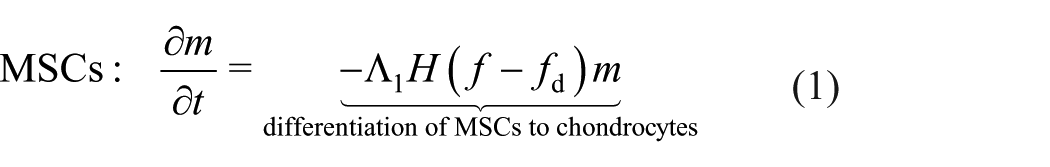Abstract
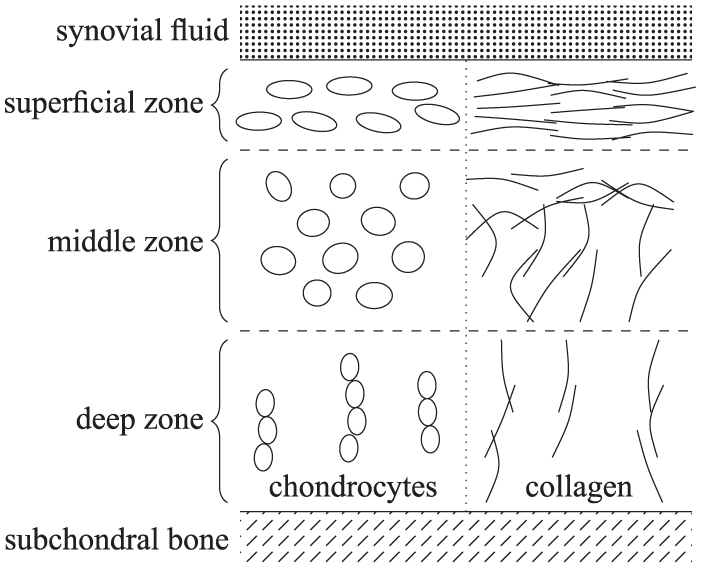
A key step in the tissue engineering of articular cartilage is the chondrogenic differentiation of mesenchymal stem cells (MSCs) into chondrocytes (native cartilage cells). Chondrogenesis is regulated by transforming growth factor-β (TGF-β), a short-lived cytokine whose effect is prolonged by storage in the extracellular matrix. Tissue engineering applications aim to maximise the yield of differentiated MSCs. Recent experiments involve seeding a hydrogel construct with a layer of MSCs lying below a layer of chondrocytes, stimulating the seeded cells in the construct from above with exogenous TGF-β and then culturing it in vitro. To investigate the efficacy of this strategy, we develop a mathematical model to describe the interactions between MSCs, chondrocytes and TGF-β. Using this model, we investigate the effect of varying the initial concentration of TGF-β, the initial densities of the MSCs and chondrocytes, and the relative depths of the two layers on the long-time composition of the tissue construct.
Introduction
Articular cartilage acts as a lubricating surface in synovial joints, preventing bone contact. This thin tissue layer is composed of a dense network of extracellular matrix (ECM) components (principally collagen and proteoglycans), which is maintained by native cartilage cells, known as chondrocytes. 1 As shown in Figure 1, the orientation and density of collagen varies with cartilage depth. In a synovial joint, the cartilage lies between the synovial fluid and the subchondral bone, and is typically described as comprising three zones. The highest density of collagen and chondrocytes is in the superficial zone, where both are aligned in the direction of the surface of the cartilage. In the middle zone, the cells are more rounded and the collagen alignment is less organised, with the density of both lower than in the superficial zone. In the deep zone, the cells are organised in column-like structures, with both these columns and the collagen aligned perpendicular to the subchondral bone. 1 The zonated structure and interaction between its components endow cartilage with the mechanical properties necessary to withstand the high stress environment of a synovial joint.
Schematic diagram of the zonated, depth-dependent structure of articular cartilage.
Articular cartilage is an avascular tissue. This property, combined with the low proliferation and motility of the chondrocytes, means that it has a low capacity to regenerate when it degrades or is damaged through injury. Consequently, chronic conditions such as osteoarthritis require surgical intervention, typically involving a full joint replacement in end-stage disease. An approach which involves replacing damaged tissue with artificially engineered cartilage implants would be an attractive alternative to these invasive surgical procedures.
Artificially engineered cartilage implants aim to mimic the function of natural tissue. From a biomechanical perspective, they will ideally have a distribution of cells and matrix components similar to that shown in Figure 1.
A recent tissue engineering approach to develop such an implant 2 has exploited additive manufacturing techniques to seed cells within a hydrogel construct reinforced with a three-dimensional (3D)-printed lattice of polymer fibres. The seeded cell population consists of mesenchymal stem cells (MSCs) and/or chondrocytes which are then biochemically and/or mechanically stimulated to promote chondrogenic differentiation of the MSCs and enhance matrix deposition by the chondrocytes. Both processes are regulated by transforming growth factor-β (TGF-β). 1 Understanding how this chemokine drives MSCs to differentiate into chondrocytes as it diffuses through a tissue engineering construct is the focus of the present study.
TGF-β plays an important role in both natural cartilage and in approaches to engineer artificial cartilage. It is produced in vivo by chondrocytes and binds rapidly to the ECM for storage. After undergoing an activation process, an active form of TGF-β is released from this stored state and can freely diffuse. It then acts to stimulate chondrocytes to synthesise ECM components, including collagen type II and proteoglycans (particularly aggrecan). Importantly for tissue engineering applications, it also drives MSCs to differentiate into chondrocytes. There are a number of subtleties to the biochemistry of TGF-β (as shown in Figure 2). TGF-β is endogenously secreted as part of a large molecular complex, this latent form comprises a ligand (the active component) bound to a protein and a peptide. As a result, the active component is unable to bind to receptors on the outer membrane of cells (in this study, MSCs and chondrocytes) until it is activated by being cleaved from this complex.

