Abstract
Nanoparticle-based therapeutics delivery holds great promise for the treatment of intractable diseases. The high loading of drug molecules and their precise delivery to target sites are needed to gain optimal therapeutic functions of the nanoparticle delivery system. In this communication, we highlight, among other properties of nanoparticles (e.g. size, shape, surface chemistry, and degradation), the nanoscale topography, which has recently been shown to be an important parameter, ultimately determining drug loading, cell penetration, and body clearance. This nanotopographical aspect is considered to offer a new effective strategy to the development of nanoparticles for drug and gene delivery with enhanced therapeutic outcome.
Delivery with nanoparticles and the parameters to consider
The delivery of therapeutics (drugs, proteins, and genetic molecules) to intracellular compartments is a critical issue in current nanomedicine. Loading efficiency, cellular uptake, targeting, and release profile are key among the considerations in the delivery process to maximize the therapeutic potential of the delivery systems. Nanoparticles (NPs) are developed to load more cargo molecules and then release sustainably;1–3 NPs are modified to target cells or subcellular components (e.g. mitochondria, nucleus);4–7 NPs are functionalized to respond specifically to environment (e.g. pH, temperature, enzyme, and electricity);8–10 and NPs are also tailored to allow imaging for tracking and diagnosis purposes.3,11–13
Over the last three decades, many NP-based delivery systems, consisting of organics (liposomes, dendrimers, and synthetic polymers), inorganics (silica, iron oxide, gold, and quantum dots), and their composites/hybrids, have been developed.14–16 Typically, NP parameters including shape, size, and surface chemistry have been carefully designed to optimize the loading and delivery capacity. For example, gold NPs with different sizes have been shown to penetrate cells at different rates. 17 Among the sizes examined, 45-nm Au NPs exhibited the highest uptake which was considered optimal in minimizing cell membrane energy. 17 Also, the variable shapes and sizes of gold NPs showed different cell penetration mechanisms. 18 The mesoporous silica nanoparticles (MSN) with a high aspect ratio were shown to be taken up by cells more easily than those with a low aspect ratio.19,20 NPs were also often modified to have pores, channels, or internal cavity to increase the loading of therapeutic molecules.21–23 Silica NPs are the exemplar carriers that exhibit tunable pore structure and thus merit effective molecular loading.
Among other designs of NPs, the surface modification has been a powerful tool to effectively load cargo molecules, to penetrate cell membrane, to escape lysosomal degradation, and to target intracellular component space. For example, MSNs modified with poly-
Therefore, the physical and chemical properties of NPs are of utmost importance in determining the delivery capacity of drugs and genes and their therapeutic efficacy. Among other physicochemical parameters that can affect the cellular uptake and delivery capacity of NPs (as illustrated in Figure 1 and Table 1), recently, the surface nanoscale topography of NPs has gained special attention; it is considered to control the interactions with biological entities, such as bioactive molecules and cells, in the process of molecular loading and cell penetration.30–43 While the nanotopographical impact of scaffolds and matrices on cell responses has been substantially disseminated,44–48 such aspect in the cellular interactions with NPs has recently emerged. In this short communication, we aim to highlight the nanoscale topological effects of NPs on the therapeutics loading and delivery into cells that may advance the capacity of NP-based delivery systems for the treatment of diseased tissues.

Schematic drawing of the physicochemical parameters of NPs that can affect the cellular uptake and delivery efficiency.
A brief summary of NP parameters (e.g. size, shape, elasticity, and chemistry) that influence the delivery of therapeutic molecules.

NP: nanoparticle; MSN: mesoporous silica nanoparticles; PEI: polyethyleneimine; DNA: deoxyribonucleic acid; DNP: DNA nanoparticle; DOX: doxorubicin; SNP: silica nanoparticles; R-MSHS: rough hollow silica with pollen-like structure; NIR: near-infrared; HPMO: hollow periodic mesoporous organosilica; BBB: blood–brain barrier; PDT: photodynamic therapy; MRI: magnetic resonance imaging; FL: fluorescence.
