Abstract
Introduction:
The pathogenesis of post-COVID interstitial lung disease, marked by lung tissue scarring and functional decline, remains largely unknown.
Objectives:
We aimed to elucidate the temporal cytokine/chemokine changes in bronchoalveolar lavage (BAL) from patients with post-COVID interstitial lung disease to uncover potential immune drivers of pulmonary complications.
Design:
We evaluated 16 females diagnosed with post-COVID interstitial lung disease, originating from moderate to severe cases during the second epidemic wave in the Autumn of 2020, treated at the Pneumology Department of the Arad County Clinical Hospital, Romania. Their inflammatory response over time was compared to a control group.
Methods:
A total of 48 BAL samples were collected over three intervals (1, 3, and 6 months) and underwent cytology, gene, and protein expression analyses for pro/anti-inflammatory lung cytokines and chemokines using reverse transcription polymerase chain reaction and enzyme-linked immunosorbent assay.
Results:
One month after infection, there were significant increases in the levels of IL-6 and IL-8. These levels decreased gradually over the course of 6 months but were still higher than those seen in control. Interferon-gamma and tumor necrosis factor alpha exhibited similar patterns. Persistent elevations were found in IL-10, IL-13, and pro-fibrotic M2 macrophages’ chemokines (CCL13 and CCL18) for 6 months. Furthermore, pronounced neutrophilia was observed at 1 month post-COVID, highlighting persistent inflammation and lung damage. Neutrophil efferocytosis, aiding inflammation resolution and tissue repair, was evident at the 1-month time interval. A notable time-dependent reduction in CD28 was also noticed.
Conclusion:
Our research provides insight into the immunological processes that may lead to the fibrotic changes noted in the lungs following COVID-19.
Plain language summary
The objective of this pilot study was to investigate changes in lung cytokine pro- and anti-inflammatory profiles among patients with interstitial lung disease after COVID-19 infection. Background: Post-COVID lung disease represents a significant health concern that demands comprehensive research. The pathogenesis of post-COVID interstitial lung disease, marked by lung tissue scarring and functional decline, remains largely unknown. Methods: We evaluated 16 females diagnosed with post-COVID interstitial lung disease, originating from moderate to severe cases during the second epidemic wave in the Autumn 2020, treated at the Pneumology Department of the Arad County Clinical Hospital, Romania. Their inflammatory response over time was compared to a control group. A total of 48 BAL samples were collected over three intervals (1, 3, and 6 months) and underwent cytology, gene, and protein expression analyses for pro/anti-inflammatory lung cytokines and chemokines using RT-PCR and ELISA The interrelationships between the expression levels of various pro-inflammatory and anti-inflammatory cytokines and chemokines by Pearson’s correlations was investigated. Results: One month after infection, there were significant increases in the levels of IL-6 and IL-8. These levels decreased gradually over the course of six months but were still higher than those seen in control. IFN-γ and TNF-α exhibited similar patterns. Persistent elevations were found in IL-10, IL-13, and pro-fibrotic M2 macrophages’ chemokines (CCL13 and CCL18) for six months. Pronounced neutrophilia was observed at 1 month post-COVID, highlighting persistent inflammation and lung damage. Neutrophils efferocytosis, aiding inflammation resolution and tissue repair, was evident at the 1-month time-interval. A notable time-dependent reduction in CD28 was also noticed. Conclusions: Our research provides insight into the immunological processes that may lead to the fibrotic changes noted in the lungs following COVID-19.
