Abstract
Background:
The effectiveness and side effects between different medical treatments in patients with primary hyperaldosteronism have not been systematically studied.
Objective:
To analyze the efficacy between different mineralocorticoid receptor antagonists (MRAs) and epithelial sodium channel (ENaC) inhibitors in a network meta-analysis (NMA) framework, while also evaluating adverse events.
Design:
Systematic review and NMA.
Data sources and methods:
The systematic review and NMA was reported according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. PubMed, MEDLINE, the Cochrane library, and Excerpta Medica database (EMBASE) were searched for randomized controlled trials (RCTs) involving adult patients with primary hyperaldosteronism until 23 June 2023. Studies that compared the efficacy and side effects of different medical treatments of primary hyperaldosteronism were included. The primary outcomes included the effect on blood pressure, serum potassium, and major adverse cardiovascular events. The secondary outcomes were adverse events related to MRAs (hyperkalemia and gynecomastia). Frequentist NMA and pairwise meta-analysis were conducted.
Results:
A total of 5 RCTs comprising 392 participants were included. Eplerenone, esaxerenone, and amiloride were compared to spironolactone and demonstrated comparable effect on the reduction of systolic blood pressure. In comparison to spironolactone, eplerenone exhibited a less pronounced effect on reducing diastolic blood pressure [−4.63 mmHg; 95% confidence interval (CI): −8.87 to −0.40 mmHg] and correcting serum potassium (−0.2 mg/dL; 95% CI: −0.37 to −0.03 mg/dL). Spironolactone presented a higher risk of gynecomastia compared with eplerenone (relative risk: 4.69; 95% CI: 3.58–6.14).
Conclusion:
The present NMA indicated that the blood pressure reduction and potassium-correcting effects of the three MRAs may demonstrate marginal differences, with confidence levels in the evidence being very low. Therefore, further research is needed to explore the efficacy of these MRAs, especially regarding their impact on mortality and cardiovascular outcomes.
Trial registration:
PROSPERO (CRD: 42023446811).
Introduction
Primary hyperaldosteronism is a common cause of secondary hypertension and is associated with hypokalemia.1,2 Compared to essential hypertension, primary hyperaldosteronism is associated with a higher risk of cardiovascular events and adverse effects in different target organs. These effects encompass organ fibrosis, 3 cardiovascular diseases, 4 osteoporosis, 5 metabolic syndrome, new-onset diabetes mellitus, 6 anxiety, 7 and obstructive sleep apnea. 8 The prevalence rate varies from 3.9% in stage 1 hypertension 9 to 22% in patients with resistant hypertension. 10 Accurate diagnosis and timely treatment of primary hyperaldosteronism not only cure this form of secondary hypertension but also reduce associated end-organ damage.8,11,12 However, the real-world screening rate for primary hyperaldosteronism is low 13 and forms a major limitation to effective treatments of the disease. Surgical adrenalectomy is the recommended treatment for lateralized primary hyperaldosteronism.1,2 Chen et al. 14 reported that the adrenalectomy group had a lower risk for mortality [odds ratio (OR) = 0.33, 95% confidence interval (CI): 0.15–0.73] and cardiovascular events (OR = 0.55, 95% CI: 0.40–0.74) compared with medical treatment group. For patients with bilateral adrenal hyperplasia or who are not eligible for surgery, mineralocorticoid receptor antagonists (MRAs) are commonly used to improve hypertension and correct hypokalemia. Recent guidelines1,2,15,16 have recommended spironolactone as the first-line medical treatment, based on early randomized controlled trial (RCT) evidence. 17 However, some other RCTs have not shown significant differences in blood pressure control between spironolactone and eplerenone.18,19 In addition, epithelial sodium channel (ENaC) inhibitors have being considered as potential medical agents for hyperaldosteronism. 20
Over the past two decades, numerous studies have explored primary hyperaldosteronism, 21 with recent advancements in genetic research,22,23 diagnostic testings,24 –26 and prognostic factor evaluations,11,22 which have deepened our understanding of the disease. However, despite these advancements, there remained a notable gap in the literature regarding the comparative efficacy of different medical treatments. Specifically, systematic examinations and comparisons of the effects of various MRAs such as spironolactone, eplerenone, and esaxerenone, along with ENaC inhibitors, have not been comprehensively conducted to date.
