Abstract
Stem cell-based therapy plays a significant role in the repair of bone defects. However, traditional stem cell transplantation strategies in bone tissue engineering are characterized by low survival rates and unstable treatment outcomes. In this study, we propose a timely delivery strategy for inflammatory changes in the setting of bone injury to improve the survival rate of transplanted cells and bone repair. The results of cell tracing in vivo showed that this strategy could effectively improve the survival rate of low-dose exogenous transplanted cells in bone defect areas, and CD31 immunofluorescence and histological sections suggested that this strategy effectively promoted vascularization and new bone formation in the calvarial defect area. Subsequently, we analyzed the mechanism of action of the “Two-step” strategy from the perspective of inflammatory microenvironment regulation, and the results suggested that the first batch transplanted stem cells caused localized and transient increases in tissue apoptosis levels and inflammatory factors, and recruited macrophage chemotaxis, and the second batch of cells may promote pro-inflammatory - anti-inflammatory transformation of the tissue. Finally, mRNA sequencing results suggest that the first batch cells in the “Two-step” strategy are important initiators in bone repair, which not only actively regulate the immune microenvironment at the bone defect, but also guide richer cellular activity and more positive biochemical responses. Therefore, the “Two-step” strategy leads to efficient inflammatory environment regulation and superior bone repair effects, which may provide an alternative option for the treatment of bone defects in the future.

Keywords
Introduction
Skeletal system injuries represent a highly prevalent health concern, with ~15 million fractures reported worldwide each year, of which approximately 1.9%–4.9% result in non-union. 1 Despite the regenerative capacity of the bone tissue, extensive bone defects resulting from high-energy trauma, severe infection, vascular injury, or resection of tumor lesions often lead to non-union or skeletal deformity. In clinical practice, autologous bone grafting remains the gold standard for the treatment of bone defects. However, limitations such as limited supply, secondary injury, chronic pain, and infection hinder its further application and development.2,3 Therefore, over the last decade, bone graft substitutes (BGSes), which offer the advantages of reproducible production, individualization, and non-immunogenicity, have gradually become a research topic of immense interest in the fields of bone biology and material science.4,5
Mesenchymal stem cell (MSC)-based therapies are considered an important component of BGSes, and bone mesenchymal stem cells (BMSCs) have become the most widely studied and applied method in bone repair because of their higher expression of osteogenic differentiation genes.6,7 Most previous studies have suggested that BMSCs are directly involved in the process of tissue regeneration, due to their strong proliferative capacity and ability to directionally differentiate into osteoblasts or chondrocytes in vitro.8,9 However, increasing evidence suggests that the therapeutic efficacy of MSCs is mainly attributed to their ability to modify the host microenvironment rather than their ability to differentiate and integrate into host tissues.10,11 Studies have shown that BMSCs can support the survival of surrounding tissues by releasing certain paracrine factors. For example, factors released by BMSCs can recruit macrophages and endothelial lineage cells to the wound site, thereby promoting wound healing. 12 In addition, BMSCs also possess immunomodulatory capacity and elicit immunosuppressive effects in most cases.13,14 Because of the low expression of class II major histocompatibility complex and co-stimulatory molecules on the cell surface, these are not only immune-privileged cells, but also interfere with different immune response pathways by means of direct cell-cell immune responses.15,16 It has been reported that BMSCs communicate extensively with a variety of inflammatory cells. For example, MSCs can prevent a variety of immune cell functions: cytokine secretion and cytotoxicity of T and natural killer cells; B cell maturation and antibody secretion; dendritic cell maturation and activation; and antigen presentation.17,18 Thus, the effects of BMSCs application on bone repair may theoretically involve a dual role, one that supports both regeneration and immune regulation.
Despite the encouraging preliminary results, the clinical application of stem cells still faces many difficulties. For example, the optimal timing and route of cell delivery for stem cell transplantation remains uncertain in bone defect. Initial studies delivered stem cells to recipients via the vasculature, which had the advantage of being minimally invasive and safely delivering large numbers of cells, but stem cells easily arrested in other organs and were easily attacked by the host immune system, resulting in minimal numbers of stem cells reaching target organs.19,20 Therefore, the current practice of stem cell transplantation mainly adopts local delivery in which cells are injected directly or combined with biomaterials to the targeted site to reduce cell loss during delivery. However, the survival rate of the delivered stem cells is extremely low due to the local hypovascular environment and inflammatory storm at the injury site. Freitas et al. 21 showed that the transplanted stem cells declined sharply within 10 days and were undetectable in critical calvarial defects after 25 days. This unexpectedly low survival rate limits the tatal repair function of stem cells and poses a significant challenge in elucidating the biological role of MSCs. Currently, relevant studies have reported the importance of the timing of stem cell transplantation for tissue regeneration in different disease models. For example, Hu et al. 22 compared the effects of BMSC transplantation at 1 h, 1 week, and 2 weeks after myocardial injury on the prognosis of a rat myocardial infarction model and concluded that rats receiving stem cell therapy after 1 week of myocardial infarction displayed the greatest benefit, due to scar formation and reduced inflammation. In addition, Wang et al. 23 found that BMSCs transplantation at 3 and 24 h after focal ischemia, but not 7 days, significantly reduced the lesion size and improved the motor deficits in an ischemic stroke model. The seemingly contradictory results described above may suggest significant tissue or organ differences in the optimal timing of stem cell transplantation. Therefore, it is highly desirable to optimize the timing of stem cell therapy for bone defects. At the same time, the deeper mechanism of action of stem cells needs to be further elucidated.
It is well known that bone tissue regeneration goes through three successive and overlapping phases: inflammation, callus formation, and remodeling. 24 The inflammatory phase, which is the starting point of the cascade, is critical for the initiation of bone tissue repair. During normal bone repair, inflammation begins in the early stages of injury and subsides almost immediately, to stimulate secretion of osteogenic and angiogenic growth factors as well as chemotaxis of target cells, ensuring proper tissue repair and optimal bone regeneration. 25 However, in nonunion and bone defects, the inflammatory state is often dysregulated (aggravated or chronic), which seriously hinders the process of bone repair.26,27 In this study, we first investigated the changes of inflammatory environment in calvarial bone defects, based on which stem cells were applied at different inflammatory stages (acute or subacute stages) to evaluate the effect of cell transplantation timing on the repair of calvarial defects, while the effect of transplanted stem cells on the local inflammatory environment was reverse assessed, hoping to explore a cell transplantation strategy specific for bone defects in order to improve the efficiency of stem cell transplantation and bone repair potential.
Materials and methods
Fabrication of collagen scaffolds
In this study, the collagen scaffolds were prepared using a freeze-drying technique, as described previously. 28 First, a 0.1 M acetic acid solution was prepared and chilled for 15 min in a refrigerator, at 4°C. Following that, 50 mg of collagen (type І collagen, Solarbio, China, Cat.C8061) was weighed and added to 5 mL of the aforementioned acetic acid solution, following which it was thoroughly dissolved using a vortex oscillator, to obtain a 10 mg/mL collagen gel solution. The collagen gel was centrifuged at 3000 rpm for 5 min in a high-speed centrifuge, to remove air bubbles. Subsequently, a suitable amount of the aforementioned collagen solution was added to custom-made molds (with an inner diameter of 5 mm and height of 10 mm) and frozen at −80°C for 15 min. After the collagen solution solidified completely, the molds containing the solidified collagen were rapidly placed in a freeze-dryer (ZL-20TD, ZOLLO, China) at −80°C. After 48 h of freeze-drying, the dried collagen scaffold was removed from the molds and cut into cylindrical scaffolds with a diameter of 5 mm and height of 2 mm. The scaffolds were then exposed to ultraviolet light for 48 h and set aside for further use. After 48 h of lyophilization, the dried collagen scaffolds were removed from the mold and immersed in EDC solution for cross-linking for 4 h, followed by repeated rinsing with distilled water to remove residual EDC. After lyophilization for another 48 h, collagen scaffolds were cut into cylindrical scaffolds with a diameter of 5 mm and a height of 2 mm. Finally, the scaffolds were exposed to ultraviolet light for 48 h and left for future use.