Keywords
Introduction
Post-COVID lung disease represents a significant health concern that demands comprehensive research. Although much remains to be understood about the long-term consequences of COVID-19, there have been documented cases of individuals developing a condition called post-COVID lung disease. This condition is characterized by the presence of lung tissue scarring and thickening, leading to a decline in lung function commonly referred to as ‘COVID lung fibrosis’.1,2 The underlying mechanisms responsible for the development of COVID lung fibrosis are not yet fully understood, but it is believed to involve a dysregulated immune response to the virus, resulting in chronic inflammation and tissue damage. 3
The pathophysiology of COVID lung fibrosis remains largely unknown, but insights have been gained from studying a humanized model of long COVID using single-cell transcriptomics of lung tissue. This research revealed elevated expression of CD47, IL-6, and phospho-JUN (pJUN), which correlated with disease severity and the presence of pathogenic fibroblast populations. In addition, a combined approach targeting both inflammation and fibrosis not only improved fibrotic conditions but also restored innate immune equilibrium. These findings hold potential implications for the clinical management of COVID lung fibrosis in patients. 2
Limited research has been conducted on the long-term symptoms that can persist for several weeks or even months. A nationwide clinical study conducted in the UK, involving 3290 individuals, revealed that 40% of patients experienced 5–6 ongoing symptoms following a COVID-19 infection, including breathing difficulties. 4 Similarly, another study involving 2113 patients who did not require intensive care unit admission reported fatigue and dyspnea as prominent symptoms persisting after a period of 3 months. 5 In another clinical study involving 1142 patients, it was found that 61% experienced fatigue and 55% suffered from dyspnea during physical activity at the 7-month mark following the infection. 6 In addition, 23.5% of patients experienced dyspnea even at rest. However, despite these clinical observations, there is currently a dearth of results regarding the molecular immune pathways that drive the development of post-COVID fibrosis or diffuse parenchymal lung disease, which ultimately contribute to the symptoms evaluated in multicenter clinical studies.
To address this knowledge gap, we have designed a pilot study aiming to assess the temporal changes in the expression profiles of pro-inflammatory versus anti-inflammatory cytokine/chemokines gene and protein levels, as well as cytopathological analysis in bronchoalveolar lavage fluid (BALF) obtained from patients clinically diagnosed with post-COVID interstitial lung disease. The study will investigate three time points (1, 3, and 6 months after viral exposure) and compare the results with those of healthy volunteers. By examining these cytokine/chemokines profiles, the study hopes to gain insights into the underlying immune mechanisms involved in post-COVID lung complications.
Materials and methods
Patients
In all, 48 bronchoalveolar lavage (BAL) fluid samples were collected from 16 females with a mean age of 64.8 ± 8.9 years, diagnosed with post-COVID interstitial lung disease, originating from moderate to severe cases during the second epidemic wave in the Autumn of 2020, treated at the Pneumology Department of the Arad County Clinical Hospital, Romania. For a comprehensive understanding, samples were collected at 1, 3, and 6 months post-infection. By contrast, 10 BAL fluid samples from healthy females (mean age: 47.8 ± 15.2 years) were included as controls. Prior to the study, neither the patients nor the controls presented interstitial lung disease. Furthermore, any individuals with a history of smoking were not included in either group for this study. To delineate the temporal evolution of cytokines post SARS-CoV-2 infection, the same patients were sampled at intervals: 1, 3, and 6 months. To eliminate the possibility of undetected asymptomatic COVID-19 infections among the controls, the presence of antibodies to the SARS-CoV-2 nucleocapsid protein was assessed.
Eligibility for inclusion was extended to patients diagnosed with restrictive ventilatory failure 1 month post-infection, characterized by forced vital capacity measurements that were between 60% and 70% of the predicted values. Persistent symptoms, such as notable asthenia, dyspnea, dry cough, hair loss, and memory anomalies, were reported by patients across the study duration. For diagnostic clarity, tri-monthly chest CT scans were conducted. Abnormalities, including ground glass opacities, reticulofibrillary lesions, and bronchiectasis, persisted even half a year post-infection, as confirmed by radiological evaluation.
Supplementary health data, like comorbidities and functional metrics, were collected retrospectively into a digital database. All participants provided written informed consent, and the study was authorized by the Ethics Committee of the Emergency Clinical County Hospital in Arad (Approval no. 19471/26.05.2022).
