Abstract
Background:
Myasthenia gravis (MG) is a chronic, fluctuating disease whose unique natural history complicates designing therapeutic trials. The evidence on which many MG management recommendations are based is limited.
Objective:
To determine the degree of knowledge, agreement, and application of internationally recognized recommendations for managing MG in actual practice.
Design:
A cross-sectional survey was conducted among Spanish neurologists specializing in MG.
Methods:
The survey assessed knowledge, agreement, and application of recommendations, along with the degree of agreement with international consensus definitions from the Myasthenia Gravis Foundation of America.
Results:
Fifty-three neurologists (mean age 45.4 years, 53% men) attended an average of 3.6 patients with MG daily. In 40 out of 61 recommendations, at least 90% of the neurologists were aware of the recommendation. The least known recommendations dealt with juvenile MG and MG in pregnancy. There was no majority agreement in two recommendations (for MG in pregnancy and the use of intravenous immunoglobulin in mild or ocular MG). The implementation of the recommendations was high except for the ones related to plasma exchange, thymectomy, methotrexate, or eculizumab.
Conclusion:
The recommendations are well-known, agreed upon, and applied. However, the evidence, consensus, and knowledge dissemination need reinforcement regarding aspects such as the management of juvenile MG, MG in pregnancy, or the use of certain treatments.
Plain language summary
Myasthenia gravis (MG) is a long-term disease that comes and goes, making it hard to design treatment studies. Many treatment guidelines for MG are based on limited scientific evidence. This study asked neurologists in Spain how well they know, agree with, and use international MG treatment recommendations in their daily work. Fifty-three neurologists who regularly treat MG patients completed a survey. Most were familiar with the guidelines—over 90% knew about 40 of the 61 recommendations. However, some topics, like MG in children and during pregnancy, were less well-known. There was also some disagreement about when to use certain treatments, like intravenous immunoglobulin in mild cases. Most recommendations were widely followed, but a few, such as those involving plasma exchange, thymectomy, or certain drugs like methotrexate and eculizumab, were used less often. In summary, most neurologists know and follow the MG treatment guidelines, but more research and clearer guidance are needed for specific situations like treating children, pregnant women, and using certain therapies.
Keywords
Introduction
Myasthenia gravis (MG) is a chronic, fluctuating, antibody-mediated autoimmune disease directed against postsynaptic antigens of the neuromuscular junction. 1 In about 85% of cases, the auto-antibodies target the nicotinic acetylcholine receptor, disrupting neuromuscular transmission. 2 Other functionally important immunological targets are muscle-specific tyrosine kinase (MuSK) and low-density lipoprotein receptor-associated protein 4 (LRP4). 2 MG manifests with a highly variable clinical spectrum ranging from mild eyelid ptosis to profound weakness of ocular, bulbar, limb, or respiratory muscles.1,3 It is a relatively rare disease, with an estimated annual incidence of 1.7–21.3 cases per million population and an estimated worldwide prevalence of 15–179 cases per million population.4,5 In Spain, the prevalence is consistently above 100 cases per million inhabitants, with some regions such as Osona (Barcelona) and Ourense (Galicia) reporting even higher rates. 6 The prevalence of MG has been increasing since the mid-20th century, probably due to better recognition of the disease, population aging, and longer life expectancy of affected patients.4,7
Traditionally, four main therapeutic approaches have been used in the management of MG: (1) symptomatic treatment by acetylcholinesterase inhibitors (ChEIs) to increase the amount of acetylcholine (ACh) in the neuromuscular junction; (2) glucocorticoids and immunosuppressants to treat the underlying immune dysregulation; (3) immunomodulatory treatments of rapid action but short duration such as plasma exchange (PLEX) or intravenous immunoglobulin (IVIg); and (4) surgical treatment (thymectomy).1,8 However, treatment decisions are highly individualized and not strictly sequential. In recent years, newer targeted therapies with more specific mechanisms of action and improved safety and efficacy profiles have been developed and are now recognized in current guidelines as additional options, particularly for patients who are refractory to conventional immunosuppression or experience significant adverse effects (AEs).1,8
Given the unique natural history of the disorder, designing therapeutic trials for MG has been rather complex. The evidence in favor of several of the conventional agents is weak,9,10 which generates a wide variation in clinical practice in terms of drug choice, making it difficult to systematize therapeutic protocols. The main guidelines for the management of MG are based on an international consensus promoted by the Myasthenia Gravis Foundation of America, published in 2016 and updated in 2020 in which recommendations with different levels of evidence are offered and definitions of concepts such as remission, ocular MG, or refractory MG are agreed upon.9,10 The 2020 recommendations included updates regarding issues such as the use of methotrexate, rituximab, or eculizumab in MG or the indications for thymectomy. 10
This study explored the degree of knowledge of international recommendations for the management of MG among neurologists specializing in the management of this disease and the agreement level and implementation of these recommendations. The aim was to make a diagnosis of the situation regarding the clinical management of MG under conditions of routine clinical practice in Spain from the perspective of professionals with expertise in the disorder and responsible for the treatment of patients. This could help to identify shortcomings and opportunities for improvement in the management of MG.