In light of this gap, our study aimed to analyze the effect on blood pressure, serum potassium, and major adverse cardiovascular events (MACE) between different MRAs and ENaC inhibitors in a network meta-analysis (NMA) framework. Additionally, we analyzed and compared the occurrence of adverse events such as hyperkalemia and gynecomastia associated with these medications.
Materials and methods
Literature search strategy
The current study was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Extension for NMAs (Supplemental Table 1) and the protocol was registered in PROSPERO (CRD: 42023446811).
Two independent investigators (W.-Y.H. and J.-J.C.) conducted an independent search for studies published prior to 23 June 2023 in the databases of PubMed, MEDLINE, the Cochrane library [including ClinicalTrials.gov and International Clinical Trials Registry Platform (ICTRP)], and Excerpta Medica database (EMBASE) without language limitation. Search strategies targeted published RCTs that compared the efficacy and side effects of different medical treatments of primary hyperaldosteronism. The comprehensive exploration approach and results of the investigation were presented in Supplemental Table 2. We also screened for relevant trials and the references of review articles.
Study eligibility and excluding criteria
Studies were included if they met the following criteria:
(1) Enrolled adult patients diagnosed with primary hyperaldosteronism using prespecified, acceptable criteria per study protocol (as listed in Table 1).
(2) Allocated patients randomly to a minimum of two intervention groups to compare the efficacy of different drugs.
(3) Reported either one of the primary outcomes (effect on blood pressure, serum potassium, or MACE) or secondary outcomes (hyperkalemia or gynecomastia).
(4) Prospective RCT.
Characteristic of enrolled studies.
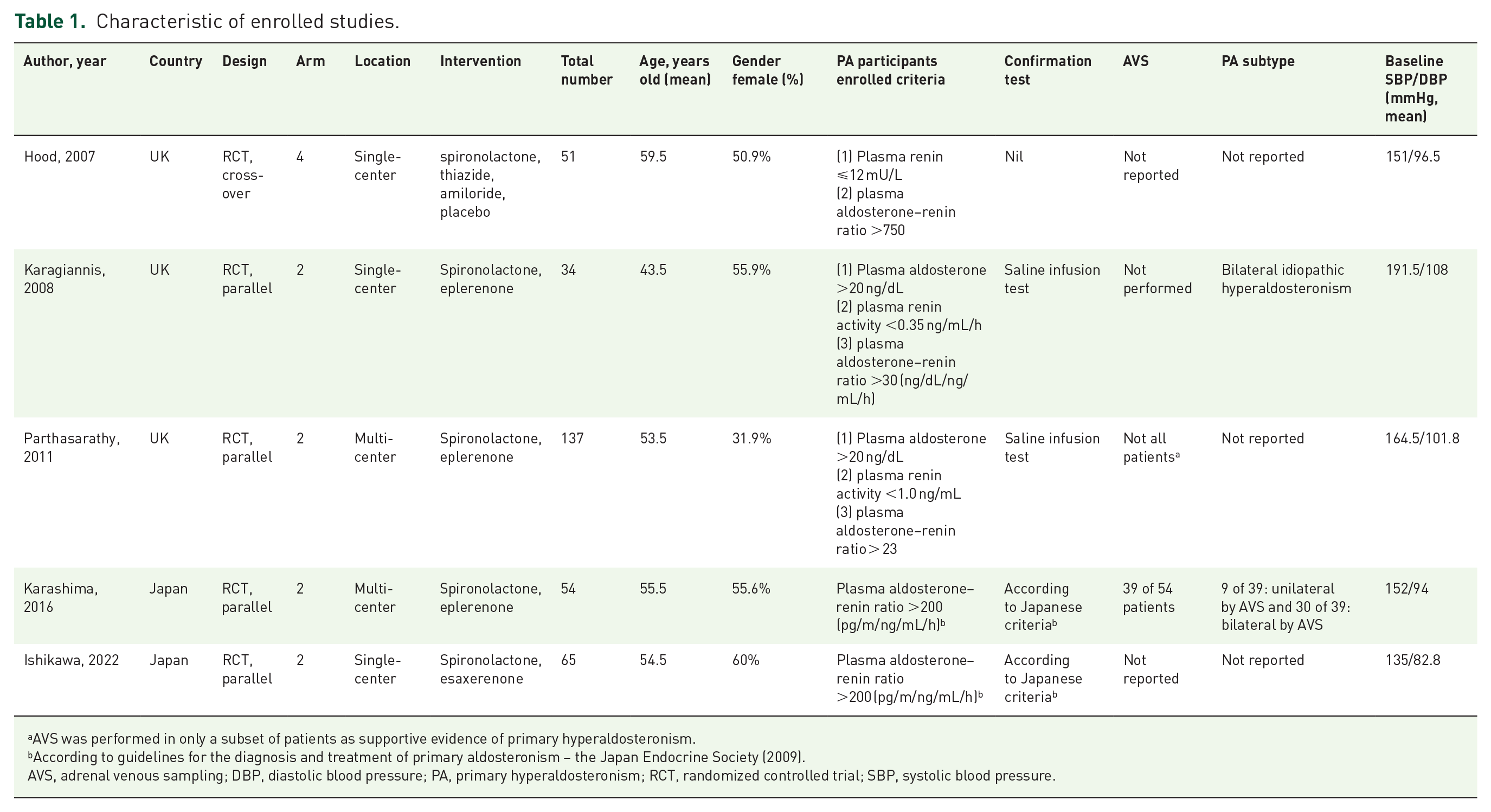
AVS was performed in only a subset of patients as supportive evidence of primary hyperaldosteronism.
According to guidelines for the diagnosis and treatment of primary aldosteronism – the Japan Endocrine Society (2009).
AVS, adrenal venous sampling; DBP, diastolic blood pressure; PA, primary hyperaldosteronism; RCT, randomized controlled trial; SBP, systolic blood pressure.
The titles and abstracts of references found during the search process were initially independently screened by two investigators (W.-Y.H. and J.-J.C.) to exclude clearly irrelevant studies. Full texts of relevant articles were obtained to determine whether the studies were eligible. A third investigator (C.-C.H.) was consulted to resolve disagreements on eligibility and categorization of studies.
Data extraction
