Abstract
Reconstructing critical-sized craniofacial bone defects is a global healthcare challenge. Current methods, like autologous bone transplantation, face limitations. Bone tissue engineering offers an alternative to autologous bone, with traditional approaches focusing on stimulating osteogenesis via the intramembranous ossification (IMO) pathway. However, IMO falls short in addressing larger defects, particularly in clinical scenarios where there is insufficient vascularisation. This review explores redirecting bone regeneration through endochondral ossification (ECO), a process observed in long bone healing stimulated by hypoxic conditions. Despite its promise, gaps exist in applying ECO to bone tissue engineering experiments, requiring the elucidation of key aspects such as cell sources, biomaterials and priming protocols. This review discusses various scaffold biomaterials and cellular sources for chondrogenesis and hypertrophic chondrocyte priming, mirroring the ECO pathway. The review highlights challenges in current endochondral priming and proposes alternative approaches. Emphasis is on segmental mandibular defect repair, offering insights for future research and clinical application. This concise review aims to advance bone tissue engineering by addressing critical gaps in ECO strategies.
Keywords
Introduction
Reconstructing large craniofacial bone defects, including mandibular bone defects caused by trauma, congenital disorders, cancer and infection poses a significant healthcare challenge worldwide.1,2 Current clinical practice to encounter oral cancer principally involves radical surgery and radiotherapy, which not only cause difficulties with eating and speaking, but also impairs the ability of neighbouring tissues to heal and regenerate. The jawbone removed during radical surgery is reconstructed by transplanting a patient’s own bone, for example – bone harvested from patient’s leg, for tissue replacement and repair.3,4 Additionally, this complex procedure, which can last over 14 h, often fails to replicate the intricate jaw structure with autologous bone, impacting both aesthetics and functionality. However, this approach is associated with significant drawbacks, including complications at the donor site, risk of infection and limited availability of donor tissue.5,6 Moreover, the use of titanium to secure transplanted bone to the jaw can lead to unwanted stress shielding, radiation dose intensification and imaging artifacts which cause implant failure and hinder cancer surveillance.7–9 Whichever method is applied for bone defect reconstruction, be it autografting, allografting, xenografting or alloplastic replacement, each has its own specific uses and limitations.7–9 Bone tissue engineering (BTE) is an attractive alternative solution for repairing critical-sized bone defects. BTE involves a combination of cells, biomaterials with specific biochemical and physicochemical properties and growth factors to facilitate bone regeneration. BTE has the potential to address the limitations associated with autologous bone transplantation 10 , offering several advantages including the ability to produce unlimited and customised graft material, reduced morbidity and faster healing times. 11
Traditional BTE encompasses several crucial elements. Firstly, an osteoconductive scaffold is employed.12,13 This scaffold provides a supportive framework for bone regeneration. However, the biomaterials used for manufacturing scaffolds to bridge or reconstruct bone defects are mostly biologically inactive and may not induce bone ingrowth. 14 Furthermore, after cancer surgery, there is only a 6-week healing period before radiotherapy’s anti-osteogenic effects halt bone regeneration. 15 This critical 6-week window is insufficient for complete bone bridging with biomaterials-based scaffolds alone. Therefore, accelerating new bone formation within the scaffold necessitates osteoinduction. Hence, mesenchymal stem cells (MSCs), derived from diverse sources, capable of differentiating into a variety of cell types, including osteogenic cells, as well as cultured osteoblasts are introduced to the graft site, capable of initiating bone formation or creating a microenvironment that stimulates resident cells. 16 These cells play a vital role in promoting the generation of new bone tissue. Additionally, osteoinductive molecules are utilised to trigger implanted or resident cells to actively participate in the formation of bone tissue. 17 Lastly, the establishment of sufficient vascularisation is crucial to meet the nutritional requirements of the newly formed tissue as well as facilitate waste removal.18,19 To date, BTE has focused on replicating the intramembranous ossification (IMO) pathway, where stromal cells and MSCs are directed to differentiate directly into osteoblasts responsible for depositing a mineralised bone matrix. 20 To enhance the growth of mineralised bone-like tissue, MSC-seeded scaffolds are cultured in bioreactors, which provide mechanical signals to the cells.21,22 These mechanical signals play a role in promoting the development of mineralised bone-like tissue within the scaffolds. Flat bones of the craniofacial region, including mandible mostly follow IMO-based repair process during fracture healing. 23 While the IMO approach is effective for repairing smaller or stable bone defects, it may not be suitable for large bone defects that are hypoxic due to insufficient vascularisation.24,25 Additionally, insufficient vascularisation during the initial phases of implantation at the defect site can result in cell death due to a lack of oxygen and nutrients. Furthermore, pre-mineralised tissues, often employed in bone regeneration, pose hurdles for host cell infiltration and blood vessel formation. 26 Hence, researchers are investigating alternative approaches such as hypoxia-inducible factors (HIFs), mechanical modulation and therapeutic angiogenesis to improve vascularisation. 27 HIFs play a crucial role in enhancing angiogenesis by promoting the expression of pro-angiogenic factors. Despite their beneficial effects, excessive activation of HIFs can result in abnormal vessel growth and potentially contribute to tumor progression. 28 Mechanical forces, such as shear stress, stimulate endothelial cells and enhance scaffold integration, yet challenges remain in identifying optimal parameters and designing specialised bioreactors for this purpose. 29 While targeted delivery of pro-angiogenic factors holds significant clinical potential, it is also essential to address issues related to short-term effects and the risk of uncontrolled growth.
An alternative approach could involve directing bone regeneration through the endochondral ossification (ECO) pathway, which is stimulated by hypoxic conditions. Physiologically, many defects in long bones are repaired following the ECO pathway. This process involves mesenchymal progenitor cells initially creating a cartilaginous matrix, which subsequently undergoes transformation into bone tissue.30,31 Briefly, following a fracture, a hematoma forms and surrounding tissues, including the periosteum, stabilise it. 32 The inner cambium layer of the periosteum generates chondrocytes from progenitor cells and forms a cartilaginous template reminiscent of the ECO process seen in normal bone development.31,33 This cartilaginous template undergoes ossification, ultimately leading to the formation of a hard and soft callus that replaces the fractured bone.31,33 However, while the concept of using ECO strategies for BTE is promising, there is still insufficient knowledge regarding its application; Several crucial aspects such as the selection of cell sources, biomaterials and endochondral priming protocols require further elucidation. Therefore, this review considers the application of scaffold biomaterials and cellular sources that can be primed for chondrogenesis to mimic the ECO pathway. In addition, this review will shed light on the challenges in endochondral priming and discuss potential alternative approaches to successfully translate in vitro findings into animal models and clinical settings, with a focus on repairing critical-sized craniofacial bone defects, such as segmental mandibulectomy.
Endochondral ossification
ECO is vital for skeletal development, fracture healing and bone defect repair. 34 It involves a series of well-coordinated steps that contribute to the formation of new bone tissue (Figure 1). The process begins with the condensation and differentiation of mesenchymal cells into chondrocytes, which secrete a cartilage matrix composed of collagen and proteoglycans. 34 This cartilage template serves as a framework for subsequent bone formation. As the cartilage grows, chondrocytes undergo proliferation and hypertrophy, resulting in the elongation and expansion of the cartilage template. 34

Schematic of endochondral ossification process. Step 1: Formation of a hyaline cartilage template, around which a bone collar develops. Step 2: Subsequently, the cartilage matrix degrades, creating cavitation of the cartilage template. Step 3: Blood vessels from the periosteal bud invade these cavities, bringing osteoprogenitor cells, forming spongy bone. Step 4: Continued ossification results in the formation of the medullary cavity and the emergence of secondary ossification centres in the epiphyses. Step 5: Eventually, only the epiphyseal plate (growth plate) and articular cartilage remain cartilaginous, allowing for continued bone growth in length until skeletal maturity is reached.