Construction of BMSCs-collagen scaffold complexes
In this experiment, the static seeding method was employed to seed BMSCs onto the collagen scaffold. 29 To enhance the osteogenic properties of BMSCs (BMSCs, Cyagen, China, Cat.RASMX-01001), all cells underwent osteogenic induction (Osteogenic differentiation medium, Cyagen, China, Cat.HUXMX-90021) for 14 days before transplantation. After digestion with trypsin, the cells were resuspended in growth medium(Cyagen, China, Cat.RAXMX-90011) and counted, and cell suspensions with densities of 2 × 107 and 4 × 107 cells/mL were prepared. The collagen scaffold was pre-soaked in phosphate-buffered saline (PBS), dried with sterile gauze, and then placed in a 24-well plate. A volume of 12.5 µL of the cell suspension was seeded onto each collagen scaffold, followed by static incubation for 4 h (37°C, 5% CO2, and 95% humidity) before further use.
Animal surgery
All animal experiments was approved by the Institutional Animal Care and Use Committee of Guangzhou Huateng Bio-pharmaceutical Technology Co., Ltd. (No. HTSW210906) and followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals. According to the regulations and guidelines of the laboratory animal center, male Sprague-Dawley rats (8 weeks old, 300–350 g) were maintained under specific pathogen free (SPF) conditions provided by Guangzhou Huateng Bio-pharmaceutical Technology Co., Ltd.
A critical calvarial bone defect model in healthy SD rats was prepared under sterile conditions, as described previously. 30 Prior to surgery, routine skin disinfection was performed. Anesthesia was induced using 1% pentobarbital sodium (40 mg/kg). After anesthesia, the skin was incised ~2.0 cm along the midline of the rat’s head, and subcutaneous tissue and periosteum were carefully separated to expose the parietal bone. With the assistance of pre-chilled sterile physiological saline for cooling, a 5-mm bone defect was created in the parietal bone, using a hollow drill. After adequate hemostasis, blank or cell-loaded collagen scaffolds were placed in the circular defect and sutured layer by layer. The wound was disinfected with iodophor, and the rats exhibited good activity post-operation. For the injection of the second batch of cells, rats were first anesthetized again with sodium pentobarbital, then 12.5 µL of cell suspension (2 × 107 or 4 × 107 cells/mL) was aspirated using a microsyringe (21 G), and finally, cell suspension was injected 3 mm subcutaneously into the calvarial defect area with finely control of the depth of the needle tip (Scheme 1).

Experimental process and grouping.
Live imaging-based detection of stem cells
In vivo imaging techniques have been used to trace transplanted stem cells in areas of calvarial defects. Lentiviruses carrying the firefly luciferase gene (JiKai, China, Cat.GV260) were used as vectors to construct bioluminescent stem cells (Luc+ BMSCs). Firstly, logarithmic-phase P3 BMSCs were subjected to bioluminescent lentivirus transfection. BMSCs were seeded into 6-well plates, at a density of 50 × 104 cells/well, and allowed to adhere for 4 h. The multiplicity of infection was set to 50, and 5 µL of lentivirus (at a final concentration of 1 × 108 transducing units/mL) was added to each well, along with a final concentration of 25 µg/mL of the enhancing agent polybrene. After 12 h of infection, the regular growth medium was replaced and the cells were continuously cultured for 72 h, with the addition of puromycin (1 µg/mL) to screen out unsuccessfully transfected cells. For detecting the survival rate of Luc+ BMSCs in the rats, the rats were placed in an anesthesia box and anesthetized using isoflurane gas (oxygen flow rate = 2 L/min) for 3 min, followed by maintenance anesthesia (oxygen flow rate = 1 L/min). After the rats were anesthetized, they were injected with a solution of D-luciferin potassium salt (AAT Bioquest Inc, USA, 150 mg/kg) via the abdominal cavity and then placed in a dark box and supplied with isoflurane for maintaining anesthesia. Quantitative statistics were performed using the IVIS® software system (PerkinElmer, USA) set to Auto mode for continuous imaging, with each detection selecting the image representing the peak of bioluminescent expression. Imaging began the day after the surgery and continued once a week, until the bioluminescent signal was no longer visible.
Immunofluorescence and histological staining
Immunofluorescence was used to evaluate the expression of markers for vascular regeneration and macrophages in the calvarial samples. In brief, the samples were first fixed in 4% paraformaldehyde for 24 h and decalcified in 0.5 M neutral Ethylenediaminetetraacetic acid. The decalcified bone tissue was then made into 5-µm paraffin sections. The tissue sections were deparaffinized, cleared, hydrated, and transferred to Tris-buffered saline (TBS) containing 10% goat serum for 20 min. Following serum removal, sections were incubated overnight at 4°C in primary antibodies working solutions for CD31, CD68 + inducible nitric oxide synthase (iNOS), or CD68 + CD206, respectively. The following day, after rinsing with TBS, 50 µL of the fluorescent secondary antibody was added to each slice and incubated at room temperature for 65 min, followed by another TBS rinse. DAPI-containing anti-quenching sealing solution was used for nuclear restaining, and the coverslips were placed and sealed. Images were captured under a fluorescence microscope (Olympus, Japan).
The following antibodies were used in the experiment: Rabbit anti-CD31(1:100, Abcam, Britain, Cat.ab222783), CD31 conjugated to Alexa Fluor 488 (1:200, Abcam, Britain, Cat.ab150117), Mouse anti-CD68(1:50, Abcam, Britain, Cat.ab201340), Rabbit anti-iNOS (1:100, Abcam, Britain, Cat.ab15323), Rabbit anti-CD206 (1:100, Abcam, Britain, Cat.ab64693), CD68 conjugated to Alexa Fluor 647 (1:400, Abcam, Britain, Cat.ab150115), conjugated to Alexa Fluor 488 (1:400, Abcam, Britain, Cat.ab150077).
Tissue sections were stained with hematoxylin and eosin (H&E) and Masson’s trichrome staining (Solarbio, China, Cat.G1120 and G1340), to visualize the cellular and bone tissue regeneration structures. The staining methods were performed according to the instructions provided by the reagent supplier. Images were captured using a bright-field microscope.
Micro-computed tomography (micro-CT)
At 12 weeks post-surgery, the euthanized rats were dissected to obtain calvarial samples. These samples were fixed in 4% paraformaldehyde for 24 h and then scanned using a micro-CT system (SkyScan1276, Bruker, USA). The specimens were scanned at a pixel resolution of 18 µm, with a voltage of 100 kV and current of 80 µA. Using the software CTAn provided by the manufacturer, the 2D images were reconstructed into 3D models with a threshold of 250. The calvarial defect area was selected as the region of interest for analyzing and comparing the new bone volume among the different groups.
