Abstract
Glatiramer acetate (GA) has been a pivotal therapy for relapsing multiple sclerosis (MS) due to its favorable safety profile. Long-term data spanning decades demonstrate its continued use in diverse patient populations. Adverse events include manageable localized injection site reactions, lipoatrophy or necrosis, and rare cases of liver injury. GA has minimal effects on immune function, and does not increase the risk of opportunistic infections, making it suitable for MS patients at risk for infections or reactivation of latent infections. GA’s immunomodulatory properties may pose a lower infection risk than other disease-modifying treatments. Progressive multifocal leukoencephalopathy risk with GA is low, and screening for latent infection is unnecessary before treatment. Vaccination is important for preventing infections in MS patients. GA does not compromise vaccine efficacy and is compatible with both inactivated and live attenuated vaccines. Special populations that may benefit from the characteristics of GA include older adults and patients with comorbidities and/or polypharmacy. MS patients often have comorbidities, necessitating careful management of potential drug interactions and side effects. Drug interactions with GA are not predicted, and clinical data suggest that the risk is low. GA is not contraindicated during pregnancy and exhibits a reassuring safety profile during breastfeeding, with no increased risk of adverse outcomes identified. Regulatory restrictions on GA use during breastfeeding have been removed. In summary, GA remains a safe and well-established therapy for MS patients, including those in special populations. Its favorable safety profile, compatibility with vaccination, and reassuring outcomes solidify its role in MS treatment.
Plane language summary
Glatiramer acetate is a relatively safe DMT with minimal effects on immune function that does not compromise vaccine efficacy, making it suitable for MS patients at risk for infections or requiring vaccination. A reassuring safety profile and minimal drug interactions make it a viable treatment option for special populations common among MS patients, such as older adults and patients with comorbidities or polypharmacy. Use during pregnancy is not contraindicated, and should consider the benefit-risk assessment, while regulatory restrictions on its use in breastfeeding have been removed.
Keywords
Glatiramer acetate
Glatiramer acetate (GA) was shown to reduce relapse rate and MRI disease activity in controlled clinical trials,1–4 leading to its approval for reducing multiple sclerosis (MS) relapse rates and MRI disease activity in patients with relapsing MS in the United States in 1996, 5 and in Europe in 2003. 6
GA is a mixture of polypeptides with an average molecular weight of 5–9 kDa, comprising four naturally occurring amino acids. This non-biological complex drug (NBCD) is manufactured by the copolymerization of l-glycine, l-lysine, l-alanine, and l-tyrosine in a molar ratio of 1.9:4.6:6.0:1.0. Omission of any one amino acid resulted in loss of effect. 7 In contrast to the original version, GA is currently produced with a molar ratio of 4.2:3.4:1.4:1, although the reason for this change remains unreported. 8 The large number of possible components with different molecular weights and sequences precludes definitive structural elucidation by component analysis 9 ; however, modern analytical and quantitative methods enabled reverse engineering of the synthesis procedure for the development of generics, ensuring equivalence with the originator. The development and production of follow-on GAs (FOGA) require a strictly controlled manufacturing process to avoid unanticipated consequences from even minor differences in the distribution of molecular weights or in the composition of antigenic polypeptide sequences, which can have an impact on the efficacy, toxicity, and immunogenicity profiles of the FOGA. 10
GA reduces inflammation by shifting reactive T cells toward a Th2 pattern, enhancing T regulatory cells (Tregs) and increasing anti-inflammatory cytokine production, while reducing the production of inflammatory cytokines.11–14 This Th2 skewing of the T cell response is one widely accepted mechanism of action for GA; however, a newer concept involves additional immunomodulatory effects on cells of the myelo-monocytic lineage through induction of anti-inflammatory “type II” antigen-presenting monocytes.15,16 In recent studies, GA treatment was linked to decreased number of plasmablasts, B cells, and memory B cells, in addition to a shift of B cells from a pro-inflammatory to an anti-inflammatory phenotype. 17 GA enhances NK cell cytolysis of dendritic cells,18–22 perhaps by enhancing interaction through NK cytotoxicity receptors and reducing MHC class I expression on dendritic cells (Figure 1).