Methods
A descriptive, cross-sectional, observational study was carried out by means of a structured, self-completed survey by neurologists with expertise in MG in Spain. The study had a naturalistic orientation that neither modifies in any way the usual clinical practice of the participating services or professionals, nor the care provided to patients.
Participants and scope of study
We requested the voluntary collaboration of neurology specialists with a particular and dedicated interest in MG (target population of neurologists: 277 neurologists belonging to the Neuromuscular Diseases Study Group (GEEN)) of the Spanish Society of Neurology. They were practicing in Spanish public or private hospitals. The following inclusion criteria were used: (1) practicing neurologists of either sex and any age; (2) linked to public or private health centers as healthcare personnel; (3) routinely involved in the care of patients with MG as part of their regular clinical practice; and (4) agreed to collaborate, and their personal information and opinions could be used anonymously and aggregated for the exclusive research purposes stated in the study protocol. Data collection was performed between February and June 2023.
Instrumentation and variables
The survey was presented in a closed self-completion electronic form that was made available to participating investigators through a web page designed specifically for this study. The questionnaire collected the personal and professional profile of the participants and asked about MG-related definitions and management recommendations drawn from the 2016, and updated in 2020 international consensus9,10 together with some recommendations from the guidelines developed by the Euro Myasthenia Network. 11 The selection of guidelines was made by consensus among the authors, based on their relevance and recognized influence in European clinical practice. In addition to the definitions included in the international consensus, 9 the scientific committee created an ad-hoc definition of controlled MG.
The degree of agreement with each of the definitions was evaluated using a five-point ordinal scale (1: total disagreement, 2: disagreement, 3: neither agreement nor disagreement, 4: agreement, and 5: total agreement). Additionally, the participants were asked to give their opinion on the definition of controlled MG, considering the presence and severity of AEs.
The questions on the recommendations were divided into eleven blocks: diagnosis, symptomatic and immunosuppressive (IS) treatment, use of IVIg and PLEX, thymectomy, juvenile MG, MG with MuSK antibodies (MuSK-MG), MG in pregnancy, early immunosuppression in ocular MG, other treatments, immune checkpoint inhibitors (ICIs), and follow-up. For each recommendation, we assessed the degree of knowledge of the recommendation by using a dichotomous yes/no variable, the degree of agreement by using a five-point scale (1: strongly disagree to 5: strongly agree), and the actual degree of implementation of the recommendation by using a four-point scale (1: never, 2: occasionally, 3: usually, and 4: always). A pilot test was conducted with 15 professionals to assess the readability, design, and completion time of the questionnaire.
Sample size
It was estimated that 52 neurologists’ participation was necessary to ensure high precision in the main study estimates (maximum error of ±8%) for a confidence level of 95%, assuming a 90% proportion in the most supported response category in the central binomial distribution variables of the study. A convenience sample was used, selecting the first 52 neurologists who met the inclusion criteria and consented to participate.
Analysis of results
The relative frequency distribution (%) of each option is shown for qualitative variables. The mean with 95% confidence intervals (95% CI) and the median are shown for quantitative variables. Possible associations between the participants’ personal or professional characteristics (age, care setting, professional experience, origin, etc.) and their responses to the questionnaire were analyzed using the appropriate statistical tests for each case (chi-square, Student’s t-test, analysis of variance, or their nonparametric equivalents).
Ethical aspects
This study was conducted according to the ethical requirements of the Declaration of Helsinki, Fortaleza revision (Brazil, October 2013), for research involving human subjects. All data included in the survey were processed anonymously, following the current legislation on data protection. Ethical committee approval was not required, as the study did not involve any intervention on patients, and the collection of professional opinions was voluntary and anonymous.
