Abstract
Objective
To investigate the safety and efficacy of laquinimod in treating relapsing-remitting multiple sclerosis (RRMS).
Methods
An extensive electronic search was conducted across PubMed, Embase, Cochrane Database of Systematic Reviews, and ClinicalTrials.gov to identify suitable studies. Risk of bias was assessed using Cochrane’s Risk of Bias tool. Statistical analysis was performed using RevMan 5.4.1.
Results
The meta-analysis of four randomized controlled trials including 3665 patients found that laquinimod significantly reduced the annualized relapse rate compared with placebo (mean difference = −0.08, 95% confidence interval [CI] = −0.12, −0.04, I2 = 0%). For disability progression confirmed at 3 months, laquinimod provided a significant advantage over placebo (hazard ratio [HR] = 0.75, 95% CI = 0.59, 0.96, I2 = 25%), whereas no benefit was achieved at 6 months (HR = 0.69, 95% CI = 0.45, 1.06, I2 = 66%). Laquinimod was also significantly better than placebo in maintaining a relapse-free status (risk ratio [RR] = 1.14 95% CI = 1.06, 1.22, I2 = 10%). Laquinimod had a comparable safety profile as placebo (RR = 1.06, 95% CI = 0.81, 1.39, I2 = 33%).
Conclusions
These findings support the efficacy of laquinimod in managing RRMS but necessitate careful monitoring during treatment.
Keywords
Introduction
Multiple sclerosis (MS) is a degenerative autoimmune disease that leads to demyelination of the neurons in the brain and spinal cord, causing axonal and myelin damage. Over time, MS can result in irreversible neuron damage, cognitive and physical disability, and reduced quality of life.1,2 Approximately 2.8 million people worldwide have MS, with a prevalence rate of approximately 35.9 per 100,000. MS can be diagnosed at any age, but the typical age at diagnosis is 32. Approximately 30,000 children, including teenagers, have MS. Relapsing-remitting MS (RRMS) constitutes 85% of all cases, whereas progressive MS accounts for 12% of cases. 3 The prevalence of this figure has risen in all global areas since 2013, indicating an expanding awareness and identification of the illness. Women have a 2-fold higher likelihood of MS than men. 4
The current treatment for MS involves the use of disease-modifying therapies such as interferon beta, natalizumab, mitoxantrone, glatiramer acetate, and fingolimod. 5 Studies revealed that the prolonged use of disease-modifying therapies, including injectable treatments, provides lasting benefits by slowing MS progression. These therapies reduce inflammation and prevent further central nervous system damage. 6 Because of the inconsistent results and potential negative effects of current disease-modifying therapies, there is a need for new treatments. Patients with advanced MS in particular lack effective options. Exploring innovative therapies such as laquinimod might address some of these limitations. 7
Laquinimod is an oral immunomodulatory medication specifically developed for RRMS, a condition characterized by unpredictable symptom flare-ups and periods of improvement, significantly affecting patients’ quality of life.8,9 Whereas patients often regain near-normal function after each episode, there is a risk of irreversible disability progression, known as secondary progressive MS, over time. Early intervention might potentially delay this progression. 10 Laquinimod works by regulating immune responses, reducing inflammatory cell infiltration, and providing neuroprotection. Its favorable safety profile, with manageable side effects, makes it suitable for long-term MS treatment.11,12
Empirical data suggest that laquinimod can effectively reduce recurrence rates and delay disease progression. However, individual trial outcomes vary, necessitating further clarification of its overall efficacy and safety. Our meta-analysis, consolidating data from randomized controlled trials, provides a clearer evaluation of laquinimod’s therapeutic efficacy and safety profile.