Effects of glatiramer acetate on immune cell populations. (a) Glatiramer acetate increases the biological activities of NK cells, particularly their cytolytic potential. aHøglund et al., 20 bAl-Falahi et al., 19 cSand et al., 18 dMaghazachi et al., 21 and eSellner et al. 22 (b) Glatiramer acetate increases anti-inflammatory and reduces proinflammatory cytokine levels. Glatiramer acetate-treated B memory and regulatory cells promote the formation of Th2 T cells and regulatory cells. aAmrouche et al., 23 bHäusler et al., 24 cJackson, 25 dMaghazachi et al., 21 eSellner et al., 22 fCarrieri et al., 26 gSellebjerg et al., 27 and hHarp et al. 28 (c) Glatiramer acetate induces differentiation into type II (M2) myeloid cells and increases their phagocytic capacity. aVieira et al., 29 bJung et al., 30 cKim et al., 31 dWeber et al., 32 eBurger et al., 33 fCarpintero et al., 34 gChabot et al., 35 hPul et al., 15 and iMolnarfi et al. 36
Possible neuroprotective effects of GA (seen mainly in animal models) may be associated with increased levels of neurotrophic factors secreted by Th2-biased immune cells. Effects of GA on the neuro-proinflammatory and degenerative processes implicated in MS-associated cognitive decline may help preserve nerve cell function and integrity. 37 In experimental autoimmune encephalomyelitis (EAE), GA enhances the proliferation, migration, and differentiation of oligodendroglial and neuronal progenitor cells that foster repair, remyelination, and neurogenesis.38,39 GA protects neurons by increasing levels of neurotrophic factors and may reduce glutamate-mediated neurotoxicity.40–42 GA treatment reduced serum neurofilament light chain levels by 81% in EAE, 43 suggesting that it may serve as a prognostic marker during GA treatment.
Follow-on GAs
Access to disease-modifying therapies (DMTs) for MS is limited in many regions, especially in low- and middle-income countries, 44 and stakeholders suggest that biosimilars/generics may improve affordability and access to DMTs for MS patients.45–48 Patent protection for Copaxone® 20 mg daily (originator GA) ended in 2014. There has been debate on how to define and determine similarity between originator and FOGAs.49–54 The manufacturing process is fundamental to the production of NBCDs that maintain the characteristics of the originator product. 55
Regulatory agencies have recognized several FOGAs as equivalent to the originator in terms of safety and efficacy. In April 2015, the US Food and Drug Administration (FDA) approved a generic form of GA (Glatopa™, originally produced by Momenta Pharmaceuticals), based on the synthesis and purification procedures used, and on its physicochemical, biological, and immunological properties in EAE. 56 Subsequently, the FDA approved 20 and 40 mg formulations of a second generic GA originally produced by Natco Pharma in October 2017. These approvals were based on the Abbreviated New Drug Application process, which did not require additional clinical data.
In contrast, the European Medicine Agency (EMA) considered GA to be an NBCD and advised a third sponsor, Synthon, to demonstrate equivalent efficacy, safety, and tolerability of its generic GA product with new clinical data. These data were collected in the 9-month randomized, placebo-controlled, double-blind, Glatiramer Acetate Clinical Trial to Assess Equivalence With Copaxone (GATE) study, 57 and 15-month extension. 58 Patients aged 18–55 years old with relapsing MS (⩾1 relapse in the previous year and 1–15 gadolinium-enhancing MRI lesions) were randomly assigned to receive daily sc. injections of originator GA 20 mg (n = 357), FOGA 20 mg (n = 353), or placebo (n = 84). The study had an equivalence design and a primary endpoint of MRI-related outcome as a surrogate for relapses. Safety and tolerability were similar in both groups. Study discontinuation for any reason occurred in 3.4% of patients receiving FOGA and 1.1% receiving originator GA; however, discontinuation for any reason does not necessarily reflect a higher rate of adverse events, but could also result from a subjective tolerability issue. In the same study, 57 the occurrence of any event was reported by 51.0% of subjects receiving the FOGA and 54.3% of subjects receiving the originator GA. Similarly, the percentages of subjects experiencing any serious adverse event (3.4% and 4.8%) and any severe event (4.0% and 2.8%) in the FOGA and originator GA, respectively, suggest that these variables are comparable between the two treatments.
All participants then received FOGA in the 15-month open-label extension, 58 during which safety and tolerability, as well as the development of anti-drug antibodies, were found to be comparable in both incidence and titer (Figure 2).

Glatiramer antibody (ADA) titer: mean (∓SD) ADA titer values in patients treated with generic glatiramer acetate (GTR) over 24 months and patients treated with brand glatiramer acetate (GA) for 9 months then switched to GTR for 15 months.