Results
Participants profiles
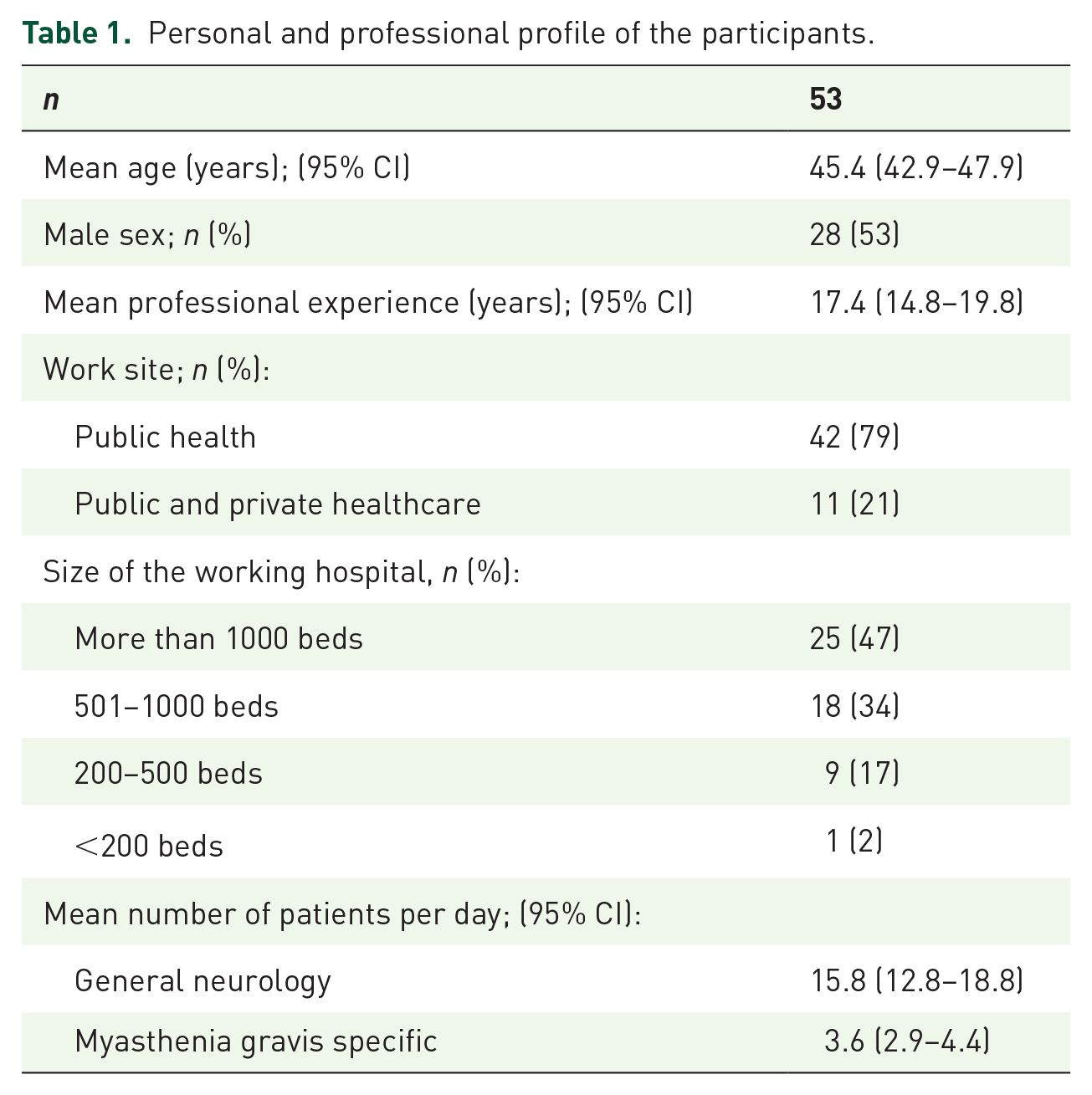
Fifty-three neurology physicians (53% male) with a median age of 44 and an age range of 30–65 participated in the survey. Their professional experience ranged from 4 to 42 years, with a median of 15.5 years. A total of 79.2% worked exclusively in public healthcare, and 81.2% in hospitals with more than 500 beds. They attended an average of 3.6 patients with MG per day (95% CI: 2.9–4.4). The personal and professional profiles of the participants are summarized in Table 1.
Personal and professional profile of the participants.
Opinion on current definitions in the field of MG
The proposed definitions are shown in Table 2. The degree of agreement with these definitions is shown in Table 2 and Figure 1. There is a high degree of agreement on all the definitions except for the one considering that the patients have controlled MG if they do not require intravenous immunoglobulin or periodic PLEX in the last year (Table 2 and Figure 1). In addition, participants were asked to give their opinion on the definition of controlled MG, considering the presence and severity of AEs. Thirty-four percent considered that the definition of controlled MG is independent of the presence of AEs and their severity, 5.7% reported that in controlled MG, there are no AEs or these are mild or moderate, 49.1% that there are no AEs or these are mild, and 11.3% that there are no AEs related to medications.
Definitions related to MG and degree of agreement.

Mean or median degree of agreement with the definitions on a scale from 1 to 5 (1 = total disagreement; 5 = total agreement).
MG, Myasthenia Gravis; MG-ADL, Myasthenia Gravis Activities of Daily Living scale; MGFA-PIS, Myasthenia Gravis Foundation of America-Post Intervention Status.