Materials and methods
Protocol registration
The present systematic review and meta-analysis followed the guidelines outlined in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. 13 The study protocol was registered with PROSPERO (ID CRD42024576774).
Search strategy
An extensive electronic search was conducted across PubMed, Embase, the Cochrane Database of Systematic Reviews, and ClinicalTrials.gov for studies published up to 25 July 2024 without imposing any search restrictions. Detailed information about the search strategy is available in Supplementary File 1.
Eligibility criteria and study selection
All references from the search were imported into EndNote v20, and duplicates were removed. Two reviewers (AT and HK) reviewed titles and abstracts. The studies that did not meet our PICO criteria were excluded. The full texts of the remaining studies were then examined for eligibility. Discrepancies were resolved by a third reviewer (AM). The included studies were limited to randomized controlled trials comparing laquinimod 0.6 mg with placebo in patients with RRMS.
Data extraction and outcomes
Data from the included studies—such as study details, patient numbers, and treatment group information—were independently extracted by two reviewers (SK and HK) using a standardized form in Microsoft Excel (Microsoft, Redmond, WA, USA). Discrepancies were resolved by a third reviewer (AA). Patient-specific data included age, sex, time since first MS symptom, treatment dose, time since MS diagnosis, number of relapses in the previous year, Expanded Disability Status Scale score, MRI findings, and prior disease-modifying treatments. The primary efficacy outcomes were the annualized relapse rate, number of relapse-free patients, and disability progression at 3 and 6 months. Secondary outcomes focused on safety, including serious adverse events, arthralgia, elevated alanine transaminase levels, and death.
Risk of bias assessment
The risk of bias in the included studies was assessed by two independent reviewers (SK and MA) using the Cochrane Risk of Bias tool (RoB 2.0) 14 for randomized controlled trials (RCTs). The evaluation focused on domains including randomization, deviations from interventions, missing outcome data, outcome measurement, and reporting bias. Studies were classified as having a low, moderate, or high risk of bias. Discrepancies were resolved with input from a third reviewer (AA).
Statistical analysis
Statistical analyses were conducted using Review Manager (version 5.4.1; The Cochrane Collaboration, Copenhagen, Denmark). Risk ratios (RRs) were calculated for dichotomous outcomes, whereas mean differences were used for continuous outcomes, both with 95% confidence intervals (CIs). Hazard ratios (HRs) were calculated for time-to-event outcomes. Adjusted means with standard errors were meta-analyzed using a random effects model, considering standardized mean differences and standard errors for variance determination and study weighting. The DerSimonian and Laird procedure, using a random-effects model, was applied, and the results were displayed as forest plots. Heterogeneity was assessed using the I2 statistic following the guidelines from the Cochrane Handbook for Systematic Reviews of Interventions. 15
Results
Search results
The preliminary search yielded 1041 articles. After filtering out 141 duplicates, we evaluated the remaining 900 articles to determine their eligibility. In total, 786 publications were removed after reviewing their titles and abstracts. We obtained the complete texts of the remaining 114 articles and verified them using the eligibility criteria. The final analysis included four RCTs and 3665 patients.16–19 Figure 1 provides an in-depth summary of the screening method.

Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram.
Study characteristics
The years of publication of the included studies spanned from 2008 to 2021. The mean patient age was 18 to 50 years. The treatment dose of laquinimod was 0.6 mg. Table 1 summarizes the characteristics of the included studies. Table 2 summarizes the baseline characteristics of the study participants.
Study characteristics of the included trials.

Laquinimod 1.2 mg was discontinued before the endpoint because of cardiovascular toxicity.
RCT, randomized controlled trial; EDSS, Expanded Disability Status Scale; MSFC, Multiple Sclerosis Functional Composite.
Baseline characteristics of participants in the included trials.

Value is presented as the median.
Value is not reported as the mean or median.
T, treatment; C, control; NR, not reported.
Risk of bias in included studies
The risk of bias in the included studies was assessed using RoB 2.0. Three of the included studies displayed a moderate risk of bias.16–18 The domains of measurement of the outcome and selection of the reported results were responsible for this risk of bias. The remaining study was assessed to have a low risk of bias. 19 The traffic light plot for risk of bias assessment is displayed in Figure 2.

Traffic light plot for the risk of bias assessment.
Meta-analysis of primary outcomes
Annualized relapse rate
Three studies including 3457 patients (1711 on laquinimod, 1746 on placebo) revealed that laquinimod significantly reduced the annualized relapse rate versus placebo with a mean difference of −0.08 (95% CI = −0.12, −0.04; P = 0.0002; I2 = 0%; Figure 3).
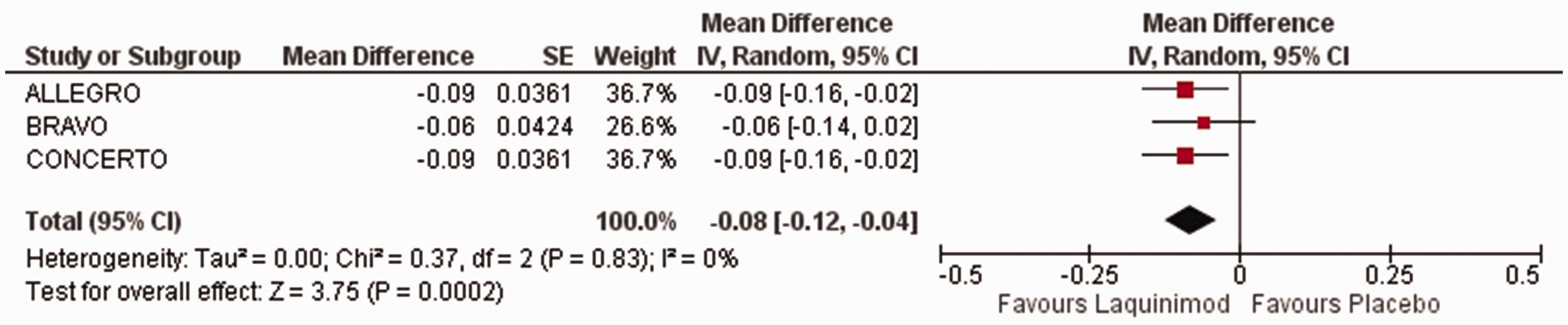
Forest plot of the annualized relapse rate. CI, confidence interval; SE, standard error.
Disability progression at 3 and 6 months
Disability progression confirmed at 3 months was reported in three studies including 3457 patients (1711 on laquinimod, 1746 on placebo). Laquinimod significantly outperformed placebo, achieving an HR of 0.75 (95% CI = 0.59, 0.96; P = 0.0002; I2 = 0%; Figure 4).

Forest plot of disability progression confirmed at 3 months. CI, confidence interval; SE, standard error.
For disability confirmed at 6 months, laquinimod had a comparable effect as placebo (HR = 0.69, 95% CI, 0.45, 1.06; P = 0.09; I2 = 66%; Figure 5).

Forest plot of disability progression confirmed at 6 months. CI, confidence interval; SE, standard error.
Relapse-free patients during the study
In the analysis of relapse-free patients, which included 2198 patients (1090 on laquinimod, 1108 on placebo), laquinimod displayed significant superiority over placebo (RR = 1.14, 95% CI = 1.06, 1.22, P = 0.0004, I2 = 10%; Figure 6).

Forest plot of relapse-free survival. CI, confidence interval; SE, standard error.
Sensitivity analysis
To evaluate the impact of individual studies on heterogeneity, we performed a sensitivity analysis by sequentially excluding each of the four included RCTs and reassessing the heterogeneity for each outcome. Notably, excluding the CONCERTO trial resulted in a substantial reduction in heterogeneity for disease progression at 6 months, with I2 value decreasing from 66% to 0% (Figure 7). For the remaining outcomes, heterogeneity remained consistently low to moderate irrespective of which study was excluded.

Forest plot of disease progression at 6 months after excluding the CONCERTO trial. CI, confidence interval; SE, standard error.
Meta-analysis of safety profile
Serious adverse events
Serious adverse events in four studies involving 3663 patients (1816 on laquinimod, 1847 on placebo) were similar between the groups (RR = 1.06, 95% CI = 0.81, 1.39, P = 0.68; I2 = 33%; Figure 8).