A schematic showing the simplified life cycle of TGF-β used in the model. The steps in this life cycle are (1) secretion of latent TGF-β by chondrocytes, (2) binding of latent TGF-β to the extracellular matrix, (3) activation (and release) of bound TGF-β by interaction with an activating chemical species and (4a, b) binding of active TGF-β to the receptors on MSCs and chondrocytes.
When first secreted, the latent TGF-β may freely diffuse, an arrangement (as part of a larger complex) which enables latent TGF-β to bind to and thus be stored throughout the ECM. Activation (i.e. the cleaving of just the active ligand from the complex) then occurs when chondrocyte stimulation is required (in response to ECM damage, for instance). TGF-β can be activated via mechanical interaction with cell integrins 3 or via chemical interactions with proteases, thrombospondin-1, reactive oxygen species or extremes in pH. 4 Once activated, it can freely diffuse but has a short half-life of only a few minutes, as compared to the timescale of days over which cell differentiation occurs. 5 As a consequence, active TGF-β must bind rapidly to cell receptors to have an effect on the cells. In particular, if TGF-β binds to surface receptors of MSCs in sufficient quantity, they are driven to chondrogenic differentiation. 1
For tissue engineering applications, there are two dominant sources of TGF-β: it may be secreted by chondrocytes or it may be added exogenously, where exogenous supplies may be replenished regularly to ensure a sustained effect on the cells. The key modelling question that we address in this article is to understand how these two sources of TGF-β act, either alone or in combination with each other, to direct stem cell differentiation. In particular, we seek to establish whether synergistic effects are possible where such a combination is more effective than relying solely on stimulation from either the TGF-β produced by the seeded chondrocytes or exogenous TGF-β.
In a previous study, 6 we modelled chondrogenesis in well-mixed cell populations for which the distribution of the various forms of TGF-β were assumed to be spatially uniform. In that study, attention focussed on scenarios for which MSCs could be induced to differentiate into chondrocytes either by the addition of exogenous TGF-β or by co-culturing them with harvested chondrocytes. Analysis of an ordinary differential equation model representing these scenarios predicted that differentiation of the entire MSC population is induced if either sufficient concentrations of TGF-β is added or if the MSCs are co-cultured with a sufficiently high density of chondrocytes which themselves secrete TGF-β. Further simulations revealed and suggested synergistic effects between the two scenarios, so that experimental efficiencies could be achieved by adding exogenous TGF-β to a MSC/chondrocyte co-culture, as compared to using either strategy in isolation.
The current work is motivated by recent experimental efforts to develop regenerative articular cartilage implants using a combination of additive manufacturing techniques and purpose-designed biomaterials. 2 This includes experiments involving tissue engineering constructs where the initial seeded populations of MSCs and chondrocytes are spatially separated, see Figure 3. These constructs are comprised of cells embedded in a disc of PCL-reinforced hydrogel, 7 and cultured in a bioreactor for a period of 28 days to promote the chondrogenic differentiation of the MSCs and the production of ECM. While in the bioreactor, the cell-seeded hydrogel disc is either free standing or housed in a sample of porcine tissue to mimic being implanted in a joint. Throughout this period, the construct is bathed in culture medium which contains exogenously added TGF-β replenished at regular intervals.

A schematic diagram of a layered MSC/chondrocyte co-culture stimulated with exogenous TGF-β, along with a microscope image from an experiment to develop a regenerative articular cartilage implant performed at University Hospital Würzburg.
This layered construct was devised to mimic natural articular cartilage. The upper layer of chondrocytes correspond to the superficial zone of the cartilage, while the MSCs in the lower layer, once differentiated, are intended both to mimic its deep zone and to integrate with the subchondral bone (since MSCs may also be driven to an osteogenic rather than chondrogenic lineage). These initial experiments demonstrate that the constructs are biocompatible with the seeded cells. Furthermore, ECM components associated with cartilage are deposited in the upper, chondrocyte-seeded layer (the brown staining in Figure 3 is for aggrecan). These ECM components are not present in the lower layer, which suggests that chondrogenic differentiation of the MSCs had not occurred. Understanding these results motivates the present in silico study.
A schematic diagram of an idealised version of the cell seeding strategy used in these experiments is shown in Figure 3, with a layer of chondrocytes lying above a layer of MSCs and TGF-β stimulation coming above. This spatially simplified situation corresponds to only the central most part of the hydrogel disc, where stimulation from the sides of the construct may be neglected. Under this assumption, we only consider one spatial dimension in our mathematical model (parallel to the depth of the construct) and so focus our investigation on the effect of stimulating this central region from above.
We will consider a variety of strategies that combine zonated cell seeding and the addition of exogenous TGF-β, and determine the efficacy of each strategy to differentiate the seeded MSCs. The three seeding strategies under consideration and shown schematically in Figure 4, are
(1) layered MSC (lower) and chondrocyte (upper) cell seeding without exogenous TGF-β
(2) pure MSC cell seeding with exogenous TGF-β
(3) layered MSC (lower) and chondrocyte (upper) cell seeding with exogenous TGF-β