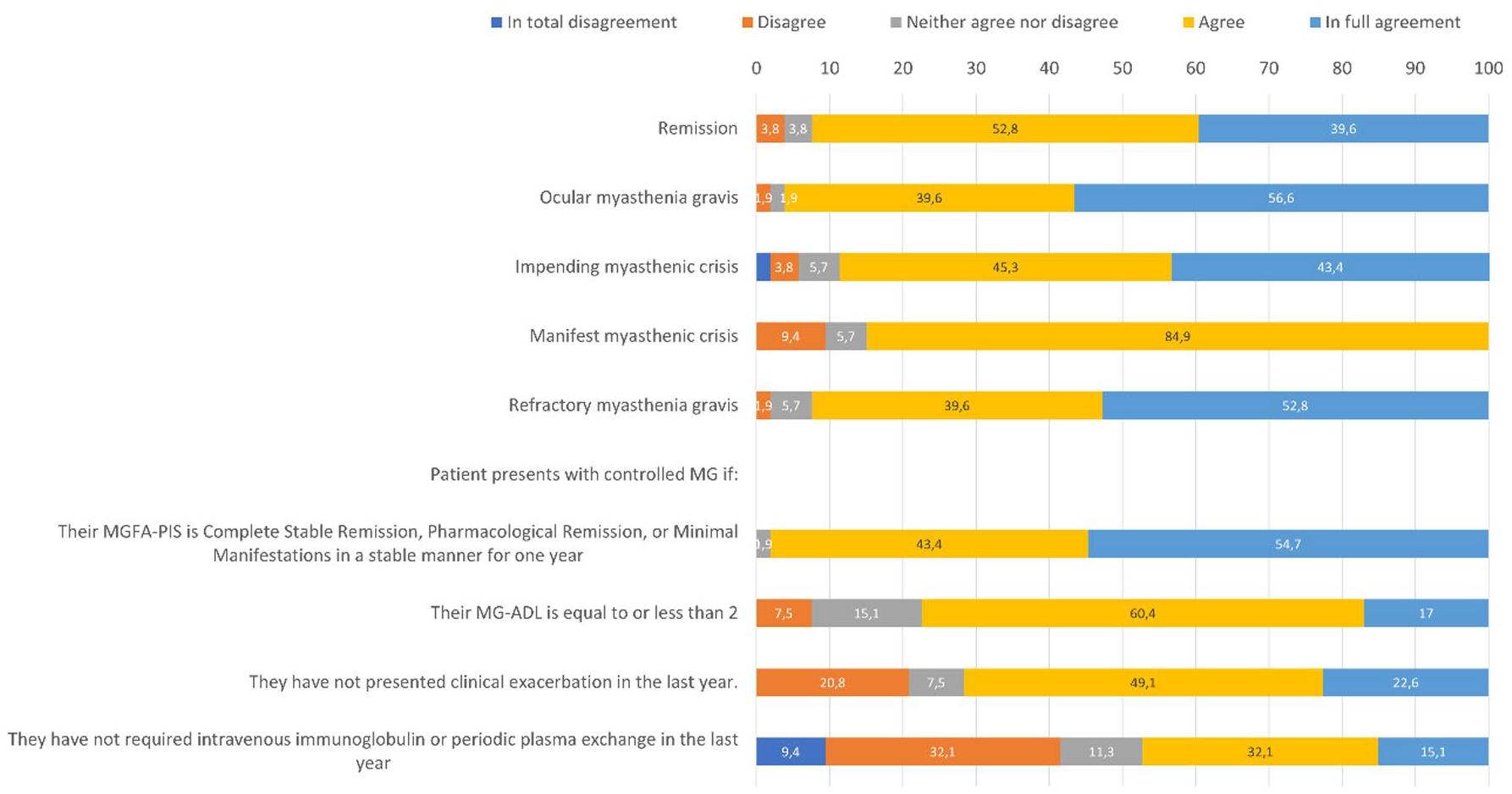
Degree of agreement on definitions related to myasthenia gravis.
Level of knowledge of the recommendations
A total of 61 recommendations were assessed and divided into 11 blocks. Tables S1–S11 show the specific wording of the recommendations and the level of knowledge about them. At least 90% of the neurologists were aware of 40 out of the 61 recommendations. The least known recommendations were R38 regarding using cyclosporine in pregnancy and R28 for managing children with acquired autoimmune ocular MG (50.9% and 69.8% knowledge by participants, respectively).
Diagnosis
The recommendation to consider MG when characteristic signs and symptoms are present, along with specific antibodies (R1), was widely known (96.2%). The recommendation to rule out MG in patients with autoimmune diseases, thyroid abnormalities, or thymoma (R2) had a knowledge level of 73.6%.
In R3, which had a knowledge level of 94.3%, respondents were aware that a firm diagnosis of MG should generally rely on a combination of characteristic clinical history and physical examination supported by diagnostic tests. The presence of antibodies, such as Anti-acetylcholine receptor antibodies (anti-AChR) anti-MuSK, or anti-LRP4, is central to the diagnosis. Electrophysiological studies, such as repetitive nerve stimulation at 2–3 Hz, are recommended to support the diagnosis, although they are not always necessary if antibodies are present. In cases where both antibody and electrophysiological tests are negative, 100% of respondents recognized the value of relying on the clinical response to IS therapy or PLEX/IVIg, as well as a positive Tensilon or neostigmine test, to confirm the diagnosis (R4). However, caution is needed due to potential side effects and the possibility of false positives.
Symptomatic and immunosuppressive treatment
A total of 100% of respondents recognized the recommendation to use pyridostigmine as part of the initial treatment in most patients (R5) and to consider its discontinuation as a treatment goal indicator (R6). Similarly, 100% agreed that corticosteroids or other IS therapies should be introduced when pyridostigmine is insufficient (R7), and nonsteroidal IS agents should be used when steroids are contraindicated or the patient is corticodependent (R8, 81.1%). For R9, all respondents (100%) demonstrated knowledge of the different nonsteroidal IS agents that can be used in the treatment of MG, including azathioprine, cyclosporine, mycophenolate mofetil, methotrexate, and tacrolimus. Similarly, in R10, all respondents (100%) agreed that patients with refractory MG should be referred to specialized centers. These centers offer advanced therapies such as chronic IVIg, PLEX, cyclophosphamide, or rituximab for patients who do not respond to conventional treatments. Regarding dosage and tapering of IS therapies (R11), 94.3% of respondents understood that tapering must be done gradually, and that immunosuppression may need to continue for years, sometimes for life.