Vascularisation is a critical end-product of ECO. Blood vessels penetrate the cartilage, delivering osteoprogenitor cells and other precursor cells necessary for bone formation. 34 These osteoprogenitor cells differentiate into osteoblasts, which deposit osteoid, an unmineralised bone matrix, onto the cartilage scaffold. 34 The osteoid consists of collagen fibres and mineral salts, providing the initial structural framework and rigidity to the developing bone tissue.
Subsequently, the osteoid undergoes mineralisation as calcium and phosphate ions are deposited, transforming into mature bone tissue. 34 This mineralisation process imparts strength and hardness to the bone structure, which undergoes constant remodelling throughout life. The newly formed bone is continually shaped and refined through a dynamic balance between bone resorption and bone formation. Osteoclasts are responsible for resorbing old or damaged bone tissue, while osteoblasts deposit new bone matrix. 35 This remodelling process helps maintain bone integrity and adapt to changing mechanical demands.
Understanding the intricacies of ECO is vital for the development of effective strategies to promote bone regeneration and repair in various clinical scenarios. By harnessing the underlying principles of this process, researchers can explore novel approaches for enhancing fracture healing, addressing bone defects and advancing regenerative medicine applications.
Endochondral ossification-based bone fracture healing process
Endochondral ossification during bone fracture repair is a multifaceted process consisting of distinct stages, each critical for the successful healing of bone fractures. This process is governed by the intricate interplay of various cell types and molecular signals.
Fracture and inflammatory phase
Following a fracture, the trauma causes blood vessels within the bone to rupture, leading to the formation of a blood clot known as a fracture hematoma. This hematoma acts as a temporary scaffold, facilitating the influx of inflammatory cells and the formation of granulation tissue. 36 The inflammatory phase is crucial for clearing debris and pathogens from the fracture site, setting the stage for healing. Immune cells release cytokines and growth factors, such as interleukins, tumor necrosis factor-alpha (TNF-α) and various chemokines, which collectively support the migration and proliferation of repair cells. 37
Soft callus formation
Within a few days of the fracture, new capillaries penetrate the hematoma, delivering essential nutrients and oxygen to the repair site. Fibroblasts and chondroblasts, primarily from the periosteum and endosteum, migrate into the fracture area. These chondroblasts produce a fibrocartilaginous matrix that bridges the fracture gap, forming a soft callus. This phase is critical as it provides mechanical stability and serves as a scaffold for subsequent bone formation. The soft callus is composed of collagen fibres and cartilage, which have not yet been mineralised. 38
Hard callus formation
In the weeks following the soft callus formation, the callus begins to ossify, transforming into a hard callus. Osteoblasts, also originating from the periosteum and endosteum, start depositing a mineralised bone matrix over the fibrocartilaginous scaffold. This leads to the formation of woven bone, which is initially disorganised and mechanically weak. However, this woven bone provides the necessary rigidity to stabilise the fracture site as it undergoes further remodelling. 39
Remodelling phase
The final phase of fracture healing is the remodelling phase, which can last several months to years. During this phase, osteoclasts resorb the excess woven bone, while osteoblasts lay down new lamellar bone in a more organised structure. This conversion process transforms the initially weak woven bone into strong, resilient lamellar bone, consisting of both compact and cancellous bone. The remodelling phase is essential for restoring the bone’s original shape, structure and mechanical strength, ensuring the functional integrity of the healed bone. 40
Molecular regulation
The process of endochondral ossification is tightly regulated by a variety of growth factors and cytokines that direct cellular activities at each stage of bone repair. Key regulatory molecules include: Transforming Growth Factor-beta (TGF-β): TGF-β enhances mesenchymal stem cell (MSC) activity, promotes extracellular matrix production, and attracts osteoblasts, the bone-forming cells. TGF-β1 specifically stimulates cartilage formation within the callus, which is crucial for proper bone regeneration. Overall, TGF-β1 orchestrates critical events during fracture healing, including callus formation, bone strengthening, and remodeling, ensuring efficient and effective bone repair.
41
Bone Morphogenetic Proteins (BMPs): BMPs, particularly BMP-2, BMP-4, and BMP-7, play a significant role in the initiation and promotion of MSC differentiation into osteoblastic and chondroblastic lineages. BMPs are essential for both the early and late stages of bone healing, from soft callus formation to hard callus mineralization.
42
Insulin-like Growth Factors (IGFs): IGFs enhance the proliferation of osteoblasts and chondrocytes and stimulate the synthesis of bone matrix proteins. The release of active transforming growth factor-β (TGFβ) and insulin-like growth factor 1 (IGF1) after resorption of the bone matrix triggers osteoblast differentiation to replace the resorbed matrix with new bone matrix.43,44
The role of hypertrophic chondrocytes in endochondral ossification
Hypertrophic chondrocytes (HyCs) play a central role in endochondral ossification. HyCs are a distinct population of cells located in the hypertrophic zone of the growth plate. They undergo a process called hypertrophy, characterised by an increase in cell size and changes in gene expression patterns. Cellular enlargement is accompanied by the expression of type X collagen (COL X), which is indicative of the HyC phenotype. 34 Figure 2 illustrates the differentiation of HyCs from MSCs followed by transformation into osteoblasts, leading to ECO.