TdT-mediated dUTP nick end labeling (TUNEL) staining
To detect the effect of different transplantation strategies on apoptosis within the bone defect, TUNEL staining (Cayman Chemical, USA, Cat.KGA7071) was performed on tissue sections from each group. The staining method followed the procedure instructions provided by the reagent supplier (Cayman Chemical). In brief, after routine deparaffinization and hydration of the paraffin sections, 50 µL of TdT enzyme reaction solution was added to the sections, for a 30-min reaction. The sections were then rinsed with PBS three times. Next, 100 µL of Proteinase K working solution was added to the tissue sections and kept in the dark for 60 min. After another round of PBS wash, 50 µL of Streptavidin-Fluorescein labeling solution was added to the tissue sections and kept in the dark for 30 min. The sections were washed with PBS three more times, and DAPI-containing sealing solution was used for nuclear restaining. Subsequently, coverslips were placed and sealed, and the samples were observed and photographed using a fluorescence microscope, at an excitation wavelength of 488 nm (Olympus, Japan).
Enzyme-linked immunosorbent assay (ELISA)
To investigate the effect of stem cell transplantation on the inflammatory state of local injured bone tissue after bone injury, pro-inflammatory and anti-inflammatory factors were analyzed by ELISA. The detection method followed the instructions provided by the reagent supplier (Cusabio, China). Blood collection was performed as follows: after anesthesia, the rats were placed in a supine position, routine disinfection was carried out, and a cloth was placed. The skin was longitudinally incised about 3 cm along the midline of the abdomen, fully exposing the abdominal cavity. The abdominal organs were pushed to the right, exposing the bottom of the abdominal cavity. The fascia was separated along the left side of the spine to expose the inferior vena cava. A 3-mL blood sample was collected from 5 mm below the left renal vein using a sodium citrate anti-coagulation tube, while ensuring that the blood was inverted and mixed thoroughly to prevent coagulation. After centrifugation at 1000 × g for 5 min, the supernatant was used for ELISA detection or stored at −20°C for later use.
RNA sequencing (RNA-seq) analysis
We performed RNA-seq analysis of skull tissues from the defect area on day 7, to assess the effect of day 0—transplanted stem cells on the local location of the bone defect. Three independent biological replicates were sequenced for each group of samples. Total RNA was extracted using TRIzol™ reagent kit (Invitrogen, USA), according to the manufacturer’s protocol. RNA quality was assessed on an Agilent 2100 Bioanalyzer (Agilent Technologies, USA) and checked using RNase-free agarose gel electrophoresis. After the total RNA was extracted, eukaryotic mRNA was enriched by means of oligo(dT) beads and then prokaryotic mRNA was enriched by removing rRNA using the Ribo-Zero™ Magnetic Kit (Epicentre, USA). Following that, the enriched mRNA was fragmented using fragmentation buffer and reverse-transcribed into cDNA, using the NEBNext® Ultra™ RNA Library Prep Kit for Illumina (New England Biolabs, USA).The purified double-stranded cDNA fragments were subjected to end-repair, A base-addition, and ligation to Illumina sequencing adapters. The ligation reaction was purified using AMPure XP Beads (1.0×) and amplified using polymerase chain reaction. The resulting cDNA library was sequenced by Gene Denovo Biotechnology (Guangzhou, China), using the Illumina NovaSeq 6000 system. Finally, gene libraries were subjected to bioinformatics analyses such as principal component, correlation, differentially expressed gene (DEG), gene ontology (GO), and Kyoto Encyclopedia of Gene and Genomes (KEGG) enrichment analyses.
Statistical analysis
Parametric data from multiple groups were statistically analyzed using one-way analysis of variance, followed by Tukey’s test. All analyses were performed using Prism 7.0 software (GraphPad, San Diego, CA, USA) and all data are presented as mean ± SD. Significance was defined as p < 0.05.
Results
Investigation of the low-inflammatory window in the bone defect environment, to develop a cell transplantation strategy
We determined the low inflammatory phase by investigating changes in local environment and systemic inflammatory factors following skull injury in rats (Figure 1(a)). On day 1, a few apoptotic cells and tumor necrosis factor alpha (TNF-α)+ cells started to appear in the initial phase of traumatic inflammation. By day 3, the infiltrating TNF-α+ and TUNEL+ cells in the scaffold space increased synchronously and reached a peak. On day 7, inflammation gradually subsided and apoptosis levels decreased. By day 14, the level of inflammation had decreased even further (Figure 1(b)–(d)). Importantly, we observed that the granulation tissue started to grow into scaffolds by day 7, whereas collagen scaffolds started to degrade by day 14, with the collagen scaffold pores getting replaced by dense connective tissue. In addition, the effect of bone damage on inflammatory factors in the vasculature appears to be milder than in local tissue. The results showed that pro-inflammatory factors in the vasculature system increased only 1 day after bone tissue injury and then gradually decreased on days 3, 7, and 14 (Figure 1(d)–(g)).

To evaluate the changes of inflammation in rat calvarial bone defect and design experimental procedures. (a) Experimental implementation process, (b) HE, TUNEL, and TNF-α immunohistochemical staining were performed on the local tissue of the defect on days 1, 3, 7, and 14 after manufacturing the skull defect (scale bar (HE and TUNEL): 200 μm, TNF-α: 50 μm). (n = 3), (c and d) quantification of apoptotic+ cells and TNF-α+ cells, and (e–g) changes of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6 in serum during acute period of skull injury (n = 3). Values are given as the mean ± SEM.
Therefore, we believe that the seventh day of bone injury is an appropriate low inflammation time window and can be used as a reference time point for subacute cell transplantation corresponding to the acute phase of inflammation. Because both local and systemic inflammation have already decreased to low levels by this time, and fibrous tissue has not yet fully grown can provide sufficient space for cell transplantation.
The “Two-step” strategy effectively improves the survival rate of the transplanted cells
To investigate the effect of different strategies on the survival of the transplanted stem cells, in vivo imaging techniques were used for continuous tracing of these cells. We observed that the bioluminescence signal in the defect area decreased sequentially from the center to the periphery, and the distribution range exceeded the size of the calvarial defect, suggesting that the transplanted cells spread to adjacent tissues in the local area of the defect. Interestingly, bioluminescence quantification of the primary transplanted cells (Seeding and Injection groups) peaked on day 3, increasing instead of decreasing, as compared to that observed on day 1. We believe that this is due to the incomplete bioluminescence reaction caused by the local hypoxic microenvironment on day 1, as the bioluminescence reaction requires the participation of sufficient oxygen and adenosine triphosphate. As the hypoxia improved, the bioluminescence signal could fit the survival number of stem cells. Over time, the bioluminescence signal gradually weakened and concentrated in the center of the calvarial defect in each group, suggesting that the demise of transplanted stem cells may progress from the surrounding area to the central area. In the Seeding group, the biological signal persisted for 35 days after cell transplantation, while the Injection group showed a more dramatic rate of decline, with the bioluminescence signal becoming undetectable at day 28 after injection. In contrast, the Two-step group extended the bioluminescence signal to day 42, by timely supplementing the system with a second batch of cells, that is, on day 7 (Figure 2(a)). The above results showed that the “Two-step” strategy can significantly improve the survival rate of transplantation in calvarial bone defects.