IVIg and plasma exchange use
All respondents (100%) recognized that PLEX and IVIg are appropriate short-term treatments in MG patients with life-threatening symptoms such as respiratory insufficiency or dysphagia (R12) and when a rapid response is needed (R14). Additionally, 98.1% acknowledged their use in preparation for surgery in patients with bulbar dysfunction (R13), and 98.1% were aware of their use when other treatments are insufficiently effective (R15).
A total of 84.9% of respondents knew their role before corticosteroid therapy to prevent exacerbations (R16). Although clinical trials suggest equal effectiveness of PLEX and IVIg in treating myasthenic crises, 77.4% recognized that PLEX may be faster based on expert consensus (R17).
Regarding treating severe generalized MG, 100% of respondents were aware that PLEX and IVIg are equally effective (R18). However, 88.7% acknowledged the uncertainty regarding IVIg efficacy in milder or ocular MG (R19). Finally, 96.2% recognized IVIg as maintenance therapy for refractory MG or when IS agents are contraindicated (R20).
Thymectomy
For children with generalized AChR-Ab+ MG, 66.0% of respondents demonstrated knowledge that thymectomy should be considered when pyridostigmine and IS therapy are insufficient (R21). In comparison, 62.3% knew it should also be considered to avoid complications from IS therapy (R22). Among adults aged 18–50 with nonthymomatous generalized MG and AChR-Ab+, 96.2% of respondents knew thymectomy should be considered early to improve outcomes and reduce the need for immunotherapy and hospitalizations (R23). Regarding the timing of thymectomy, 94.3% understood it should be performed electively when the patient is stable to avoid postoperative complications (R24). Knowledge was also high (86.8%) regarding the recommendation to strongly consider thymectomy in patients who do not respond adequately to immunotherapy or have intolerable side effects (R25). A total of 94.3% of respondents were aware that endoscopic and robotic thymectomy approaches are increasingly performed with similar outcomes to more invasive procedures (R26), and 90.6% recognized that thymectomy is not recommended for patients with MuSK, LRP4, or agrin antibodies (R27).
Juvenile MG and MuSK-MG
For juvenile MG, 69.8% of respondents recognized that children with acquired autoimmune ocular MG are more likely to experience spontaneous remission and thus can be treated initially with pyridostigmine, with immunotherapy introduced if treatment goals are not achieved (R28). A larger proportion (88.7%) demonstrated knowledge of the risks of steroid side effects in children, such as growth failure, poor bone mineralization, and infection, and understood that the lowest effective dose should be used long term (R29). Additionally, 84.9% knew that maintenance PLEX or IVIg are alternatives to IS drugs in children (R30).
For MuSK-MG, 94.3% of respondents understood that those patients often respond poorly to ChEIs and that conventional pyridostigmine doses frequently cause side effects (R31). Moreover, 77.4% recognized that PLEX appears to be more effective than IVIg in treating MuSK-MG (R32).
MG in pregnancy
Most respondents (98.1%) demonstrated knowledge that pregnancy planning should start well in advance to optimize the patient’s condition and minimize fetal risks (R33). Additionally, 96.2% recognized that women whose MG is well-controlled before pregnancy tend to remain stable, though symptoms may worsen postpartum (R34). Prednisone was widely known as the IS agent of choice during pregnancy, with 90.6% showing knowledge of this recommendation (R36), while 79.2% of respondents understood that pyridostigmine is the first-line treatment, though its intravenous form should be avoided due to uterine contraction risks (R35).
A majority (86.8%) were aware that azathioprine is relatively safe during pregnancy (R37), though knowledge about cyclosporine was lower, with 50.9% acknowledging its relative safety (R38). A total of 92.5% of respondents understood that mycophenolate mofetil and methotrexate are contraindicated due to their teratogenic risks (R39), and 81.1% were aware that magnesium sulfate should not be used for eclampsia management in MG (R40). Finally, 98.1% of respondents recognized the need for newborns of myasthenic mothers to be examined for transient myasthenic weakness and have access to neonatal critical care (R41).
Ocular MG
All respondents (100%) recognized that ophthalmoparesis or ptosis unresponsive to ChEIs should be treated with IS agents if symptoms are functionally limiting (R42). Similarly, 98.1% knew that corticosteroids should be the initial treatment, with steroid-sparing agents used when corticosteroids are ineffective or not tolerated (R43). 96.2% of respondents were aware that low-dose corticosteroids can be effective for ocular MG, reducing side effects associated with high-dose use (R44). A smaller proportion (73.6%) understood that thymectomy may be an option for AChR-Ab+ patients who do not respond to ChEIs and prefer to avoid or are refractory to IS therapy (R45).
Other treatments
Most respondents (98.1%) demonstrated awareness of the use of rituximab as an early option in MuSK Ab+ MG patients unresponsive to initial immunotherapy (R46). While 96.2% recognized its uncertain efficacy in refractory AChR-Ab+ MG but supported its use when other treatments fail (R47), 84.9% of respondents recognized the use of methotrexate as a steroid-sparing agent, despite limited data (R48).