Two investigators (W.-Y.H. and J.-J.C.) extracted relevant information from each selected study independently. Data on study characteristics, enrolled participant demographics (age and gender, country, study design, definition of primary hyperaldosteronism, and baseline blood pressure), detailed information on interventions, and outcomes of interest were extracted. A third investigator (C.-C.H.) was consulted to resolve disagreements on data extraction.
Outcome measures
The primary outcomes were the effect on systolic and diastolic blood pressure, serum potassium, and MACE. Secondary outcomes were adverse events related to treatment (hyperkalemia and gynecomastia). The definition of hyperkalemia varied slightly across three trials. Karagiannis et al. 18 defined hyperkalemia as serum potassium levels ⩾5.6 mmol/L, Parthasarathy et al. 17 considered it as >5.5 mmol/L on two occasions or >6.0 mmol/L once, while Ishikawa et al. 27 defined significant hyperkalemia as a level ⩾6.0 mmol/L.
In this study focusing on continuous outcomes, namely systolic and diastolic blood pressure, as well as serum potassium level, we extracted the mean difference (MD) and standard deviation (SD) of the MD before and after the intervention. Additionally, we identified and extracted the number of adverse events reported in the studies.
For studies19,27 that did not report the MD and SD of the MD before and after the intervention, but provided data on pre-intervention mean blood pressure, SD of pre-intervention mean blood pressure, post-intervention mean blood pressure, SD of post-intervention mean blood pressure, and the p value of comparison, we implemented a statistical approach. Specifically, we calculated the t-statistic from the given p value, enabling us to obtain the standard error of the MD before and after the intervention. This allowed us to include these studies in our analysis despite the absence of direct measurements for MD and SD of MD. 28
Statistical analysis
To assess the effects of different MRAs and ENaC inhibitors on systolic and diastolic blood pressure, as well as serum potassium levels, MD was used. Relative risk (RR) was utilized for binary outcomes such as hyperkalemia and gynecomastia. Frequentist NMA with random effect model was conducted via the netmeta package in R, version 4.0.2. We accessed the heterogeneity using the I2 statistic and summarized the result from both NMA and direct comparison in a league table. The P-score method was employed to measure the probability of an MRA or ENaC inhibitor being superior to a competing medication. Incoherence was evaluated using a design-by-treatment interaction test.29,30 A p value >0.1 indicated no concern regarding incoherence. For pairwise meta-analysis with binary outcomes, we utilized the metabin function in the R package meta (version 4.18-2).