Several studies using sophisticated methodologies have demonstrated differences in physico-chemical characteristics between originator GA and other glatiramoids.59,60 Studies have also reported different and sometimes contrasting effects on biological pathways relevant to the pathogenesis of MS (such as increased pro-inflammatory cytokine secretion by FOGAs) and effects on gene expression, raising concern about non-equivalent efficacy and antigenicity of FOGAs if not tested in appropriate clinical trials.53,61–63 In a genome-wide expression study performed in THP-1 cells by the company Teva (Teva Pharmaceutical Industries Ltd, Petach Tikva, Israel), originator GA modulated the expression of 6890 genes after 6 h of treatment, while treatment with the FOGA Probioglat® (Probiomed, Mexico City, Mexico) resulted in differential expression of 136 genes at the same time point. 63 The authors claim that Probioglat induced a more pro-inflammatory response compared to the originator GA. Similar shifts in expression of the genes identified (CCL5, CCL2, MMP9, MMP1, CXCL10, CD14, ICAM1, and BIRC3) were not observed in the study described above (CXCL10 was down-regulated by both originator and FOGA). 63
A study by Hasson et al. 61 showed differences between originator GA and the FOGA Polimunol® (Synthon Bago, Autonomous City of Buenos Aires, Argentina). However, this study compared a single Polimunol batch vs. three Copaxone batches, and used a gene expression difference cut-off of 1.1-fold, which is not considered biologically relevant. Instead, the genome-wide expression study conducted by Kolitz et al. compared five FOGA batches with five originator GA batches, using 1.4-fold change cut-off value at a p-value of <0.05, which could be expected to provide more biologically meaningful results. 63
Furthermore, Bakshi et al. conducted an investigation using splenocytes isolated from mice that had been immunized with the GA reference standard (Teva) and subsequently exposed ex vivo to various glatiramoids. Their comparison between GA-Teva and GA-Natco revealed significant differences in the expression of 98 genes (fold change ⩾ 1.3), which are primarily associated with T cell differentiation, immune suppression, antigen presentation, induction of Tregs, inflammatory responses, and cell adhesion. 62 In a related study, Towfic et al. developed computational methods to assess the immunological effects of branded versus generic medications. 49 Their results indicated a notable variability in gene expression following activation by the generic GA-Natco compared to GA-Teva batches. While Copaxone effectively induced the upregulation of Treg-related genes, the GA-Natco generic was associated with increased inflammatory markers linked to monocytes and macrophages (e.g., CD14, TLR2, IL1B). 49 It is important to note that these studies were performed in a healthy mouse model, which may not fully capture the complex dynamics of human MS. Nevertheless, these findings highlight the need for translational studies using human cells to better understand the distinctions between FOGA and Copaxone and their implications for treatment.49,64
The scientific discussion in the EMA public assessment report of 2016 includes a review of the above study findings, including those related to gene expression. 65 Based on all data presented, the Board did not seek further clarification during approval of FOGAs, due to methodological issues with these studies, and concluded that Mylan GA 20 mg/mL can be regarded as therapeutically equivalent (efficacy and safety) to the reference product. 65 Moreover, these studies notwithstanding, the EMA approved the 20 mg once-daily formulation of the Mylan FOGA in 2016, 66 and the 40 mg thrice-weekly formulation in 2017 67 based on the results of the GATE study, which demonstrated equivalent efficacy, safety, and tolerability of Viatris GA to the originator GA. 57
In addition to these products, FOGA alternatives have been developed under various trade names, depending on the pharmaceutical companies that produce and distribute them (Table 1).68,69
Glatiramoid class members approved as DMT for for relapsing MS.
