Abstract
Titanium (Ti) plays a predominant role as the material of choice in orthopaedic and dental implants. Despite the majority of Ti implants having long-term success, premature failure due to unsuccessful osseointegration leading to aseptic loosening is still too common. Recently, surface topography modification and biological/non-biological coatings have been integrated into orthopaedic/dental implants in order to mimic the surrounding biological environment as well as reduce the inflammation/infection that may occur. In this review, we summarize the impact of various Ti coatings on cell behaviour both in vivo and in vitro. First, we focus on the Ti surface properties and their effects on osteogenesis and then on bacterial adhesion and viability. We conclude from the current literature that surface modification of Ti implants can be generated that offer both osteoinductive and antimicrobial properties.
Introduction
Titanium (Ti) and its alloys are commonly used materials in orthopaedic and dental implants due to their mechanical and chemical properties; these include high strength to weight ratio and high yield and fatigue strength along with a relatively low Young’s modulus counteracting the effects of stress shielding. An instantaneously forming passive oxide layer leads to corrosion resistance and biocompatibility.1–5 Moreover, Ti is amenable to alterations in physical and chemical properties, including changing the surface oxide composition, thickness and topography, together making Ti a suitable material for enhancement via surface modification. 6 The biocompatibility of Ti and its alloys are related to the capacity of the Ti oxide layer to react with water ions and serum proteins as well as the resistance to corrosion that provided by the oxide layer.7–10
Scaffold surface features need to be biocompatible, bioactive and perhaps biodegradable as they are replaced by natural tissue during the regenerative process. Replicating the key structures of the extracellular matrix (ECM) and providing stem cell environments are powerful bioactive strategies that material scientists can copy and exploit. 11 Although Ti materials have many favourable properties, there are known potential shortcomings. For example, aluminium in Ti alloys may be associated with neurological disorders. 12 In addition, intra-articular injection of Ti dioxide (TiO2) nanoparticles in rats has been noted to cause toxicological effects in lungs with follicular lymphoid hyperplasia and inflammatory cells aggregated around the bronchia. 13 Moreover, ionic Ti may have a mutagenic effect on cells either directly by damaging DNA via free radicals or indirectly by inhibiting the DNA repair 14 and may also induce some allergic reactions.4,15 The biological response to orthopaedic and dental implants is determined by the physical and chemical features of the implant surface. These include surface topography, surface free energy, oxide thickness and oxide composition. The interaction between cells and the interface will be affected by one or more of these factors and any change in one will affect the other parameters.16–19 Surface topography has the ability to regulate the cell behaviour in a reproducible manner. 20 Furthermore, advances in topographical fabrication are making nanoscale topographical features achievable in a large scale on more complex materials (traditionally only flat surfaces and small surface areas have been able to be patterned at the nanoscale). 21 The use of topography to guide mesenchymal stem cells (MSCs) may, in fact, play a key role in bone tissue engineering as, unlike chemical and mechanical alterations, topographical modifications do not affect the bulk properties of materials and orthopaedic materials need to be able to support load. The stem cells’ ability to adhere and spread into specific surfaces has shown a dramatic effect in cellular development. 22 Osseointegration is the direct contact between bone and the implant, with histological evidence suggesting that new bone is forming around the inert object. The quality and amount of osseointegrated bone around the implant, in addition to other factors such as the degree of inflammation, an excessive force, may affect their stability and consequently their failure rates. 23 Osseointegration and subsequent mineralization is dependent on the initial adhesion of fibrin in blood-mediated osseointegration of osteoblasts or MSCs onto the implant surface.12,24,25 Failure to achieve osseointegration will lead to premature implant failure and this integration is required to be maintained throughout the implant’s lifespan to ensure longevity 26 although patient and surgical related technical/environment factors may also contribute to failure. 27 For instance, among patient factors, male gender, smoking, autoimmune disease and penicillin allergy showed a trend towards greater failure rates.28–30 Late-stage failure tends to occur as a result of implant overloading, wear and peri-implantitis. 31
Moreover, implant infection is the most serious issue after surgery. Biomaterial centred infections (BCI) and prosthetic implant infections (PIIs) have a significant contribution in prosthetic implant failure and aseptic loosening32,33 with the average rate 2%–5%. 34 Host defence mechanisms and current antibiotic treatments become ineffective when bacterial biofilms build up.35,36 However, Ti is generally considered a very safe and highly biocompatible material that has had extensive clinical use for many decades.