Risk-of-bias and quality assessments
The risk of bias (RoB) was assessed using the Revised Cochrane Risk of Bias tool (RoB2). 31 Two independent reviewers (C.-C.H. and J.-J.C.) assessed the RoB, and in the case of any disagreement, a third reviewer (W.-Y.H.) was consulted to reach a decision. The certainty of evidence of the primary outcomes was assessed using the Confidence in Network Meta-Analysis (CINeMA) framework. 30 We defined a clinically significant difference in systolic and diastolic blood pressure as 10 and 5 mmHg, respectively, based on previous studies.32 –34 For the effect on potassium levels, we considered a difference of 0.5 mEq/L to be clinically significant based on traditional standards.35,36
Results
Study selection and characteristics of enrolled studies
The search process and list of excluded studies were provided (Supplemental Tables 2 and 3). After removing duplicate references, a total of 146 articles were screened based on their titles and abstracts. Among these, 22 articles were selected for full-text retrieval. Additionally, by reviewing references from meta-analyses or review articles, four more relevant references were identified. Following the application of enrollment criteria, five publications involving 392 participants were deemed eligible and included in the analysis (Figure 1).17 –19,27,37 The characteristics of the enrolled studies are summarized in Table 1. Three MRAs (spironolactone, eplerenone, and esaxerenone) and one ENaC inhibitor (amiloride) were enrolled. Four enrolled studies were two arm studies and one study (Hood 37 ) was a four-arm study (spironolactone, amiloride, thiazide, and placebo). In this four-arm study, only results from spironolactone, amiloride, and placebo treatment arms were included. The mean age ranged from 43.5 to 59.5 years old across the enrolled studies, with females accounting for 31.9–60% of participants. No study compared the effectiveness of different MRAs in terms of reducing MACE in current systematic review.

PRISMA flow diagram.
Primary outcome: blood pressure reduction and serum potassium correction
