discussion
In the present study, our results showed Al-BMSCs have higher osteogenic differentiation than Lon-BMSCs. Al-BMSCs can be induced to cell sheet with Vit C, with the similar histological structure and containing ECM protein (fibronectin and integrin β1) to Lon-BMSC. But Al-BMSCs sheet showed higher osteogenic differentiation capacity and bone defect reconstruction in rabbit calvarial defect model. Like other neural crest derived MSCs, Homeobox genes, such as
Canonical stem cell based bone engineering is dependent on stem cells and materials. After further research, many drawbacks have been found. This strategy needs culturing and expanding cells, which would destroy the ECM and intercellular connections between cells. ECM contains many necessary bioactive molecules for cells and tissues homeostasis,21,22 and the destruction of ECM can impair tissue regeneration efficiency
To date, cell sheets have been widely used in tissue engineering, including craniofacial bone 8 and dental tissue.18,28 Like PDLSCs and DPSCs have been successfully used for periodontal regeneration.29,30 Our previous studies have shown that PDLSC sheet can also be used for bio-root regeneration.31,32 MSC sheets first used for the regeneration of osteogenic tissue were described in 2006. 7 Subsequently, many research groups have reported bone formation or regeneration by the BMSCs sheet itself. 33 In order to enhance the bone regenerated capacity of MSCs sheet, many methods have been used to combine with MSCs sheet, 34 like HA/TCP, chitosan/hyaluronic acid nanoparticles plus microRNA-21, nanoscale HA combined with autologous platelet-rich fibrin, as well as various biological agents. 9
Lon-BMSCs were the major MSCs used in previously MSCs sheet based bone reconstruction.
35
Recently, numerous types of neural crest derived-MSCs have been isolated and characterized, including PDLSCs, DPSCs, stem cells from exfoliated deciduous teeth, Al-BMSCs.
36
All of these MSCs exhibit self-renewal, multilineage differentiation potential, and immunomodulatory properties.
37
Compared to other MSCs, they are highly proliferative, survival properties, and significantly higher osteogenesis
Multiple immune cells, such as Th1, Th2, Th17, macrophages, infiltrated into the transplanted area is inevitable, certainly immune cell infiltrated were necessary for tissue regeneration or wound-healing. 40 At the early stage of 1 week to 1 month during tissue regeneration, it was infiltrated by neutrophils, followed by dense infiltration of monocytes. 41 At the late stage, it was turned to anti-inflammatory cells, like M2 macrophages. 42 The results of the present study showed the Th1, M1 and M2 have no significantly difference in different groups. This indicating that the immunoregulation function of Lon-BMSCs and Al-BMSCs have no differentiation. Study showed that Al-BMSCs exhibited immunosuppressive effects on monocyte activation and T cell activation and proliferation similar to Lon-BMSCs. Both Al-BMSCs and Lon-BMSCs drove macrophages into an anti-inflammatory M2 phenotype. 43
In order to uncover the mechanism of the difference between Al-BMSCs and BMSCs, researchers have performed gene array analyses.
44
Although lots of differential genes have been found, it was hard to identify the key gene. Thus, we overlapped the differential genes with another gene expression data derived from S Yu et al.,
20
which compared the gene expression between another neural crest MSCs, PDLSCs and UCMSCs. The results showed that the Homeobox genes, like
Conclusion
In conclusion, the results showed Al-BMSCs sheet have higher osteogenic differentiation and bone defect reconstruction capacity than Lon-BMSCs sheet. Homeobox genes were upregulated in Al-BMSCs than Lon-BMSCs, demonstrated that Homeobox genes may be the key gene regulated Al-BMSCs biology property. All the results showed that Al-BMSCs sheet based tissue regeneration would be a promising strategy for craniofacial defect repair in future clinical applications.
Supplemental Material
suppl_figs – Supplemental material for Bone regeneration capacities of alveolar bone mesenchymal stem cells sheet in rabbit calvarial bone defect
Supplemental material, suppl_figs for Bone regeneration capacities of alveolar bone mesenchymal stem cells sheet in rabbit calvarial bone defect by Yanan Liu, Haifeng Wang, Huixin Dou, Bin Tian, Le Li, Luyuan Jin, Zhenting Zhang and Lei Hu in Journal of Tissue Engineering
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the grant from Beijing Municipal Administration of Hospitals’ Youth Program (QML20191504), Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201828).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
