Abstract
Teriflunomide is an oral, once-daily disease-modifying therapy (DMT) approved in the USA, Australia, and Argentina for the treatment of relapsing forms of multiple sclerosis (RMS). Teriflunomide reversibly limits the expansion of activated T and B cells associated with the inflammatory process purportedly involved in multiple sclerosis pathogenesis, while preserving lymphocytes for routine immune surveillance. In an extensive clinical development program, teriflunomide demonstrated consistent benefits on both clinical and magnetic resonance imaging outcomes. In long-term studies, teriflunomide treatment was associated with low rates of relapse and disability progression for up to 8 years. The safety profile of teriflunomide has been well characterized, with adverse events generally mild to moderate in nature and infrequently leading to permanent treatment discontinuation. The evidence reviewed here indicates that teriflunomide is an effective addition to the current DMTs used to treat RMS.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic, progressive autoimmune disease that can lead to neurological impairment and long-term disability [Loma and Heyman, 2011]. The goals of MS therapy include modifying the course of the disease by reducing the frequency and severity of attacks, treatment of individual attacks, and the treatment of specific MS-related symptoms [EMA, 2006]. Insight into the pathophysiology of MS has led to the development of new disease-modifying therapies (DMTs) in the hope of obtaining more effective agents that are better tolerated and safer when given long term. Teriflunomide (Aubagio®, Genzyme Corporation, Cambridge, MA, USA) was one of the first oral agents to be developed for the treatment of relapsing MS (RMS), starting with clinical trials over a decade ago. It offers a well-tolerated, efficacious, oral alternative to current DMTs.
Teriflunomide, a once-daily oral immunomodulatory DMT, has been recently approved for the treatment of patients with RMS [Genzyme Corporation, 2012]. This review provides an overview of the mechanism of action, pharmacokinetics, efficacy, safety, and tolerability of teriflunomide, as demonstrated in the extensive clinical development program.
Mechanism of action
Teriflunomide is the principal active metabolite of leflunomide (Arava®, sanofi-aventis, Bridgewater, NJ, USA), used for the treatment of active rheumatoid arthritis in adults since 1998. The chemical structures of both compounds are shown in Figure 1. Although teriflunomide and leflunomide are closely related, they are each different chemical entities with different indications.

Chemical structures of leflunomide and teriflunomide.
Teriflunomide selectively and reversibly inhibits dihydroorotate dehydrogenase (DHODH), a mitochondrial enzyme necessary for
Inhibition of tyrosine kinases, NF-κB activation, and COX-2 activity have been demonstrated
Preclinical studies
In the Dark Agouti (DA) rat model of experimental autoimmune encephalomyelitis (EAE) teriflunomide was shown to reduce the functional deficits associated with inflammation, demyelination, and axonal loss [Merrill et al. 2009]. Prophylactic administration of teriflunomide (10 mg/kg orally) prevented disease development, while therapeutic dosing minimized signs of disease. Teriflunomide treatment reduced the number of T and B cells in the spinal cord and brain in DA rats with EAE [Petty et al. 2010; Ringheim et al. 2011]; these outcomes are consistent with inhibition of clonal expansion and migration of inflammatory cells as proposed above.
Pharmacokinetics
The clinical pharmacokinetic profile of teriflunomide has been well characterized. In an analysis of healthy volunteers and patients with MS, the median elimination half-life (
The biotransformation pathways for teriflunomide rely on hydrolysis, oxidation, N-acetylation, and sulfate conjugation. Compared with leflunomide, teriflunomide is only moderately metabolized with minimal involvement of cytochrome P450 [Genzyme Corporation, 2012], which lowers the potential for drug–drug interactions [Ogu and Maxa, 2000].