Key example studies are also referenced.
Effects of surface nanoscale topography
For the improved therapeutic efficacy of delivery systems several properties of the nanocarriers are important, which include (1) higher loading capacity, (2) better interaction with cell membrane, (3) better cell penetration and favorable internalization pathway, (4) better lysosomal escape, and (5) higher integration with genome (for certain genetic molecules). The topography of NPs is considered to affect the interactions with biological systems. In this part, we discuss the topography-dependent properties and the related biological responses, including biomolecules loading, cell membrane adsorption, cell uptake and pathway, and body clearance.
Biomolecular loading
The surface nanoscale topography of particles has significant effect on the biomolecular loading. Recently, the effect of PEI-modified silica nanoparticles (SNP) with different surface roughness on pDNA binding was reported for gene delivery (Figure 2). 34 Three different surfaces (rambutan with spike, raspberry with hemisphere, and flower-like with bowl-type) were designed, and in particular, the surface-roughened nanostructures could be generated by the assembly of resorcinol–formaldehyde resin and silica primary particles under Stöber synthesis condition. The outer surface was then PEI-modified. Interestingly, the rambutan-like NPs (Ram-SNPs-PEI) with spiky surface showed the highest pDNA absorption capacity (133 ng/μg) and the resultant high transfection efficacy of 88%, compared to other NPs. The unique surface nanoscale topography was illustrated to protect the DNA molecules effectively against nuclease degradation with respect to other structures. Another report showed the effect of surface roughness of SNPs with octadecyl hydrophobic modification (C18) on the protein loading. 35 The rough surface NPs (C18-RSN) showed the adsorption capacity of RNase A biomolecules at significantly higher levels (~twice) than the smooth ones. Moreover, the rough surface NPs showed more sustained release profile. In another study, the rough hollow silica with pollen-like structure (R-MSHSs) was demonstrated to load lysozyme protein more (~4 times) than the smooth ones, which further exhibited sustained lysozyme release (up to 72 h) and effective antimicrobial activity toward Escherichia coli at day 3. 36

Effects of surface nanoscale topography on the pDNA loading efficiency: (a) schematic representation of 3D model images of nanotopography design for plasmid DNA delivery; rambutan, raspberry, and flower-like morphologies developed; (b) TEM images of different NPs; and (c) pDNA loading capacity of different NPs.
Among the nanotopological surface designs, virus-like surfaces have gained great attention as drug carriers. For example, MSNs with virus-like topography exhibited higher loading of drug compared with smooth ones and thus better therapeutic efficiency. 30 The virus-like NPs loaded with doxorubicin (DOX) displayed higher tumor cell killing efficiency (42%) compared to conventional NPs (28%). 31 The carrier-free nanodrug system with DNA decoration was designed to mimic virus surface. 32 The nanodrug could load DOX more efficiently (97.5%), suggesting to be effective for gene/chemo codelivery system in cancer therapy. The virus mimicking surface topography was also demonstrated to enhance the adsorption of genetic molecules, including oligoDNA and siRNA, 33 regardless of the surface charge and cell type. Taken together, the NP surfaces roughened at the nanoscale might have enhanced surface area that allows more interactive sites with genetic molecules and drugs, ultimately leading to increased loading capacity. In this case, the size and charge of the molecules to load should be carefully taken into account in the design of NPs.