Surface properties
Albrektsson and Wennerberg 37 subdivided the implant surface quality into three categories: mechanical properties, topographical properties and physicochemical properties. They conclude that these characteristics are related and by altering any of these groups, the others will also be affected. With Ti, altering the mechanical properties within the physiological range is hard to achieve and so chemistry and topography are the main focus. 37
Biological (in the bone forming sense) materials can be roughly classified into three categories: (1) biotolerant materials where a thin fibrous tissue interface is formed; (2) bioinert materials, like Ti, that can have direct bone contact under osteopermissive conditions; and (3) bioactive materials like calcium phosphate ceramics which can have high degree of direct contact bond with the surrounding bone which is believed to be due to the presence of free calcium and phosphate at the implant interface. 38 More recently, these have been re-categorized as first generation (structural, biocompatible), for example, Ti, second generation (bioactive), for example, hydroxyapatite (HA), bioglass and third generation (reproducible molecular control), for example, nanotopography. 39
Biocompatibility is important to prevent an immune response and foreign body reaction when the material is introduced into the human body. 40 The primary interaction between material and host starts with a thin interface zone, which includes rapid protein adsorption and interaction with the connective tissues. This first interaction is controlled by physical and chemical properties such as roughness, structure, defects and oxide thickness and is critical for long-term implant success.6,41
In this review, we will discuss the importance of Ti surface properties on the bioactivity of implants.
Surface wettability
Wettability is measured by contact angle measurement, usually of water, at the solid/liquid interface while surrounded by a gas phase or another liquid phase and provides gross surface characterization. A low contact angle of less than 90° indicates a hydrophilic surface; the liquid will subsequently spread over the surface. A large contact angle of more than 90° signifies that the surface is hydrophobic leading to droplet of liquid forming on the interface. However, this reaction is controlled by the molecular interaction between the different phases.42,43 Other factors such as surface tension and surface energy are also determined by surface wettability. 44
Liquids can interact with two different types of solid surfaces: high and low energy solid surfaces. Metals, glass and ceramics are examples of solid surfaces with high energy (hard solids) where molecular liquids achieve complete wetting on these solids. The weak solids like fluorocarbons and hydrocarbons have a low energy, where liquid molecules would take a very low energy to break them providing a complete or partial wetting depending on the liquid chosen.42,45
However, increasing the surface wettability may enhance the fibrin adhesion and provide contact guidance for osteoblast migration along the surface. 46 Moreover, any change in surface wettability will affect protein adsorption which consequently changes cell adhesion through integrins and non-integrin receptors. 47
Surface chemistry
The surface chemistry is an important factor in improving the osseointegration. The chemistry of the surface will dictate the interaction of cells with surface proteins in a number of ways: (1) chemical adsorption including covalent bonds and ionic bonds, (2) electrostatic forces found in electrokinetic potential or zeta potential, (3) hydrogen bonds involved in hydrophilic groups, (4) hydrophobic interaction and (5) van der Waals forces. 48 It is known, for example, that osteoblasts are sensitive to subtle differences in surface chemistry. 49
For instance, the fluorine-modified implant surface accelerated osseointegration in the early stage of healing which improved the growth of peri-implant tissue, enhanced the adhesion strength and influenced the osteogenesis gene level.50,51 Also, through control of the oxide chemistry and surface charge, charged antimicrobials may be applied to help fight potential infections. 52 Shibata et al. 53 showed that the TiO formed on the Ti–Cl surface enhanced cell extension and cell growth through a larger adsorption of fibronectin (FN) compared with control, while the TiCl3 contributed to the antibacterial activity of Ti–Cl.
Oxide thickness
Ti, in the presence of air or water, reacts with oxygen to form a protective, chemically stable oxide layer that has the capability to reform immediately after any disturbance. This oxide layer gives the implant increased corrosion resistance, a low rate of ion release and good biocompatibility via plasma protein interactions (e.g. fibrin, fibronectin, vitronectin).6,54,55 An interesting characteristic of the oxide layer is that it can be induced to provide antibacterial behaviour through light excitation without affecting mammalian cell cytocompatibility.