Teriflunomide is eliminated through direct biliary excretion of unchanged drug and renal excretion of metabolites. After 21 days of oral administration in healthy volunteers, 37.5% and 22.6% of the teriflunomide dose was excreted
Teriflunomide is excreted by the liver into the small intestine, where it is re-absorbed by the intestinal mucosa and returned to the liver
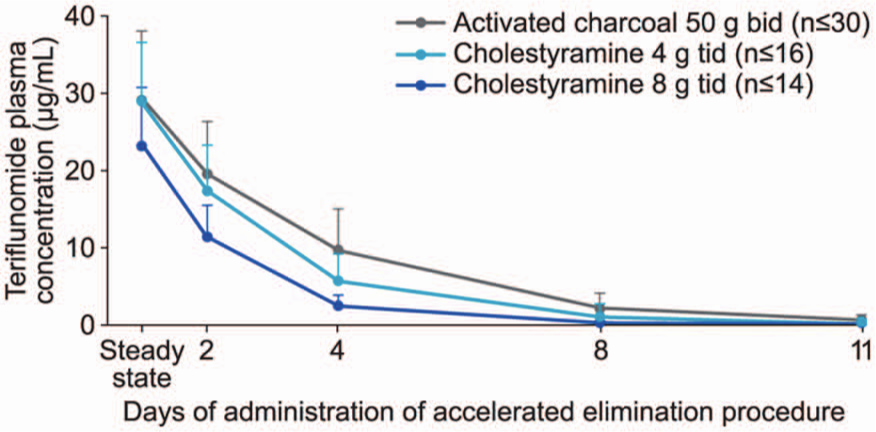
Teriflunomide plasma concentrations following an accelerated elimination procedure.
Efficacy findings from the clinical development program
The clinical development program for teriflunomide is summarized in Table 1.
The teriflunomide clinical program

GA, glatiramer acetate; IFNβ, interferon beta.
Phase II ‘proof of concept’
This 36-week, double-blind trial [ClinicalTrials.gov identifier: NCT01487096] was the first study to evaluate the efficacy and safety of teriflunomide in subjects with RMS. Both teriflunomide doses resulted in significant reductions in the mean number of combined unique active lesions per magnetic resonance imaging (MRI) scan, the primary endpoint (mean number of lesions: 0.2 and 0.3,
TEMSO
The Teriflunomide Multiple Sclerosis Oral trial [TEMSO; ClinicalTrials.gov identifier: NCT00134563] was the first teriflunomide phase III study, and was pivotal in the US approval of teriflunomide [Genzyme Corporation, 2012]. The full methodology has been described previously [O’Connor et al. 2011b]. Briefly, patients aged 18–55 years who met the 2001 McDonald criteria for MS [McDonald et al. 2001], and had a relapsing clinical course (two or more relapses in the previous 2 years or one or more relapses in the last year) with or without progression, were randomized to once-daily placebo, or teriflunomide 7 mg or 14 mg. The study population was well balanced across treatment groups; the mean age was 37.9 years, the majority of subjects were female (72.2%), and > 25% had been treated previously with MS medication. Roughly 35–38% of patients had >1 Gd-enhanced lesions at baseline. At the end of the 108-week treatment period, adjusted ARRs (95% CI) for teriflunomide 7 mg and 14 mg were 0.37 (0.32, 0.43) and 0.37 (0.31, 0.44), respectively, compared with 0.54 (0.47, 0.62) for placebo, representing relative risk reductions
Teriflunomide treatment improved several MRI outcomes in the TEMSO trial. The change from baseline in total lesion volume (the key MRI endpoint) was significantly lower in both teriflunomide groups compared with placebo (7 mg,
Magnetic resonance imaging outcomes from the TEMSO trial.

Data are based on a mixed-effects model and repeated-measures analysis, with the use of a cube-root transformation of the volume data.
This measure is the portion of the total lesion volume that appears hyperintense on T2-weighted images (dual echo spin density and fluid-attenuation inversion recovery images), but does not appear hypointense on T1-weighted images obtained after the administration of gadolinium.