Figure 2(a) demonstrates the network plot of four potassium-sparing diuretics (spironolactone, eplerenone, esaxerenone, and amiloride), involving 392 participants from five RCTs, which compared the effectiveness of systolic blood pressure reduction. In this NMA, compared to spironolactone, eplerenone, esaxerenone, and amiloride did not show statistically significant differences in SBP reduction [Figure 2(b); I2 = 44.1%, 95% CI: 0.0–83.3%; Supplemental Table 4]. The P-score for spironolactone, amiloride, esaxerenone, and eplerenone was 0.69, 0.63, 0.51, and 0.17, respectively.
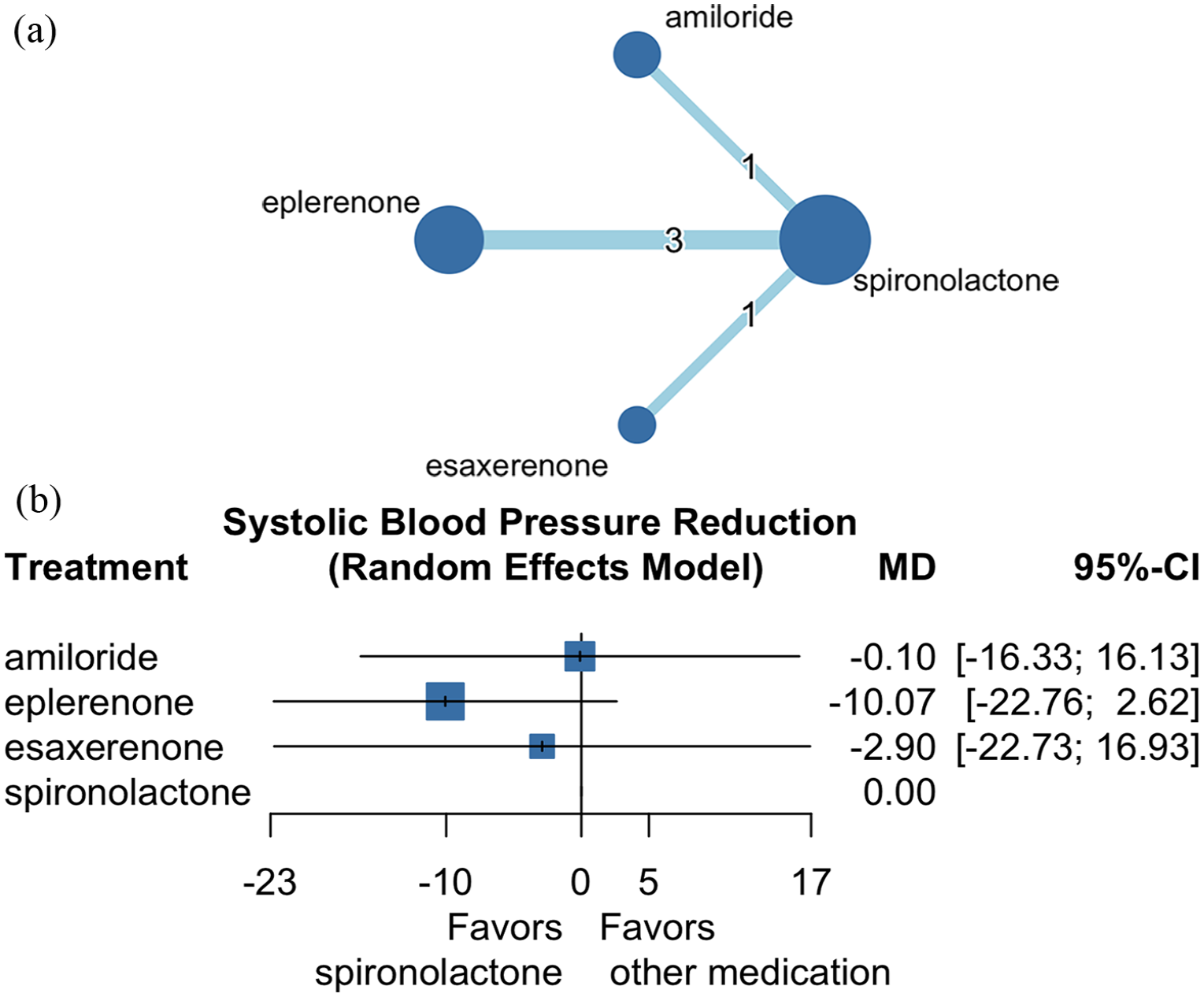
Network plot of comparisons among MRAs and ENaC inhibitors for systolic blood pressure reduction (a) and forest plot for systolic blood pressure reduction (b).
A total of four RCTs (comprising 3 MRAs and 290 participants) compared the effectiveness of reducing diastolic blood pressure (Supplemental Figure 1). In comparison with spironolactone, eplerenone exhibited a lesser effect on diastolic blood pressure reduction [Figure 3(a); −4.63 mmHg, 95% CI: −8.87 to −0.40 mmHg; Supplemental Table 5], with high heterogeneity detected (I2 = 76.1%, 95% CI: 21.3–92.7%). The P-score for esaxerenone, spironolactone, and eplerenone was 0.73, 0.70, and 0.07, respectively.

Forest plot among MRAs in diastolic blood pressure reduction (a) and serum potassium elevation (b).
Four RCTs, involving 290 participants, compared 3 MRAs for their impact on serum potassium (Supplemental Figure 2). In comparison with spironolactone, eplerenone exhibited a marginally, yet significantly, reduced effect on serum potassium elevation [Figure 3(b); −0.2 mg/dL, 95% CI: −0.37 to −0.03 mg/dL; Supplemental Table 6] with low heterogeneity (I2 = 0%, 95% CI: 0.0–89.6%). The P-score for spironolactone, eplerenone, and esaxerenone was 0.93, 0.32, and 0.25, respectively.
Secondary outcome: hyperkalemia and gynecomastia
Three RCTs, involving 236 participants, compared 3 MRAs for the risk of developing hyperkalemia (Supplemental Figure 3). In comparison with spironolactone, eplerenone and esaxerenone did not exhibit statistically significant differences in the risk of hyperkalemia [Figure 4(a); RR = 0.51, 95% CI: 0.05–4.98 and RR = 1.03, 95% CI: 0.01–113.55, respectively; Supplemental Table 7]. The P-score for eplerenone, esaxerenone, and spironolactone was 0.66, 0.44, and 0.39, respectively.