Knowledge about eculizumab was high, with 98.1% of respondents acknowledging its use for severe refractory AChR-Ab+ MG (R49) and its evolving role in treatment (R50). Additionally, 86.8% were aware of the importance of following immunization guidelines for meningococcal meningitis before initiating eculizumab (R51).
Immune checkpoint inhibitors
Most respondents (86.8%) knew that the risk of MG and other immune-mediated neurologic conditions should be discussed with patients considering ICIs (R52). Fewer respondents (73.6%) demonstrated knowledge about the lack of evidence for AChR antibody testing in patients without MG before initiating ICIs (R53), and 75.5% were aware that MG associated with ICIs tends to be severe, with a high risk of respiratory crises (R54). A total of 77.4% of respondents recognized that pre-existing MG is not an absolute contraindication for ICIs, although combined therapies should be avoided to reduce adverse events, and close monitoring is essential (R55). Finally, 86.8% demonstrated knowledge that high-dose steroids combined with PLEX or IVIg might be necessary for treating MG exacerbations during ICIs (R56).
Follow-up
Almost all respondents (98.1%) recognized the importance of non-pharmacological measures and regular follow-ups with a neurologist and family physician for managing MG (R57). All respondents (100%) understood the necessity of monitoring MG symptoms, treatment side effects, and concurrent conditions (e.g., autoimmune diseases, infections, and surgeries) during follow-up visits (R58). 94.3% of respondents acknowledged the need for closer supervision of patients at higher risk of MG crises, including early referral to MG centers or hospitals (R59). A total of 98.1% were aware of the need to monitor newly diagnosed patients closely for improvements and medication side effects during initial treatment (R60), and 96.2% recognized the importance of regular follow-up for patients with long-standing stable MG, particularly when reducing medication to prevent relapse (R61).
Degree of agreement with the recommendations
Figure 2 shows the mean values (95% CI) of the responses regarding the degree of agreement with the 61 recommendations. The proportion of participants who voted for each of the categories (from “strongly disagree” to “strongly agree”) is shown in Tables S1–S11.

Mean degree of agreement with the recommendations on the management of myasthenia gravis.
Out of the 61 recommendations reviewed, only three had a mean response below 3.5 on a scale from 1 to 5: R2 (we should rule out MG in patients with thyroid abnormalities, other autoimmune diseases, or thymoma); R19 (the efficacy of IVIg is less well supported in milder MG or ocular MG); and R38 (current information indicates that cyclosporine is relatively safe in pregnant women who are uncontrolled or intolerant of corticosteroids) (Figure 2). R19 and R38 are the only recommendations where the majority response from the participants is not “agreement” or “total agreement” (Tables S1–S11).
Degree of implementation of the recommendations
The mean values (95% CI) of the responses regarding the degree of implementation of the 61 recommendations are shown in Figure 3. The proportion of participants who voted for each of the categories (from “never apply the recommendation” to “always apply the recommendation”) is shown in Tables S1–S11.

Mean degree of implementation of the recommendations for the management of myasthenia gravis.
Out of the 61 recommendations reviewed, seven had a mean response of 2.5 or less on a scale from 1 to 4: R17 regarding the use of IVIg and PLEX; R21 and R22 regarding thymectomy in children; R38 regarding the use of cyclosporine in pregnancy; R45 regarding thymectomy in patients with ocular MG; R48 regarding the use of methotrexate; and R49 regarding the use of eculizumab (Figure 3).
Associations between the professional profile and the degree of knowledge, agreement, and implementation of the recommendations
Tables S12–S14 show associations between knowledge, agreement, and implementation of the recommendations and personal or professional factors such as age, sex, professional experience, number of total MG patients seen daily, and public or private workplace. Age only influenced knowledge in one out of the 61 recommendations (R35), and years of experience influenced knowledge in three (R16, R38, and R55). The total number of patients seen daily influenced the knowledge of four recommendations (R37, R38, R45, and R61), although only sometimes associated with greater knowledge for each. The number of patients seen with MG daily was related to lower knowledge of two recommendations (R2 and R35). There was also little correlation between these factors and the degree of agreement with the recommendations, as no Spearman coefficient exceeded four. Female gender was associated with greater agreement in six recommendations (R4, R10, R13, R15, R19, and R39) and greater implementation in seven recommendations (R1, R6, R7, R10, R15, R19, and R39). Working in public healthcare was associated with greater agreement and implementation of three recommendations (R7, R14, and R24).
Regarding the number of total patients seen per day and the implementation of the recommendations (Table 3), eight recommendations had a statistically significant weak inverse relationship. For one recommendation, the relationship was direct. In two recommendations, there was a weak inverse relationship between the number of patients seen per day with MG and the implementation of the recommendation.
Association between the years of professional experience and the volume of patients seen per day with the degree of implementation of the recommendations.

The wording of each recommendation has been adapted for clarity in the table; the full original wording is available in Tables S1–S11.