56
For example, light irradiation of amoxicillin gold nanoparticle composites (
Surface roughness and nanostructure
Surface roughness has a vital role in bone healing and enhancing the biomechanical properties by increasing the mechanical retention (interdigitation) and providing good stress distribution. Surface roughness can be divided into three levels: macro-roughness (Ra scale around 10 µm), micro-roughness (Ra scale around 1 µm) and nano-roughness (Ra scale < 200 nm). Ra is an arithmetic average of the absolute values of vertical deviations from a mean plane.46,59
Implant roughness can also be classified depending on feature morphology such as concave textures, for example, HA coating/titanium plasma spraying and convex textures, for example, etching and blasting treatments. 60 Another classification of implant roughness is the orientation of surface irregularities such as isotropic surfaces where topographies are independent of direction and anisotropic surfaces that have a clear direction4,61 (Figure 1).

The difference between anisotropic and isotropic surfaces. (a) Anisotropic surfaces have clear directionality, differ considerably in roughness and the materials properties are not the same at all points or directions. (b) Isotropic surfaces have the same topography independent of measuring direction and the physical property is the same at any point/direction through the material.
In cases of poor bone quality and reduced bone volumes, surface roughness is often used in clinical situations to help accelerate and enhance osseointegration and bone interlocking. 46 Previous studies have shown that the optimal Ra needs to be around 1–1.5 µm; otherwise, the implant fixation would be weakened. 62 Increasing surface roughness can, however, via increased surface area, increase the potential of microbial colonization and provide a shelter to bacteria, hence avoiding removal by antibiotics.26,63 However, previous studies have shown that surface roughness below 0.2 µm was less likely to promote bacterial adhesion as most bacteria are larger in size.64,65 The above studies, however, really only deal with topography for mechanical integration rather than cellular integration (osseoinduction). Surface topography can indeed influence the rate at which bone is formed next to the surface, and perhaps more so than the surface oxide thickness or microstructure 66 as will be further discussed.
There has been some success with improving secondary cellular fixation, using topographical modifications. Most techniques that are used to produce nanotopography on Ti such as sand-blasting, 67 acid etching,68,69 cluster deposition, 70 layer-by-layer assembly 71 and anodization 72 generate less defined features, lacking precise control and tunability of the topographies. 73 While such nanoscale features may lead to changes in the cell number, size, focal adhesion arrangements, cytoskeletal and nucleoskeletal organization, reproducible changes may be hard to achieve because of batch to batch variations. More precise control over nanofeatures traditionally requires lithographical techniques that are hard to use with materials such as Ti. 74 However, techniques such as through mask anodization have allowed reproducible features to be created.73,75,76 Such surfaces can be used to produce highly reproducible cell effects. These surfaces can reduce 73 or increase cell spreading and MSC differentiation in vivo or in vitro.77–81 Such precise nanotopographical tools will help to dissect the rules of cell-topographical interactions and how they can be useful for work with Ti more simply than using, for example, roughness or random patterns.