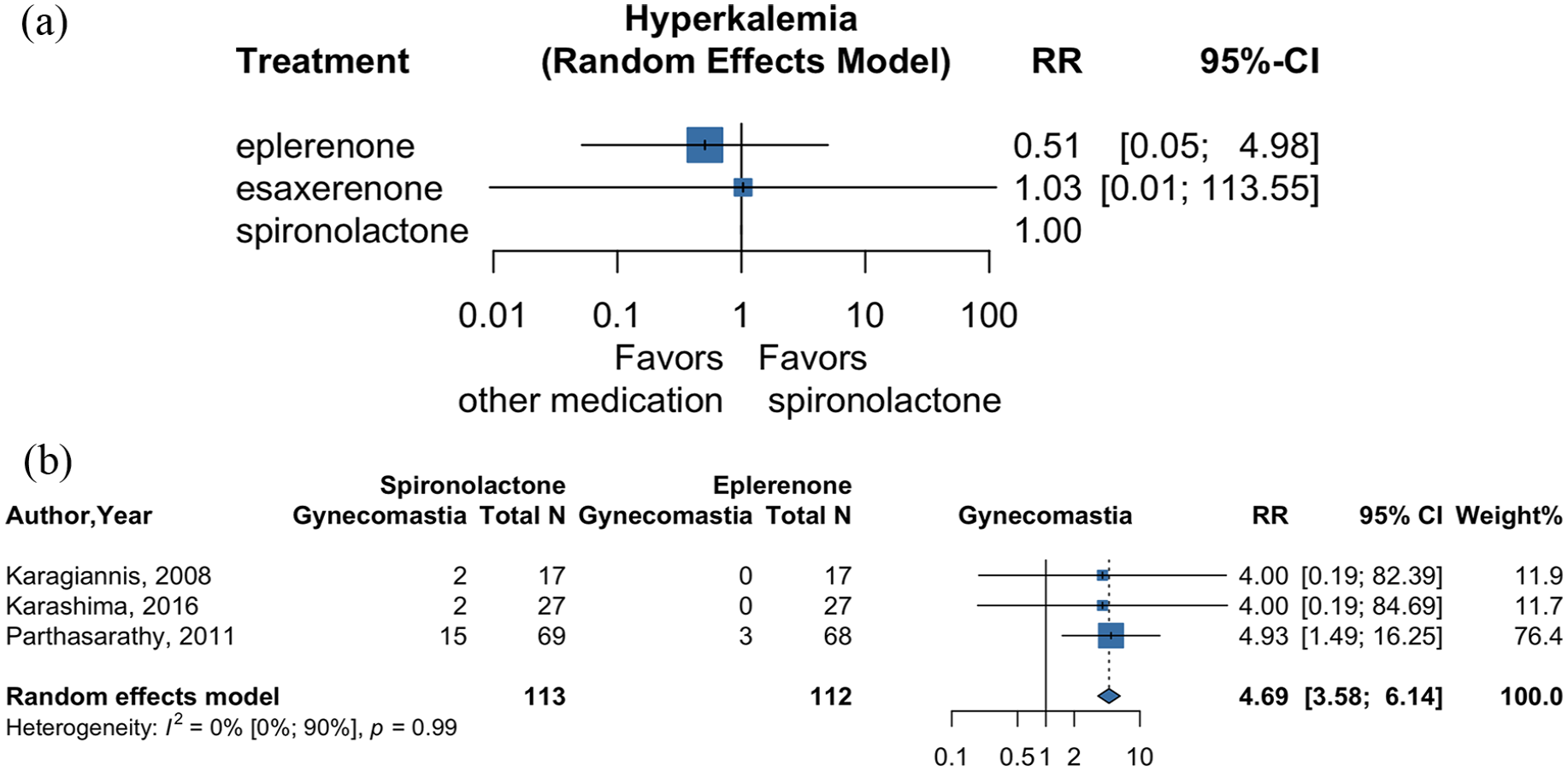
Forest plot among MRAs for hyperkalemia risk (a) and gynecomastia risk (b).
Another three RCTs, involving 225 participants, compared spironolactone and eplerenone for the risk of developing gynecomastia. Spironolactone showed a higher risk of gynecomastia compared to eplerenone [Figure 4(b); RR = 4.69, 95% CI: 3.58–6.14] with low heterogeneity (I2 = 0%, 95% CI: 0.0–89.6%).