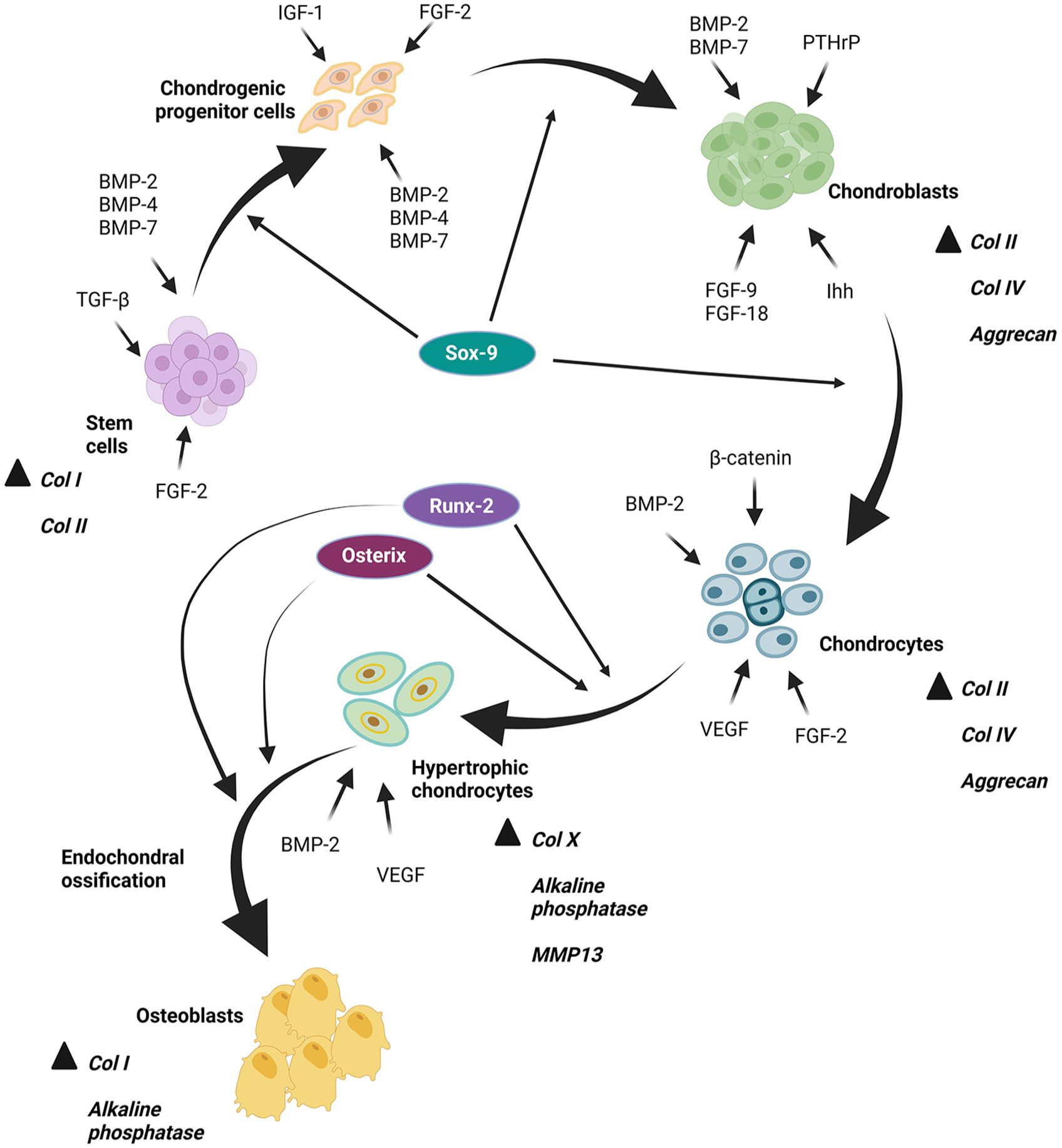
In vitro simulation of MSC differentiation into HyCs, followed by further transformation into osteoblasts, simulating endochondral ossification process. Initially, MSCs undergo differentiation into chondrogenic progenitor cells under the influence of TGF-β and BMPs, with Sox-9 serving as a critical regulator in early chondrogenesis. Further differentiation into chondroblasts is facilitated by IGF-1, FGF-2, BMP-2, -4 and -7. The transition of chondroblasts into chondrocytes is regulated by BMP-2 and -7, FGF-9 and -18, PTHrP and Ihh. During the hypertrophic phase, characterised by chondrocyte enlargement, β-catenin, BMP-2, VEGF and FGF-2 promote differentiation into HyCs. With additional stimulation from BMP-2 and VEGF, HyCs undergo osteoblastic transformation, simulating the endochondral ossification process.
One of the key functions of HyCs is their involvement in matrix mineralisation. During hypertrophy, these cells release matrix vesicles, which are small extracellular vesicles containing various enzymes and regulatory factors necessary for mineral deposition. 45 Matrix vesicles provide a scaffold for the deposition of calcium and phosphate ions, leading to the formation of a calcified cartilage matrix. This calcification process provides structural support and serves as a template for subsequent bone formation. 46
In addition to matrix mineralisation, HyCs also play a critical role in angiogenesis, which is essential for the development and repair of bone tissue. HyCs secrete vascular endothelial growth factor (VEGF), a potent angiogenic factor that stimulates the formation of new blood vessels. 47 VEGF promotes the migration and proliferation of endothelial cells, facilitating the invasion of blood vessels into the developing bone. The establishment of a vascular network is crucial for the delivery of nutrients, oxygen and osteoprogenitor cells, which are vital for the survival and function of developing bone tissue. 48
HyCs are also involved in the recruitment and differentiation of osteoprogenitor cells. They produce signalling molecules such as Indian hedgehog (IHH) and parathyroid hormone-related protein (PTHrP), which regulate the balance between chondrogenesis and osteogenesis. 49 IHH promotes the differentiation of MSCs surrounding HyCs into osteoblasts, initiating bone formation. PTHrP acts as a negative regulator, preventing premature differentiation and maintaining the chondrocyte phenotype. 50
HyCs were traditionally believed to undergo apoptosis, resulting in cell death. However, recent studies have challenged this notion and proposed that HyCs can adopt alternative fates. Transdifferentiation, a process where differentiated cells convert into another cell type due to intrinsic or extrinsic factors, has been suggested as one possible fate for HyCs.51,52 In the context of endochondral bones, early investigations in mouse and rat growth plates indicated that HyCs can assume different cellular fates, including apoptosis or transdifferentiation into osteoblasts. 53 Morphometric studies employing 5-Ethynyl-2′-deoxyuridine (EdU)-labelling by Roach suggested that terminally differentiated HyCs can re-enter the cell cycle and differentiate into osteoblasts at the ossification front. 54 Additionally, HyCs have been shown to have the potential to undergo osteogenic differentiation in response to extrinsic factors present in the bone microenvironment, such as gradients of signalling molecules and high concentrations of peptides, ions and glycans.55–57 These findings collectively indicate that not all HyCs are destined to die and suggest the possibility of transdifferentiation into osteoblasts.32,58,59 Understanding the pivotal role of HyCs in matrix mineralisation, angiogenesis and osteoprogenitor cell dynamics enables researchers to design tailored strategies, including scaffolds, that provide a conducive environment supporting the HyCs phenotype, with the goal of harnessing their potential in tissue engineering.
The role of hypertrophic chondrocytes in osteo-angiogenesis duo
HyCs play a crucial role in the process of osteogenic-angiogenic coupling, which refers to the coordinated formation of new blood vessels and bone tissue during ECO. This process is essential for the successful development and repair of bone tissue. During ECO, HyCs undergo a series of complex phenotypic changes. These cells exhibit increased volume, secrete various signalling molecules and express specific genes that contribute to both angiogenesis and osteogenesis. VEGF acts as a paracrine factor, stimulating the migration and proliferation of endothelial cells and leading to the formation of a vascular network. 48 Additionally, HyCs produce other angiogenic factors, such as fibroblast growth factor (FGF) and platelet-derived growth factor (PDGF), which further contribute to angiogenesis.60,61
The interaction between HyCs and endothelial cells is bidirectional. While HyCs release angiogenic factors that promote blood vessel formation, the newly formed blood vessels provide oxygen and nutrients essential for the survival and function of HyCs. Oxygen tension within the hypertrophic zone is critical for the regulation of chondrocyte function. Studies have shown that low oxygen tension (hypoxia) in the hypertrophic zone stabilises the transcription factor hypoxia inducible factor-1 alpha (HIF-1α), which in turn upregulates the expression of VEGF and other angiogenic factors.62,63 This hypoxia-inducible angiogenic response ensures a sufficient blood supply to the developing bone tissue. The role of hypoxia in the endochondral ossification process is described in further detail in the next section.
When the extracellular matrix (ECM) of HyCs undergoes mineralisation, leading to the formation of calcified cartilage, calcium ions and other mineralisation factors are released that promote the recruitment and differentiation of osteoblasts. Osteoblasts, in turn, secrete factors like bone morphogenetic proteins (BMPs) and transforming growth factor-beta (TGF-β), which promote angiogenesis and contribute to the formation of a functional vasculature within the developing bone tissue.47,61 This intricate interplay between HyCs, endothelial cells and osteoblasts ensures the coordinated development of the vascular network and bone tissue during ECO.