Schematic diagram of the three cell seeding and/or TGF-β stimulation strategies considered in this study: (a) Case study 1, (b) Case study 2 and (c) Case study 3.
These strategies are analogous to the strategies considered in our previous model, 6 with the addition of spatial dependence. Here, case study 3 corresponds to the experiments described above, where there was no observed lower layer differentiation, and so we aim to use our modelling approach to devise improved protocols for future in vitro studies. The goal is to make the most efficient use of the available chondrocytes, since these are in limited supply, and to use exogenous TGF-β, which is expensive, at as low a concentration as possible while still achieving chondrogenesis.
Methods
Mathematical model and background
We develop a mechanistic mathematical model to investigate how the spatial distributions of populations of MSCs and chondrocytes embedded in a layered hydrogel evolve over time in response to diffusion of TGF-β. We neglect cell motility since the cells are embedded in a dense hydrogel and are not observed to move. We assume that the chondrocytes neither proliferate nor dedifferentiate; these are reasonable assumptions for chondrocytes that have been derived from articular cartilage and that are being cultured in vitro.8–10 Over the timescale of interest (approximately 1–10 days), it is also reasonable to neglect cell death.
10
For simplicity, we consider cell populations and TGF-β concentrations which vary with one spatial dimension
The variables used to represent the various cell densities and forms of TGF-β are summarised in Table 1. We consider two distinct cell populations; we denote by
Description of the model variables, along with their units.
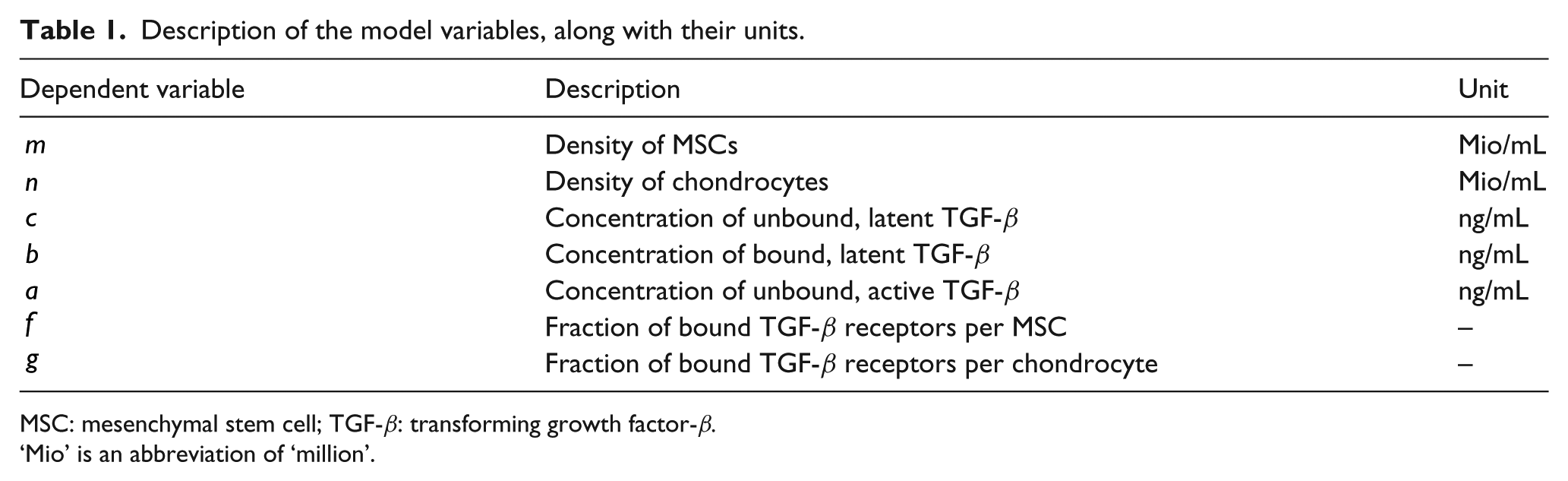
MSC: mesenchymal stem cell; TGF-β: transforming growth factor-β.
‘Mio’ is an abbreviation of ‘million’.
In equations (1) and (2),
Summary of the dimensional parameters that appear in equations (1)–(7), together with estimates of their values.
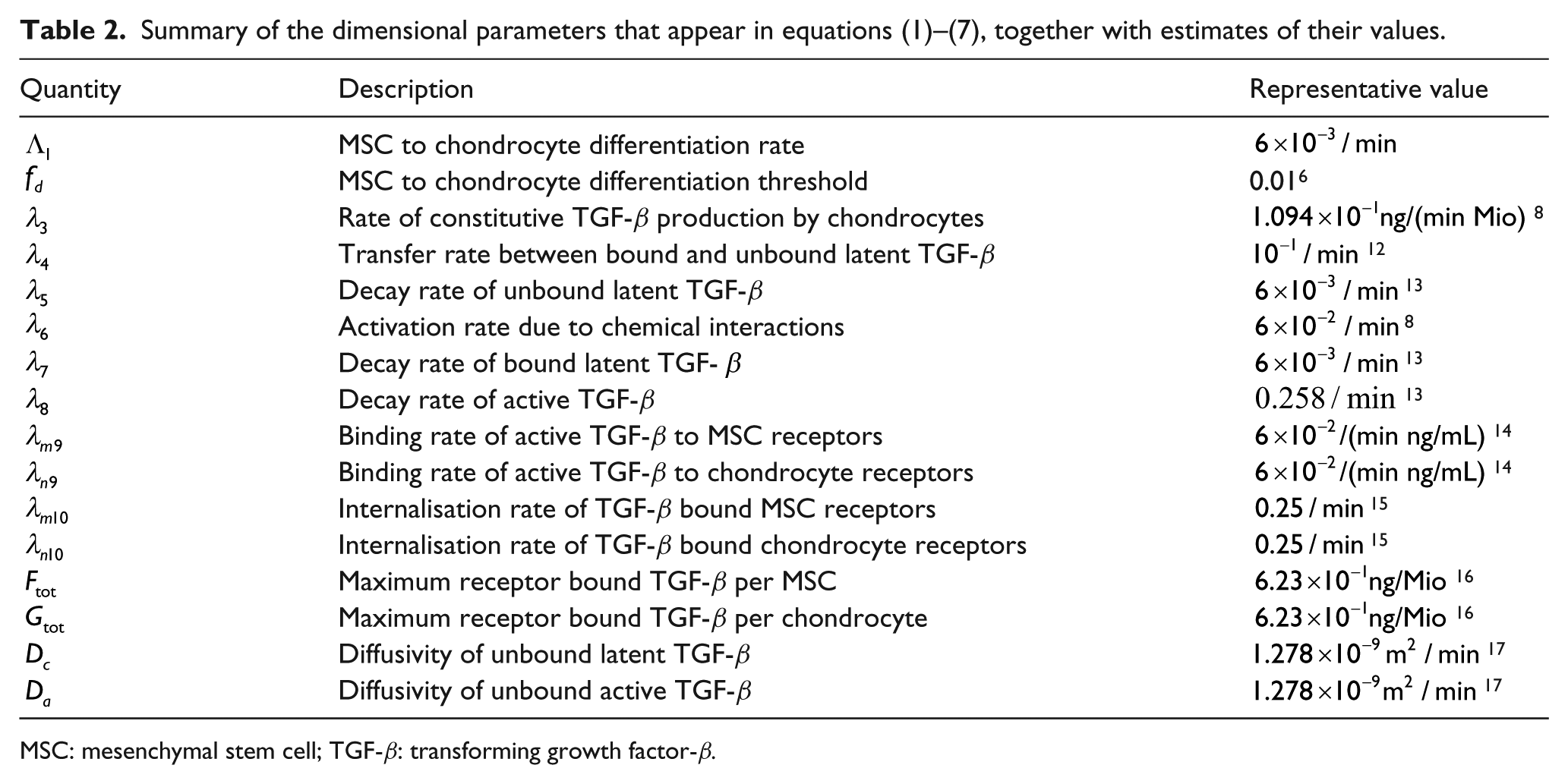
MSC: mesenchymal stem cell; TGF-β: transforming growth factor-β.
As shown in Figure 2, the life cycle of TGF-β is complex; it can exist in a variety of forms. 4 For simplicity and following, 6 we distinguish three forms of TGF-β:
endogenous, unbound latent TGF-β, denoted
latent TGF-β, bound and stored in the ECM (or, in this application, the hydrogel), denoted
active TGF-β, either exogenous or cleaved (and activated) from the ECM, denoted
In its active form, TGF-β can bind to cell receptors on the outer membrane of MSCs and chondrocytes. We track the proportion of bound TGF-β receptors/cell for each cell type, and denote them by
Endogenous TGF-β,