Effect of different transplantation strategies on the survival rate of transplanted stem cells (n = 4): (a) in vivo imaging techniques showed the survival time of stem cells under different transplantation strategies and (b) quantitative bioluminescence analysis.
The “Two-step” strategy effectively promotes angiogenesis in the early stage of the bone repair environment
We detected CD31 immunofluorescence to observe the effects of different cell transplantation methods on angiogenesis in the defect area. On post-operative day 8, the Blank and Injection groups displayed similar vascularization. CD31+ cells formed linear structures covering the upper surface of the defect area and were sporadically distributed in the bottom layer and margin of the bone defect, while there was little vascular tissue in the central area (Figure 3(a)). In the Seeding and Two-step groups, the number of new blood vessels in the marginal region of the defect increased significantly. However, more vascular tissue formation (p < 0.05) was stimulated in the Seeding group than in the Two-step group, suggesting that angiogenesis may be positively correlated with the amount of primary cell seeding (Figure 3(b)). On post-operative day 14, the vascular tissue in the Blank group remained on the surface of the defect area, and only a single blood vessel was visible in the central area of the defect, while the CD31+ cells in the Injection group increased significantly, as compared to that on the eighth day, but mainly covered the upper third of the defect area. In the Seeding and Two-step groups, CD31+ cells extended from the edge to the central region of the defect, evenly covering the entire longitudinal section of the bone defect. Impressively, the number of blood vessels in the Two-step group exceeded that in the Seeding group (p < 0.05).

Effect of different transplantation strategies on early angiogenesis in skull defects (n = 4). (a) Neovascular tissue labeling by CD 31 (green) in calvarial defects on days 8 and 14 post-surgery (scale bar: 500, 100, and 50 μm). Emphasis was placed on the subperiosteal area on day 8 and on the central area of the defect on day 14. Yellow and red boxes represent locally enlarged images. (b) Quantitative statistics of CD31 positive area. Values are given as the mean ± SEM.
The “Two-step” strategy effectively promotes the repair of a critical calvarial bone defect
H&E and Masson staining were utilized to evaluate the tissue repair of calvarial defects. At 4 weeks, the defect area in the Blank group was filled with a large amount of fibrous tissue, and only a small amount of bud-like new bone tissue was observed at the edges of the two broken ends of the bone defect. In the Injection group, there was a significant increase in the new bone tissue, as compared to that in the Blank group, and the new bone tissue on the lateral side of the bone defect formed an expanded “clubbing” structure, while the medial callus grew a “budding” to the central area of the defect, leaving most of the non-union areas. In the Seeding group, there was an increase in the new bone tissue, as compared to that in the Injection group, and a further reduction in the extent of fibrous tissue. Interestingly, we observed that the medial “budded” new callus crawled faster and covered more defect area than the lateral “clubbed” new bone tissue. In the Two-step group, the medial “budded” new callus reached the lateral “clubbed” callus, achieving bony bridging at both ends of the defect and covering the entire defect area. At the same time, the new bone tissue presented a well-demarcated upper and lower layered structure: an upper lamellar structure composed of layered osteoid and scattered osteocytes and a lower irregular woven structure composed of deeply stained multicellular aggregates. At 12 weeks, most of the non-union areas remained in the Blank group. Both the Injection and Seeding groups had different volumes of new bone tissue renewal, with the Seeding group significantly outperforming the Injection group. In the Two-step group, a sturdier lamella-like structure was formed, and reconstructed medullary canal structures were observed in some areas.
Micro-CT was performed on rat skull samples at 4 and 12 weeks after surgery and the 3D reconstruction and corresponding coronal and transverse images are shown in Figure 5(a). In the Blank group, neonatal bone tissue was limited and distributed only at the edge of the defect area, leaving most of the blank area in the central area of the defect. After stem cell transplantation in different ways, the volume of new bone tissue showed varying degrees of growth and a tendency to grow from the defect margin to the central area. In the Two-step group, the defect area was essentially filled with new bone tissue, and the amount of osteogenesis was significantly better than that in the Seeding and Injection groups. Interestingly, the medial half of the circular defects had significantly more abundant new bone tissue than the lateral half, consistent with the observations upon H&E and Masson’s staining, suggesting that the transplanted stem cells effectively attracted and recruited autologous cells from the medial sagittal suture for bone repair. Quantitative analysis showed that at 12 weeks, the new bone volume increased by 144.27%, 81.46%, and 50.81% in the Two-step group, as compared to that in the Blank, Injection, and Seeding groups, respectively. In summary, the “Two-step” strategy demonstrated a superior bone-promoting repair effect.
The “Two-step” strategy for transplantation actively regulates the inflammatory microenvironment in the early stage of bone injury
To determine the effect of different transplantation strategies on the microenvironment at the early stage of bone injury, we further investigated changes in apoptotic levels and inflammatory factors in bone defects. TUNEL assay showed that cell transplantation resulted in increased expression of apoptotic factors in the injured environment. On the eighth day after surgery, the levels of apoptosis in the Seeding and Two-step groups were significantly higher than those in the Blank group. However, on the 14th day after surgery, the apoptosis level in all groups was lower than that on the 8th day, with a more significant decrease in the Two-step and Seeding groups, while the apoptosis level in the Injection group decreased slowly. We found that the same number of stem cells caused a significantly milder apoptotic response at day 7 compared to day 0, suggesting that the level of histiocyte apoptosis is influenced by the time of transplantation. Meanwhile, apoptosis levels in the Two-step group were comparable to those in the Seeding group.
To further investigate the regulatory effect of stem cell transplantation on local inflammation, we examined anti-inflammatory and pro-inflammatory factors in bone defect tissues. In terms of anti-inflammatory factors, IL-4 and IL-10 were significantly increased in the Seeding and Two-step groups compared with the Injection and Blank groups on postoperative day 8, suggesting that early cell transplantation could promote anti-inflammatory factor secretion, while on postoperative day 14, the Two-step group had higher anti-inflammatory factor secretion compared with the Seeding group (p < 0.05). In terms of pro-inflammatory factors, we observed a synchronous rise in pro-inflammatory factors (TNF-α, IL-1β, and IL-6) with anti-inflammatory factors in the Two-step and Seeding groups at 8 days after surgery, however, by 14 days after surgery, these pro-inflammatory factors were significantly decreased in all groups. This suggests that early transplanted stem cells promote secretion of pro-inflammatory factors, but this effect is transient and has decreased to low levels before day 14 after surgery. The data presented above suggests that stem cell transplantation promotes a shift from a pro-inflammatory to anti-inflammatory state in the local bone injury microenvironment, whereas the Two-step strategy is more successful in pro-inflammatory - anti-inflammatory transformation due to higher secretion of anti-inflammatory factors.