The role of hypoxia in endochondral bone formation
ECO governs the embryological development of the majority of bones, including the long bones of both the axial and appendicular skeleton.34,46 This process initiates with the condensation of mesenchymal cells at a specified anatomical locale, culminating in the formation of cartilaginous templates termed cartilaginous anlagen, ultimately progressing into fetal growth plates. Within these growth plates, distinctive avascular mesenchymal tissues are hypoxic during their expansion phase. 62 Notably, chondrocytes residing within these structures exhibit exceptional adaptability and differentiation in response to hypoxia, a phenomenon influenced in part by the regulatory actions of HIFs. 62 Concurrently, chondrocytes play a pivotal role in orchestrating localised vascularisation at the cartilage periphery, which is imperative for the sustained development and growth of bone tissue. 64
Within the nascent cartilaginous moulds and later within the fetal growth plates, chondrocytes actively biosynthesize an extracellular matrix characterised by a conspicuous prevalence of type II collagen (COL II). Exhibiting heightened proliferative capacity, these cells organise into stratified columnar layers, as illustrated in Figure 3. The terminal cells within these layers undergo cell cycle arrest and hypertrophy, transitioning into HyCs responsible for the synthesis of COL X and subsequent mineralisation of the surrounding matrix. Significantly, the proliferative chondrocytes exhibit resistance to vascular invasion, owing to the active secretion of angiogenic inhibitors, including chondromodulin1 and tenomodulin. 65

In embryonic development, the oxygenation of avascular cartilage, particularly in the central region of the fetal growth plate, undergoes regulation by HIF-1α and VEGF. As the growth plate expands, the central portion becomes hypoxic (depicted in blue). The induction of VEGF expression by chondrocytes within the growth plate, stimulated partly by hypoxia and mediated through HIF-1α, triggers angiogenesis, thereby enhancing the oxygen supply to chondrocytes. Additionally, HIF-1α plays VEGF-independent roles in regulating the survival of hypoxic chondrocytes, including the activation of the glycolytic metabolic pathway. Both HIF-1α and VEGF exhibit diverse effects at various stages of endochondral bone development, although the precise molecular mechanisms governing these pleiotropic functions remain largely unknown.
As the fetal growth plate undergoes expansion, the central core of the cartilage mould remains hypoxic due to the absence of vascularisation in this specific anatomical region.62,66 In contrast, the transition of chondrocytes into HyCs is associated with angiogenic stimuli, notably VEGF, that recruit blood vessels into the region. 64 Concurrently, osteoblast precursors and specialised osteoclasts, identified as chondroclasts, infiltrate the region occupied by terminal HyCs, instigating the controlled erosion of the underlying cartilage.34,47 Subsequently, differentiating osteoblasts deposit a bone matrix, predominantly comprised of type I collagen, onto the calcified matrix left by the HyCs.34,47 Collectively, these events culminate in the establishment of the primary ossification centre within the central region of developing long bones.34,47
The transformation from avascular cartilage to highly vascularised bone and marrow tissues involves a series of vascularisation processes.34,46,47,64 Primary ossification is triggered by the invasion of blood vessels from the surrounding perichondrium.34,46,47,64 Subsequently, progressive capillary infiltration along the metaphyseal border of the growth plate facilitates accelerated bone lengthening.34,46,47,64 Simultaneously, a network of blood vessels overlays the avascular cartilage at the bone ends (epiphyses), expanding on the cartilage surface during its growth phase.34,46,47,64 Postnatally, these vessels penetrate the epiphyseal cartilage, marking the initiation of secondary ossification centres at each end of the long bones, serving as a precursor to the termination of longitudinal bone growth.27,66
The role of hypertrophic chondrocytes in bone resorption
While HyCs are primarily associated with the process of endochondral bone formation, emerging research suggests their involvement in regulating osteoclastogenesis; the formation and activation of osteoclasts, which are the cells responsible for bone resorption. Several studies have demonstrated that HyCs express various factors that can influence osteoclast differentiation and activity. It has been suggested that HyCs may secrete factors that attract osteoclast precursor cells and promote their differentiation and fusion into mature osteoclasts. 67 One such factor is the receptor activator of nuclear factor-kappa B ligand (RANKL), a key cytokine known to stimulate osteoclast formation and function.68,69 Additionally, HyCs produce osteoprotegerin (OPG), a decoy receptor for RANKL that acts as an inhibitor of osteoclastogenesis. 70 The balance between RANKL and OPG expression by HyCs may influence the formation and activity of osteoclasts. Furthermore, HyCs secrete factors that can indirectly modulate osteoclastogenesis, such as macrophage colony-stimulating factor (M-CSF) and various matrix metalloproteinases (MMPs), required for osteoclast precursor cell survival, differentiation, proliferation and bone resorption.71,72
In vitro simulation of hypertrophic chondrocyte-mediated bone regeneration
Strategies aimed at recapitulating ECO generally involve a two-step process. 7 Firstly, a cartilaginous intermediate is engineered in vitro, mimicking the initial stage of bone development. Secondly, the engineered cartilaginous template is implanted at the site of the bone defect to induce bone regeneration. 7 In vitro simulation approaches have been developed to mimic the process of HyC-mediated bone regeneration, providing insights into the cellular and molecular mechanisms involved and guiding the development of regenerative strategies. These approaches involve the use of cell culture systems and biomaterial scaffolds that replicate the native microenvironment of HyCs and their interactions with other cell types.
A widely employed strategy involves culturing HyCs in three-dimensional (3D) scaffolds that replicate the composition and structure of the growth plate ECM. These scaffolds offer a supportive milieu for the growth and differentiation of HyCs and can be engineered to incorporate various factors that regulate HyCs behaviour, including growth factors and signalling molecules. 73 To illustrate, natural biomaterials such as collagen or synthetic polymers can be modified to create scaffolds capable of delivering specific cues that enhance HyC proliferation, matrix synthesis and osteogenic differentiation.25,73,74 Moreover, the incorporation of co-cultures involving HyCs and other cell types, such as osteoblasts or endothelial cells, in a multi-layered system can replicate the intricate cellular interactions observed during endochondral bone formation. This enables the examination of paracrine signalling between different cell populations and the investigation of their collective impact on tissue regeneration. Co-culture systems can be established through techniques like cell sheet engineering, where distinct cell types are cultured in multiple layers, or microfluidic platforms that provide precise control over cell-cell interactions and the creation of soluble factor gradients.75,76
In vitro simulation approaches also encompass the utilisation of bioreactors to deliver mechanical stimulation and ensure proper nutrient supply to the scaffolds seeded with HyCs. Bioreactors can apply mechanical forces, such as compression or fluid shear stress, replicating the physiological loading conditions experienced by HyCs during bone development. These mechanical stimuli play a crucial role in influencing HyCs differentiation, matrix synthesis and the formation of functional tissue constructs. Additionally, bioreactors provide an environment conducive to the growth and development of engineered tissues by maintaining optimal nutrient levels and promoting tissue maturation.77,78
Potential cell sources to generate hypertrophic chondrocytes for endochondral ossification
Differentiating various cell sources into HyCs is a critical step in mimicking the process of ECO for BTE. Several cell sources have been investigated for their potential to differentiate into HyCs. Primary chondrocytes derived from articular cartilage or growth plate cartilage have been utilised as a cell source for HyC differentiation. Chondrocytes can be isolated and expanded in culture and under appropriate conditions, induced to progress to the hypertrophic stage.79,80 However, the availability and proliferation capacity of primary chondrocytes are limited, making them less feasible for large-scale tissue engineering applications. Articular cartilage is a unique tissue that naturally resists hypertrophy, angiogenesis and neo-ossification. 81 In its normal physiological state, articular chondrocytes remain quiescent without undergoing proliferation or terminal differentiation; however, in the context of osteoarthritis, articular chondrocytes can undergo processes resembling ECO. 7 This leads to abnormal proliferation and disruption of their phenotype, characterised by hypertrophy and the expression of genes such as COL X, MMP-13 and alkaline phosphatase (ALP). 81 Exploiting these inherent characteristics, attempts have been employed utilising osteoarthritic (OA) articular chondrocytes to engineer constructs that undergo ECO-like processes upon implantation, either subcutaneously or orthotopically. 81 In a recent study, it was shown that the administration of TGF-β1 during the ex vivo expansion of human articular chondrocytes redirects them towards a hypertrophic phenotype. 82 While this effect may be undesirable in traditional chondrocyte culture or cartilage tissue engineering, it can be advantageous in the context of endochondral bone engineering. Besides articular cartilage, nasal septal cartilage can also be potentially differentiated into HyCs. 83 However, generating tissue-engineered cartilaginous grafts using osteoarthritic chondrocytes poses several challenges. These challenges encompass obtaining osteoarthritic chondrocytes and inducing osteoarthritis before initiating the endochondral ossification process. The clinical translation of engineered constructs using articular chondrocytes or nasal septal chondrocytes is currently constrained by challenges such as maintaining phenotype stability, potential donor site morbidity and limitations in in vitro expansion techniques. Further research and development are needed to overcome these obstacles and advance the clinical application of primary chondrocyte-based approaches.