Spearman’s correlation coefficient. The degree of implementation was measured on a scale from 1 to 4 (1 = the recommendation is never implemented; 4 = the recommendation is always implemented).
AChR-Ab+, anti-acetylcholine receptor antibody positive; ChEIs, cholinesterase inhibitors; IVIg, intravenous immunoglobulin; MG, myasthenia gravis; PLEX, plasma exchange.
Discussion
This work reflects the opinion of neurologists who treat patients with MG on key definitions of the disease, their degree of knowledge and agreement with the most recent recommendations for its management, and the degree of application of the recommendations in actual practice in Spain. This survey can serve to make a diagnosis of the situation regarding the clinical management of MG in routine practice conditions, describe their clinical habits, and detect areas that deserve further research and consensus.
Despite the scarce evidence with conventional drugs or the limited experience with new drugs, our survey allowed us to conclude that the majority of physicians are aware of the recommendations, agree with most of them, and usually apply them in clinical practice, with exceptions especially concerning juvenile MG and the use of cyclosporine in pregnancy.
Regarding definitions related to MG, such as remission, ocular MG, impending myasthenic crisis, manifest myasthenic crisis, or refractory MG, there was a high degree of agreement with them despite not being revised since 2016. 9 On the other hand, there is no clear and homogeneous definition of controlled MG in the literature, and multiple efficacy parameters are used in clinical trials. 12 Therefore, the scientific committee created an ad-hoc definition, including the Myasthenia Gravis Foundation of America-Post Intervention Status (MGFA-PIS) scale, 13 and the Myasthenia Gravis Activities of Daily Living scale (MG-ADL). The highest degree of agreement of the participants was on the use of the MGFA-PIS scale and the least on the use of IVIg or PLEX in the last year. On the other hand, 59% of the participants considered that an essential criterion for controlled MG is not the presence of medication-related AEs or mild AEs. In contrast, 34% considered that controlled MG is a concept independent of the presence of AEs and their severity. A greater degree of consensus would be necessary to homogenize the definition of control in MG. Our group is working on a project in this direction.
Regarding the recommendations for the management of MG proposed by the guidelines,9–11 the level of knowledge of these recommendations was high. In two-thirds of the recommendations, the level of knowledge was greater than 90%. The profile of the participants indicated that they had experience in the management of MG and treated an appreciable volume of patients, so it was not surprising that the level of knowledge of the recommendations was high. The lesser-known recommendations deal with the management of MG in pregnancy (especially R38 regarding the use of cyclosporine in pregnancy) and the management of children with acquired autoimmune ocular MG (R28), which are relatively infrequent clinical situations in consultation.14–16 In addition, recommendations for these two types of patients have not been updated since 2016 in the international consensus on the management of MG.9,10 It would be necessary to create new recommendations for these populations, including new drugs or therapeutic strategies under development.17,18
Regarding the degree of agreement with the recommendations, in 59 out of 61 recommendations, the majority of participants’ responses fell within the “agree” or “strongly agree” range. The least supported recommendations were R19 regarding the efficacy of IVIg in milder MG or ocular MG, and, again, R38 regarding the use of cyclosporine in pregnancy. In this regard, there are no well-controlled clinical trials in pregnant women using cyclosporine. 19 A recent meta-analysis involving transplanted pregnant women on cyclosporine treatment showed an increased risk of preeclampsia, preterm delivery, and low birth weight that were related to the underlying pathology rather than to cyclosporine per se. 20 According to the technical data sheet, the use of cyclosporine during pregnancy is not recommended unless the potential benefits for the mother outweigh the risks for the fetus. 19 Our results suggest that more clinical experience and a broader consensus are needed on how to manage MG during pregnancy.
The degree of implementation of the recommendations was generally high except for seven recommendations: one was related to the use of PLEX (R17), three were related to thymectomy (R21, R22, and R45), one to cyclosporine in pregnancy (R38) and two to the use of methotrexate (R48) or eculizumab (R49). However, in six out of seven of these recommendations (all except R38 regarding cyclosporine in pregnancy), most participants agreed or strongly agreed with the recommendation. The discrepancy between the degree of agreement with the recommendation and its lesser use in clinical practice may mean that these are procedures or treatments (such as PLEX, thymectomy, or the use of eculizumab) that are not routinely used in actual practice.
Studies analyzing methotrexate efficacy in MG are limited, yielding insufficient evidence to support its widespread use in clinical settings. 10 Therefore, its limited use in daily practice is not surprising. Additionally, the implementation of clinical practice guidelines often varies and can sometimes be suboptimal across various specialties, including neurology, due to factors such as time constraints, limited applicability to individual patients, and external pressures.21–28 In our study, the lack of robust evidence supporting certain recommendations—such as those related to cyclosporine use in pregnancy, methotrexate, PLEX in some indications, or thymectomy in specific subgroups—may further contribute to decreased adherence in these clinical scenarios. Therefore, we believe that strengthening the underlying evidence is a key strategy for improving the implementation of guidelines. However, the lower knowledge or implementation of guidelines in areas such as juvenile MG, MG in pregnancy, or the use of therapies like PLEX, thymectomy, or eculizumab may partly reflect limited clinical exposure or familiarity among practitioners. Efforts to enhance knowledge dissemination and clinical education are also crucial for improving the implementation of these recommendations.