Types of coating
There are three ways to change the physical, chemical and mechanical properties of surfaces: (1) by adding a new layer to the surface, (2) by changing the surface itself by exposure to physical or chemical agents like plasma or wet chemicals or (3) by subtraction or attrition process to modify the mechanical surface. However, to achieve the nanoscale modifications it should be able to reach all the topography device surface, change it to reach the commercial scale to be finally industrially integrated.2,82–84
In mechanical modification, the changes are required to improve the adhesion, bonding and bio-mineralization by increasing the surface area. 85 Surface mechanical attrition treatment (SMAT) is a novel technique developed to provide a surface roughness at the nanoscale. This increase in the surface roughness leads to increases in the adhesion energy which have a positive reflection on cellular response.86,87 The cons behind this technique is the flexibility limitation in controlling the intracellular response beyond local adhesion energy. 2 There are three ways for chemical modification for metal surfaces: (1) physiochemical adsorption, (2) molecule covalent binding and (3) peptide inclusion into a carrier material. However, different methods including anodization, oxidative, biochemical functionalization, acid/alkaline treatment, chemical vapour and sol-gel process can affect biologically active moieties onto the surface by controlling the relative densities or arrangement that in turn may have an effect on cell signalling.2,88–90 Physical modification mainly involves the physical spraying of coating or atomic rearrangement with ion implantation. 91 The common techniques used to change the physical components of the substrate include plasma and vapour deposition, ion implantation, thermal oxidation and laser irradiation. Plasma is the fourth type of matter that highly excites the atoms, ions or radial species. The vacuum deposition is using vacuum condensation of a thin material to coat the substrate, while during the ion implantation, selected ions can be deposited on the material surface. Moreover, the involvement of temperature leads to alteration of the crystal structure of the Ti oxide layer which generates a superficial stress or changing in the previous surface nanostructure. 2 Previous studies showed the effect of various nanosurface modifications on enhancing osteoblast activity, spreading, proliferation, differentiation and osteoconduction.91–97
There are three ways of coating: organic, inorganic and combination of both. Organic coating such as polymers, biomimetic and bioinspired films like a component of natural cell surroundings and inorganic components such as calcium phosphate (CaP), HA, titanium oxide (TiO2) and nitride coating. 82 The combination coating is also divided into many types: by their mode of action, type of biological reagent incorporated with (e.g. antibiotics), type of coating (e.g. biodegradable polymers, hydrogel or bioceramic), coating deposit (layer-by-layer, vacuum deposit or electrophoresis) and the coating function. The antibacterial combination coating was reported and discussed in detail in previous study. 98 Tobin, 99 in his review, discussed the three types of combination device coating: (1) reduced infection either by controlling the kinetics release or coating with low potential to induce microbial resistance, (2) enhanced device integration or (3) reduced infection and enhanced integration. A number of reviews have been published on the combination of different coating on orthopaedic implants98–101 and dental implants.46,82