Effect of the “Two-step” strategy on the macrophage phenotype in the early bone injury environment
To observe the local macrophage expression in the defect, we used the surface markers CD68 (red) and iNOS (green) to label M1 macrophages. On post-operative day 8, only a few M1 macrophages were sporadically distributed at the edge of the defect area. In the Injection group, however, the number of M1 macrophages increased, as compared to those in the Blank group, and showed a tendency to migrate toward the central region of the stent. In the Seeding and Two-step groups, there was a significant increase in the number of M1 macrophages, suggesting that early cell transplantation has a recruiting effect on M1 macrophages. M1 cells were predominant in the Seeding group, whereas a small number of CD68+iNOS− cells appeared in the Two-step group, suggesting that the phenotype of macrophages may have transformed. On post-operative day 14, the number of M1 macrophages decreased in the Blank group, while M1 macrophages increased slightly in the Injection group, as compared to those observed on day 8. In the Seeding group, macrophages were predominantly of the M1 phenotype and presented with few CD68+iNOS− cells, whereas in the Two-step group, the M1 phenotype decreased and the proportion of CD68+iNOS− cells increased. We further validated the phenotype changes in the M2 macrophages (yellow) by detecting the cell surface markers CD68 (red) and CD206 (green). On Post-operative day 8, the macrophage phenotype was predominantly CD68+CD206− in the Blank, Injection, and Seeding groups, whereas the number of yellow-stained M2 cells was minimal. In contrast, there was a significant increase in the M2 cells in the Two-step group. Macrophages showed a more pronounced phenotype transformation on Post-operative day 14. The number of M2 macrophages in the Blank and Injection groups did not change significantly from day 8 after surgery. In the Seeding group, local M2 macrophages increased in number, as compared to those observed on day 8, but more M1 macrophages were mixed. However, the proportion of M2 macrophages increased in the Two-step group, as compared to that in the Seeding group.
Transcriptome sequencing reveals potential functions of first transplanted cells of the “Two-step” strategy
RNA-seq is crucial for deciphering the structure and function of the genome and identifying changes in relevant signaling pathways. We utilized RNA-seq to further explore the early immune response and related mechanisms after stem cell implantation. Therefore, we transplanted varying quantities of stem cells into rat cranial bone defects for 7 days and extracted total cellular mRNA from the defects, for transcriptome sequencing.
DEG analysis revealed 241 up-regulated DEGs and 81 down-regulated DEGs in the Two-step group compared with the Blank group, while 33 up-regulated DEGs and 47 down-regulated DEGs in the Seeding group compared with the Two-step group. Of these, only seven differential genes were co-expressed genes. Volcano map further showed significant differences in DEGs between the Two-step and Blank groups, indicating that stem cell transplantation significantly affected gene expression in the bone defect area, while DEGs between Two-step and Seeding groups were significantly reduced compared with that between the Two-step and Blank groups, demonstrating that differences in the number of transplanted stem cells at the early stage of bone injury may have little effect on gene expression (Figure 8(a)–(d)).
DEG analysis showed a large number of differentially expressed genes between the Two-step and Blank group. However, the biological functions resulting from these differences remain unclear. Therefore, we performed GO enrichment analysis (Figure 8(e)–f)). At the level of biological process (BP), the Two-step group showed a significant up-regulation of chemokines and cell surface receptors compared with the Blank group, suggesting that there is a varied communication activity between transplanted stem cells and host cells, which can be confirmed in the chemotaxis of macrophages. In addition, the inflammatory response, immune response, and positive regulation of immune system in DEGs observed synchronous up-regulation, which indicated that transplanted stem cells stimulated positive regulation of inflammation and pro-inflammatory—anti-inflammatory transition while causing tissue inflammatory response. Subsequently, KEGG enrichment analysis was conducted to further explore the potential signaling pathways leading to the functional changes (Figure 8(h) and (i)). In the Two-step group, the expression of signaling pathways such as cytokine-cytokine receptor interaction, Th1 and Th2 cell differentiation, cell adhesion, and antigen processing and presentation were upregulated, while the expression of the IL-17 signaling pathway, adipocyte differentiation, osteoclast differentiation and mineral absorption were significantly downregulated.
Furthermore, the STRING database was employed to analyze the protein-protein interaction network between the Two-step and Blank groups (Figure 8(g)). The results showed that several key proteins formed a tightly connected network; the enriched protein were primarily involved in T cell activation (such as Zap70, Gata3, Txk, Foxp3, and Itk) and glycolysis (such as Pygm, Eno3, and Trib3), which may play important roles in regulating immune homeostasis and tissue regeneration.
Discussion
Bone defect is a difficult point in the treatment of clinical orthopedic diseases, which often leads to nonunion due to large bone loss combined with infection, soft tissue injury, vascular injury, and other reasons. 31 Stem cell therapy is the most important part of bone tissue engineering besides scaffolds and cytokines, but the low survival rate caused by adverse factors such as hypoxia or nutritional deficiency, severe inflammation, mechanical stress, and immune rejection greatly affects the therapeutic effect.32,33 At present, many studies have carried out multi-faceted discussion on improving the survival rate of stem cell transplantation and achieved good results, such as: gene programming, preconditioning, bioaffinity material encapsulation, and so on.34,35 In this study, a collagen scaffold with a single component (Figure S1) was applied and a tissue engineering strategy was proposed to optimize the timing of stem cell transplantation based on inflammatory changes in calvarial bone defects.
Bioluminescent imaging results showed that application of this strategy resulted in stem cells surviving more than 42 days in bone defects. At present, several studies have demonstrated that the effect of stem cell therapy and cell survival time have a significant dose-dependent effect. For example, Kawabori et al. 36 demonstrated that high-dose stem cells (100 × 104) achieved good engraftment rates and brain function recovery in both the subacute and chronic phases of cerebral infarction treatment, while low-dose stem cells (10 × 104) achieved the same therapeutic effect only when transplanted at a specific time. 37 Actually, we have observed that high doses of cells (100–500 × 104 cells) are now widely used in preclinical studies of bone repair models,21,38,39 whereas in contrast, the “Two-step” strategy achieved higher cell survival rates using lower doses of stem cells (50 × 104 cells). This strategy effectively improves stem cell utilization, which is crucial for shortening the in vitro expansion time of stem cells and timely replantation into patients during the clinical treatment. We believe that cells transplanted in the acute phase provide conditional support for their own survival by actively responding to microenvironment changes, while stem cells timely supplemented in the subacute phase achieve the treatment effect of “1 + 1 > 2.” Delivery of transplanted stem cells in the acute phase of bone injury (Seeding group) resulted in a higher survival rate of cells than that in the subacute phase (Injection group). We attribute this phenomenon to the ability of stem cells to regulate the acute phase microenvironment. RNA-seq results suggest that cell therapy produces more abundant chemokine and cell surface receptor signals than the cell-free transplantation state (Figure 8(e)), suggesting extensive cell-to-cell crosstalk and abundant biochemical responses between transplanted cells and host cells in the acute phase. These responses may promote survival of transplanted stem cells themselves, as also supported by the decrease in negatively regulated genes for cell growth observed in RNA-seq results (Figure 8(f)). In addition, stem cells in the Injected group showed a faster rate of disappearance after delivery than in the Seeding group, possibly suggesting that the environment in the subacute phase may be unsuitable for cell survival. However, it should be noted that the low cell survival rate in the Injected group may be related to the mode of cell transplantation. Compared to the mode of in vitro seeding, the injected cells have lower affinity and adhesion to the scaffold material, which may indirectly affect the survival rate.32,40 However, despite the low affinity of the injection procedure, the survival rate of the second batch of cells was sufficiently prolonged under the “Two-step” strategy, thus, this again confirms that the improvement of the host environment by the first batch of cells has an important supporting effect on the survival of the second batch of cells. Therefore, this strategy simultaneously overcame the defects of insufficient repair cells in the Seeding group and unfriendly environment in the injected group.