MSCs are the most commonly used cell source in BTE due to favourable characteristics such as rapid proliferation, multipotency, self-renewal capacity and secretion of various cytokines and growth factors.84,85 The secreted bioactive molecules possess properties against inflammation and immune response, enabling MSCs to regulate inflammation and facilitate tissue regeneration. 86 One significant advantage of utilising MSC-based strategies is their immune privilege, as they do not elicit an immune response.86,87 Their multifunctional capabilities and immunomodulatory effects make MSCs a valuable cell source for BTE applications. MSCs play essential roles in the ECO process, contributing to the formation and maturation of HyCs. 86 Studies have demonstrated that MSCs can undergo chondrogenic differentiation and progress through the stages of chondrocyte maturation, ultimately reaching the hypertrophic phenotype.88,89 In vitro chondrogenic differentiation of bone marrow-derived MSCs (BMSCs) resembles the process of ECO. During chondrogenesis, BMSCs gradually produce COL II and COL X, mirroring the sequential expression of these collagens observed in ECO. 90 While the majority of investigations generate a cartilaginous scaffold in vitro that subsequently undergoes mineralisation upon in vivo implantation, some studies demonstrated the capacity for pre-mineralisation of these constructs within in vitro environments.90,91 The addition of a phosphate donor to the culture medium can induce mineralisation within the engineered constructs, simulating the mineralisation process observed during ECO.90,91 However, in vitro pre-mineralisation reduces the pro-angiogenic potential of the cartilaginous template, 91 which may affect the vascularisation process after in vivo implantation. In contrast, when chondrogenically primed constructs are implanted in vivo, they have the potential to undergo bone formation, deposit mineralised matrix and allow for blood vessel ingrowth. 92 Various chondrogenic induction protocols involving growth factors, such as TGF-β and BMPs, have been employed to promote the differentiation of MSCs into HyCs.89,93,94
Adipose tissue-derived stem cells (ADSCs) possess multilineage differentiation potential, including chondrogenic differentiation. ADSCs have been induced to differentiate into chondrocytes and progress to the hypertrophic stage, exhibiting characteristics similar to native HyCs. 95 Various growth factors and culture conditions have been employed to promote chondrogenesis in ADSCs. 96 The addition of TGF-β to ADSCs promotes the expression of hypertrophic markers, including COL X and ALP, indicating their differentiation into HyCs. 95 ADSCs, when cultured in a 3D environment, develop cartilage-like tissue, and upon treatment with L-thyroxin and β-glycerophosphate, they demonstrate an increased expression of hypertrophic markers, further confirming their potential to differentiate into HyCs.97,98 Moreover, combining ADSCs with other cell types may enhance their differentiation into HyCs. Co-culturing ADSCs with chondrocytes promotes chondrogenic differentiation, with further culture in hypertrophic differentiation conditioned medium resulting in the formation of hypertrophic cartilage-like tissues.99,100
Induced pluripotent stem cells (iPSCs) have emerged as a pivotal tool in regenerative medicine, particularly in the generation of hypertrophic chondrocytes for endochondral ossification. 101 iPSCs are reprogrammed from adult somatic cells and possess the ability to differentiate into various cell types, including chondrocytes, which can then undergo hypertrophy – a critical stage in endochondral ossification. Recent advancements have enabled the differentiation of iPSCs into chondrocytes that exhibit hypertrophic characteristics, akin to those found in the growth plate cartilage during skeletal development. 102 These hypertrophic chondrocytes express markers such as type X collagen and can mineralise their extracellular matrix, thus mimicking the natural progression towards ossification. 102 The ability to generate these cells in vitro provides a unique opportunity to study skeletal disorders and develop new treatments. The differentiation process typically involves the use of growth factors and cytokines to guide iPSCs through stages of mesenchymal progenitor cells and chondroprogenitors before reaching the hypertrophic phase. 103 This process can be optimised to enhance the yield and quality of the hypertrophic chondrocytes, which is crucial for their application in simulating the endochondral ossification process. 104
Embryonic Stem Cells (ESCs), derived from the inner cell mass of an embryo, possess the ability to differentiate into cells of all three germ layers. When subjected to appropriate differentiation cues, ESCs can be directed towards the chondrogenic lineage and subsequently induced to undergo hypertrophy.105–107 The practice of co-culturing embryonic and other stem/progenitor cells alongside mature cells or tissues is becoming more common as a method to guide their specialisation into desired cell lineages.39,108,109 In the past, the differentiation of ESCs into chondrocytes in vitro has been most successful with mouse cells. This was accomplished by adding specific factors, including TGF-β1, BMP-2 and BMP-4, or by co-culturing with embryonic limb buds.110,111 The in vitro chondrogenic induction of mouse ESCs leads to the deposition of cartilage matrix on ceramic scaffolds, and upon subcutaneous implantation in subcutaneous sites or critical-sized cranial defects, these constructs demonstrate simulation of endochondral bone formation. 112
Human embryonic stem cell-derived mesenchymal stem cells (ESC-MSCs; unlike ESCs, ES-MSCs do not differentiate into hematopoietic cells and they belong to the mesenchymal lineage) possess the specific differentiation potential of MSCs but exhibit enhanced proliferative and immunosuppressive capabilities. 113 Studies have shown that ESC-MSCs have the capacity to recapitulate the process of ECO and repair bone defects without preimplantation differentiation into osteo- or chondroprogenitors.114–116 These findings suggest the potential of ESC-MSCs as a semiautonomous approach for bone regeneration and highlight their advantages over other cell sources.
Periosteum, a tissue surrounding the outer surface of bones, contains a population of progenitor cells in the inner cambium layer that can differentiate into chondrocytes. 117 These progenitor cells demonstrate multipotent qualities similar to MSCs. 118 However, they exhibit higher clonogenicity, enhanced differentiation potential and a greater capacity for bone regeneration. 119 Periosteum derived progenitor cells can be primed to differentiate into chondrocytes, followed by the formation of intermediate cartilage tissue. 120 Therefore, these cells, can be isolated from periosteal tissues and cultured in a chondrogenic medium supplemented with appropriate growth factors followed by culturing them in a hypertrophic differentiation medium to generate HyCs with the potential to simulate the ECO process for bone formation.