In equation (3), we have introduced several parameters whose definitions, along with representative values, are given in Table 2. We have also assumed that the density of the ECM/hydrogel to which the TGF-β binds is constant (and therefore absorbed into the parameter
In order for the TGF-β bound to the ECM and/or hydrogel to become accessible to cells, it must be activated. Here we assume that the dominant mechanism of activation is chemical (due to extremes in pH, or interaction with proteases, thrombospondin-1, reactive oxygen species). It follows that the differential equation governing
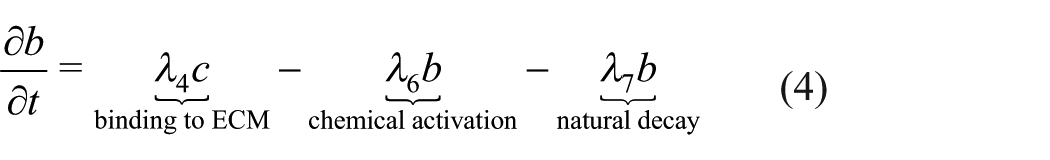
Active TGF-β can bind to cells and diffuse but has a very short half-life. The equation governing the concentration of active TGF-β,

In this work, we consider a simpler model of receptor-ligand dynamics than that proposed by other authors.
18
We assume that all receptors are identical and once bound to cell surface receptors, TGF-β is rapidly internalised. We also assume that when an MSC differentiates any bound TGF-β will be bound to the newly created chondrocyte. Furthermore, we assume that the two cell types have a fixed number of receptors per cell, with the total possible mass of TGF-β bound to an MSC or chondrocyte denoted


As stated earlier, descriptions and typical values of the parameters that appear in equations (1)–(7) are given in Table 2. Where possible parameter values are taken from previous modelling studies of related biological systems, others are estimated from in vitro experiments involving TGF-β.
We solve equations (1)–(7) in a domain that represents a culture system comprising a cell-seeded scaffold which is bathed in culture medium (see Figure 4). The scaffold and culture medium are each assumed to be of height

The initial conditions used for the three strategies depicted in Figure 4 are stated in terms of dimensionless variables below.
Non-dimensionalisation, numerical solution technique
We non-dimensionalise equations (1)–(7) by scaling the cell densities and concentrations of TGF-β as follows (where stars denote dimensionless quantities)

where

These scales are chosen to be




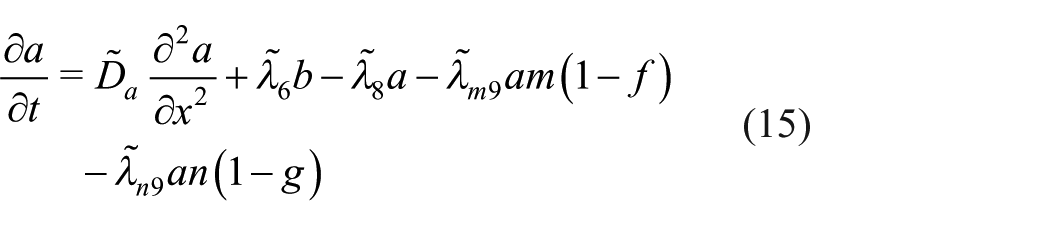

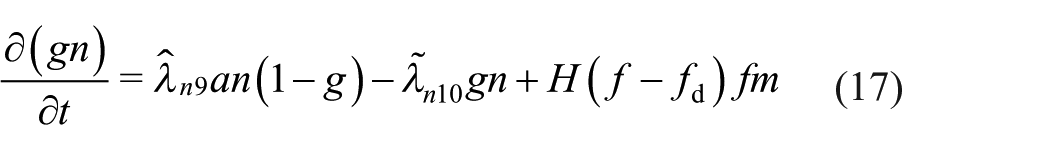
Suitably non-dimensionalised, the boundary conditions (8) are

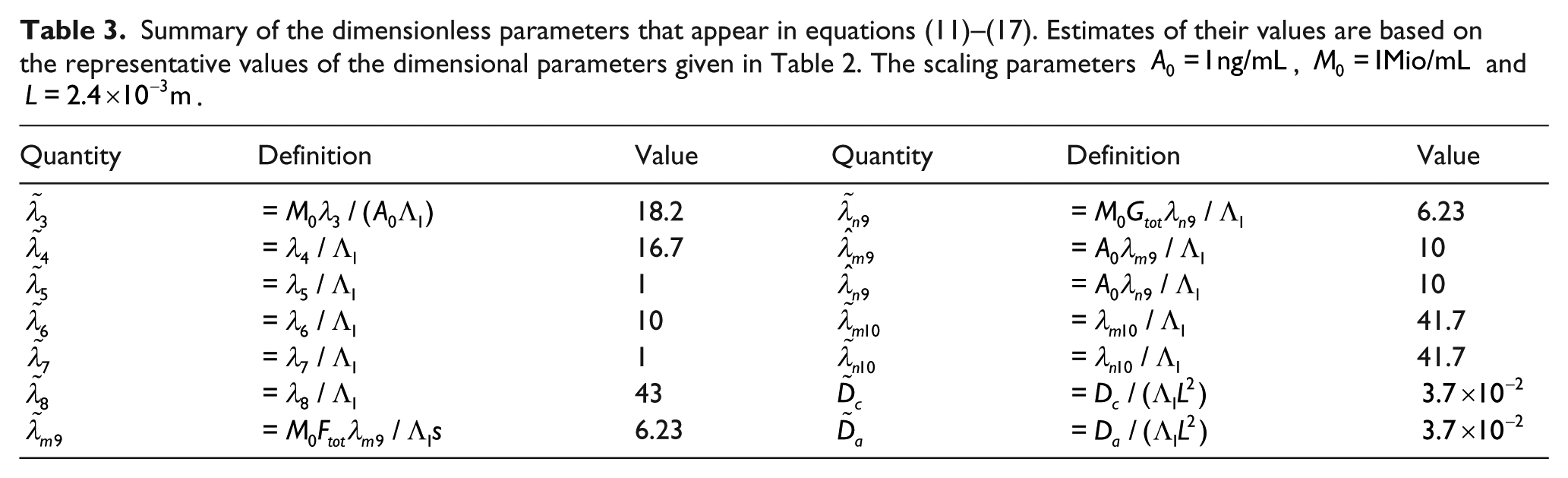
Equations (11)–(17) contain a number of dimensionless parameters; they are defined in terms of the dimensional parameters in Table 3, along with estimated values based on the representative values of the dimensional parameters given in Table 2.
Summary of the dimensionless parameters that appear in equations (11)–(17). Estimates of their values are based on the representative values of the dimensional parameters given in Table 2. The scaling parameters
Initial conditions
We close equations (11)–(17), subject to boundary conditions (18), by prescribing suitable initial conditions for the three case studies of interest.
Case study 1
As shown in Figure 4(a), the initial cell densities for this case are