Unique active lesions were defined as the number of gadolinium-enhanced lesions on T1-weighted images, or new or enlarged lesions on T2-weighted images, without double counting. Values were calculated with the use of a Poisson regression model adjusted for treatment, Expanded Disability Status Scale score, and number of lesions at baseline and geographic region, with the log of the number of magnetic resonance imaging scans serving as an offset variable.
Data were missing for 17 patients in the placebo group, 15 patients in the lower dose teriflunomide group, and 18 patients in the higher dose teriflunomide group.
Brain parenchymal fraction was calculated as the inverse of the normalized cerebrospinal fluid volume and assessed with the use of a mixed-effects model with repeated-measures analysis.
Plus–minus values are least-square means ± standard error.
CI, confidence interval.
(Adapted from O’Connor et al. [2011b] Copyright © [2011] Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.)
An extension of TEMSO showed that the adjusted ARR remained low 5 years after initial randomization [O’Connor et al. 2011c], reflecting the outcomes observed in subjects treated for up to 8 years in a long-term extension of the phase II study [Confavreux et al. 2012].
The overall effects of teriflunomide treatment on clinical outcomes in the entire TEMSO cohort were evaluated in specific patient populations in a prospectively defined subgroup analysis. Subjects were stratified according to baseline demographics, disease characteristics, MRI parameters, and prior use of DMTs. Improvements in ARR and disability progression with teriflunomide 7 mg or 14 mg were consistent across all subgroups [Miller et al. 2012c].
An objective in the treatment of immune-mediated inflammatory diseases is sustained disease-free activity (the absence of clinical relapses, no sustained disability progression, and no MRI activity) [Havrdova et al. 2009; Giovannoni et al. 2011].
Separate findings from the TEMSO study population suggest the benefits of teriflunomide on relapses extend beyond simply a reduction in relapse frequency (i.e. ARR). Severe relapses, such as those characterized by neurological sequelae, or those requiring steroid treatment or hospitalization, have a significant impact on patients and are associated with an increased consumption of healthcare resources, and thus healthcare costs [Lad et al. 2010; Naci et al. 2010; Jennum et al. 2012]. In
TOWER
