Abstract
Background:
Hepatocellular carcinoma (HCC) is a major global health issue, in which the underlying liver disease aetiology has shifted towards non-viral causes, particularly metabolic (dysfunction)-associated fatty liver disease (MAFLD). While traditionally associated with cirrhosis, a subset of HCC cases arises in patients with MAFLD but without cirrhosis, whose characteristics remain poorly understood.
Objectives:
The study aims to explore the clinical, tumour and genetic characteristics of non-cirrhotic MAFLD-related HCC when compared to those that develop in the context of cirrhosis.
Design:
A multi-centre, retrospective study of 89 MAFLD-related HCC patients enrolled between 2009 and 2023 was performed.
Methods:
We conducted a study of well-defined MAFLD-related HCC patients to explore their MAFLD-related clinical and genetic associations. Statistical analysis was undertaken to compare the underlying cirrhosis and non-cirrhosis groups for HCC features, adjusting for relevant confounders.
Results:
Patients with HCC arising in cases of MAFLD without cirrhosis exhibited a lower body mass index, higher triglyceride levels and increased smoking prevalence compared to their counterparts with cirrhosis. Despite arising in the absence of cirrhosis, these patients had more aggressive tumour features, including larger tumour size, multifocality and portal vein thrombosis. Logistic regression confirmed non-cirrhosis status to be an independent predictor of larger tumour size and increased lesion number.
Conclusion:
Non-cirrhotic MAFLD-related HCC presents with distinct clinical and tumour characteristics, suggesting the existence of unique disease drivers that are yet to be discovered.
Plain language summary
Liver cancer (HCC) is a serious global health problem. Recently, the causes of liver disease leading to HCC have shifted from viral infections to metabolic issues, especially metabolic-associated fatty liver disease (MAFLD). While HCC is usually linked to liver scarring (cirrhosis), some cases occur in MAFLD patients without cirrhosis, and these cases are not well understood. We studied 89 patients with MAFLD-related HCC from multiple centers to understand their clinical and genetic traits. We compared patients with and without cirrhosis to identify differences in HCC features, using statistical methods to account for other influencing factors. Patients with HCC from MAFLD without cirrhosis had lower body mass index (BMI), higher triglyceride levels, and smoked more than those with cirrhosis. Despite not having cirrhosis, these patients had more aggressive tumors, including larger sizes, multiple tumors, and vein involvement. Statistical analysis showed that not having cirrhosis was linked to larger and multiple tumors. HCC in MAFLD patients without cirrhosis has unique clinical and tumor characteristics, indicating there may be unknown factors driving this disease.
Introduction
Hepatocellular carcinoma (HCC) places a significant health and economic burden on society, being the third leading cause of global cancer-related mortality. Annual deaths from HCC are projected to rise dramatically from 800,000 in 2020 to 1,300,000 by 2040. 1 The underlying causal factors for HCC have shifted from viral hepatitis to non-viral aetiologies, 2 mirroring the advances in the management of hepatitis infections and the escalating prevalence of metabolic (dysfunction)-associated fatty liver disease (MAFLD). 3
MAFLD is now the most common type of chronic liver disease, whose prevalence is expected to increase even among non-obese individuals.4,5 Approximately 30% of the world’s population is affected by MAFLD 6 and the prevalence is projected to rise to 56% by 2030. 7 MAFLD is a well-recognised risk factor for liver cancer and given its prevalence and the rising burden of overweight and obesity, the proportion of MAFLD-related HCC is likely to increase over time.8,9 Currently, total MAFLD-related HCC prevalence is estimated at 48.7%, with single-MAFLD HCC accounting for 12.4% of cases.8–10
Most HCC cases typically occur on a background of liver cirrhosis, while the occurrence of HCC in a liver without cirrhosis is thought to be uncommon. Recent studies, however, have demonstrated that MAFLD patients without liver cirrhosis (i.e. non-cirrhotic MAFLD) also show an increased risk for HCC compared to individuals with other liver diseases. 11 Based on a recent meta-analysis, the prevalence of HCC in non-cirrhotic metabolic-associated steatohepatitis (the inflammatory form of MAFLD) patients is 38%, whereas the prevalence of HCC in patients with other non-cirrhotic liver diseases is 14.2%. 12 The projected rise in the proportion of HCC cases linked to MAFLD poses a challenge for cancer surveillance and for early diagnosis. In this regard, a better understanding of the clinical characteristics of non-cirrhotic MAFLD-related HCC is the initial step for the development of management guidelines. In the present study, we explored the clinical, genetic and tumour characteristics of HCC in the context of non-cirrhotic MAFLD in a well-characterised cohort of HCC patients.