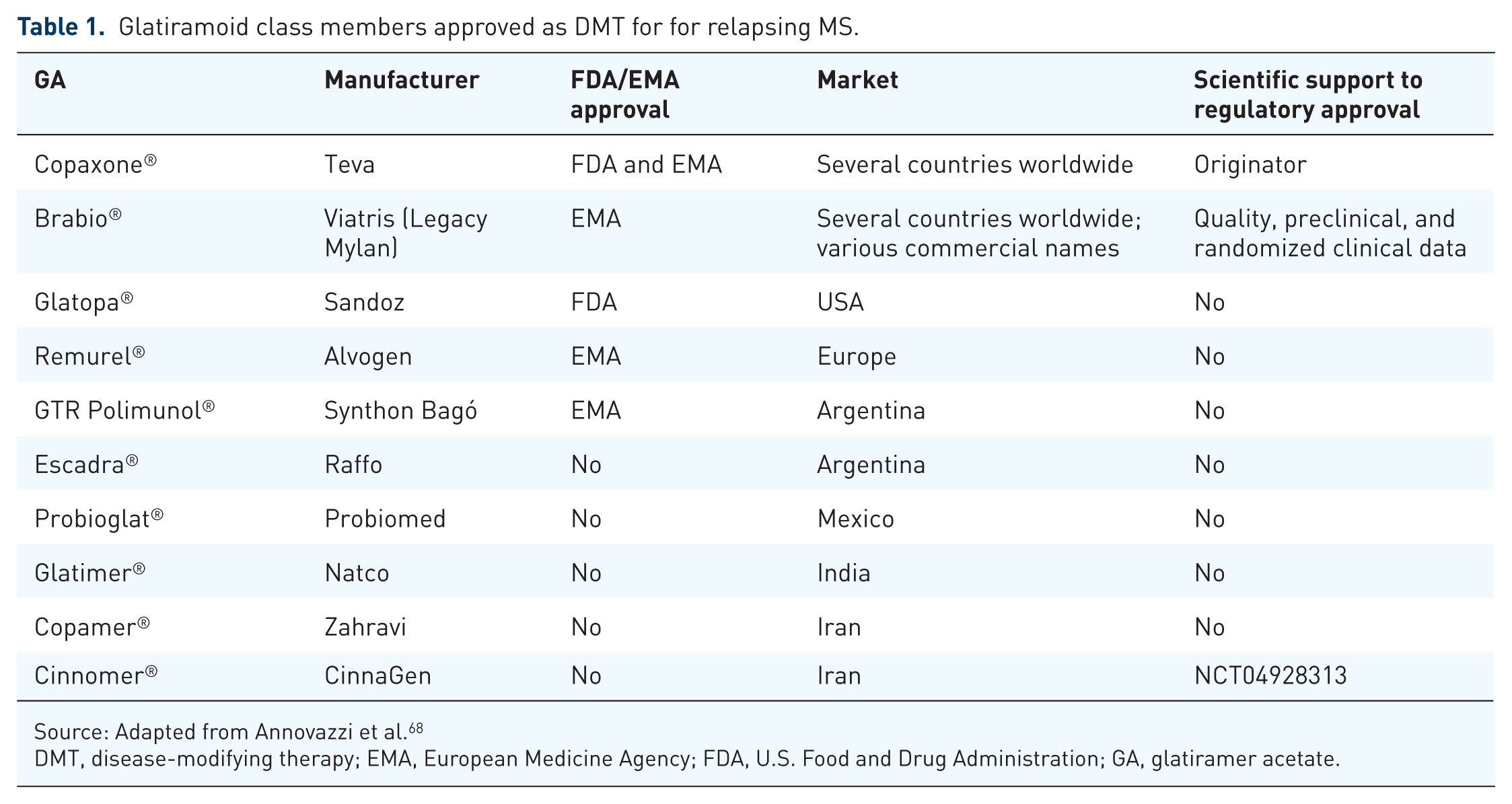
Source: Adapted from Annovazzi et al. 68
DMT, disease-modifying therapy; EMA, European Medicine Agency; FDA, U.S. Food and Drug Administration; GA, glatiramer acetate.
The lack of a centralized regulatory pathway for NBCDs and their follow-on versions 70 may affect the perception of their efficacy and safety by clinicians and patients; therefore, it is important to collect clinical data on efficacy and safety, and to conduct post-marketing surveillance to provide a better understanding of performance in real-world clinical practice. This is particularly relevant because variations in the GA production process have been associated with changes in efficacy, safety, immunogenicity, and biological activity compared to the originator GA,51,53,59,60 including effects on gene expression in biological pathways relevant to the pathogenesis of MS.61–63
This narrative review will assess the long-term safety and tolerability data for GA originator and FOGAs in the management of patients with MS. It will also present data from the Ad-hoc Safety Review Report on Viatris GA.
GA safety
After three decades and millions of patient years, GA is still a widely prescribed DMT for treating patients with relapsing MS, despite the introduction of more efficacious DMTs. 71 This is likely due to its ease of management, which does not require extensive monitoring, its favorable safety profile, including in special populations including older adults, patients with comorbidities, or those planning for pregnancy and breastfeeding. 72
Currently, the longest continuous follow-up data for GA comes from the US Glatiramer Acetate Trial conducted by Ford et al., which has 27 years of safety data on 52 patients.73,74 The extensive phase IV clinical data collected following the initial approval of originator GA support the continuing efficacy and safety of GA in relapsing-remitting MS,71,72 and is used, together with post-marketing surveillance, pharmacovigilance data, scientific publications, input from expert panels, and regulatory guidance, to curate the manufacturers’ prescribing information.