Cell membrane adsorption
There is growing interest in how the surface topography of NPs interacts with cell membrane as the cell surface interaction is the first step for NP uptake by the cell. The physicochemical properties of NPs such as size, shape, and surface charge have been demonstrated as the typical parameters that prompt cellular interaction and adhesion. Among others, the surface nanoscale topography provides different surface area and energy to the NPs, influencing the interactions with biological entities including cell membrane molecules. In the study by Song et al., 36 the silica nanopollens with engineered rough surface were designed to enhance the bacterial adhesion (Figure 3). By introducing resorcinol, formaldehyde, and tetraethyl orthosilicate into a typical Stöber synthesis solution, a nanocomposite was assembled by selectively forming a resorcinol–formaldehyde core first, followed by a co-condensation of silica and resorcinol–formaldehyde interpenetrating shell. After calcination, silica nanopollens with accessible inner cavity and spiky surface could be obtained. The nanopollen surface was considered to have multivalent interactions induced by the surface spikes during the contact with the hairy bacteria surface (the number of particle contacts to bacteria was ~10 times higher in nanopollen surface). Because of the stronger adhesion via a large number of contacts, lysozyme was released more slowly, being enriched on the bacterial (E. coli) surface. This silica nanopollen-based lysozyme formulation exhibited potent antibacterial activity. In another report, MSNs with three different surface topologies, including smooth, wrinkle, and rough spiky surface, were developed as nanocarriers and modified with aminopropyl groups to load Ag for antibacterial application. The wrinkle and rough spiky surfaces exhibited higher antibacterial activity because of direct interaction of Ag NPs with the bacterial cell membrane. 49

Silica nanopollens developed for bacterial membrane adhesion and antibacterial activity: (a) schematic showing the synthesis of silica nanopollens and the lysozyme-loaded nanopollens adhesive to bacterial surface; (b) SEM images of smooth and rough NPs adhered to Escherichia coli surface; and (c) NP amount adhered to bacteria measured by ICP-OES analysis.
As to the nature of the rough particle–cell membrane contact, nonspecific binding forces were proposed by Nel and colleagues. 50 Based on simulation, the nanotopographical surface roughness was demonstrated to decrease repulsive interactions (hydrophilic and electrostatic) significantly, thereby promoting adhesion and cellular uptake. 51 Similarly, another group reported that the surface roughness of self-assembled block copolymer NPs significantly enhanced the cell adhesion and entry into cells due to the significantly reduced repulsive interactions. 38 The incorporation of cyclodextrins in poly(anhydride) NPs also increased the bio-adhesive capacity which was correlated with the enhanced surface area. 52 Therefore, the increased contact of particles with cell membrane molecules is considered as the primary reason for the higher cell binding of the NPs with nano-roughened surfaces; however, the detailed binding mechanism is yet to be elucidated, which requires further investigation.
Intracellular uptake and pathway
The intracellular uptake rate and the endocytic cellular pathway of NPs are affected by many physicochemical properties of NP. While studies have mainly focused on the size, shape, surface charge, functional group, and hydrophilicity of NPs, the effect of nanoscale surface topography of NPs on cellular uptake and pathway has recently been highlighted. Among the endocytosis pathways (clathrin-mediated, caveolae-mediated, clathrin/caveolae-independent, and micropinocytosis), the clathrin-mediated pathway is known to bring about ineffective drug delivery due to the lysosomal degradation of particles. Interestingly, the rough surface NPs have been shown to effectively avoid this pathway by adopting the other pathways. The MSNs designed to be virus-like displayed higher cellular internalization than the conventional MSN, 31 which utilized the caveolae-mediated endocytosis and macropinocytosis, different from the conventional ones. In another study, the carrier-free nanodrug system was developed based on the virus-mimicking surface decorated with DNA-captured NPs (PEI/DNA@DNPs). The NPs were demonstrated to enhance the gene transfection efficiency, 32 showing an energy-dependent process and the clathrin- and caveolae-independent internalization pathways. Recently, the DNA-loaded nanocomplexes were designed to have virus-mimic surface by decorating with Au NPs. 37 The plasmid condensed with PEI (PEI800) was used as the organic core and the citrated Au NPs were electrostatically decorated to produce virus-surface mimic hybrid NPs. The designed PEI/pDNA/Au nanohybrid significantly enhanced cellular uptake and transfection efficacy (100-fold) when compared with the PEI/pDNA alone without Au NPs and ultimately increased gene/photothermal therapeutic effects (Figure 4). In another study on gene delivery with roughened NPs, the intracellular pDNA trafficking was observed by confocal microscopy. 34 Among the different nanoscale topographies of MSNs, the rambutan-like spiky surface NPs enabled effective protection against nuclease degradation in cells. This work highlights the effect on degradation protection of genetic molecules implemented by the surface nanotopography. Recently, one intriguing study reported the effect on red blood cells. 41 The MSNs engineered with asymmetrical head–tail morphology showed enhanced cellular uptake and higher hemocompatibility due to reduced membrane deformation of red blood cells and decreased level of reactive oxygen species. Taken the studies together, the virus-like or nanoroughened surfaces were demonstrated to significantly enhance the gene transfection efficiency either by altering the intracellular trafficking pathway or by protecting the genetic molecules from an enzymatic degradation.