Assessing RoB
The overall RoB assessment for the enrolled studies was summarized [Supplemental Figure 4(A) and (B)]. Two of the enrolled studies were rated as ‘some concerns’18,27 due to issues in domain 1 (randomization without concealment). Two other enrolled studies were classified as ‘high risk’17,37 because of missing outcome data. Specifically, in the study by Hood et al., 6 out of the 57 enrolled participants did not report the primary outcome of blood pressure control, while in the study by Parthasarathy et al., 40 out of the 141 enrolled participants did not report this primary outcome. One study 19 was judged as ‘low risk’ in all five domains. In the overall RoB assessment, 40% of the enrolled RCTs were categorized as having a high RoB, 40% as having some concerns, and 20% as having a low RoB. The confidence of evidence within the current network, regarding spironolactone compared to other MRAs or amiloride in terms of systolic blood pressure reduction or serum potassium elevation, was assessed using the CINeMA framework. The first domain assessed was within-study bias. After evaluating the RoB for each enrolled study, the RoB between each comparison was evaluated. The greater the contribution from a study with a high or moderate RoB, the higher the concern about the comparison. Two studies17,37 comparing the effectiveness of amiloride, spironolactone, and eplerenone were deemed to have high RoB. The comparisons involved in these two studies were categorized as ‘some concerns’ or ‘major concerns’. The second domain assessed was reporting bias. If there was concern about small study bias, reliance on primarily industry-funded trials, or potential failure to include unpublished data, the comparison was considered at risk of reporting bias. In our NMA, the second domain for all comparisons was considered to be ‘low risk’. The third domain, indirectness, evaluated the relevance of the enrolled studies to our research questions. For all comparisons in our NMA, the third domain was judged as ‘no concerns’. The imprecision domain was assessed based on whether the 95% CI of the treatment effect crossed the upper or lower limit of effect equivalence. The range of equivalence was defined by a prespecified clinically important effect size. For instance, in our study, we considered a reduction in systolic blood pressure of 10 mmHg and an elevation in serum potassium of 0.5 mEq/L as clinically important. Due to the limited number of studies included in the NMA and the wide 95% CI of the estimated treatment effect, the imprecision domain was judged as ‘some concerns’ or ‘major concerns’. The heterogeneity domain was assessed based on whether the prediction interval and 95% CI had the same direction regarding the range of equivalence. Most comparisons were ranked as ‘no concerns’ in this domain. The final incoherence domain was evaluated using the design-by-treatment interaction model and node-splitting analysis. Given the limited number of studies included and the absence of concurrent direct and indirect comparisons between different medical treatments, this domain was considered to be ‘major concerns’. Due to limited evidence with concern about imprecision, incoherence, and RoB, all comparisons were rated as having ‘very low’ confidence of evidence (Supplemental Tables 8 and 9).
Discussion
Regarding the selection between various MRAs and ENaC inhibitors in patients with primary hyperaldosteronism, our study highlighted three key findings. Firstly, spironolactone appeared to offer slightly better reduction in diastolic blood pressure compared to eplerenone, but the difference was subtle. Secondly, spironolactone exhibited slightly greater serum potassium elevation compared to eplerenone, but it may have had no clinical significance. Thirdly, no significant differences were observed among the three MRAs in the development of hyperkalemia, but spironolactone showed a greater risk of gynecomastia.