Viatris, the manufacturer of a FOGA, hereafter referred to as Viatris GA, maintains a safety surveillance database of cases reported from 12 countries in Europe. Data sources include post-marketing surveys, spontaneous reports, regulatory authorities, and publications. Cumulative exposure data from Viatris for all European countries where the product has been marketed are calculated from sales data available up to March of 2022. Cumulative patient-time exposure to both of the glatiramer formulations from the launch date to June 30, 2022 is estimated at 428,168 patient years (98,955 for the 20 mg/mL dose, and 329,213 for the 40 mg/mL dose).
As of June 30, 2022, Viatris has received a total of 5366 cases from the global safety database. In 192 cases, the manufacturer of the GA product could not be established. Based on review of the cumulative data, no new safety or efficacy concerns were identified in patients switching from originator to Viatris GA, consistent with the results from patients switching from originator to Viatris GA, in the GATE Extension study, 58 and with reported results from patients switching from originator to the FOGA Copamer®. 75 Overall, cumulative post-marketing experience with Viatris GA has been consistent with the known safety profile of originator GA. More than 80% of the reported adverse events were non-serious in nature.
Injection site reactions
Injectable MS DMTs commonly cause localized injection site reactions and a range of dermatological adverse effects, most of which are mild and, although they do not lead to discontinuation, they may impact quality of life. 76 The most frequent treatment-emergent adverse events in pivotal trials of GA were local injection site reactions,2,4 consisting of transient erythema, pain, edema, and pruritus that occur with decreasing frequency over time. 77 A pooled analysis of safety and tolerability among all MS patients exposed to GA in clinical trials before 2017 (10,017 patient-years, median exposure 1.8 years) confirmed that injection site reactions were the most common adverse events 78 ; this was also consistent with the results from long-term extension studies. No late safety issues emerged in the extension studies.4,73,74 Injection site reactions were less frequent with the 40 mg/mL formulation administered three times weekly in the open label GLACIER study. 79 Patients in the US Glatiramer Acetate Trial had the option of switching from daily injections to three times weekly injections when the 40 mg/mL formulation became available, but many chose to continue with daily injections, suggesting that tolerability was acceptable. 73
Lipoatrophy or necrosis
Injection site lipoatrophy or necrosis is a more serious adverse event that was reported in approximately 2% of patients who received GA in placebo-controlled trials, and patients should practice injection site rotation, as described in the prescribing information. 6
Immediate post-injection reaction
Immediate post-injection reaction may manifest as flushing, chest tightness/pain, palpitations, tachycardia, anxiety, dyspnea, throat constriction, and/or urticaria occurring within 1 h of injection. This is a rare occurrence, with a single or a few episodes occurring during long periods of treatment in 16% of treated patients. 6 The exact nature of these reactions is unknown, and they are generally transient and self-limiting, with dyspnea occurring with the highest frequency (12%). Caution is warranted, however, as a recent direct healthcare professional communication from the EMA warns about the possibility of anaphylactic reactions occurring months to years after treatment initiation. While these reactions are uncommon (⩾1/1000 to <1/100), symptoms may overlap with those of post-injection reactions, potentially delaying their identification. 80
Other adverse events occurring with a frequency >10%
Consistent with findings reported in the prescribing information,5,66 a post hoc pooled analysis of safety and tolerability identified rashes (15%), headaches (14%), infections (12%), and episodes of vasodilation (11%). 78 The rate of headache was similar to the reported rate of 15.8% in the general population on any given day. 81 Infections were defined broadly, encompassing common and minor events, and the absence of a comparator arm makes interpretation difficult. 78 Transient episodes of idiopathic chest pain, including episodes not associated with other symptoms, have also been reported. While leukopenia can be an issue with some DMTs, it occurs infrequently with GA and is generally mild if present. 82 This is likely due to its mechanism of action, which involves anti-inflammatory cytokine shifts in CD4+ and CD8+ T cells, restoration of Tregs, and reductions in memory B and T cells.