Moreover, implant coating must meet a number of significant challenging requirements to achieve a successful clinical implementation: for instance, a sufficient mechanical integrity, minimization of the local/systemic cytotoxicity and genotoxicity, sufficient amount of the pharmaceutical or biologic agent in the excipient coating matrix, optimization of diffusion kinetics that are not impeded by attachment of proteins to the implant, broad spectrum of antibiotics against biofilm formation without indication a bacterial resistance, and for the technical parts: the coating should be produced with a low coat, easy to manufacture, easy handling, long shelf life and ability to sterilize using conventional sterilization techniques without damaging the incorporated drug or biologic agent. However, there is no current coating system that fulfils all of these requirements. 100
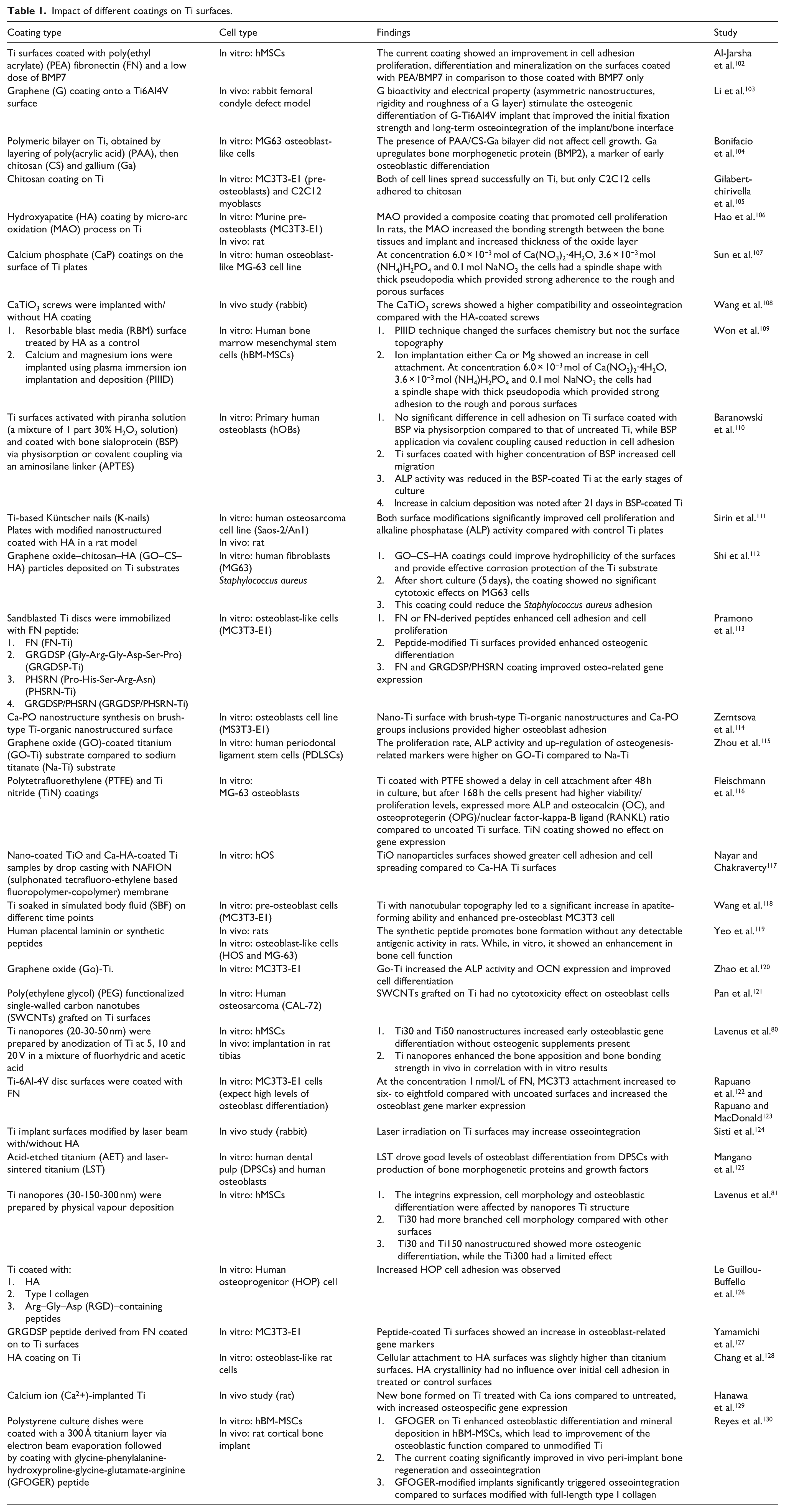
Nevertheless, due to the differences in cell variability related to the species (rat, mouse or human) and cell type (stem cell, osteoblast, etc.), the ability to assess a different kind of coating on the cell structure and function could be challenging. To address this, in the next section (Table 1), we provide selected examples of the impact of different types of coating (organic/non-organic, physical and chemical coating) on Ti surfaces in vivo or in vitro studies.
Impact of different coatings on Ti surfaces.
Surface modification and bacteria
Ti implants are designed to last around 20–25 years but around 10% fail prematurely with the most common cause being bacterial infection in the first year of implantation. 131 Bacteria such as Staphylococcus aureus, Staphylococcus epidermidis, Klebsiella pneumoniae and Pseudomonas aeruginosa for orthopaedic implants and Prevotella intermedia, Porphyromonas gingivalis and Fusobacterium nucleatum for dental implants are understood to play a major role in tissue inflammation and subsequent bone recession through peri-implantitis, osteolysis and osteomyelitis leading to premature implant failure.132,133
Many of these bacteria have the capability to form biofilms which can potentially occur within hours of initial bacteria attachment to an interface (Figure 2(a)). Primary colonizers such as Streptococci attach and proliferate to form microcolonies and secrete self-produced extracellular polymeric substances (EPS) such as proteins, extracellular DNA and exopolysaccharides to form a protective film or matrix. Once a surface has been populated, secondary colonizers, such as Porphyromonas gingivalis, are able to adhere to the sessile cells within the biofilm via receptors. Further aggregation, proliferation and EPS production result in a mature, multi-species biofilm containing a range of environments with varying nutrients and oxygen levels, allowing the bacteria to persist for long periods on the surface, causing chronic complications and becoming resistant to antibiotic treatment. 134

(a) The process of biofilm formation. Initially, cells attach, proliferate and coadhere to form microcolonies. They then continue to expand in similar fashion, together with production of EPS, to form a mature biofilm community. (b) Two possible ways to reduce implant infection: (1) provide no place for bacteria due to a continuous cell layer on the substrate and (2) use an antiadhesive coating that prevent bacterial attachment.