Suitable biomaterials for hypertrophic chondrocyte construct generation
The generation of functional HyCs constructs for BTE requires the selection of biomaterials that can provide an optimal microenvironment for cell attachment, proliferation and differentiation. Various biomaterials have been investigated, including natural and synthetic polymers, and composite materials, each offering unique advantages and characteristics.
Natural polymers
Natural polymers derived from ECM components have gained attention due to their biocompatibility, bioactivity and ability to mimic the native tissue environment. Collagen, gelatin, hyaluronic acid and chitosan are commonly used natural polymers in BTE. These materials offer cell-adhesive properties and can facilitate chondrogenic differentiation.121–130 They can be processed into scaffolds with suitable porosity, mechanical properties and degradation rates, promoting HyC growth and tissue development.
Collagen
Collagen is the most abundant protein in the ECM of bone and cartilage. 131 It offers excellent biocompatibility, biodegradability and structural similarity to the native tissue environment, making it an attractive choice for tissue engineering applications. Collagen scaffolds can support cell attachment, proliferation and differentiation, including hypertrophic differentiation of chondrocytes.120,132,133 Additionally, collagen scaffolds can be functionalised with growth factors or other bioactive molecules to enhance the differentiation and maturation of HyCs.25,134 Porous collagen sponges were first used to generate cartilage in vitro and have recently also been used to generate endochondral bone.30,135,136
Gelatin
Gelatin is derived from collagen through a process of hydrolysis. It shares many properties with collagen and has been widely used in tissue engineering, including bone regeneration. Gelatin plays a significant role in 3D cell culture and has been used alone and in combination with other materials to improve endochondral bone regeneration. 137 Similar to collagen scaffolds, gelatin scaffolds can also support cell adhesion, proliferation and differentiation, inclusive of ECO. 138 Gelatin methacryloyl (GelMA) hydrogels, synthesised from gelatin and methacrylic groups, have been loaded with MSCs and chondroinductive particles to stimulate endochondral bone formation in a subcutaneous rat model.139,140
Hyaluronic acid
Hyaluronic acid is a naturally occurring glycosaminoglycan present in the ECM of many tissues. It possesses excellent biocompatibility, biodegradability and hydrophilicity. Hyaluronic acid scaffolds provide a favourable microenvironment for the differentiation of chondrocytes into HyCs and can be functionalised with growth factors or other bioactive molecules to promote HyC differentiation and tissue regeneration. 141 Regulating the chondrogenic and hypertrophic characteristics of MSCs through their interaction with scaffold materials is crucial when creating tissue-engineered cartilage grafts suitable for clinical use 141 ; Cells that have undergone chondrogenic differentiation placed within hyaluronic acid hydrogels, adopt a rounded shape. This rounded morphology is beneficial as it ensures a more consistent chondrogenic differentiation process.74,142 The mechanical properties of hyaluronic acid-based scaffolds can be tailored to promote chondrocyte hypertrophy and mineralisation during the differentiation culture of MSCs. 79 Hyaluronic acid can enhance the expression of chondrogenic markers, such as COL II and aggrecan, and induce the expression of hypertrophic markers, including COL X.143,144
Chitosan
Chitosan is a polysaccharide derived from the deacetylation of chitin, a component found in the exoskeleton of crustaceans. It possesses biocompatibility, biodegradability and antimicrobial properties, making it suitable for tissue engineering applications. 145 Chitosan provides a favourable microenvironment for chondrocyte proliferation and induces chondrogenesis and ECM synthesis. 146 Incorporation of hyaluronic acid-chitosan nanoparticles into a pellet co-culture of human infrapatellar fat pad-derived MSCs with osteoarthritic chondrocytes has been shown to enhance chondrogenic differentiation. 147 When these MSCs are seeded on 3D-printed chitosan scaffolds and cultured in chondrogenic media containing TGF-β3 and BMP-6, they attach, proliferate and differentiate into chondrocyte-like cells, promoting the formation of cartilaginous tissue in vitro.148,149
Alginate
Alginate is a natural polysaccharide derived from brown algae. It forms hydrogels that can encapsulate cells and provide a supportive microenvironment for their growth and differentiation. Alginate is a promising biomaterial for chondrogenic induction. 150 Combination of alginate microspheres and hyaluronic acid hydrogel serves as a carrier for MSCs and TGF-β, retaining the bioactivity of the growth factor within the scaffold. 151 This composite system promotes chondrogenesis of MSCs, facilitating the differentiation of MSCs into chondrocytes and supporting cartilage tissue regeneration. 151 Alginate hydrogels can be further functionalised with growth factors or other bioactive molecules to enhance HyC differentiation and tissue regeneration. Moreover, the mechanical properties of alginate hydrogels can be modified by crosslinking agents or blending with other polymers to tailor the scaffold properties according to the specific requirements of tissue engineering construct generation.152,153
Fibrin
Fibrin, derived from the monomer fibrinogen, is a biopolymer that is vital for blood clotting, cell-matrix interactions, inflammation and wound healing. 154 Fibrin hydrogels provide a biocompatible and biodegradable scaffold that closely mimics native ECM. They offer favourable mechanical properties, cell adhesion sites and the ability to sequester and release growth factors, making them suitable for supporting chondrogenic differentiation and HyC construct generation. Fibrin hydrogels facilitate MSC-mediated vascularisation, endochondral bone formation and bone marrow development. 74 They have also been shown to support the attachment, proliferation and differentiation of chondrocytes.155,156 Additionally, fibrin hydrogels can be combined with other biomaterials or synthetic scaffolds to enhance their mechanical properties and provide a more robust scaffold for HyC construct generation. 7
Synthetic polymers
The mechanical strength and degradation rate of natural polymers often pose limitations. Synthetic polymers have been extensively explored for supporting cellular differentiation in tissue engineering including for endochondral bone tissue regeneration owing to their chondrogenic conduction, good biocompatibility, slow degradation and tunable mechanical strength. 149 Some of the commonly studied synthetic polymers having great potential in generating HyCs construct include:
Poly(lactic-co-glycolic acid) (PLGA)
PLGA is a biocompatible and biodegradable synthetic polymer widely used in tissue engineering. It offers tunable mechanical properties, making it suitable for various tissue engineering applications. The porous structure of PLGA scaffolds facilitates cell infiltration and nutrient transport, allowing for the growth and differentiation of chondrogenic cells.157–159 Incorporating extracellular matrix derived from human amniotic mesenchymal cells into the PLGA scaffold could provide a favourable growth environment for MSCs and promote chondrogenic differentiation. 160 PLGA scaffolds loaded with either chondrogenically predifferentiated rat BMSCs or nasal chondrocytes have been demonstrated to heal bone defects in rats by mimicking the endochondral pathway of bone regeneration.161,83 Nevertheless, while the porous PLGA scaffold facilitates cellular penetration and nutrient delivery, its hydrophobic nature may pose a potential obstacle to optimal cellular adhesion and proliferation.162,163
Polyethylene glycol (PEG)
PEG is a hydrophilic polymer that offers tunable mechanical properties and excellent biocompatibility. It has been employed in conjunction with methacrylated chondroitin sulphate, a constituent found in native cartilage and its ECM. This naturally occurring material is well recognised for its positive impact on the proliferation and differentiation of chondrocytes. The incorporation of this material with PEG is utilised to fabricate composite scaffolds conducive to chondrogenic differentiation. 164 PEG hydrogels have also been incorporated with hyaluronic acid to encapsulate cells and provide a suitable microenvironment for chondrogenesis. 165 In another study, it was demonstrated that PCL-PEG-PCL (PECE) films seeded with ADSCs supported cell adhesion, proliferation and cartilage formation. However, this synthetic polymer is yet to be investigated for differentiating HyCs. 166