Liver injury
Cases of liver injury, including hepatitis and liver failure, have been reported in patients treated with GA, occurring days to years after starting treatment. 6 Meunier et al. have cataloged 15 cases of GA-associated liver injury, 6 of which occurred in patients who had switched to GA from interferon (IFN) due to elevated liver enzymes (Table 2). 83 Among these 15 cases, only 4 were negative for anti-nuclear and anti-smooth muscle autoantibodies, and had not previously switched to GA due to elevated liver enzyme abnormalities associated with the previous treatment. 83
Cases of GA-associated liver injury.

Source: Reprinted from Meunier et al. 83 Open Access under Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/). No special permission is required to reuse all figures or part of article published by MDPI.
ANA, anti-nuclear antibodies; ASMA, anti-smooth muscle antibodies; Cho, cholestasis; CTC, corticosteroids; Hep, hepatitis; IFN, interferon; MPDN, methylprednisolone.
Meanwhile, Villani et al. described 11 cases of autoimmune hepatitis in untreated patients with MS, 98 and Tsouris et al. identified similar frequencies of autoantibodies to hepatic antigens in treatment-naïve and treated patients with MS. 99 It is important to consider the type of liver injury involved, and consider that, while case reports can provide hints about a safety signal, they require systematic assessment for confirmation. 100
While it is not possible to estimate the frequency of drug-induced hepatotoxicity based on these cases, there were no reported cases in pivotal trials of GA originator, 101 which may indicate that they are not common. Post-marketing surveillance in clinical practice is useful for monitoring drug safety and detecting safety signals. Spontaneous reporting systems like the FDA Adverse Event Reporting System can detect rare events like drug-induced liver injury. 102 Disproportionality analysis of FDA Adverse Event Reporting System data acquired from 2004 through 2016 (18,591 total reports on GA) did not identify a safety signal for overall liver injury or for severe liver injury with GA, reporting odds ratios (95% CI) 0.70 (0.62–0.79), and 1.01 (0.88–1.16), respectively. 103
In the Viatris safety surveillance database, 55 cases (65 events) suggestive of hepatic injury have been reported. Ages ranged from 23 to 90 years, and 46 patients were female. Thirty-one of 55 cases were non-serious, and 53% consisted of liver enzyme elevation. Reported forms of severe liver injury included serious drug-induced hepatotoxicity, hepatitis, autoimmune hepatitis, liver cancer, and one case of liver failure. The case of liver failure occurred in the context of multi-organ failure secondary to gastrointestinal infection, thrombosis, and pulmonary embolism, excluding a direct causal role of GA in its occurrence and fatal outcome. Of the three reported cases of autoimmune hepatitis, only one was supported by a positive anti-nuclear antibody titer, whilst information was limited in the other two. The two reported cases of liver cancer were not evaluable for causal association to GA based on limited information.
GA prescribing information recommends monitoring for signs of hepatic injury, instructing patients to seek immediate medical attention in case of symptoms of liver injury, and consider discontinuing GA in case of clinically significant liver injury.6,67
Opportunistic infections
GA modulates the immune system rather than suppressing it, promoting anti-inflammatory and regulatory pathways, while avoiding an increase in the risk of infections. DMT-associated risk of opportunistic infections is considered lowest with the injectable therapies IFN beta and GA.78,104–106
GA treatment is associated with a reduction of pro-inflammatory B cell functions 107 ; however, leukopenia and leukocytosis are rare. 108 Progressive multifocal leukoencephalopathy (PML), triggered by reactivation of latent John Cunningham virus, is an important safety concern when treating MS. 109 Among DMTs, natalizumab is associated with the highest risk of PML, with rare cases also reported for ocrelizumab, fingolimod, and dimethyl fumarate. In contrast, GA has not been associated with an increased risk of PML or hepatitis B virus reactivation compared with the general population.110,111 Infection risk is a key consideration when initiating or switching treatments, 112 and ongoing monitoring for infections is an essential practice with some DMTs.
Meanwhile, post-marketing surveillance has not shown a risk for opportunistic infections (including PML),78,104 and there are no requirements for infection screening or monitoring. 5 In the Viatris database, 148 infections excluding the SARS-CoV-2 virus were reported. Of these, 75% were non-serious, and included influenza, herpes zoster, oral herpes, Staphylococcal infections, and fungal infections. Most events were consistent with the immune-modulating mechanism of action of GA, and no fatal outcomes were reported.