Forest plot of serious adverse events. CI, confidence interval; SE, standard error.
Mortality
Mortality data from four studies involving 3663 patients (1816 on laquinimod, 1847 on placebo) revealed similar mortality between the groups (RR = 0.55, 95% CI = 0.11, 2.78, P = 0.47, I2 = 0%; Figure 9).

Forest plot of mortality. CI, confidence interval; SE, standard error.
Alanine aminotransferase (ALT) assessment
Data from three studies including 2026 patients (994 on laquinimod, 1032 on placebo) indicated that laquinimod was more likely to cause ALT elevation (RR = 1.86, 95% CI = 1.53, 2.27, P < 0.00001, I2 = 31%; Figure 10).

Forest plot of alanine aminotransferase elevation. CI, confidence interval; SE, standard error.
Arthralgia
Arthralgia data from three studies involving 2781 patients (1383 on laquinimod, 1398 on placebo) indicated that the drug did not influence the risk of arthralgia (RR = 1.38, 95% CI = 0.95, –2.03, P = 0.09, I2 = 18%; Figure 11).

Forest plot of arthralgia. CI, confidence interval; SE, standard error.
Discussion
Laquinimod, as an oral immunomodulator, has attracted attention as a possible therapy for RRMS. Unlike traditional immunosuppressive therapies, laquinimod is designed to modulate the immune response without significantly suppressing immune activity, potentially permitting a more balanced approach to managing MS. This characteristic is important because it might reduce the risk of infections and other complications associated with more aggressive immunosuppressive treatments. 20 The significance of our meta-analysis lies in its comprehensive evaluation of the efficacy and safety of laquinimod across four RCTs involving 3665 patients, providing a clearer understanding of its therapeutic potential and safety profile in a clinical setting.
In our meta-analysis, laquinimod induced a notable decline in the annualized relapse rate and a notable improvement in disability progression confirmed at 3 months. Although the results for disability progression at 6 months were not significant, laquinimod achieved a higher rate of relapse-free survival during the study. Regarding safety assessed as a secondary outcome, laquinimod had a similar safety profile as placebo regarding serious adverse events and mortality. However, it was associated with a higher risk of ALT elevation.
The annualized relapse rate, disability progression confirmed at 3 and 6 months, and relapse-free survival during the study were assessed as the primary outcomes. Laquinimod was associated with a significantly lower annualized relapse rate than placebo across 3457 patients. This outcome was consistent with the ALLEGRO trial, in which the mean annualized relapse rate for laquinimod was 0.30 ± 0.02, compared with 0.39 ± 0.03 for placebo (P = 0.002). 16 In a prior analysis, laquinimod 0.6 mg daily was linked to a 21% decrease in the annualized relapse rate using the pooled ALLEGRO and BRAVO datasets. 21
A recent retrospective study from the UK reported an annualized relapse rate of 0.08 for patients with RRMS, which was lower than previously reported rates in RCTs. This suggests that although laquinimod displays efficacy in clinical trials, real-world annualized relapse rates can be influenced by various factors, including patient demographics and treatment adherence. 22
Laquinimod significantly reduced disability progression at 3 months compared with placebo, whereas the disability progression results were similar between the groups at 6 months. These results align with the ALLEGRO trial, in which laquinimod reduced the risk of disability progression at 3 months by approximately 29% (HR = 0.64). 16 However, the CONCERTO trial did not report significant effects for laquinimod 0.6 mg, reporting an HR of 0.94 (P = 0.706) for 3-month confirmed disability progression, indicating variability in results across different studies. 18 At 6 months, the results indicated a trend of reduced disability progression for laquinimod without achieving significance. The I2 value of 66% suggests considerable heterogeneity among the studies included in this analysis. This is notable because the BRAVO study also reported non-significant reductions in disability progression at similar time points, highlighting the inconsistency in outcomes across different trials. 17 This suggests that the effect of laquinimod on 6-month disability progression was more variable across the included studies than the 3-month outcome.