One of the superior qualities of Ti is its ability to absorb calcium, phosphate and serum proteins that are understood to accelerate and support osseointegration. However, such beneficial characteristics may also promote unfavourable processes such as bacterial adhesion. 135
As implants have no resident microbiota to provide colonization resistance, they are susceptible to attachment by incoming microbes. Ideally, a surface should be designed to have selective activity against different cell types, mammalian cells or bacterial cells. Antiadhesive coatings have been created to repel bacteria from the surface and prevent attachment, thus inhibiting biofilms at the first stage. If this surface was then conversely encouraging host stem cells to adhere, proliferate, mature and differentiate, producing a continual cell layer before bacteria are able attach to the surface, implant infection and biofilm growth will be reduced or inhibited altogether (Figure 2(b)). However, it is important to note that the antiadhesive coating may reduce the mammalian cell attachment; hence, surface modifications combining antiadhesive polymers with cell adhesive motifs (e.g. FN, RGD) would be the ideal solution.102,113,126,127
There are two arguments about the effect of surface roughness on bacterial adhesion. The first scenario is that more bacteria adhere as surface microscale roughness increases due to the increased surface area that provides more binding sites and protection. The other argument is that increasing the surface roughness on the nanoscale may provide an unfavourable situation for the bacteria to adhere since the bacteria size is in microscale.
Regarding topography, however, after seminal reports showing that high aspect ratio topographies can kill bacteria, surfaces that can promote osteogenesis and prevent infection are being sought.136–143 The use of such high aspect features has been demonstrated in Ti in several new reports.142,143 In fact, it is becoming clear that both physical and chemical parameters play a role in potentially controlling bacterial adhesion (Table 2).
Examples of the effect of different coatings on bacterial adhesion.

Anti-bacterial, high aspect ratio topographies, in fact, exist in nature. For example, cicada and dragonfly wings have topography that has been shown to be able to disrupt the bacterial membrane leading to cell lysis. 136 Chemical and physical methods are now being developed to fabricate such topographies on clinically relevant materials like titanium, making the prospect of limiting implant infections while inside the body possible and reducing the rates of revision surgery and antibiotic treatment.
A titanium alloy, Ti-6Al-4V, has been developed using thermal oxidation to create a range of titanium dioxide nanostructures and through fluorescence studies, scanning electron microscope (SEM), transmission electron microscope (TEM) and focused ion beam scanning electron microscopy (FIB-SEM) has been shown to disrupt the bacterial membranes, ultimately leading to 40% E. coli cell death after 2-h incubation on the surface.161,162 Furthermore, previous studies showed that the TiO2 nanowires interact with the lipopolysaccharide and proteins, which are held together by electrostatic interactions with divalent cations. These interactions are essential to stabilize the outer membrane helping the TiO2 nanowires to form a molecular linkage at the cell surface allowing it to disturb bacteria membrane function which lead to the lysis of the bacteria. However, this is not the case in Gram-positive bacteria, where no antimicrobial activity has been observed as there might be no interaction of TiO2 nanowires with lipoteichoic acid that is present in the outer membrane of Gram-positive bacteria.163–165
Hydrothermal etching has been used to create topography in the micron range to produce a hierarchically ordered array shown to physically rupture S. aureus and P. aeruginosa cells leading to loss of viability seen in P. aeruginosa; 47.1% death compared to S. aureus with 19.8% death after 18 h of incubation. 166 A chlorine base etching process has also been reported to form anisotropic nanostructures on the surface of titanium with a height of approximately 1 µm. The morphology of P. aeruginosa and S. aureus was significantly altered which correlated well with fluorescence studies showing high bactericidal activity for the Gram-negative bacteria with 98% ± 2% for P. aeruginosa and 95% ± 5% for E. coli after 4 h. For the Gram-positive bacteria S. aureus, there was less killing, with 22% ± 8% non-viable cells after 4 h, but this increased to 76% ± 4% after 24 h. 139
Alkaline hydrothermal processes use sodium hydroxide, high temperatures and pressures to form titanium dioxide nano- and microscale topography on titanium substrates. Using electron microscopy, bacterial cell envelopes have been shown to be pierced by these spikes. 161 Using fluorescence microscopy, loss of viability has been reported when in contact with this nanotopography.141,142,167 Diu et al. 167 reported that motile bacteria (P. aeruginosa, E. coli and B. subtilis) were more liable to lysis with more than 50% cell death in the first hour while non-motile bacteria (S. aureus, E. faecalis and K. pneumoniae) experienced less than 5%. Not only has membrane disruption been seen but anti-biofilm activity has also been shown. Different structures of nanotopography have also been formed using alkaline hydrothermal method, a ‘spear-type’ topography and ‘pocket-type’ topography. After 6 days, there was half as much growth on the spear-type and five times less on the pocket-type compared to a control of flat-polished titanium. 141 Tsimbouri et al. 142 reported ~30% bacterial death after 1-h incubation of P. aeruginosa, which increased to 58% after 18 h incubation.