Vaccination
Vaccines are not associated with an increased risk of relapses or disability in MS patients, regardless of treatment status, and their benefit greatly outweighs potential risks, as infections are frequent triggers for relapse113–117. Nonetheless, it may be prudent to avoid vaccination in patients who are experiencing an active relapse. 118 Inactivated vaccines can be safely used in MS patients regardless of treatment, whereas live attenuated vaccines should be limited to untreated patients or those receiving immunomodulatory therapy with GA or IFNs. 119
Most DMTs for MS modulate or suppress components of the immune system, potentially influencing the ability to mount an immune response to vaccines. Several DMTs are known to attenuate vaccine responses (e.g., sphingosine-1-phosphate receptor modulators, anti-CD20 monoclonal antibodies, natalizumab), and care should be taken to update vaccines before initiating these treatments, observe the optimal interval from the last DMT dose when administering annual vaccinations, and consider administering additional vaccine doses. 119 It is recommended that vaccine-induced antibody titers should be confirmed 1–2 months after the last dose of the hepatitis B, measles, mumps, tetanus, or varicella vaccines in patients who are already receiving immunosuppressive DMTs. 119 GA does not appear to compromise response rates to influenza, tick-born encephalitis, or SARS-CoV-2 vaccines in patients with MS.120–123
Special populations
Older adults
Older adults were not well represented in pivotal clinical studies of DMTs for MS, which generally presents in young adults124,125; therefore, extra attention may be warranted when prescribing DMTs in older adults, because risk–benefit considerations may change with age. This is important because as survival has improved and the population ages, the peak MS prevalence has currently increased to between 55 and 65 years.126–128 Disease characteristics change as patients with MS age. Older MS patients have lower relapse rates, 129 less inflammatory activity,130,131 and may have less need for highly effective DMTs. 132 Older adults with MS generally have higher rates of comorbidities, which are associated with higher risk of hospitalization, 133 a higher risk for adverse events, 125 and an apparent increase in MS disability progression on the Expanded Disability Status Scale. 134 Large cohort studies have shown that DMT efficacy decreases with age, 135 raising the question of whether de-escalation or discontinuation should be considered in older patients with no clinical or radiological disease activity. Such choices must be made in the context of shared decision-making. In a survey of older patients in the North American Research Committee on Multiple Sclerosis Registry who had been receiving the same DMT for at least 5 years, 88.1% of 377 respondents reported being unlikely to consider DMT discontinuation if their MS was stable 136 ; therefore, de-escalation may be a more attractive option to discontinuation in this situation. Moreover, the results of the DISCOMS (discontinue disease-modifying therapies) and DOT-MS studies do not support the discontinuation of DMTs in older patients.137,138 However, the DISCOMS study had a non-inferiority design, and the difference was very small at 7.5 percentage points. 137 Thus, decisions should be made on an individual basis considering impact on quality of life and patient preference. 139
Patients with comorbidities and polypharmacy
Meta-analysis of 249 studies on comorbidities in MS identified depression, anxiety, hypertension, hypercholesterolemia, and chronic lung disease as the most prevalent comorbidities overall, 140 while the most prevalent autoimmune comorbidities were thyroid disease and psoriasis. 141 Number of comorbidities may also impact the choice of MS DMT favoring GA. 142 Patients with comorbid systemic autoimmune conditions may benefit from GA as a treatment option. Clinical trials investigating the potential benefit in other autoimmune disorders are not available. There is only a case series about nine patients with MS and systemic lupus erythematosus, two of whom were treated with GA and experienced no exacerbation of the lupus-associated symptoms. 143 In another case series, successful treatment of uveitis with GA was reported. 144
Administering DMTs for MS, together with symptomatic drugs and therapy for comorbid conditions can result in polypharmacy,145,146 which has been shown to increase the risk of pharmacodynamic and pharmacokinetic drug–drug interactions in patients with MS. 147 Drug–drug interactions can cause side effects or alter the effectiveness of drugs, increasing the risk of treatment failure. In addition to toxicity, polypharmacy may be associated with a higher risk of falls in patients with MS, particularly in patients receiving anti-cholinergic agents, 148 as well as fatigue and cognitive impairment. 149 A useful strategy for avoiding this includes identifying potential interactions using a database (e.g., Hecker et al. 150 ), followed by elimination of unnecessary drugs and supplements, and considering alternative agents with lower potential for interaction, when possible.