representing a layer of MSCs of density 1 and height
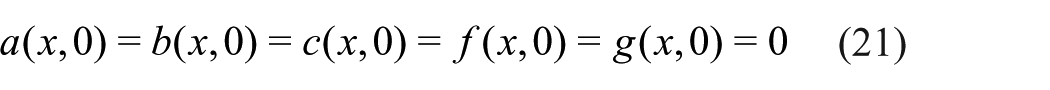
Consequently, any MSC differentiation will be due to TGF-β constitutively produced by chondrocytes. Our simulations focus on the effect of varying
Case study 2
As shown in Figure 4(b), the initial MSC density and active TGF-β concentration for this scenario are


so that initially only MSCs are present and the culture medium is filled with active TGF-β at concentration

The focus of this case study is to investigate the effect on the MSC layer of varying
Case study 3
As shown in 4(c), the initial cell densities and active TGF-β concentration are now



thus combining the layered cell seeding of case study 1 and the TGF-β stimulation of case study 2. All other forms of TGF-β are initially absent so that

The focus here is to investigate possible synergistic effects between the two sources of TGF-β by varying both
Numerical solution procedure
Equations (11)–(17) with boundary conditions (18) are solved numerically subject to the initial conditions associated with a particular case study. To solve the governing equations, we partition the spatial domain into a grid of
Results/discussion
We describe below the key results from our investigations of the cell seeding and/or TGF-β stimulation strategies depicted in Figure 4. We also discuss briefly the dynamics that lead to chondrogenesis.
To summarise the output of our simulations, we introduce the quantity
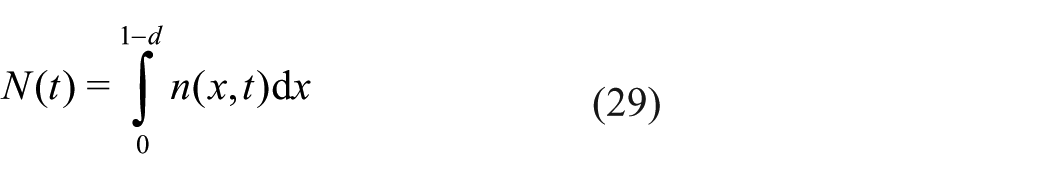
We also introduce
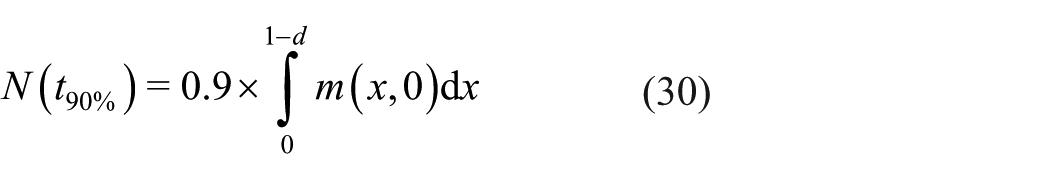
Case study 1
MSC/chondrocyte co-culture with equal height layers
We have found that there are two possible outcomes for this strategy: if the chondrocytes are seeded at too low a density, then none of the MSCs in the lower layer differentiate; if they are seeded above a critical density of
Figure 5 shows how the long-term state of the system depends on