Bronchoalveolar lavage
BAL was performed on patients after obtaining their informed consent, following previously described protocols. BAL fluid (BALF) was collected using a flexible bronchoscope (Olympus, Tokyo, Japan) by instilling 100 ml of saline solution and collecting 80 ml. To remove mucus from the lavage fluid, the samples were strained through double-layered sterile gauze pads into sterile conical tubes. A portion of the collected BALF was used on the same day for cell counts and cytological examination. The remaining BAL cells were aliquoted and stored at −80°C in 15 ml conical tubes for subsequent reverse transcription polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA) analysis.
BAL cytology
For cytological examination, the cellular components were separated by centrifugation at 1000 rpm for 5 min. The cells were then washed twice with phosphate-buffered saline (PBS) and resuspended in PBS at a concentration of 2 × 106 cells/ml. Slides were prepared by centrifuging 50 µl of the prepared cell suspension (containing 100,000 cells/slide) using a cytospin apparatus (Hettich, Tuttlingen, Germany) at 1000 rpm for 10 min. The cytospin slides containing the BAL cells were stained with May-Grünwald-Giemsa (MGG; Merck, Darmstadt, Germany) for cell differentiation. At least 500 cells were counted.
The presence of the iron-laden macrophages in lung tissue was assessed using the Perls Prussian blue stain, as follows. After centrifugation (Cytospin; Shandon Southern Instruments, Runcorn, UK) for 10 min at 2000 rotations per minute (rpm), we obtained a cell pellet. After the slides were air-dried, they were incubated for 10 min in a stain that contains potassium ferricyanide and hydrochloric acid, then counterstained with eosin and hematoxylin Mayer. At magnification of 400×, 100–300 alveolar macrophages (depending on the quality of the slide) were examined for the number of cells that stained with Perls Prussian blue stain, and a percentage score was established by dividing the number of Prussian-blue-positive cells by the total number of macrophages counted (hemosiderin-laden macrophage index).7,8
To obtain the Golde score, each macrophage was scored for hemosiderin content using the grading system described in Table 1. The cumulative score obtained was extrapolated to represent the average across a sample size of 100 cells.7,9,10
Golde score description and classification of the iron-laden macrophages found in BAL fluid obtained from patients who underwent COVID-19.

RT-PCR
The BAL cell samples taken from the patients were stored at −80°C until the analysis time. Total RNA extraction was performed using the Direct-zol RNA MiniPrep Plus kit (Cat. No.: R2072) manufactured by Zymo Research according to the manufacturer’s instructions. The quantity and quality of total extracted and purified RNA were assessed using the NanoDrop One spectrophotometer. Then, the RNA was reverse transcribed into cDNA required for the subsequent studies using First-Strand cDNA Synthesis Kit (Cat. No.: K1612) manufactured by Thermoscientific according to the manufacturer’s instructions, in a thermal cycler (Biometra TAdvanced 96SG, Analytik Jena, Jena, Germany). Real-time PCR was performed using the QuantiNova SYBR Green PCR kit (cat. no. 208054, Qiagen, Hilden, Germany) according to the manufacturer’s instructions on the Rotor-Gene Q real-time PCR system; all samples were run in triplicate. The sequence of the primers used is given in Table 2.
RT-PCR primers.

Cytokine and chemokine assays
IL-6 (EIAab/E0079h), IL-8 (EIAab/E0080h), IL-13 (EIAab/E0060h), CCL2 (EIAab/E0087h), CCL3 (EIAab/E0092h), CCL13 (EIAab/E0216h), and CCL18 levels EIAab/E0211h were measured by an ELISA from non-concentrated BAL fluids using kits according to the manufacturer’s instructions (Wuhan EIAab Science CO., LTD, Wuhan, China). These proteins were analyzed on a Tecan Infinite F200.