Patients and methods
Study cohort
This study was conducted between 2009 and 2023. We included 89 consecutive MAFLD-related HCC patients from Westmead and Blacktown Hospitals in Sydney, Australia. Written informed consent was obtained from all patients. The study was approved by the Human Research Ethics Committee of the Sydney West Area Health Service (Approval ID: HREC/18/WMEAD/5 (5522)). All methods were performed in accordance with the relevant guidelines and regulations, including the Declaration of Helsinki.
MAFLD was diagnosed by the presence of hepatic steatosis on liver biopsy, ultrasound or CT/MRI imaging, together with evidence of metabolic dysfunction according to the proposed criteria for MAFLD. 13 All MAFLD-related HCCs were confirmed through radiological imaging and liver histology. Patients with other concomitant aetiologies such as alcohol-related liver disease, viral infection (hepatitis B, C or D infection) and haemochromatosis were excluded. In addition, patients with a history of alcohol consumption >2 standard drinks per day and those with unknown cirrhosis status were excluded.
Clinical variables retrieved included patient demographics, body mass index (BMI), history of diabetes, hypertension, serum cholesterol, triglyceride, high-density lipoprotein (HDL), low-density lipoproteins (LDL), fasting haemoglobin A1C, baseline liver function tests, Child-Pugh-Turcotte score and liver fibrosis score (by fibrosis-4 index, FIB-4). Information regarding the presence of cirrhosis, number of tumour nodules, size of tumours, presence of portal vein thrombosis (PVT) and Barcelona Clinic Liver Cancer (BCLC) staging score was collected.
Genotyping
Tissues were processed for genotyping for seven single-nucleotide polymorphisms, including DEPDC5 (rs1012068), MICA (rs259654), MERTK (rs4374383), IFNL3 (rs8099917), MBOAT7 (rs8736), PNPLA3 (rs738409) and TM6SF2 (rs58542926). First, DNA was extracted from the frozen tissue using the FavorPrep™ Genomic DNA Mini Kit, FAVORGEN, Taiwan. Then genotyping was performed using the Allelic Discrimination Method (Applied Biosystems, Foster City, CA, USA) with TaqMan GTXpress™ Master Mix.
Statistical analysis
Continuous variables are expressed as median and range or number. Categorical variables are expressed as frequencies and percentages. The difference between groups was analysed using the Wilcoxon rank-sum test for continuous variables and Fisher’s exact test for categorical variables. A logistic regression model was used to determine the independent factors associated with HCC features (tumour size and number of lesions). Data are expressed as odds ratios (OR) and 95% confidence intervals (CI). A p < 0.05 was considered statistically significant. Data was analysed using SPSS version 29, IBM SPSS Statistics for Macintosh, Version 29.0, Armonk, NY.
Results
Clinical and genetic characteristics of patients with MAFLD-related HCC
A total of 89 well-characterised MAFLD-related, histologically-proven HCC patients were recruited for the study. The median age of participants was 71 years and 64% were male. Of these patients, 16 (18%) did not have liver cirrhosis. Clinical, biochemical and genetic characteristics of non-cirrhotic and cirrhotic MAFLD-related HCC patients are presented in Table 1.
Clinical characteristics of non-cirrhotic and cirrhotic HCC-MAFLD patients.

Values are median of min–max and the values in the parentheses indicate the percentage. p Value was calculated using Fisher’s exact test and Student’s t test. The bold values indicate statistically significant results. Values in parentheses are percentages.
ALBI Grade, albumin-bilirubin grade; ALT, alanine transaminase; AST, aspartate transaminase; BMI, body mass index; FIB-4, fibrosis-4 index; HCC, hepatocellular carcinoma; HDL, high-density lipoprotein; LDL, low-density lipoproteins; MAFLD, metabolic dysfunction-associated fatty liver disease; NFS, NAFLD fibrosis score.
Compared to the cirrhotic MAFLD-related HCC, non-cirrhotic MAFLD-related HCC patients had lower liver fibrosis stage, a higher platelet count (209 vs 114, p = 0.01), lower BMI (26.9 vs 31.1 kg/m2, p = 0.04) and higher triglyceride level (1.9 vs 1.2 mmol/L, p = 0.04). Patients with non-cirrhotic MAFLD-related HCC were more likely to be smokers (66.7% vs 37.5%, p = 0.04). However, no significant difference in age, sex, ethnicity, family history of liver cancer or other parameters of the metabolic profile was identified between the two groups (Table 1).