The results of a second phase III study, Teriflunomide Oral in People with Relapsing–Remitting Multiple Sclerosis [TOWER; ClinicalTrials.gov identifier: NCT00751881], supported the clinical outcomes observed in TEMSO [Miller et al. 2013]. The teriflunomide 14 mg dose significantly reduced ARR (36.3%;
Key clinical efficacy outcomes from the TEMSO and TOWER studies.
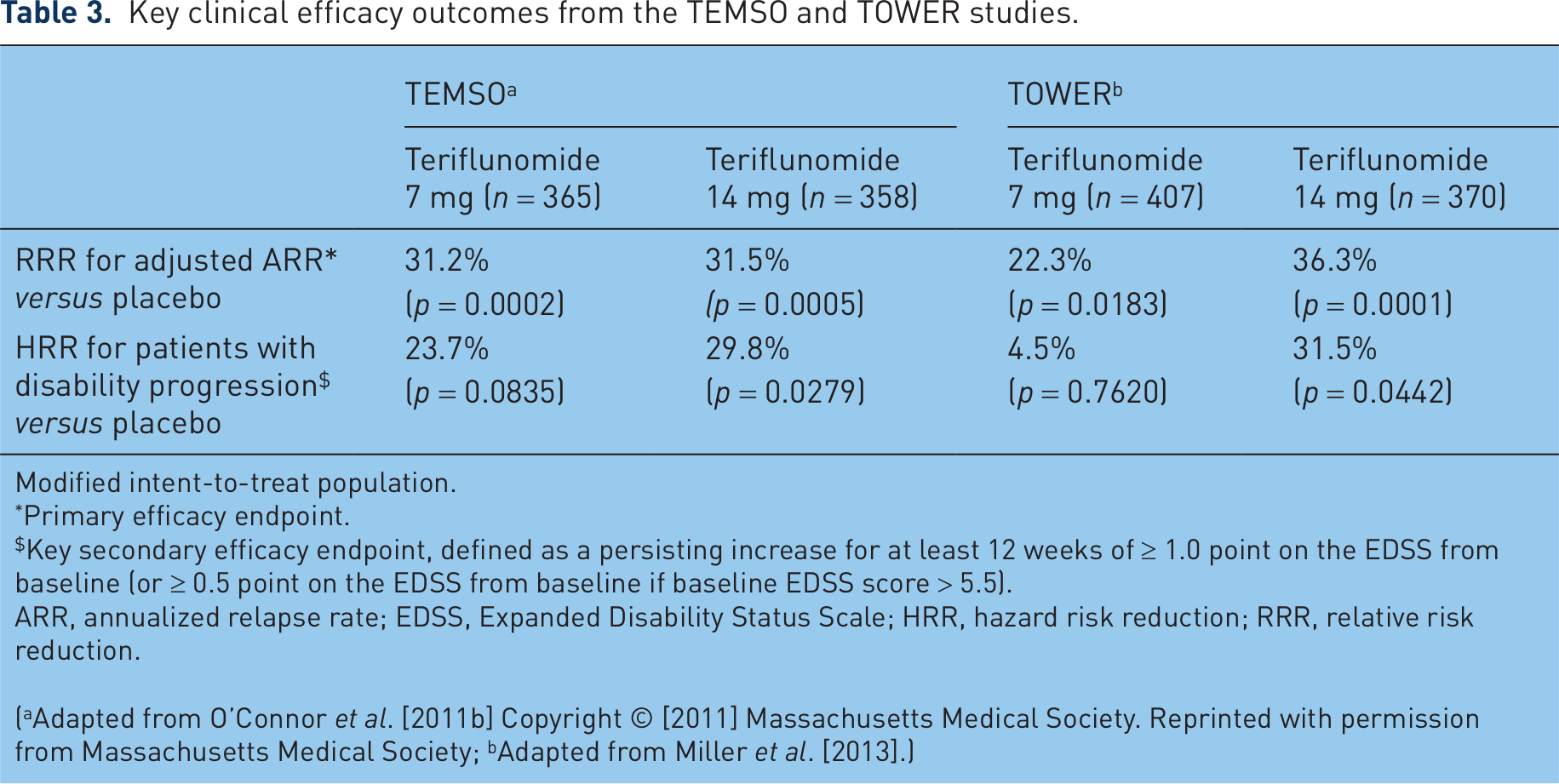
Modified intent-to-treat population.
Primary efficacy endpoint.
Key secondary efficacy endpoint, defined as a persisting increase for at least 12 weeks of ≥ 1.0 point on the EDSS from baseline (or ≥ 0.5 point on the EDSS from baseline if baseline EDSS score > 5.5).
ARR, annualized relapse rate; EDSS, Expanded Disability Status Scale; HRR, hazard risk reduction; RRR, relative risk reduction.
(aAdapted from O’Connor et al. [2011b] Copyright © [2011] Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society; bAdapted from Miller et al. [2013].)
TENERE
TENERE [ClinicalTrials.gov identifier: NCT00883337] was the first head-to-head clinical trial comparing teriflunomide treatment with interferon (IFN) β-1a 44 µg subcutaneously (Rebif®, EMD Serono, Inc., Rockland, MA, USA) in subjects with RMS [Vermersch et al. 2012]. The primary endpoint, time to failure, was defined as the first occurrence of confirmed relapse or permanent treatment discontinuation for any reason, whichever came first. No statistical superiority in time to failure was observed when comparing either teriflunomide-treatment group with the IFNβ-1a group (7 mg
Adjunctive therapy
Some patients continue to experience relapses and clinical deterioration while on treatment. An alternative therapeutic approach is to combine two DMTs with proven tolerability and distinct modes of action. Teriflunomide administered as an adjunctive therapy to IFNβ [Freedman et al. 2012e], or glatiramer acetate (GA) [ClinicalTrials.gov identifier: NCT00489489] [Freedman et al. 2011], was well tolerated, with safety profiles consistent with teriflunomide monotherapy. Teriflunomide added to ongoing IFNβ therapy significantly reduced the number of Gd-enhanced lesions compared with IFNβ alone (82.8%,
TOPIC
The possibility that early intervention with teriflunomide in patients presenting with a first clinical episode suggestive of a demyelinating event might delay or prevent conversion to clinically definite MS (CDMS) is being investigated in a placebo-controlled phase III study (TOPIC) [ClinicalTrials.gov identifier: NCT00622700]. Conversion to CDMS was defined by the occurrence of relapse (primary outcome measure) and dissemination of lesions on MRI over time (secondary outcome measure).