Polycaprolactone (PCL)
PCL is a synthetic polyester with good mechanical properties and a slow degradation rate. 167 It has been applied as a scaffold to support the differentiation of MSCs into chondrocytes, including HyCs. 168 PCL scaffolds can be fabricated through electrospinning or 3D printing, in combination with other biomaterials to create structures suitable for cartilage tissue engineering.169–171 Although PCL scaffolds provide support for stem cell differentiation, including for chondrogenic and osteogenic induction, the hydrophobicity of PCL requires surface modification through chemical or plasma treatment. 172 Hybridisation of PCL with PLGA has enabled chondrogenic differentiation of MSCs followed by endochondral bone formation in a rat model.173,174
Poly(lactic acid) (PLA)
PLA is a biodegradable and biocompatible synthetic polymer that has been widely studied and applied in cartilage tissue engineering. Several studies have demonstrated the effectiveness of PLA scaffolds in promoting chondrogenic differentiation and cartilage regeneration. PLA fibrous scaffolds have been shown to possess a robust structure that supports high proliferation rates of seeded MSCs in physiological solutions. 175 In a rabbit model, constructs consisting of MSCs seeded into PLA scaffolds formed hyaline-like cartilage tissue, indicating the potential of PLA for cartilage tissue engineering applications. 176 Furthermore, the addition of nanohydroxyapatite to PLA scaffolds has been shown to enhance cell attachment, proliferation and differentiation of the progenitor cells to bone-like tissue.177,178
Ceramics
Ceramics are widely recognised for their exceptional biocompatibility and are increasingly utilised for compressive loading. 179 Some bioceramics, such as beta-tricalcium phosphate (β-TCP), hydroxyapatite (HAp) and biphasic calcium phosphate (BCP), have been extensively studied as scaffolds for endochondral bone formation. 113 The specific type of bioceramic used can influence the capacity for endochondral bone regeneration. 74
(i) β-TCP has been shown to promote cartilage regeneration and biomineralisation 180 and support endochondral bone formation. 181 Combining β-TCP with other materials can enhance the chondrogenic and endochondral potential of scaffolds. Incorporating β-TCP particles into a 3D biomimetic hydrogel scaffold induces the expression of chondrogenic markers, such as COL II and aggrecan, as well as the hypertrophic marker COL X and the osteogenic marker ALP. 182
(ii) HAp, as the main mineral component of natural bone, has been extensively studied as a bone biomaterial.183–185 HAp exhibits good osteoinductivity, 186 high osteoconductivity187,188 and excellent biocompatibility. 7 Therefore, HAp-based scaffolds have been widely used to promote bone regeneration. HAp scaffolds have shown enhanced vascularisation and recruitment of host endothelial cells when cells progress through the endochondral route compared to the intramembranous route. 189 Incorporating HAp particles into biodegradable polymers, such as collagen 25 and poly(vinyl alcohol), 190 improves the mechanical properties of scaffolds, resulting in mechanical strength comparable to that of bone or cartilage. Additionally, HAp-coated titanium scaffolds have been shown to promote chondrogenic differentiation of MSCs and enhance bone formation when implanted into rat mandibles. 191 However, HAp has high crystallinity and stability, which can hinder its degradation in vivo. 192
(iii) Biphasic calcium phosphate (BCP) bioceramics have gained recognition as ideal bone graft substitutes due to their demonstrated safety, osteoconductivity, bioactivity, biocompatibility, abundance and potential applications in tissue engineering and drug delivery systems.193,194 BCP scaffolds, comprising different proportions of HAp and β-TCP, can exhibit diverse surface topographies, pore sizes and porosities.193,195 Combining these two materials provides the predictable biodegradability offered by β-TCP and the improved resilience provided by Hap. 7 Studies have indicated that porous BCP scaffolds can influence cellular differentiation. 196 For instance, a total porosity of 65% promotes growth/matrix production and cell differentiation, as evidenced by total protein production and ALP activity in human MSCs. Scaffolds with total porosities of 25% and 75% were less supportive. 197 Moreover, porous HAp/β-TCP granules have the ability to support endochondral bone formation at ectopic sites by enhancing biomineralisation. 198
Current endochondral ossification-based in vitro and in vivo bone tissue engineering approaches
Table 1 comprehensively discusses a range of current endochondral ossification-based in vitro and in vivo bone tissue engineering approaches. These studies collectively explore innovative strategies aimed at enhancing bone regeneration through the manipulation of hypertrophic chondrocytes and their interactions within engineered scaffolds.
Applications of various in vitro and in vivo strategies to simulate the endochondral ossification.

Future perspectives of using endochondral ossification to repair critical-sized craniofacial bone defects
The potential application of hypertrophic chondrocytes in repairing segmental mandibular defects presents a promising avenue for craniofacial regenerative medicine. Previous in vitro and in vivo studies, as detailed in our comprehensive table, have predominantly focused on the impact of hypertrophic chondrocytes or cartilaginous grafts in the repair of cranial and long bones. While long bone healing typically follows an endochondral pathway, characterised by a cartilage intermediate, this process may not be as apparent in the healing of flat bones such as those in the craniofacial region. 23 However, the evidence compiled in Table 1 demonstrates that hypertrophic chondrocytes can effectively repair cranial bone defects, which, like the mandible, are categorised as flat bones. Furthermore, Table 1 also provided evidence where hypertrophic chondrocytes or cartilaginous grafts demonstrated its potential in repairing segmental mandibular defect in small animal model. These findings suggest that the mandible may also utilise an endochondral pathway during the fracture healing process. This is significant because it indicates that hypertrophic chondrocytes could play a critical role in the regeneration of mandibular bone, potentially improving outcomes for patients with segmental defects. Therefore, the future of repairing craniofacial bone defects, including segmental mandibular defects using hypertrophic chondrocytes holds tremendous promise, representing a significant advancement in the field of tissue engineering and regenerative medicine. Hypertrophic chondrocytes, integral to the ECO process, offer a targeted and precise approach to bone regeneration. By harnessing their innate osteoinductive properties, researchers aim to guide the formation of new bone within complex craniofacial structures with greater accuracy and lower morbidity than current reconstructive approaches allow.
As discussed earlier, accelerating bone growth within the scaffold is essential to avoid the inhibitory effect of post-surgery radiotherpay in oral cancer patients and hence, incorporating HyCs into tissue-engineered constructs presents an opportunity to enhance the osteoinductive capacity of these materials. These cells secrete factors that stimulate surrounding cells to differentiate into osteoblasts, promoting bone formation within the defect site. By leveraging this inherent ability, researchers can create environments conducive to robust bone regeneration, even in challenging clinical scenarios such as hypoxic conditions induced by radiotherapy. This accelerated bone formation will facilitate more uniform and timely bone formation throughout the scaffold, enhancing the overall success of reconstructive efforts.
A key advantage of utilising HyCs lies in their ability to promote the formation of a transitional cartilage-bone interface. 34 The presence of hypertrophic cartilage template within the defect site promotes vascular invasion, which is essential for its gradual transition to bone within the defect site and maturation, eventually ensuring improved osteointegration. This integration reduces the risk of implant rejection or displacement, ensuring long-term biomechanical stability and functionality. Furthermore, such integration will also mimic the natural architecture of jawbones, further enhancing the success of tissue-engineered solutions.