No significant difference in the proportions of the well-validated MAFLD or HCC related genetic polymorphisms, namely DEPDC5 (rs1012068), MICA (rs259654), MERTK (rs4374383), IFNL3 (rs8099917), MBOAT7 (rs8736), PNPLA3 (rs738409), TM6SF2 (rs58542926) variants was identified between the two groups (Table 1).
Non-cirrhotic MAFLD-related HCC patients have more advanced tumour stages and more aggressive tumour characteristics
We explored the tumour characteristics between non-cirrhotic and cirrhotic MAFLD-related HCC patients. Interestingly, tumour size in the non-cirrhotic MAFLD-related HCC patients tended to be greater than in patients with cirrhosis. As shown in Table 2, the tumour size in 90.9% of patients with non-cirrhotic MAFLD-related HCC was ⩾3 cm, whereas only 50% of patients with cirrhotic MAFLD-related HCC had tumours ⩾3 cm (p = 0.02). Multifocal tumours (number of tumour nodules >3) and PVT were more common in non-cirrhotic MAFLD-related HCC than in those with MAFLD cirrhosis and HCC (45.5% vs 13.2%, p = 0.02; and 55.6% vs 13.4%, p = 0.009, respectively; Table 2).
Tumour characteristics based on cirrhosis status.
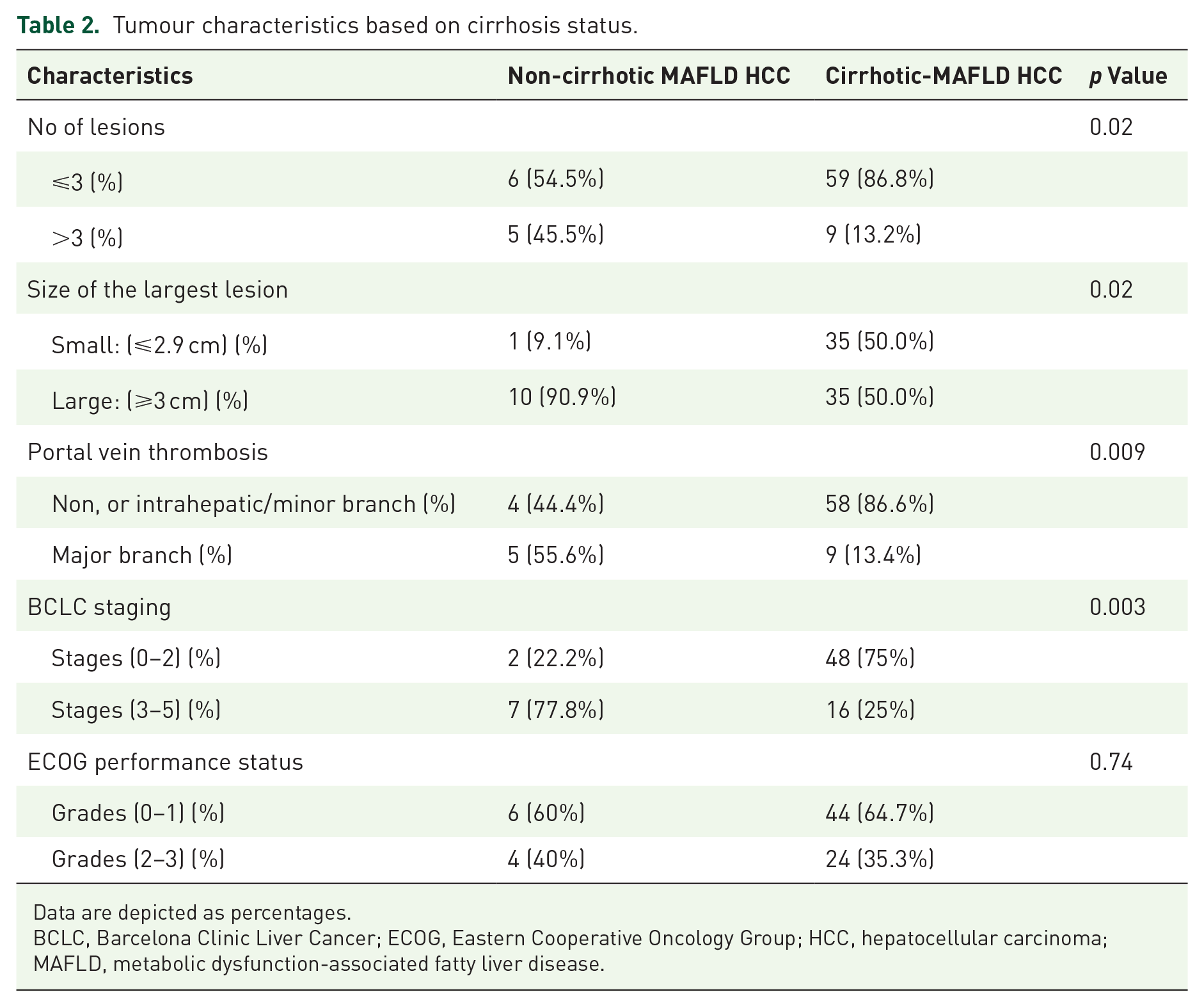
Data are depicted as percentages.