The survival rate of transplanted cells is also closely related to angiogenesis, which provides nutrients and helps scavenge reactive oxygen species and necrotic tissue, improving the tissue microenvironment to facilitate bone repair. 41 CD31 immunofluorescence showed that transplantation of stem cells during the acute phase promoted tissue angiogenesis, while a “Two-step” strategy promoted a greater number of vessels in the central area of the defect. Previous studies have shown that neovascularization in injured tissue grows slowly at a rate of several tenths of micrometers per day, so scaffold materials used for bone tissue engineering often fail due to insufficient blood supply to the central area.42,43 We observed that the proangiogenic effect of stem cells was stronger in Seeding than Two-step group at 8 days after surgery, while the opposite was observed at 14 days after surgery (Figure 3(a)). These results suggest that the survival time of the second batch of stem cells was prolonged and a more active proangiogenic effect was exerted when the first batch of cells improved the local microenvironment. Previous studies have shown that vascular networks can provide nutrients, oxygen, growth factors, cytokines, and osteogenic precursor cells and play an important role in bone growth and development, remodeling, and bone repair.44,45 We observed a similar phenomenon, with abundant neovascularization invading the central area from the periphery of the stent, and a large amount of neo-osseous tissue formed at the base of the defect in the Two-step group. Interestingly, there is a clear stratification of the newly formed bone tissue: the upper layer is lamellar bone, while the lower layer is irregular woven bone with dense nuclei, which we suspect may belong to osteogenic precursor cells (Figure 4(a)). In addition, we observed more abundant new bone tissue in the medial area of the defect (near the cranial suture) than in the lateral area of the defect (Figure 5(a)). Because the cranial suture contains abundant host endogenous stem cells,46,47 we hypothesize that a “Two-step” strategy effectively recruits endogenous osteoblasts and participates in tissue repair.

Effect of different transplantation strategies on bone regeneration (n = 4). HE and Masson staining of skull tissue was performed at week 4 and week 12 after surgery. Red boxes represent locally enlarged images (scale bar: 500 and 100 μm). Fb means fibrous tissue, Nb indicates newly formed bone, *lamellar bone, #irregular woven bone tissue, black arrows indicate medial (near cranial suture), red arrows indicate lateraland.

Micro-CT to examine bone regeneration under different grafting strategies (n = 4): (a) newly formed bone tissue in the skull defect (3D reconstruction and sagittal and transverse views shown, respectively) was assessed using micro-CT at 4 and 12 weeks postoperatively, with red dashed boxes indicating regions of interest and (b) total BV/TV analysis and BMD of skull samples. Values are given as the mean ± SEM.
Post-transplantation, the cells died mainly in a necrotic or apoptotic manner. In contrast to necrosis, apoptosis is a more controlled mode of programmed cell death within cells, and in it, the cellular contents do not spill outside the cell to cause inflammatory responses. Previous studies have shown that apoptosis plays an important role in the regulation of bone development, remodeling, or dynamic balance of regeneration, and 50%–70% of osteoblasts will undergo apoptosis during bone regeneration. 48 Zhang et al. 49 found that implanted BMSCs can further activate anti-inflammatory proteins, such as TNF-stimulated gene-6, through apoptosis, thereby playing an anti-inflammatory role and promoting tissue healing. In addition, it has been reported that apoptotic cells can promote macrophage/stem cell migration and chemotaxis, which is an important condition for initiating bone repair. However, persistently increased levels of apoptosis are unfavorable for bone repair. Komatsu et al. 50 observed that the reduced apoptosis of osteoblasts in fracture models favors bone formation and proposed that inhibition of apoptosis could be a novel therapeutic direction to promote bone regeneration. In the present study, we observed that stem cells were involved in the regulation of local apoptotic levels in bone defects, and stem cells transplanted in the acute phase elicited a more intense tissue apoptotic response on day 8 (Figure 6); however, a significant decrease in apoptotic levels was observed at day 14. Interestingly, the Two-step group induced lower levels of apoptosis than the Seeding group, both on days 8 and 14. Thus, the step-by-step approach may promote bone regeneration by stimulating limited levels of apoptosis.

Effect of transplanted stem cells on tissue apoptosis and local inflammatory factors (n = 3): (a) TUNEL staining of local skull defect tissues at 8 and 14 days after surgery (scale bar: 200 μm), (b) statistics of TUNEL+ cells, (c and d) detection of anti-inflammatory factors in skull tissue, and (e–g) detection of proinflammatory factors in skull tissue.Values are given as the mean ± SEM.
We observed a pattern similar to apoptotic changes in the detection of inflammatory factors in the defect tissue. A step-by-step approach caused simultaneous upregulation of pro-inflammatory cytokines (TNF-a, IL-1b, and IL-6) and anti-inflammatory cytokines (IL-4 and IL-10) on postoperative day 8, whereas on day 14, anti-inflammatory factors remained high as pro-inflammatory factors steadily decreased (Figure 6). The above results suggest that under the “Two-step” transplantation strategy, the first batch of transplanted stem cells caused limited inflammatory stimulation to some extent, while the second batch of stem cells contributed to the pro-inflammatory - anti-inflammatory transition. Previous studies have shown that osteogenically differentiated BMSCs have similar immunomodulatory behavior compared to undifferentiated cells, both of which lack immunogenicity because major histocompatibility complex class II and immunostimulatory molecules (e.g. CD80 and CD86) are expressed at lower levels.51–53 However, it has also been pointed out that when stem cells are exposed to IFNγ, their immunogenic markers are upregulated, which predicts the potential ability of stem cells to induce immune responses. 54 Besides, cell transplantation will exacerbate cellular oxygen consumption and cell death due to vascular loss at the early stage of injury, which will further induce inflammation. But the pro-inflammatory effect of transplanted stem cells in vivo is transient and limited as it manifests only during the first 8 days after surgery, concomitant with up-regulation of anti-inflammatory factors. The prevailing consensus is that inflammatory exposure contributes to the immunosuppressive effect of stem cells. One study showed that addition of sufficient amounts of IFN-γ and TNF-α contributed to achieving MSCs inhibition of T cell proliferation, 55 and some researchers have used pro-inflammatory factors to pretreat MSCs to enhance their immunosuppressive ability.56,57 Therefore, we hypothesize that a large number of pro-inflammatory mediators released at the initial stage of bone injury events, such as TNF-α and IL-1β, will activate the immunosuppressive phenotype of the second batch of stem cells and thus exert anti-inflammatory effects. The initial and optimal transient phases of acute inflammation are critical factors for successful and robust bone healing, and we believe that and the two batches of cells in this strategy may automatically adapt to environmental changes and play pro-inflammatory (limited) and anti-inflammatory roles, respectively, laying the foundation for successful bone repair.