TERIVA
As slowly dividing or resting cells rely more on the pyrimidine salvage pathway to meet their pyrimidine demand, which is largely unaffected by teriflunomide, it is postulated that memory lymphocytes remain available for immune surveillance. To investigate if teriflunomide might influence patients’ abilities to mount recall immune responses, antibody responses to the seasonal influenza vaccine were assessed in 82 subjects with RMS treated with teriflunomide 7 mg or 14 mg for a median duration of about 6 years (TERIVA) [ClinicalTrials.gov identifier: NCT01403376] [Bar-Or et al. 2012]. Over 77% of subjects had an effective vaccine response to all influenza strains, providing supporting evidence that teriflunomide does not significantly affect memory response to recall antigens, in this case, influenza vaccine strains.
Safety and tolerability findings from the clinical development program
Teriflunomide has a well-characterized safety profile and, to our knowledge, has the longest exposure data for an oral DMT. The incidence of treatment-emergent adverse events (TEAEs), serious TEAEs, and TEAEs leading to discontinuation of study medication was similar in the placebo and teriflunomide-treatment groups in TEMSO [O’Connor et al. 2011b]. TEAEs occurring with a greater frequency in the teriflunomide groups with an apparent dose effect were diarrhea, nausea, alopecia, and alanine aminotransferase (ALT) increase. Most cases of diarrhea and nausea were mild to moderate in nature, occurred during the first 3 months of study therapy, and resolved without corrective treatment [Freedman et al. 2012c]. No new or unexpected AEs emerged with long-term teriflunomide treatment in extension studies, and in general, teriflunomide was well tolerated during treatment of up to 9 years’ duration [Comi et al. 2011; Confavreux et al. 2011, 2012]. The relative risk of the most frequently occurring TEAEs in the teriflunomide groups from the TEMSO trial is shown in Figure 3.

Treatment-emergent adverse events in the TEMSO trial: relative risk
Seven deaths have been reported in subjects treated with teriflunomide in the clinical program to date. Four deaths occurred in long-term extension studies (phase II extension,
Adverse events of special interest
An overview of TEAEs of special interest (events reported by patients or noted by the investigator) from the TEMSO trial are shown in Table 4.
Treatment-emergent adverse events of special interest from the TEMSO trial.

Safety population. Events identified and retrieved for the analyses using MedDRA.
Malignant neoplasms were reported in three placebo patients (breast cancer, thyroid cancer, and cervical carcinoma), and one patient in the teriflunomide 14 mg group (cervical carcinoma with recovery following surgery).
MedDRA-preferred term for hair thinning/decreased hair density: alopecia. The term ‘alopecia’ is used to describe any type of hair loss. Most cases of alopecia were reported as hair thinning, decreased hair density, or hair loss.
MedDRA,
Hepatic effects
Teriflunomide is contraindicated in patients with severe hepatic impairment [Genzyme Corporation, 2012], on the basis of postmarketing reports of severe liver injury, including fatal liver failure in patients with rheumatoid arthritis treated with leflunomide [sanofi-aventis, 2012]. If patients taking teriflunomide have ALT levels more than three times the upper limit of normal (ULN) on repeated testing then treatment discontinuation is recommended, and values generally normalize rapidly. Treatment discontinuation and the accelerated elimination procedure should be performed if teriflunomide is suspected as the cause of liver injury.