The therapeutic goals for primary hyperaldosteronism include normalization of hypertension, correction of hypokalemia, and prevention of organ damage associated with hyperaldosteronism. 38 In terms of pharmacological mechanism, MRAs block the activity of aldosterone by inhibiting its binding to the mineralocorticoid receptor, and are conventionally chosen as initial medical treatments. Spironolactone, a first-generation MRA, is non-selective and inhibits both mineralocorticoid and androgen receptors, potentially leading to side effects such as hyperkalemia and gynecomastia. Eplerenone, representing second-generation MRAs, exhibits greater selectivity for the mineralocorticoid receptor, thereby reducing the incidence of anti-androgen side effects. In recent years, third-generation MRAs, characterized by their non-steroidal nature, have been developed, offering even greater selectivity and improved tolerability profiles. Examples include esaxerenone and finerenone.39,40
Among different MRAs, spironolactone has been recommended as a first-line treatment agent in guidelines or consensus statements,1,2,15,16 largely based on a RCT conducted by Parthasarathy et al., 17 which suggested that spironolactone might be superior in blood pressure control and serum potassium correction for primary hyperaldosteronism compared with eplerenone. Nevertheless, conflicting data from Karagiannis et al. 18 demonstrated that eplerenone was comparably effective to spironolactone in controlling blood pressure in individuals with primary aldosteronism. In other words, in the absence of support from large-scale RCTs directly comparing different MRAs, the strength of evidence supporting this recommendation is weak. Moreover, our systematic review revealed a shortage of prospective trials directly comparing MRAs in relation to mortality or cardiovascular events. In summary, while current guidelines recommend spironolactone as the first-line medical treatment of primary aldosteronism, the evidence from our study, which do not meet the predefined clinically significant difference criteria, suggests nearly equivalent effects on blood pressure control and serum potassium among MRAs. Additionally, the absence of evidence regarding mortality and cardiovascular benefits leaves uncertainty regarding the superiority or inferiority of MRAs. Factors like adverse event profiles, cost-effectiveness, drug availability, and patients’ preference should guide the choice of MRAs in primary hyperaldosteronism, and we believe that further evidence is needed.
Amiloride functions as a potassium-sparing diuretic. Beyond blood pressure control, prior studies have elucidated the involvement of epithelial sodium channels (ENaCs) in vascular stiffness and aortic dysfunction associated with hyperaldosteronism, 41 as well as insulin resistance. 42 Notably, a study by Izzo et al. 43 has indicated that amiloride might offer protection against vascular fibrosis associated with hyperaldosteronism. Our study found that amiloride has similar effects on systolic blood pressure control compared to spironolactone. Therefore, using amiloride for blood pressure control in patients with primary hyperaldosteronism who have contraindications or intolerance to MRAs may be reasonable; however, the comparability of the organ-protective effects of the two kinds of drugs remains uncertain. Due to limited evidence, further investigation is warranted to determine the use of amiloride as an alternative choice to MRAs.
Our study had several strengths. Firstly, we conducted a NMA to systematically compare the blood pressure effect between MRAs of three different generations and one ENAC inhibitor in patients with primary hyperaldosteronism. Secondly, we assessed the effect on serum potassium levels between MRAs. Thirdly, we conducted a comparative analysis of the adverse effects associated with MRAs.
However, there are also limitations to consider. Firstly, our analysis was constrained by a limited number of studies. Consequently, the findings should be interpreted with caution, particularly given the major concerns identified in the domains of imprecision and incoherence. Secondly, the studies enrolled were conducted in the United Kingdom and Japan, and the inherent differences between populations might have potential implications on the generalizability of our findings. Thirdly, the definitions of hyperkalemia and diagnostic criteria for primary hyperaldosteronism varied slightly among the included studies, potentially introducing bias into the results. Fourthly, while being among the medical treatment options for primary hyperaldosteronism, the lack of evidence on ENaC inhibitors also served as a research limit. Lastly, there was currently a lack of RCTs comparing different medical treatments in patients with primary hyperaldosteronism regarding mortality and cardiovascular events, highlighting the necessity for further RCTs.
Conclusion
The present NMA suggests that eplerenone have slightly less impact on reducing blood pressure and correcting serum potassium levels compared to spironolactone, but the differences are marginal. Furthermore, there is no significant difference in the risk of hyperkalemia among different MRAs, but spironolactone carries a higher risk of gynecomastia. Given these findings and the lack of evidence regarding the mortality and cardiovascular benefits of different MRAs, there is no conclusive evidence regarding the superiority or inferiority of MRAs. It is crucial to evaluate the impact of various medical treatments on hard outcomes, and future comparative studies concerning cardiovascular outcomes could be conducted, potentially utilizing a target trial emulation design based on real-world evidence from a claims database.
Supplemental Material
sj-docx-1-taj-10.1177_20406223241239775 – Supplemental material for Comparison of different medical treatments for primary hyperaldosteronism: a systematic review and network meta-analysis
Supplemental material, sj-docx-1-taj-10.1177_20406223241239775 for Comparison of different medical treatments for primary hyperaldosteronism: a systematic review and network meta-analysis by Wen-Yu Ho, Ching-Chung Hsiao, Ping-Hsun Wu, Jui-Yi Chen, Yu-Kang Tu, Vin-Cent Wu and Jia-Jin Chen in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