After tissue engineered grafts were implanted in vivo, pro-inflammatory macrophages (M1) and anti-inflammatory macrophages (M2) appeared successively at the host-implant interface to play a role in removing necrotic tissue and promoting tissue healing, respectively,58,59 while macrophage recruitment and M1–M2 phenotypic transformation were considered to have a positive bone repair effect.60,61 In this study, we observed that stem cell transplantation significantly promoted macrophage chemotaxis (Figure 7). Meanwhile, stem cells injected in the acute phase elicited a more drastic macrophage response than cells transplanted in subacute phase, but Two-step group had the most abundant M2 macrophages at day 14, suggesting a higher M1–M2 turnover rate. These data are consistent with changes in pro-inflammatory and anti-inflammatory factors. A large number of studies have now reported that MSCs release M2 inducers (such as IL-4) when co-cultured with macrophages in vitro,62–64 and we speculate that this may be a key factor in the phenotypic transformation of macrophages driven by stem cell in this strategy. At present, it is generally accepted that the phenotype of macrophages is closely related to bone injury repair, and the reasons are various. Firstly, M2 macrophages have been shown to promote MSCs survival rather than M1 macrophages. 65 Secondly, M1 and M2 cells co-orchestrate to exert a proangiogenic effect. It has been shown that M1 macrophages can promote angiogenesis by secreting inflammatory factors such as VEGF and TNF-α, however M2 macrophages stabilize angiogenesis. 66 Finally, M1 macrophages promoted BMSC migration in vitro, whereas M2 macrophages significantly promoted BMSC survival, proliferation, and osteogenic differentiation. 67 Many strategies have been derived to modulate macrophage immunophenotype to promote bone repair.58,68 Therefore, we believe there is a close communication between the surviving exogenous stem cells and host macrophages, thereby directing bone repair.
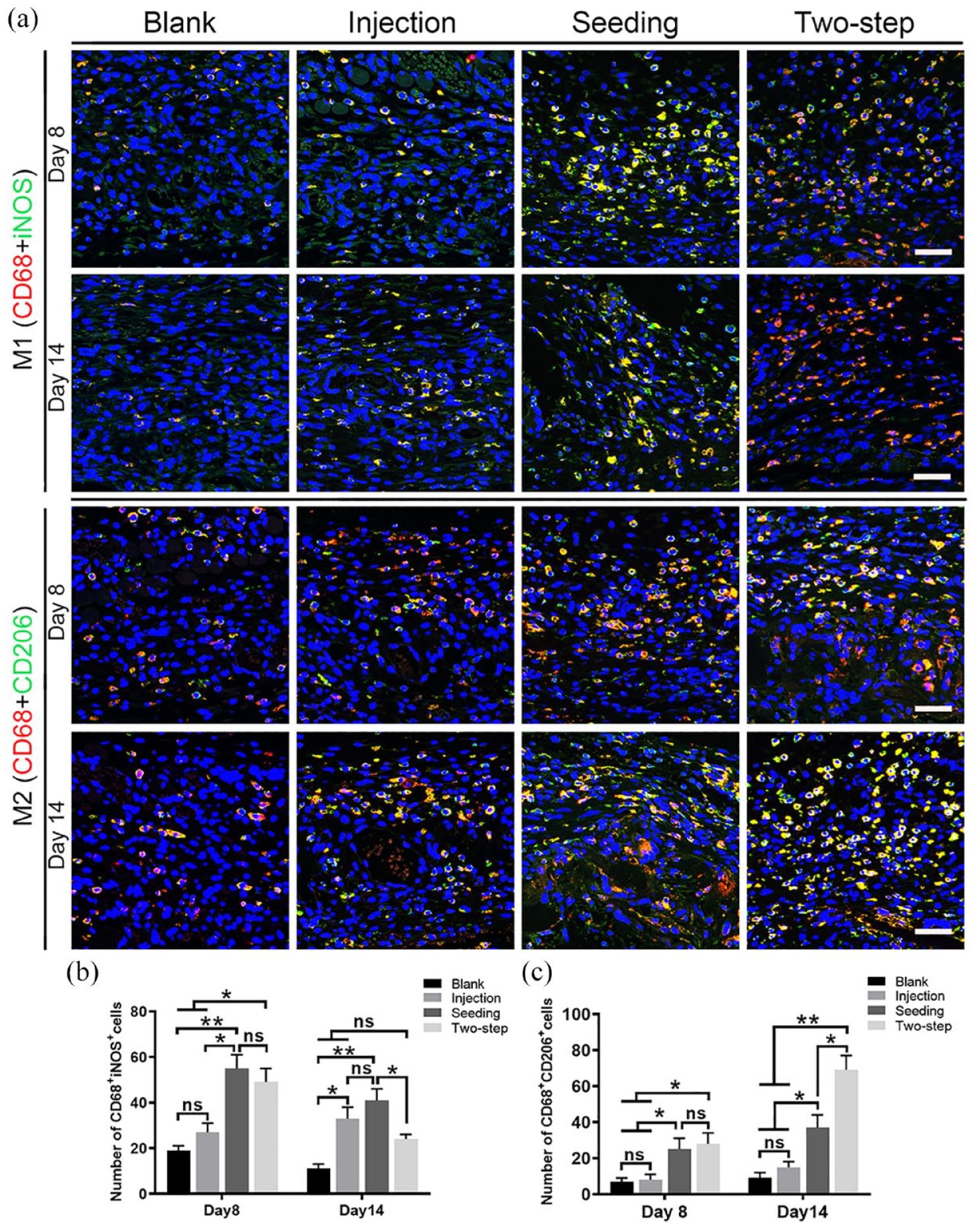
Effect of transplanted stem cells on macrophage phenotype in the defect (n = 4): (a) Immunofluorescence staining of M1 and M2 macrophage on calvarial defects on day 8 and day 14 after surgery (scale bar: 50 μm) and (b and c) number statistics for CD68 + iNOS+ cells and CD68 + CD206+ cells. Values are given as the mean ± SEM.
Bone regeneration is a large, complex, and highly coordinated process that consists of various complex biological processes and a variety of biochemical reactions. To comprehensively investigate the role of stem cells transplanted in the acute phase, we focused on the local microenvironment differences before the second batch of cell transplantation using high-throughput RNA-seq analysis techniques. Differential gene analysis showed that there was a large difference in gene expression between the Two-step d7 and the Blank d7 group, compared with the differential gene between the Two-step d7 and Seeding d7 group (Figure 8(a)). We suggest that this result may predict the outcome of critical bone defect treatment, while demonstrating high similarity between half-dose and full-dose stem cells in terms of the gene expression that underlies the success of the second batch of cells in the “Two-step” strategy. Significant differences in clot formation, cytokine profiles, and cell populations have been reported in the early bone immune environment between normal and delayed bone healing. 69 Therefore, we used GO enrichment and KEGG pathway analyses to further determine the specific cell activity in the bone defect, and found that stem cell transplantation promoted biological processes (BP) such as proliferation and differentiation, migration, cell chemotaxis, and adhesion of cells in the local environment, which predicted that this strategy induced an active cell state in the defect tissue. Correspondingly, we observed that local immune responses were also active on day 7 after stem cell transplantation. This is in line with the macrophage M1/M2 infiltration and up-regulation of anti-inflammatory factors in tissues. Similar results were observed in the study by Lienau et al., 70 who compared hematoma samples from animal models of healing versus non-healing fractures and found that genes involved in wound response, including inflammatory response, were more highly expressed in the standard healing group. In addition, a recent comparative study of normal healing fractures versus non-healing bone defects pointed out that innate and adaptive immune-related genes were significantly down-regulated in the non-healing bone defects group, suggesting that related biological processes could not be activated. 71 Therefore, it is reasonable to suggest that adequate activation of the immune system in the early stages of injury is a critical event for successful repair of bone defects.