The final yield of chondrocytes in the lower layer for case study 1 as
This behaviour is directly analogous to that seen in a well-mixed co-culture of chondrocytes and MSCs,
6
except that the value of
Note that although the density
MSC/chondrocyte co-culture where the chondrocyte layer is thin
As above, in practice, the initial number of chondrocytes, not just their density, must be considered since they are limited in supply since in the context of tissue engineering, they must be harvested from healthy tissue. A strategy that uses a relatively thin upper layer would require fewer chondrocytes than a relatively thick upper layer seeded at the same initial chondrocyte density. We suggest that such a seeding strategy may be an effective use of the available chondrocytes when they are limited in number, since they will be seeded at a higher density.
The initial number of chondrocytes in the upper layer (per cross-sectional area) is given by

Dependence of the long term outcome on upper layer depth
For instance, the crosses in Figure 6 represent cases where the same number of chondrocytes are seeded at two different layer heights; the thicker layer (rightmost cross for
Case study 2
TGF-β stimulation for a pure MSC layer
We now consider a situation in which the scaffold is initially seeded with MSCs only and stimulated by exogenous TGF-β (see Figure 4(b)). Our simulations indicate that three long-term outcomes are possible depending on the initial concentration of exogenous TGF-β. For low concentrations, no cells differentiated; for moderate concentrations, some cells are differentiated near the top of the scaffold; and above a critical initial concentration
The results of our simulations are summarised in Figure 7, where the shading of the upper plot indicates

The final yield of chondrocytes for case study 2, shown over a range of
Full differentiation is triggered if the initial TGF-β concentration
The behaviour here is qualitatively similar to the analogous well-mixed case 6 where the exogenous TGF-β is assumed to be evenly distributed throughout the scaffold at all times, but the corresponding initial TGF-β concentration required to induce chondrogenesis in the well-mixed case is an order of magnitude lower. 6 In the layered case, the exogenous TGF-β must diffuse through scaffold to have an effect on the cells during which time it degrades, whereas in the well-mixed case, it was assumed to be instantaneously available at the location of the cells. The mathematical model of the present work, which includes diffusion of TGF-β, is a more appropriate description of experiments such as that shown in Figure 3.
Case study 3
TGF-β stimulation for a co-culture with layers of equal height
There are three possible outcomes for this strategy. These outcomes are summarised in Figure 8 for a range of initial TGF-β concentrations

Dependence of the long-term state on
Full differentiation occurs if
These simulations indicate that for a layered co-culture with layers of equal height, supraphysiological concentrations of TGF-β are required to induce chondrogenesis (physiological concentrations are less than 1 ng/mL or 1 in dimensionless concentration). Here, the upper chondrocyte layer acts as a barrier to diffusion, so very large initial concentrations of TGF-β must be added to the culture medium for it to have any effect; for lower initial concentrations, it will have degraded so much as to be at a negligible concentration by the time it has diffused through to the lower MSC layer. Such large concentrations of TGF-β are not physically realistic. For reference, the dimensionless half-life of the exogenous (active) TGF-β is approximately 0.016. As indicated by the value of
TGF-β stimulation for a co-culture where the chondrocyte layer is thin
We hypothesise that if the relative height of the upper chondrocyte layer is reduced, it will no longer act as a barrier to diffusion of TGF-β from the culture medium. We have repeated our simulations for a reduced upper layer height of

Dependence of the long-term state on
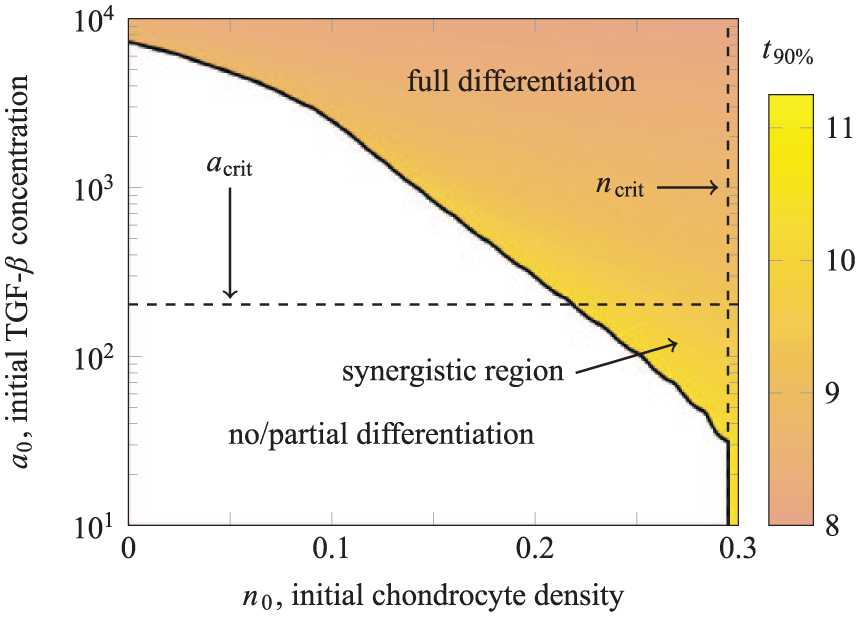
Dependence of the long-term state on
This reveals that for a relatively thin upper layer of seeded chondrocytes, it is possible to induce chondrogenesis using fewer chondrocytes and a lower concentration of exogenous TGF-β than using these strategies in isolation. The critical values
Figure 10 shows further results from our simulations, with the full differentiation region shaded for
Underlying dynamics
We conclude with a brief description of the dynamic interplay between the density of the cell populations and the various forms of TGF-β.
Case study 1
An example simulation for case study 1 is presented in Figure 11, showing the chondrocyte distribution