Along with having a bactericidal surface, it is important to ensure mammalian cells are able to attach, proliferate, mature and differentiate into desired lineages such as osteoblasts to promote successful osseointegration. Research suggests that the nanotopography is able to support osteoblast maturation through expression of osteogenic marker proteins such as Runt-related transcription factor (RUNX-2), BMP2, osteocalcin (OCN) and osteopontin (OPN).139,142,143,166,167
To improve osteointegration, various coatings have been utilized; for example, integrin-binding peptidic ligands have been functionalized onto nanotopographies and shown to significantly increase human mesenchymal stem cells (hMSCs) surface area and decrease the cell’s circularity evidence of improving surface interaction. 143
Table 2 highlights various studies where titanium surfaces have been modified to reduce bacterial adhesion. Coatings with metals such as copper, gallium and silver with well-documented antimicrobial properties have shown to reduce biofilm formation of various bacteria.104,144,147,149–151 Antibiotics such as gentamicin are widely used in the treatment of both Gram-positive and Gram-negative bacteria and have been shown to have potential to be used as a coating on titanium surfaces. 141 Antimicrobial peptides (AMPs) are recognized as promising candidates as alternatives for antibiotics due to the low chance of resistance being induced. Various AMPs have been functionalized on titanium such as HHC-36, GL13K and TBP-1 and have shown potential by reducing biofilm formation of both Gram-positive and Gram-negative bacteria.151–153
Conclusion
The main aim of bone implant industry is to mimic the normal function of tissue by enhancing the implant biocompatibility and reduce the bacterial adhesion while providing mechanical support. Recently, the coating of Ti implants has generated much interest in order to improve osseointegration and prevent unfavourable tissue reactions such as infection, inflammation and the foreign body response. Besides that, coated implants must be shown to be safe, efficient and cost-effective prior to subsequent adoption and widespread usage. Osseointegration/biofilm reduction are required goals; coating the implant with organic/inorganic components, changing the surface topography, and so on have been shown to be efficacious. In addition, the coating composition, location, thickness, uniformity and other physico-chemical variables are important to determine the efficacy and validity of the different coating.
This article aims to provide an overview of the impact of different physical and chemical modifications on Ti surface topography. Such alterations can potentially be used to enhance bone formation, provide bacterial growth inhibition or even perhaps both. That implant surface characteristics including surface roughness, surface chemistry, nanotopography to list a few, have a significant influence on osteogenesis and microbiota inhibition is emerging. Previous studies have implicated high aspect ratio nanofeatures with bactericidal potential. While an ideal implant would be both osseoinductive and antimicrobial, many aspects of these interactions require more investigation to resolve areas of uncertainty surrounding the interaction between these surfaces and MSCs when combined with bacteria. To conclude, the potential of Ti surface modifications is largely due to the ageing population placing pressure on orthopaedic treatments and Ti being the gold standard for fabrication on dental or orthopaedic implants.
Footnotes
Acknowledgements
We would like to acknowledge the mentorship and guidance of Professor Adam Curtis. Adam opened up the area of cell – topographical interactions to the world and we are grateful for his advice and friendship over the years. The authors thank Carol-Anne Smith for her technical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a studentship to L.D. from University of Jeddah, Jeddah, Saudi Arabia and EPSRC grant EP/K034898/1.