BCLC, Barcelona Clinic Liver Cancer; ECOG, Eastern Cooperative Oncology Group; HCC, hepatocellular carcinoma; MAFLD, metabolic dysfunction-associated fatty liver disease.
Comparison of tumour stage at diagnosis revealed that patients with non-cirrhotic MAFLD-related HCC were diagnosed with more advanced tumour stage than patients in the cirrhotic MAFLD-related HCC group. Over three-quarters (77.8%) of the patients with non-cirrhotic MAFLD-related HCC were in BCLC stages of C–D whereas only 25% of patients with cirrhotic MAFLD-related HCC were diagnosed in BCLC stages C–D (p = 0.003; Table 2). Overall, MAFLD-related HCC in patients without cirrhosis showed features suggestive of more advanced or aggressive tumours compared to their counterparts with cirrhosis.
Independent factors and profiles associated with tumour size and number of tumour lesions
To adjust for confounding variables, in a subsequent analysis, we compared non-cirrhotic and cirrhotic MAFLD-related HCC patients for associations with HCC features, namely the size and number of tumour lesions, using multiple logistic regression analysis.
In this analysis, in model 1, the association of the non-cirrhotic patients was significantly stronger with respect to tumour size (OR: 10.19, 95% CI: 1.2–84.6, p = 0.03) and the number of tumour lesions (OR: 5.61, 95% CI: 1.3–23.8, p = 0.02) independent of patient age, sex and race. Similar results were demonstrated in a second model adjusting for diabetes, BMI and family history of liver cancer in addition to the variables indicated above. Patients with non-cirrhotic MAFLD-related HCC had a significantly stronger association with tumour size (OR: 9.94, 95% CI: 1.1–88.7, p = 0.04) and the number of lesions (OR: 7.51, 95% CI: 1.4–4, p = 0.01) compared to those with MAFLD-related HCC and cirrhosis (Table 3).
Independent factors associated with tumour size and number of lesions analysed by multivariate logistic regression.

Non-cirrhotic HCC-MAFLD was compared to the cirrhotic HCC-MAFLD group.
BMI, body mass index; CI, confidence interval; HCC, hepatocellular carcinoma; MAFLD, metabolic dysfunction-associated fatty liver disease; OR, odds ratio.
Lean MAFLD patients exhibit enhanced aggressiveness in HCC tumours
Since patients with non-cirrhotic MAFLD-related HCC tend to have lower BMI compared to their cirrhotic counterparts, and recent data suggest that lean MAFLD might represent a distinct entity, 4 we compared the clinical characteristics of patients with lean, MAFLD-related HCC to patients with obese, MAFLD-related HCC.
As shown in Table 4, lean MAFLD-related HCC patients tended to be older and have lower bilirubin but otherwise similar characteristics to their obese counterparts. In addition, they tended to have larger tumours (>3 cm), a higher incidence of PVT involving major branches and more advanced stages based on BCLC criteria, though these did not reach statistical significance.
Clinical characteristics of MAFLD-related HCC patients regrouped as lean versus obese MAFLD.

For SNPs, Pearson Chi-square (asymptotic significance (two-sided)) has been used for the p value.
ALT, alanine transaminase; AST, aspartate transaminase; BCLC, Barcelona Clinic Liver Cancer; FIB-4, fibrosis-4 index; HCC, hepatocellular carcinoma; HDL, high-density lipoprotein; LDL, low-density lipoproteins; MAFLD, metabolic dysfunction-associated fatty liver disease.