Typical simulations for case study 1 where differentiation of the MSCs in the lower layer is triggered, the exogenous TGF-β produced by the chondrocytes in the upper layer, shown here for an initial chondrocyte density of
Initially, no TGF-β is present and so at
These newly produced chondrocytes produce TGF-β to supplement that produced by the originally seeded cells. As this diffuses deeper into the lower layer, the region in which sufficient TGF-β is bound to the MSCs to trigger differentiation (i.e. where
This process continues and by
Case study 2
As summarised in Figure 7, for case study 2, the final yield of chondrocytes depends on
Three examples of partially differentiated chondrocyte distributions

Chondrocyte densities for case study 2 (see Figure 4(b)), where MSC differentiation is induced through the initial addition of exogenous TGF-β of concentration
A case where enough chondrocytes are produced by the initial exogenous TGF-β stimulation to kick-start differentiation of the entire layer is shown in Figure 12(b) for
Case study 3
The underlying dynamics of case study 3 are a combination of those seen in case studies 1 and 2. As described in Figures 8–10, the final yield of chondrocytes depends on both

Chondrocyte densities for case study 3, where MSC differentiation is induced through the initial addition of exogenous TGF-β of concentration
As shown in Figure 13(b), when
Conclusion
We have developed a mathematical model to simulate TGF-β mediated differentiation of MSCs into chondrocytes in tissue engineering scaffolds. This model was used to investigate a variety of experimental strategies, including layered co-cultures of MSCs and chondrocytes. The model accounts for spatial variation in the distribution of the cell populations and allows for the diffusive transport of latent and active TGF-β. Our extensive simulations provide insight into the efficacy of strategies currently used to engineer cartilage in vitro and suggest directions for further experimental studies.
In case study 1, we considered a layered scaffold where differentiation of the MSCs in the lower layer was driven by the endogenous production of TGF-β by chondrocytes seeded in the upper layer. Our key findings were that
The initial density of chondrocytes must be above a threshold value
Where a fixed number of chondrocytes is available, an efficient strategy is to seed them in as thin a layer as practicable.
In case study 2. we considered a scaffold seeded with only MSCs and differentiation driven by exogenous TGF-β added to the culture medium above the scaffold. Our key findings were that
The initial concentration of exogenous TGF-β must be above a threshold value
Adding exogenous TGF-β in excess of this threshold concentration results in only a small decrease in the time taken to differentiate the seeded MSCs.
Case studies 1 and 2 serve as a foundation to examine the strategy of case study 3, a layered co-culture of MSCs lying above chondrocytes, supplemented with exogenous TGF-β. Our key findings were that
For layers of equal height, the exogenous TGF-β did not adequately diffuse through the upper chondrocyte layer to supplement that produced endogenously, and therefore had no effect in terms of promoting chondrogenesis and
When the upper layer depth is reduced, synergistic effects are possible where chondrogenesis may be triggered using a lower concentration of exogenous TGF-β and a lower density of seeded chondrocytes than would be required if these sources of TGF-β were used in isolation.
Future work may include experimental testing of scaffolds with layered cell seeding and a relatively thin chondrocyte-seeded upper layer. The mathematical model may be extended to consider scaffolds in three spatial dimensions. Other novel protocols and cell seeding strategies not previously tested experimentally may also be considered; for example, scaffolds with more than two layers which could be seeded with cell types at different densities. We could also consider the mechanism behind mechanically induced chondrogenesis by modelling TGF-β activation via integrin mediated mechanotransduction 3 and the subsequent remodelling of the mechanical environment of the scaffold as ECM components are produced by the chondrocytes.
Our simulations suggest improvements to existing experimental protocols. In particular, where layered cell seeding is used with chondrocytes lying above MSCs, there is an advantage in seeding the available chondrocytes in as thin as practicable regardless of exogenous TGF-β supplementation. In a strategy where no exogenous TGF-β is added, then for a fixed number of chondrocytes, the density will be higher if the layer is thinner, thus promoting chondrogenesis in a more efficient manner. Similarly, if layered cell seeding is used in combination with exogenous TGF-β stimulation, the upper layer should be as thin as practicable to permit diffusion of TGF-β, which in turn triggers differentiation of the seeded MSCs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research leading to these results has received funding from the European Union Seventh Framework Programme (FP7/2007–2013) under grant agreement no. 309962 (HydroZONES). S.L.W. is grateful for funding in the form of a Royal Society Leverhulme Trust Senior Research Fellowship. SLW and HMB gratefully acknowledge funding from the UK Engineering and Physical Sciences Research Council (EPSRC grants EP/R013128/1, EP/P031218/1, EP/R018472/1 and EP/N026985/1).