While the frequency of hepatic TEAEs was higher in teriflunomide-treated subjects than in those receiving placebo, incidence of serious hepatic events and the proportion of subjects discontinuing treatment because of hepatic events were low and comparable across treatment groups (Table 4). Hepatic disorders were mainly asymptomatic ALT increases that occurred within the first 6 months of treatment and often recovered even while on treatment. Figure 4 shows the percentage of subjects with serious hepatic events and ALT elevations in the TEMSO trial [O’Connor et al. 2011b]. Mild increases (ALT > 1 × ULN) were higher in the teriflunomide groups, but the frequency of ALT > 3 × ULN was low and evenly distributed across groups. Three subjects (one in each treatment group) had laboratory values in the Hy’s Law range (increased ALT ≥ 3 × ULN and total bilirubin ≥ 2 × ULN); all cases were considered as nondrug-induced liver injuries with alternative explanations provided (hepatitis C, gall bladder problems, and cytomegalovirus infection) [O’Connor et al. 2011b].
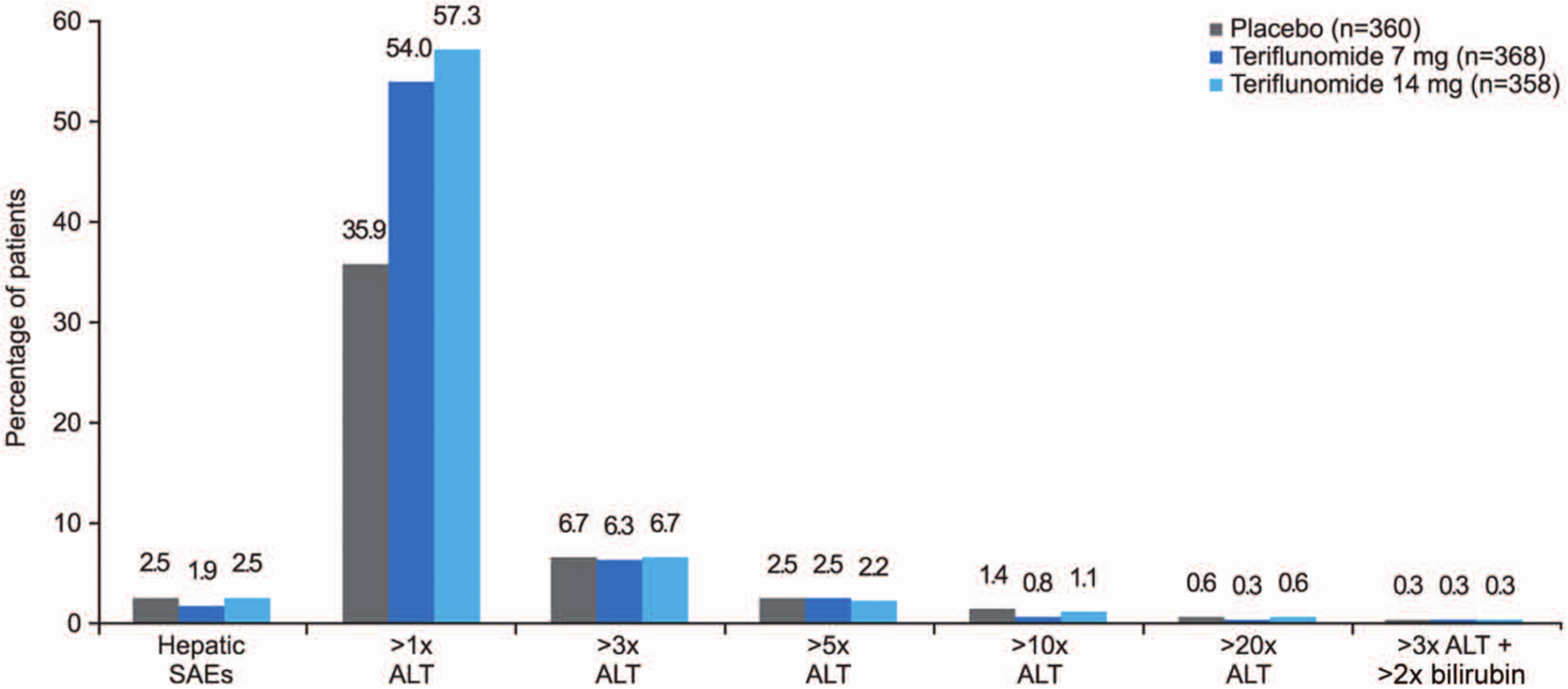
Serious hepatic adverse events and elevated alanine aminotransferase levels in the TEMSO trial.