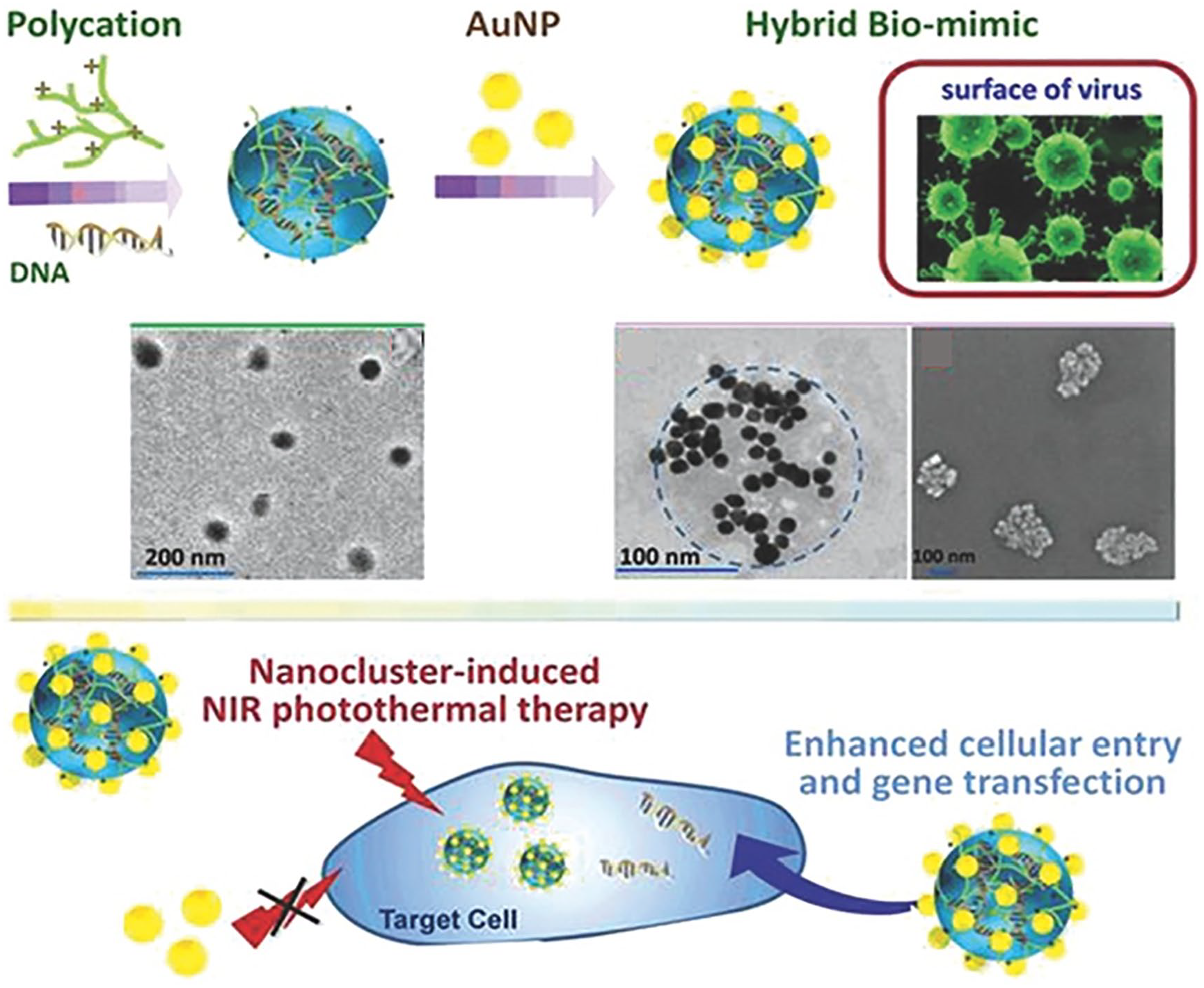
Virus surface-mimicking nanohybrid developed for enhanced cellular entry and gene transfection. Schematic showing the synthesis of DNA-loaded NPs decorated with AuNPs and the TEM and SEM images. Illustration showing the therapeutic efficacy via nanocluster-induced NIR photothermal therapy.
Blood circulation
The blood circulation of NPs with different surface nanotopographies (smooth, porous, and spiky) has been reported with the MSN system. 31 The surface nanotopographies could be obtained by a biphase reaction method with a low surfactant concentration, which allows the coassembly of reactants to take place at the oil–water interface for continuous interfacial/epitaxial growth. By changing the reaction time, the nanostructure was then controlled from porous to spiky surface (Figure 5). The spiky surface obtained with a longer reaction time showed a blood circulation time (t1/2 = 2.16 h) much more extended than the other smooth or porous NPs (0.45 h). In a similar study, the blood circulation was recorded to be much longer for the staggered lamellae NPs with respect to the other NPs of smooth disks and spheres. 38 Such an extended blood circulation was considered to be due to a good serum stability. The carrier-free nanodrug system (PEI/DNA with DOX) also demonstrated prolonged blood circulation time for the rough surface case. 32 Moreover, worm-like micelles were shown to have 1-week blood circulation time, which was approximately 10 times longer than the conventional sphere ones. 53 Thus, these studies highlight the importance of surface nanoscale topography along with particle shape in the interactions with blood and circulation time, which may help designing NPs to control the accumulation and targeted disease treatment with NPs in the future studies.