Statistical analysis
Results are expressed as a mean ± SD. One-way analysis of variance followed by Tukey’s multiple comparisons test was used to assess statistical significance using GraphPad Prism 6.0 (Boston, US). The correlation between pairs of variables was evaluated by Pearson’s correlation analysis and expressed as Pearson’s coefficient (r). p Values < 0.05 was considered statistically significant.
Results
Cytological examination
At the 1-month mark post-COVID-19 infection, a substantial quantity of mucus was found in the BAL fluid in a significant majority of the studied cases (64%). Over time, the amount of mucus content showed a consistent decrease. BAL cellularity during this phase was observed to range from 600 to 700 cells/ml. A detailed differential count of the cell types is found in Table 3. One month after infection, macrophages were the most abundant cells in the BAL fluid, followed in order by neutrophils, lymphocytes, and a small number of eosinophils. By the 6-month stage, a marked shift occurred in the BAL cell composition, with macrophages becoming the predominant cell type, demonstrating a significant increase when compared to the counts at the 1-month and 3-month intervals (p < 0.01). As a particularity, 1 month after the infection, many macrophages in the BAL were highly vacuolated, a phenotype absent in the controls [Figure 1(A, e)]. By the third month, there was a decrease in the number of macrophages exhibiting extensive vacuolation, and by the sixth month, macrophages displayed a less conspicuous vacuolated profile. Neutrophils followed a similar pattern [Figure 1 (A, f)]. The efferocytosis of apoptotic neutrophils was a common feature observed in patients’ slides a month following the viral infection [Figure 1(B)].
Cytological characteristics are reported as percentages of inflammatory cell composition by group.

p < 0.01 versus controls. ^^p < 0.01 versus 3 months. °°p < 0.01 versus 6 months.
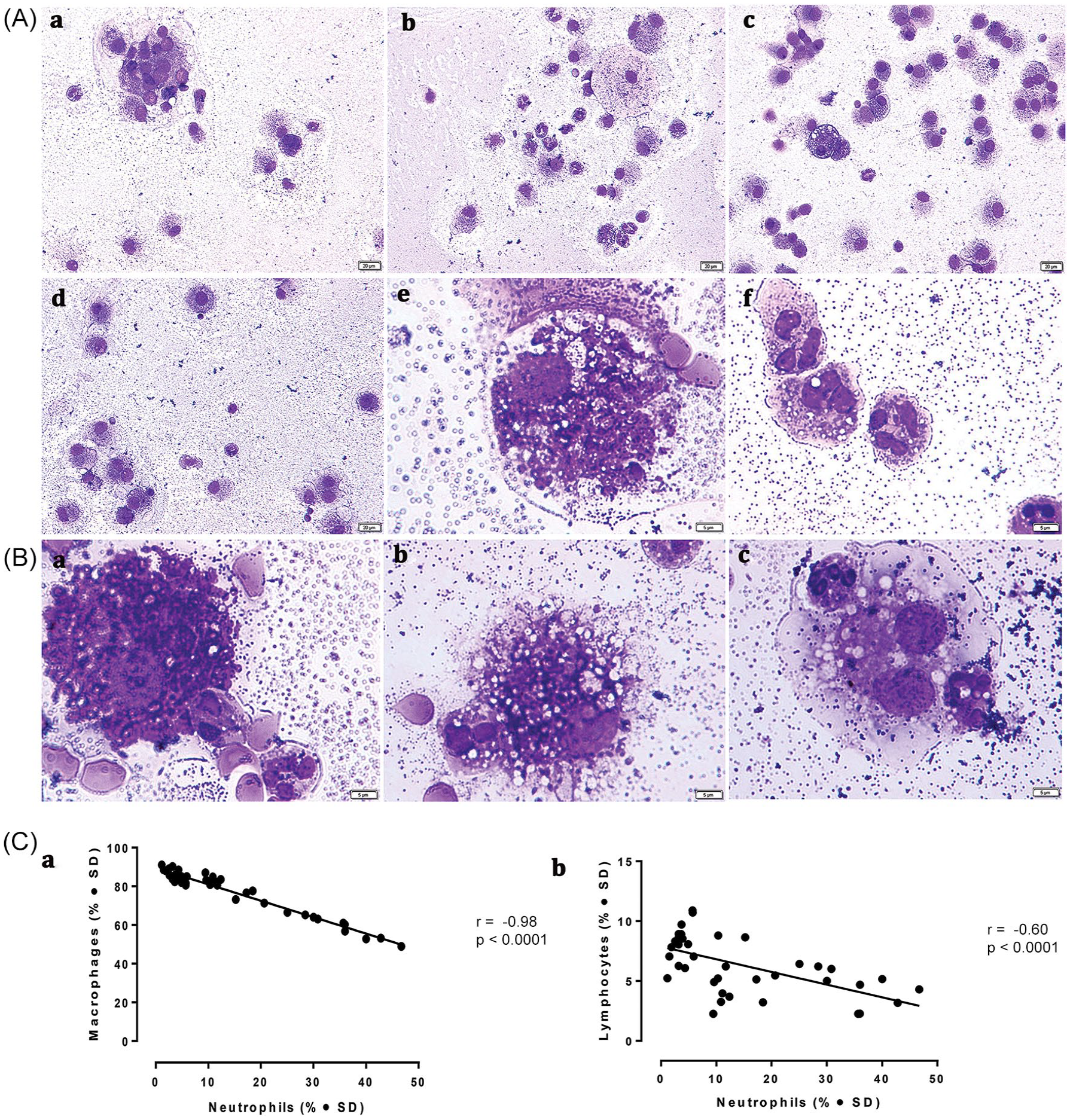