Infections/bone marrow effects
In the TEMSO trial, mean reductions in neutrophil and lymphocyte counts were small in magnitude (< 15%), and remained within the normal ranges. Importantly, there was no correlation between reduced neutrophil counts and infections [Comi et al. 2012]. A low incidence of serious infections was reported in all treatment groups (placebo, 2.2%; 7 mg, 1.6%; 14 mg, 2.5%) (Table 4), and there were no cases of serious opportunistic infection, supporting the position that teriflunomide is not an immunosuppressive agent [O’Connor et al. 2011b].
In long-term extension studies, malignancies occurred within the expected range observed in the general population, with no pattern of malignancy typical of those observed in immunocompromised patients, and no lymphoproliferative disorders reported [Confavreux et al. 2011; Comi et al. 2012].
Blood pressure effects
The proportion of patients with TEAEs related to blood pressure increase was higher in the teriflunomide treatment groups (placebo, 3.1%; teriflunomide 7 mg, 5.4%; teriflunomide 14 mg, 5.0%, respectively) (Table 4), although no event was serious or led to study treatment discontinuation [O’Connor et al. 2011b].
Peripheral neuropathy
Suspected peripheral neuropathies were confirmed by electrophysiological nerve conduction studies in four patients (1.2%) in the teriflunomide 7 mg group and six patients (1.9%) in the teriflunomide 14 mg group. All reports related to peripheral neuropathy were not serious, and most were of mild and moderate intensity. Of the 10 confirmed cases, 5 were mononeuropathies related to carpal tunnel syndrome or nerve compression, 3 cases were unrelated to study drug and resolved on treatment, and 2 were ongoing at end of study [O’Connor et al. 2011b].
Hair loss/hair thinning
Pooled data from the phase II and TEMSO studies showed that the incidence of hair loss/hair thinning was higher in the teriflunomide treatment groups (7 mg, 11.4%; 14 mg, 15.2%) compared with those receiving placebo (4.3%) [Freedman et al. 2012b]. However, hair thinning in most cases was minimal in nature (76%), and resolved without sequelae (85%), often while the subjects remained on therapy. The risk of hair loss was highest during the first 6 months of treatment. Teriflunomide-related hair thinning is different in nature and severity from chemotherapy/radiotherapy-induced hair loss, which is usually acute and severe, and leads to loss of the majority of the scalp hair [Freedman et al. 2012b].
Pregnancy
Teriflunomide is contraindicated in women of childbearing potential not using reliable contraception. To minimize any potential risk, men not wishing to father a child are also advised to use contraception [Genzyme Corporation, 2012]. Despite the requirement to avoid pregnancy and to use effective contraception in the teriflunomide clinical program, pregnancies did occur. When a pregnancy was identified, the subject was advised to discontinue treatment immediately and to undergo an accelerated elimination procedure. To date, no structural or functional deficits have been reported in the 12 newborns with teriflunomide exposure [Jung Henson et al. 2013].