Controlled surface nanoscale topography of MSNs at different reaction times: (a) 6 h, (b) 12 h, (c) 18 h, (d) 24 h, (e) 36 h, and (f) 48 h. In vivo blood circulation study with different surface nanoscale topographies; after intravenous injection, (g) time-dependent level of NPs in blood and (h) blood circulation half-life (t1/2) calculated.
Concluding remarks
Here, we communicated the importance of a surface nanoscale topography of NPs in the interactions with biological entities (biomolecules and cells). Compared to other physicochemical parameters of NPs (e.g. size, shape, elasticity, and surface chemistry), the surface nanoscale topography has more recently been highlighted. As witnessed in a body of literature, the engineered nanoscale topography of NPs has significant impact on a series of biological events, such as drug/gene loading, cell surface binding, intracellular trafficking, and the stability in blood circulation, all of which are key in determining the therapeutic effects of the NP delivery system. It is thus envisaged that the engineering of NPs with unique surface nanoscale topography may offer an effective design strategy to take full advantage of the physical property of NPs, which may ultimately be helpful for the treatment of diseases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from Dankook University (Priority Institute Support Program in 2019) and National Research Foundation (2018K1A4A3A01064257, 2017R1C1B1011387, and 2018R1A2B3003446), Republic of Korea.