Finally, regarding the correlations between the participants’ personal or professional factors and the questionnaire responses, the inverse correlation between the number of patients seen per day and the degree of application of eight recommendations stands out. In this sense, we should consider whether the overload of consultations may impact the application of the recommendations and, therefore, on the care of these patients with MG. In addition, the female participants showed a higher degree of agreement and a higher degree of implementation of several recommendations. There is little data in the literature on the influence of the physician’s sex regarding the degree of adherence to guidelines in chronic pathologies; however, some authors suggest that it is greater in women.29–31 More targeted studies would be needed to elucidate this question in the case of MG.
Following the publication of the latest 2020 guidelines, three new treatments for MG, efgartigimod, ravulizumab, and zilucoplan, have been approved. Efgartigimod is a neonatal Fc receptor (FcRn) inhibitor approved by the European Medicines Agency (EMA) for the treatment of generalized MG with positive antibodies against ACh receptors (AChR-Ab+), which will have to be evaluated in clinical practice in the future. 32 Rozanolixizumab, nipocalimab, and batoclimab are other agents of the same family under development in clinical trials. 33 Ravulizumab is a humanized monoclonal complement inhibitor antibody with a more durable effect than eculizumab, approved for use in adult patients with generalized MG who are AChR-Ab+. 34 Zilucoplan is also a recently approved complement inhibitor.1,3 Efgartigimod and ravulizumab became available in Spain around the time of our survey, but their use was still limited during this period. Zilucoplan was approved after data collection was completed. Therefore, we do not expect any of these therapies to have had a significant impact on our results. Updating therapeutic guidelines will be necessary to incorporate these new treatments.
Our study has some limitations, typical of cross-sectional surveys, such as selection or recall bias. The participants were neurologists who care for a significant number of patients with MG in all the Autonomous Communities, and we believe that they are a representative sample of patient care in Spain. However, participation was voluntary among members of the GEEN of the Spanish Society of Neurology, which may introduce self-selection bias and limit the generalizability of our findings to all MG specialists in Spain. On the other hand, international guidelines of recognized prestige were selected for the elaboration of the questionnaire, although there may be other local or national guidelines with different recommendations. Additionally, the cross-sectional nature of the study provides only a snapshot at a single point in time and does not allow for assessment of causality or changes in practice over time. Furthermore, as the study relies on self-reported knowledge and implementation, there may be recall bias, social desirability bias, or discrepancies between reported and actual clinical practice. Future research could benefit from incorporating both objective measures (e.g., chart reviews, analysis of treatment patterns) and qualitative approaches (e.g., open-ended questions, interviews) to assess clinical practice and explore the reasons for discrepancies more accurately. In addition, the ad-hoc definition of controlled MG used in this study, although intended to address a gap in the literature, may not be universally accepted and could introduce variability in interpretation. We encourage further discussion and consensus on standardized definitions in future research. Moreover, this study did not include the perspective of patients with MG, which could have provided additional valuable insights into their care experiences. Nevertheless, despite these limitations, this approach offers valuable insights into current perceptions and practices among neurologists managing MG in Spain.
Conclusion
In summary, this work offers a view regarding the opinion of neurologists responsible for managing patients with MG about definitions and recommendations for managing MG and their degree of application in real practice. The degree of knowledge, agreement, and application is high. Still, there is a need to strengthen the evidence and consensus on some aspects, such as the management of juvenile MG or MG in pregnancy or the use of treatments such as PLEX, thymectomy, or eculizumab. Efforts to improve knowledge dissemination and clinical education are also essential for enhancing the implementation of these recommendations.
Supplemental Material
sj-docx-1-taj-10.1177_20406223251368063 – Supplemental material for Practitioner’s perception of myasthenia gravis management recommendations in clinical practice: a cross-sectional survey study
Supplemental material, sj-docx-1-taj-10.1177_20406223251368063 for Practitioner’s perception of myasthenia gravis management recommendations in clinical practice: a cross-sectional survey study by Elena Cortés-Vicente, Antonio Guerrero-Sola, Fernando Caballero-Martínez, Francisco Javier Campos-Lucas, Juan Gómez-Salgado, Diana Monge-Martín and Rodrigo Álvarez-Velasco in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223251368063 – Supplemental material for Practitioner’s perception of myasthenia gravis management recommendations in clinical practice: a cross-sectional survey study
Supplemental material, sj-docx-2-taj-10.1177_20406223251368063 for Practitioner’s perception of myasthenia gravis management recommendations in clinical practice: a cross-sectional survey study by Elena Cortés-Vicente, Antonio Guerrero-Sola, Fernando Caballero-Martínez, Francisco Javier Campos-Lucas, Juan Gómez-Salgado, Diana Monge-Martín and Rodrigo Álvarez-Velasco in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We thank all the neurologists who participated in this work as Collaborating Researchers. We thank Irene Santamaría of the Universidad Francisco de Vitoria (Madrid, Spain) for her help in coordinating the study’s technical aspects. We also thank Dr. Pablo Rivas for his collaboration in editing this article.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