Safety experience in other trials
The nature and occurrence of TEAEs in the TENERE study were similar to those observed in TEMSO, with fewer discontinuations due to AEs in the teriflunomide groups than in the IFNβ treatment group [Vermersch et al. 2012]. Teriflunomide administered in addition to IFNβ or GA in the phase II combination studies was generally well tolerated, with few subjects reporting serious TEAEs (IFNβ adjunct study: teriflunomide 7 mg, 4 [10.8%]; 14 mg, 1 [2.6%]; placebo, 2 [4.9%]/GA adjunct study: teriflunomide 7 mg, 5 [11.9%]; 14 mg, 2 [4.9%]; placebo, 6 [15.0%]), or TEAEs leading to treatment discontinuation (IFNβ adjunct: teriflunomide 7 mg, 3 [8.1%]; 14 mg, 3 [7.9%]; placebo, 2 [4.9%]/GA adjunct study: teriflunomide 7 mg, 3 [7.1%]; 14 mg, 5 [12.2%]; placebo, 2 [5.0%]) [Freedman et al. 2011, 2012e].
Discussion
In clinical trials, teriflunomide reduced the frequency of clinical exacerbations and delayed the accumulation of physical disability in subjects with RMS [O’Connor et al. 2011b; Miller et al. 2013]. Beneficial effects were observed for both doses of teriflunomide on a range of MRI outcomes [O’Connor et al. 2011b]. Teriflunomide also reduced the occurrence of relapses requiring hospitalization and treatment with corticosteroids in the TEMSO trial [O’Connor et al. 2011a]. Additionally, the benefits of teriflunomide treatment were consistent across predefined subgroups stratified according to baseline demographic features or disease characteristics [Miller et al. 2012c].
Although cross-trial comparisons have obvious limitations, the relative reduction in ARR in subjects treated with teriflunomide appears to be comparable to that observed in patients with MS treated with currently available first-line injectable DMTs [IFNB Multiple Sclerosis Study Group 1993; Paty and Li, 1993; Jacobs et al. 1996; Johnson, 1996; PRISMS, 1998]. Furthermore, no significant difference in ARR was observed between teriflunomide 14 mg and IFNβ-1a in the head-to-head TENERE study.
In a phase II study, teriflunomide as adjunctive treatment to ongoing IFNβ was more effective than IFNβ alone in reducing MRI lesions, raising the possibility of teriflunomide combination therapy as an option in the treatment of patients who have a suboptimal response to current first-line monotherapy [Freedman et al. 2012e]. With greater availability of generic IFNβ, combination therapy is a realistic financial possibility.
Long-term studies have not identified new or unexpected TEAEs associated with teriflunomide treatment [Confavreux et al. 2011, 2012; Comi et al. 2011], suggesting that teriflunomide has a manageable safety profile that compares favorably to those of other oral DMTs [Giovannoni et al. 2010; Kappos et al. 2010]. Data on more than 2.3 million patient-years of exposure to leflunomide in patients with rheumatoid arthritis aids the understanding of the probable long-term safety profile of teriflunomide. In addition, the accelerated elimination procedure is a useful tool in cases of pregnancy or emerging toxicity.
These extensive studies show that teriflunomide is an effective oral monotherapy for relapsing disease. In addition, the already significant long-term safety and tolerability data concerning teriflunomide are complemented by ongoing studies that continue to collect safety information, including a pregnancy registry. All of these support teriflunomide as a first-line agent for the treatment of RMS.
Footnotes
Acknowledgements
Editorial assistance provided by Catherine Simonson, Fishawack Communications Ltd, was funded by Genzyme Corporation, a Sanofi company.
Funding
All teriflunomide studies discussed in this review were funded by Genzyme Corporation, a Sanofi company.
Conflict of interest statement
MSF has received consultancy fees from Bayer, Biogen Idec, Teva, Merck Serono, Novartis, Sanofi and Celgene; payment for lectures from Bayer, Merck Serono, Novartis, and sanofi-aventis; and payment for development of educational presentations from Novartis, Lansdowne DIME, and Medscape. His institution has received research grants from Bayer and Genzyme Corporation.