Drug interactions involving GA do not appear to be well studied; however, results from clinical trials do not suggest significant interactions with therapies commonly used in MS patients, including concurrent corticosteroids for up to 1 month.5,6
Use in pregnancy
Pregnancy is associated with reduced relapse rates and may be followed by a post-partum rebound of relapse frequency during the months after birth.151–156 Safety during pregnancy is a critical issue for MS treatment with DMTs, because many of the patients are women of childbearing age. Treatment decisions around pregnancy must consider prior disease activity and whether a woman is receiving DMTs with a high risk of disease rebound after discontinuation. 157
For this reason, several database studies have assessed the safety of GA during pregnancy. Teva’s global pharmacovigilance database (n = 5042 pregnancies with known outcomes) revealed an incidence of congenital anomalies that was compatible with the expected rate based on large population studies.158,159 A systematic review and meta-analysis of pregnancy and fetal outcomes in women receiving GA (or IFN or natalizumab) did not reveal an increase in the risk of spontaneous abortions, preterm birth, or major congenital malformations. 160 Meanwhile, analysis of data from the German Multiple Sclerosis and Pregnancy Registry also showed rates of such outcomes similar to those in the general population. 161 Exposure to GA (20 or 40 mg formulations) throughout all three pregnancy trimesters was not associated with adverse pregnancy or infant outcomes. 162
In the Viatris database, 445 exposed pregnancies were reported. Common adverse outcomes related to pregnancy included 23 instances of (spontaneous) abortion, 7 preterm births, 3 ectopic pregnancies, 3 reports of vanishing twin syndrome, and 2 cases of oligohydramnios. Congenital malformations were reported in seven cases (1.6%); however, based on the nature of post-marketing data and the evidence available from these cases, it is not possible to determine if this incidence differs from that in unexposed pregnancies. Both originator and Viatris GA can be used in pregnancy.6,66
Breastfeeding
Breastfeeding is advantageous for the infant and may be associated with a reduction in postpartum MS relapses 163 ; however, there are concerns about breastfeeding while receiving DMTs. Both originator GA and Viatris GA can be used during breastfeeding.6,66 Data from the German MS and Pregnancy Registry did not reveal evidence of adverse effects of GA exposure during breastfeeding on infant development, hospitalization, or the use of antibiotics.164–166 Maternal use of GA during breastfeeding did not adversely affect offspring safety outcomes assessed during the first 18 months of life. Published pharmacovigilance data on exposed pregnancies confirm the lack of adverse outcomes with originator GA during breastfeeding. 159
The use of Viatris GA during breastfeeding was reported in 60 cases in the Viatris database. Concurrent events reported were either injection site reactions or device or administration-related issues. No adverse events were identified in the infants that could be attributed to exposure to GA via breast milk. Currently, there are no regulatory restrictions on GA use while breastfeeding.6,67
Patients at risk for infections and reactivation of latent infections
Individuals at increased risk of infections include those receiving immunosuppressive medications to prevent rejection of a transplanted organ, patients with HIV, oncology patients who receive chemotherapy or radiation therapy, and older adults experiencing immunosenescence. Infections contribute substantially to morbidity and mortality in MS.108,167,168 Compared with the general population, people with MS have approximately twice the incidence of infections and infection-related hospitalizations, 169 most frequently involving the respiratory 170 and urinary tracts. 171 Immunosuppressive therapies may further increase infection risk. 172 All DMTs for MS, with the exception of IFNs and GA, can impair immune surveillance.104,105
Patients with MS do not appear to be at higher risk of SARS-CoV-2 infection compared to healthy subjects 173 ; however, the rate of COVID-19 development in patients with MS is lower in those receiving GA (0.51%) or IFNs (0.61%), compared to other DMTs (p < 0.001 for both), whereas anti-CD20 therapies are associated with the highest risk (3.45%; p < 0.0001). 174 Pretreatment screening for latent infections is not required with GA. 6
Conclusion
The many effective DMTs now available have improved the personalization of treatment for MS. Extensive clinical experience continues to demonstrate the enduring value of GA in the management of relapsing MS not requiring a highly effective DMT. Ease of management, good tolerability, and long-term safety data make GA an appropriate choice for diverse patient populations, including patients with comorbidities, breastfeeding individuals, older adults, and patients undergoing therapeutic de-escalation. A minimal impact on immune function makes GA a suitable option for MS patients at risk of infections, while compatibility with vaccinations ensures that essential immunizations will be effective. Localized injection site reactions, although common, do not typically lead to treatment discontinuation. More severe injection site events like lipoatrophy or necrosis are rare, and while there are reported cases of liver injury, their frequency remains low. Available evidence on product substitution from branded to FOGAs shows similar efficacy and safety reinforcing its position as a reliable and enduring therapeutic option for MS management.