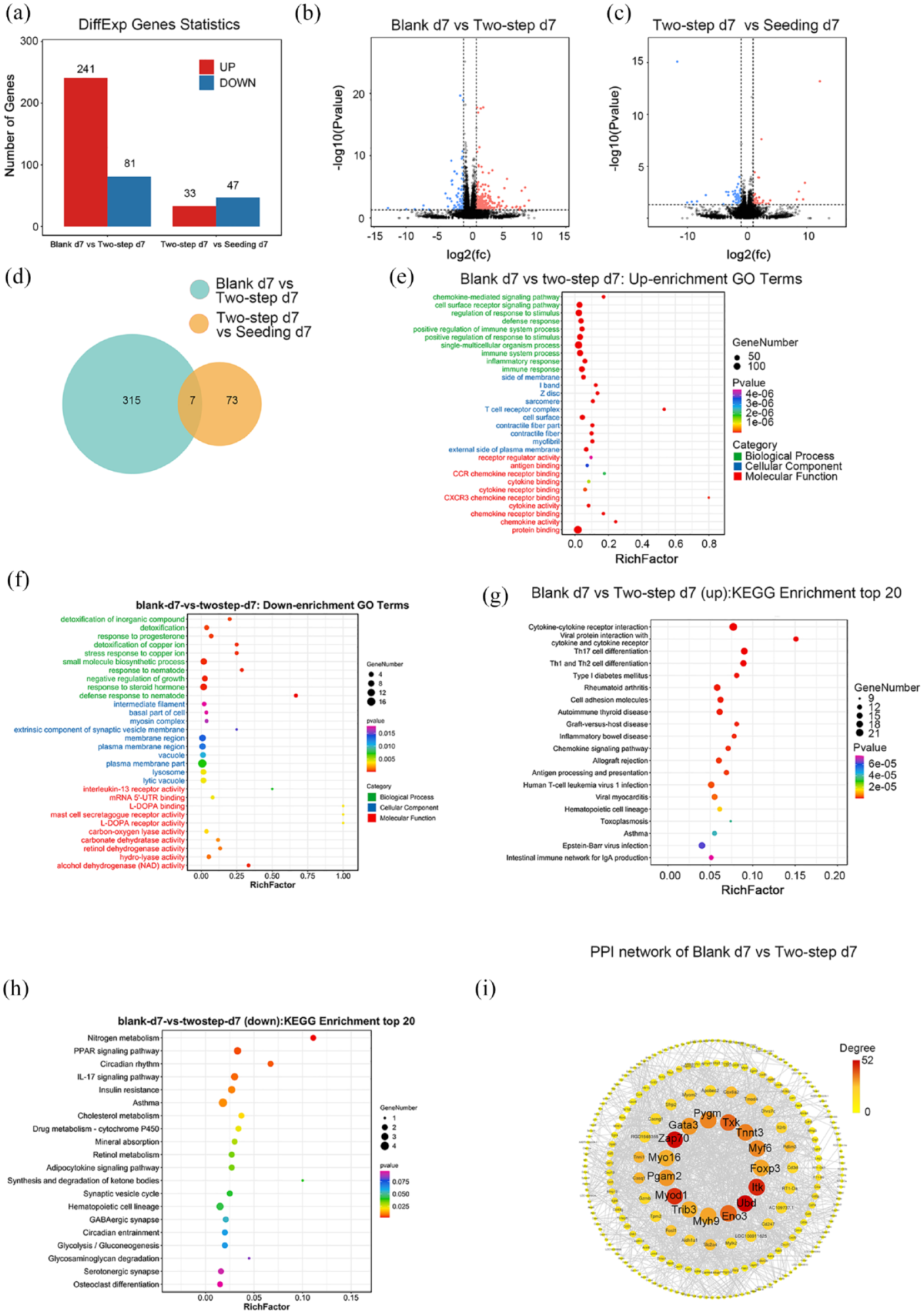
Transcriptome sequencing analysis of Blank, Two-step, and Seeding groups at 7 days post-surgery (n = 3): (a) statistics of number of differentially expressed gene (DEGs) between groups, (b) volcano map of DEGs in Blank group compared with the Two-step group, (c) Volcano map of DEGs in Two-step group compared with the Seeding group, (d) venn diagram of the DEGs between different groups, (e and f) GO enrichment analysis of DEGs, (i) The PPI network between the Blank group and Two-step group, and (h and i) KEGG enrichment analysis of DEGs.
This study has some limitations: The data confirmed that the primary transplanted cells play an important role in regulating the inflammatory microenvironment locally in the skull, but the specific role of the secondary transplanted cells still remains to be studied. Thus, future studies should be conducted exploring this angle. At the same time, the calvarial defect was repaired by means of intramembranous osteogenesis and we did not study endochondral osteogenesis as a mechanism of repair. Future studies could further verify the effectiveness of the “Two-step” transplantation strategy in the femoral defect model, which uses endochondral osteogenesis.
In conclusion, this study demonstrated that administration of BMSCs during the early acute inflammatory phase of bone injury rather than after stabilization of inflammation can better provoke the immunomodulatory effects of BMSCs. At the same time, timely supplementation of BMSCs in the subacute phase facilitates the repair of bone defects because the adverse microenvironment in the acute phase of bone injury can cause the loss of transplanted stem cells. More importantly, the survival rate and repair efficiency of low-dose stem cells can be improved by selecting personalized and accurate transplantation timing according to the changes of inflammation, thereby presenting a novel idea for the selection of the timing of bone defect treatment.
Supplemental Material
sj-docx-1-tej-10.1177_20417314241252960 – Supplemental material for Timely delivery of bone marrow mesenchymal stem cells based on the inflammatory pattern of bone injury environment to promote the repair of calvarial bone defects in rats: An optimized strategy for bone tissue engineering
Supplemental material, sj-docx-1-tej-10.1177_20417314241252960 for Timely delivery of bone marrow mesenchymal stem cells based on the inflammatory pattern of bone injury environment to promote the repair of calvarial bone defects in rats: An optimized strategy for bone tissue engineering by Yuwei Zeng, Aiju Lou, Zhenmin Zhong, Yu Cai, Yixi Yang, Haifeng Liang, Yucong Lin, Zhuoxuan He, Lei Zhou, Zhi-Yong Zhang and Le Wang in Journal of Tissue Engineering
Footnotes
Acknowledgements
Authors would like to thank Dr. Wenhao Wang and Dr. Zhanzhao Zhang for helpful suggestions, as well as we thank Guangzhou Kidio Biotechnology Co., Ltd. for providing RNA-seq related analysis support.
Author contributions
Yuwei Zeng, Aiju Lou, and Zhenmin Zhong designed the study, analyzed the data, and wrote the paper. Yu Cai, Yixi Yang, Haifeng Liang, Yucong Lin, and Zhuoxuan He conducted in vitro and in vivo experiments. Lei Zhou, Zhi-yong Zhang, and Le Wang designed the study, provided financial support, and interpreted the data for the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (Grant Nos. 82072409 and 51903087), the Natural Science Foundation of Guangdong Province (Grant No. 2019A1515012020), and the Science and Technology Plan Project of Guangzhou City (Grant No. 202102010008 and 2024A03J1000), Guangdong Medical Research Foundation (Grant No. A2024661).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