Beyond their role in osteogenesis, HyCs exert immunomodulatory effects through the secretion of immunomodulatory factors, including macrophages and T cells that can influence the local tissue microenvironment.34,215–217 By modulating immune responses and fostering a conducive healing environment, these cells play an active role in matrix degradation and vascularisation, thereby enhancing the long-term survival of tissue-engineered constructs. This immunomodulatory function is particularly advantageous in addressing critical-sized bone defects commonly observed in cancer patients, where traditional repair strategies often face challenges due to compromised tissue quality and systemic immune dysregulation. By harnessing the immunomodulatory properties of HyCs, novel mandibular defect repair strategies can be developed to enhance the efficacy and longevity of tissue-engineered constructs, ultimately improving outcomes for patients undergoing reconstruction following cancer resection or other traumatic injuries.
The translation of HyC-based therapies from laboratory research to clinical practice necessitates meticulous preclinical testing and validation. Comprehensive, long-term studies assessing safety, efficacy and functional outcomes are imperative to establish the clinical viability of these innovative approaches. Preclinical studies should encompass various aspects, including but not limited to, biocompatibility assessments, evaluation of tissue integration, analysis of host immune response and long-term follow-up to assess durability and potential adverse effects. Moreover, studies should be conducted in relevant animal models that closely mimic the human condition, considering factors such as mandibular size, biomechanics and healing capacity. Comprehensive data obtained from preclinical investigations serve as the foundation for regulatory submissions and facilitate the design of well-controlled clinical trials, which ultimately pave the way for the successful translation of HyC-based therapies into clinical practice, offering promising solutions for patients requiring mandibular defect repair. A potential application of how HyCs can be incorporated into the repair of a segmental mandibular defect is depicted in Figure 4. Furthermore, advancements in biomaterials, scaffold design and delivery methods are poised to streamline the clinical translation of HyC-based therapies for the personalised and efficacious treatment of craniofacial bone defects.
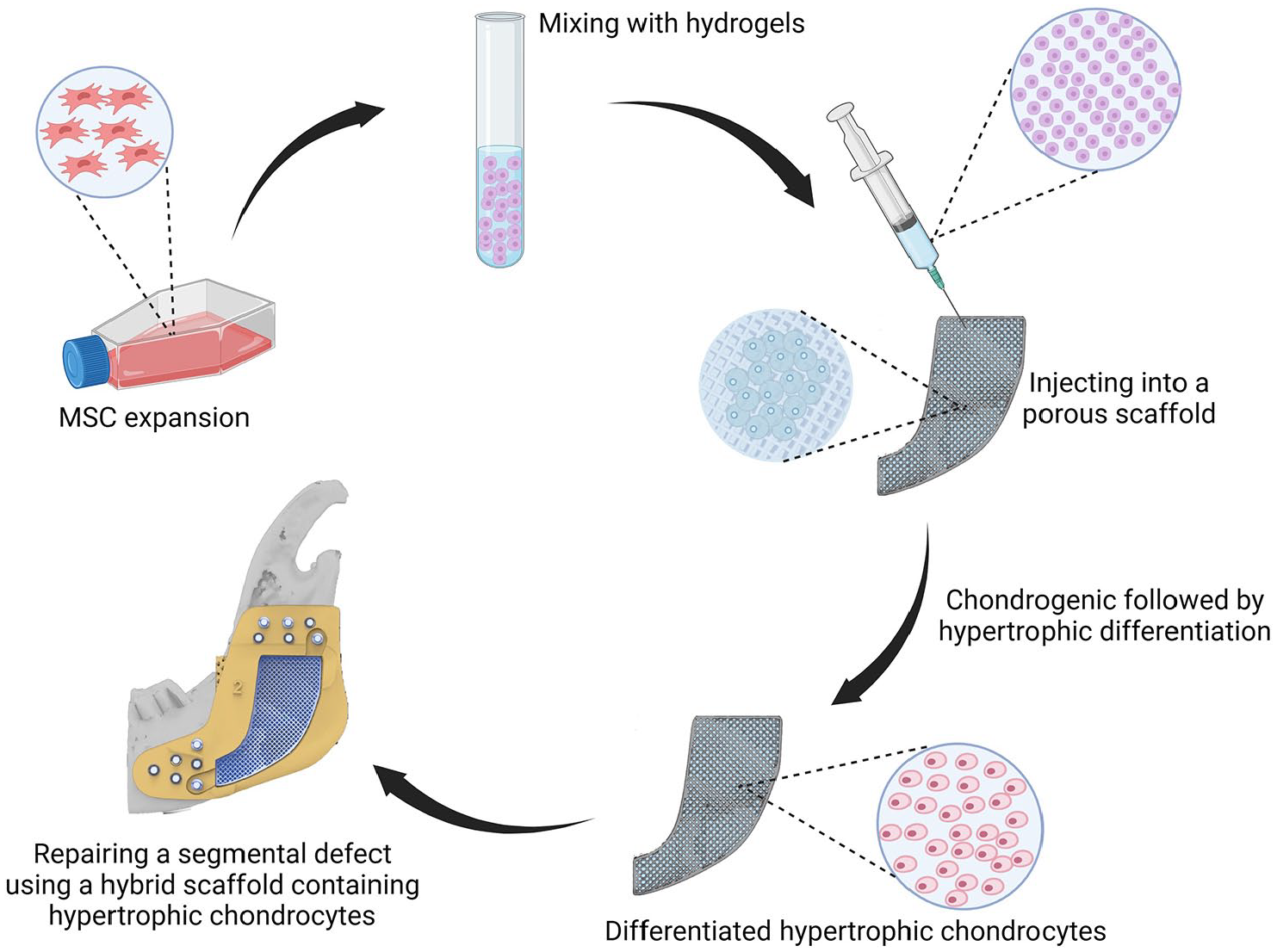
Differentiation of MSCs into HyCs within a porous scaffold followed by implantation in a segmental bone defect. MSCs are isolated and combined with hydrogels to form an injectable cell-hydrogel composite. This composite is subsequently introduced into a porous scaffold, offering structural support and promoting cell adhesion and proliferation. Within this conducive microenvironment, MSCs undergo chondrogenic differentiation, culminating in the formation of HyCs. Finally, the engineered construct containing differentiated HyCs is implanted at the site of a segmental bone defect using a hybrid scaffold, with the goal of enhancing healing and restoring functionality through the promotion of new bone formation.
Footnotes
Acknowledgements
The authors would like to thank the team members of the Integrative Prosthetic and Reconstruction team, Arto Hardy Biomedical Innovation Hub, Chris O’Brien Lifehouse, Australia and Cell, Tissue and Organ Engineering Laboratory, Biomedical Centre (BMC), Lund University, Lund, Sweden for their outstanding support in preparing this article.
Author contributions
DSAAM and HX: Conceptualisation, manuscript draft, major editing and reviewing the final manuscript; KC: Literature search, manuscript draft, figure and reviewing the final manuscript; AGG: Editing and reviewed final manuscript; MMD: Editing and reviewing final manuscript; EB-S: Editing and reviewed final manuscript; ET-C: Reviewing the final manuscript; JMC: Reviewed final manuscript; JRC: Major editing, reviewing the final manuscript and project supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to acknowledge the following sources of funding supporting this research: The Cancer Institute New South Wales (CINSW 2020/2081), the National Health and Medical Research Council (NHMRC), Sydney Local Health District, The Lang Walker Family Foundation, the Arto Hardy Family and the Federal Government of Australia through the Sarcoma and Surgical Research Centre.
Data availability statement
No datasets were generated for this review article.