The observed variability in heterogeneity might stem from factors such as sample characteristics, the drug dosage, or statistical differences, but it appears to have been primarily influenced by concerns regarding the risk of bias across three of the four included trials. To explore this further, a sensitivity analysis was performed, revealing that the CONCERTO trial was a significant contributor to heterogeneity. Specifically, its exclusion eliminated the heterogeneity for disease progression at 6 months. These findings indicate that although the primary results of the meta-analysis remain robust, the heterogeneity observed regarding disease progression outcomes at 6 months might have been particularly sensitive to the inclusion of the CONCERTO trial.
In this meta-analysis, laquinimod significantly increased the likelihood of relapse-free survival compared with placebo with low heterogeneity. In the ALLEGRO study, laquinimod achieved a relapse-free rate of 62.9%, compared with 52.2% for placebo (P < 0.001), aligning with our results. 16 Additionally, the long-term open-label extension of the ALLEGRO study recorded sustained low relapse rates and favorable safety profiles, reinforcing the findings of the initial trials. The annualized relapse rate in patients who started treatment early remained low at 0.21 after 36 months.
Other systematic reviews have also corroborated these findings, indicating that laquinimod effectively reduces the risk of relapse and disability progression in patients with RRMS. For instance, a study noted a treatment effect of 26.7% in reducing disability progression relative to placebo in relapsing patients, with even higher effects observed in relapse-free patients. 23
The findings from our meta-analysis regarding the safety profile of laquinimod, particularly concerning serious adverse events, mortality, and arthralgia, revealed important insights into its safety compared with placebo. Common adverse events reported in the laquinimod groups included myalgia, arthralgia, back pain, and a transient, dose-dependent elevation of liver enzymes, which were reversible upon treatment discontinuation. 24 Our analysis indicated that laquinimod was comparable to placebo regarding serious adverse events, consistent with the safety data reported in the pooled results of the ALLEGRO and BRAVO trials, in which serious adverse event rates were similar between the laquinimod and placebo groups (9.4% and 8.9%, respectively). 20 The serious adverse event rates in the laquinimod and placebo groups were 4.0% and 4.2%, respectively, in the BRAVO trial and 4.3% and 4.7%, respectively, in the ALLEGRO trial.16,17 These results support the pooled results of our meta-analysis and solidify the conclusion that laquinimod is well tolerated with has low incidence of major adverse events.
Similar death rates were identified between the laquinimod and placebo groups in the pooled analysis of mortality outcomes across four investigations. This result aligns with the safety profile of laquinimod reported in the ALLEGRO phase 3 trial, which reported three deaths in the placebo group (attributable to pneumonia complications, injury, and suicide) and none in the laquinimod group. 16 Lucchetta et al. conducted a network meta-analysis to assess the safety and effectiveness of laquinimod in treating RRMS. 25 Consistent with our findings, they discovered no discernible variation in mortality between the laquinimod and placebo groups (RR = 0.55, 95% CI = 0.11, 2.78, P = 0.47). The LAQ5062 trial further supported these findings, revealing some deaths in the placebo group but none in the laquinimod group. 19
The results related to arthralgia in patients receiving laquinimod compared with those on placebo provided crucial information about the drug’s safety profile. In our meta-analysis involving 2781 patients, the risk of arthralgia was similar between the laquinimod and placebo groups. This suggests that although laquinimod was associated with a numerical increase in the risk of arthralgia, it did not reach statistical significance, indicating that any difference in arthralgia occurrence might be attributable to chance rather than representing a true effect of the drug. In a comprehensive review of laquinimod therapy for multiple sclerosis, similar trends were observed as noted in a pooled analysis of the ALLERGO and BRAVO trial. 20 Arthralgia occurred in 8.5% of patients receiving laquinimod, versus 7.6% in those receiving placebo, again reflecting a marginal increase without statistical significance. A study by Polman et al. and the LAQ/5063 trial, which evaluated lower doses of laquinimod, also did not identify arthralgia as a significant adverse event.26–28 These findings reinforce the notion that although arthralgia is a reported side effect, its risk does not appear to be significantly increased by laquinimod. The risk ratio of 1.38, although suggestive of increased risk, did not provide strong evidence of a significant difference because the 95% CI crossed 1. This aligns with findings from other trials in which the safety profile of laquinimod revealed similar trends regarding the incidence of arthralgia without clear statistical significance, reinforcing the need for a cautious interpretation of these results. 18