Discussion
Using a well-characterised cohort of patients with MAFLD-associated HCC, we identified three key findings. First, MAFLD-related HCC patients without cirrhosis exhibit different clinical characteristics compared to their cirrhotic counterparts. Patients without cirrhosis had a lower BMI, higher triglyceride levels and a higher prevalence of smoking. Second, interestingly, despite being non-cirrhotic, this group displayed more aggressive tumour characteristics, including larger tumour size, a higher proportion of multifocal tumours and an increased likelihood of PVT. Third, lean MAFLD-related HCC tended to exhibit more aggressive tumour characteristics.
MAFLD has been increasingly recognised as a causal factor for HCC.14,15 Given the high population prevalence of MAFLD (one in three people), and the low prevalence of MAFLD-related cirrhosis (~1%), the non-cirrhotic cases constitute a considerable fraction of the HCC burden, posing a challenge for management and screening. 16 However, the clinical features of non-cirrhotic MAFLD-related HCC are not well characterised. We showed a predominantly similar metabolic profile between the cirrhotic- and non-cirrhotic MAFLD-related HCC patients, which is anticipated given their shared pathophysiology mediated by metabolic dysfunction. However, non-cirrhotic MAFLD-related HCC patients exhibited some differential characteristics, such as a lower BMI and a higher prevalence of smoking. Smoking is known to reduce body weight.
The lower BMI observed in non-cirrhotic MAFLD-related HCC patients is provocative. While this may relate to the higher prevalence of smoking, recent studies suggest that lean MAFLD represents a distinct entity within the spectrum of MAFLD that is shaped by differential metabolic adaptation mediated by differences in bile acids and gut microbiota.17,18 Notably, lean MAFLD patients tend to have worse long-term outcomes compared to their obese counterparts, despite their favourable metabolic and histological profiles.5,19 Our cohort of lean patients (Table 4) tended to have more aggressive tumour features compared to their counterparts who were obese. The finding that non-cirrhotic MAFLD-related HCCs tend to have larger and multiple tumours with major PVT raises intriguing possibilities. One possibility is that this group of patients represents the progression of lean MAFLD. Alternatively, it is possible that non-cirrhotic MAFLD-related HCC represents a distinct entity in which the pathogenic drivers have more to do with early oncogenic mutations rather than the milieu of cirrhosis. The third possibility is that patients without cirrhosis are more likely to be asymptomatic because of preserved liver function and hence present later with larger, multiple tumours that also have a chance to drive PVT.15,20 These findings suggest that although universal screening for HCC in all MAFLD patients might not be practical or cost-effective, there is an urgent need for the discovery of novel biomarkers and risk scores to identify non-cirrhotic patients with MAFLD who are at an increased risk of HCC development. Clearly, further detailed mechanistic studies are also warranted.
Our analysis highlights a striking disparity in smoking prevalence between the two groups. Non-cirrhotic MAFLD-related HCC patients had a notably higher likelihood of being smokers compared to their cirrhotic counterparts. Smoking is a well-known risk factor for HCC, and indeed, a recent study has shown that gut bacteria alleviate smoking-related metabolic steatohepatitis by degrading gut nicotine. 20 While the exact mechanisms linking smoking and the development of HCC in non-cirrhotic MAFLD patients remain to be elucidated, it is plausible that alterations in gut microbiota composition and function, influenced by smoking habits, contribute to MAFLD-related HCC. Further investigations into the complex interactions between smoking, gut microbiota and liver pathology are warranted.
The key strength of the current study is that stringent criteria were employed to ensure a well-defined cohort of MAFLD patients without other aetiologies, as well as a detailed characterisation of clinical and genetic variables. However, there are noteworthy limitations. First, this study has a relatively small sample size, and the results may not be generalisable to other cohorts. There might be referral bias in this study as patients were recruited from a tertiary care centre. Thirdly, the exact stage of liver fibrosis was not consistently reported in the liver pathology. However, we complemented our data with the calculation of FIB-4 and NFS scores, 21 both showed that all non-cirrhotic patients fell below the cirrhotic range for these tests. Finally, the cross-sectional nature of the study and lack of longitudinal follow-up data do not allow for comparing the outcomes and differential overall survival between the cirrhotic and non-cirrhotic MAFLD-related HCC patients.
In conclusion, these findings suggest that despite the absence of cirrhosis, non-cirrhotic MAFLD-related HCC has different clinical and tumour characteristics compared to their counterparts with cirrhosis. Future studies in larger and multi-centre cohorts with a detailed clinical, metabolic and genetic characterisation of patients with and without cirrhosis will help the development of a distinct signature that can be used for screening and early diagnosis of HCC in MAFLD patients without cirrhosis.