Laquinimod was significantly associated with ALT elevation. The ALLEGRO trial reported ALT elevation in 5.9% of patients in the laquinimod group, versus 2.7% of those in the placebo group, with 3.6% of laquinimod-treated patients experiencing elevations more than three times the upper limit of normal, compared with only 0.4% of placebo-treated patients. 16 Despite these findings, ALT elevation was usually not clinically significant, and it did not typically lead to liver failure or treatment discontinuation. Discontinuation rates attributable to elevated ALT levels were lower than 1%. 18
Comparatively, other MS therapies such as interferon beta and fingolimod also increased liver enzyme levels as common side effects.20,29 Thus, although laquinimod poses a risk of elevated ALT levels, this is not unique finding among RRMS treatments. Furthermore, understanding that elevated ALT levels can occur in a small percentage of patients receiving laquinimod might encourage proactive monitoring strategies rather than leading to undue alarm or premature discontinuation of therapy. This approach aligns with best practices for managing chronic conditions in which long-term treatment adherence is vital for achieving favorable outcomes.
Laquinimod’s introduction into the therapeutic landscape for RRMS has the potential to significantly influence daily clinical practice. Its unique mechanism of action, which modulates the immune response without extensive immunosuppression, allows for a balanced approach to managing MS. The findings from our meta-analysis, which demonstrate a notable decline in the annualized relapse rate and improvements in disability progression at 3 months, provide compelling evidence for laquinimod’s efficacy. This efficacy is further supported by the observation that laquinimod-treated patients had a higher rate of relapse-free survival during the study period, reinforcing its utility in everyday clinical settings in which maintaining patient stability is paramount. Incorporating laquinimod into treatment regimens might also facilitate personalized medicine approaches. Given its favorable safety profile, which was comparable to placebo concerning serious adverse events and mortality, clinicians might feel more confident prescribing the drug to a broader range of patients, including those who might not tolerate traditional therapies. The increased risk of elevated ALT levels, while requiring monitoring, does not appear to pose a significant barrier to its use, especially when compared with the liver enzyme elevations associated with other MS treatments. Moreover, laquinimod could improve patient compliance, as many patients prefer oral medications over injectable options. As clinicians consider laquinimod for their patients, they should also be aware of the variability in treatment responses observed across different studies, particularly regarding disability progression at 6 months.
Future research focusing on laquinimod’s long-term effects and its performance across diverse patient populations will be crucial for translating these findings into clinical significance. Understanding the effects of laquinimod in relation to various demographic factors and comorbidities will help refine treatment strategies and optimize outcomes for individuals with RRMS.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241311437 - Supplemental material for Efficacy and safety of laquinimod versus placebo in relapsing-remitting multiple sclerosis: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-1-imr-10.1177_03000605241311437 for Efficacy and safety of laquinimod versus placebo in relapsing-remitting multiple sclerosis: a systematic review and meta-analysis of randomized controlled trials by Malik Waleed Zeb Khan, Aizaz Ali, Amna Hussain, Safeena Khan, Ammara Tahir, Muhammad Haris Khan, Touba Azeem, Abdul Moeez, Arysha Monis, Aban Masaud Mian, Fazia Khattak, Moosa Ali and Jibran Ikram in Journal of International Medical Research
Footnotes
Authors’ contributions
M.W.Z.K. conceptualized, designed, and supervised the study. A.A., A.H., S.K., A.T., M.H.K., and A.M.M. searched the literature, screened studies, and extracted data. A.A. and A.H. performed data analyses and interpreted the results. S.K. and M.A. performed the quality assessment. A.M., T.A., and A.M.M. authored the introduction and discussion. J.I., F.K. and A.M.M. provided revisions to the manuscript.
Data availability statement
All data used for conducting this meta-analysis is available online.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors did not receive any funding for this research.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