BAL cytology. (A) Overview of cells found in a BAL sample from (a) control (×20); COVID-19 patient with interstitial lung disease at (b) 1 month post-infection (x20); (c) 3 months post-infection (×20); (d) 6 months post-infection (×20); (e) foamy alveolar macrophages containing vesicles of varying size and content (immersion ob.); (f) vacuolated neutrophils, (B) Efferocytosis. Several macrophages engulfing apoptotic neutrophils in BAL fluid of COVID-19 patients with interstitial lung disease at 1 month post-infection (immersion ob.). May-Grünwald-Giemsa stain. (C) Negative Pearson’s correlations between macrophages and neutrophils in BAL (a), respectively lymphocytes and neutrophils (b). The r coefficient of correlation (from Pearson’s correlation of determination) and the respective p values are shown.
The assessment of the inflammatory composition of BAL samples from COVID-19 patients at three separate time points revealed noteworthy observations. There was a significantly negative correlation between the quantities of macrophages and neutrophils (r = −0.98; p = <0.0001), suggesting that the presence of these cell types is inversely related [Figure 1(C, a)]. In addition, a moderate negative correlation was detected between lymphocytes and neutrophils (r = −0.60; p < 0.001), indicating a somewhat weaker but still notable association between these cell types [Figure 1(C, b)].
The hemosiderin content within alveolar macrophages was evaluated by considering the total cell count and classifying the type of hemosiderin using a specific iron stain (e.g. Perls’ Prussian blue) [Figure 2(A)] and the Golde Score [Figure 2(B)], as per the grading system described in Table 1. It was found a negative correlation between the percentage of hemosiderin-bearing macrophages and the overall macrophage percentage in the BAL fluid (r = −0.74; p < 0.0001 Pearson’s correlation coefficient) [Figure 2(C, a)]. On the other hand, there was a strong positive correlation between the percentage of hemosiderin-containing macrophages and the percentage of neutrophils in the BAL fluid (r = 0.78; p < 0.0001 Pearson’s correlation coefficient) [Figure 2(C, b)].
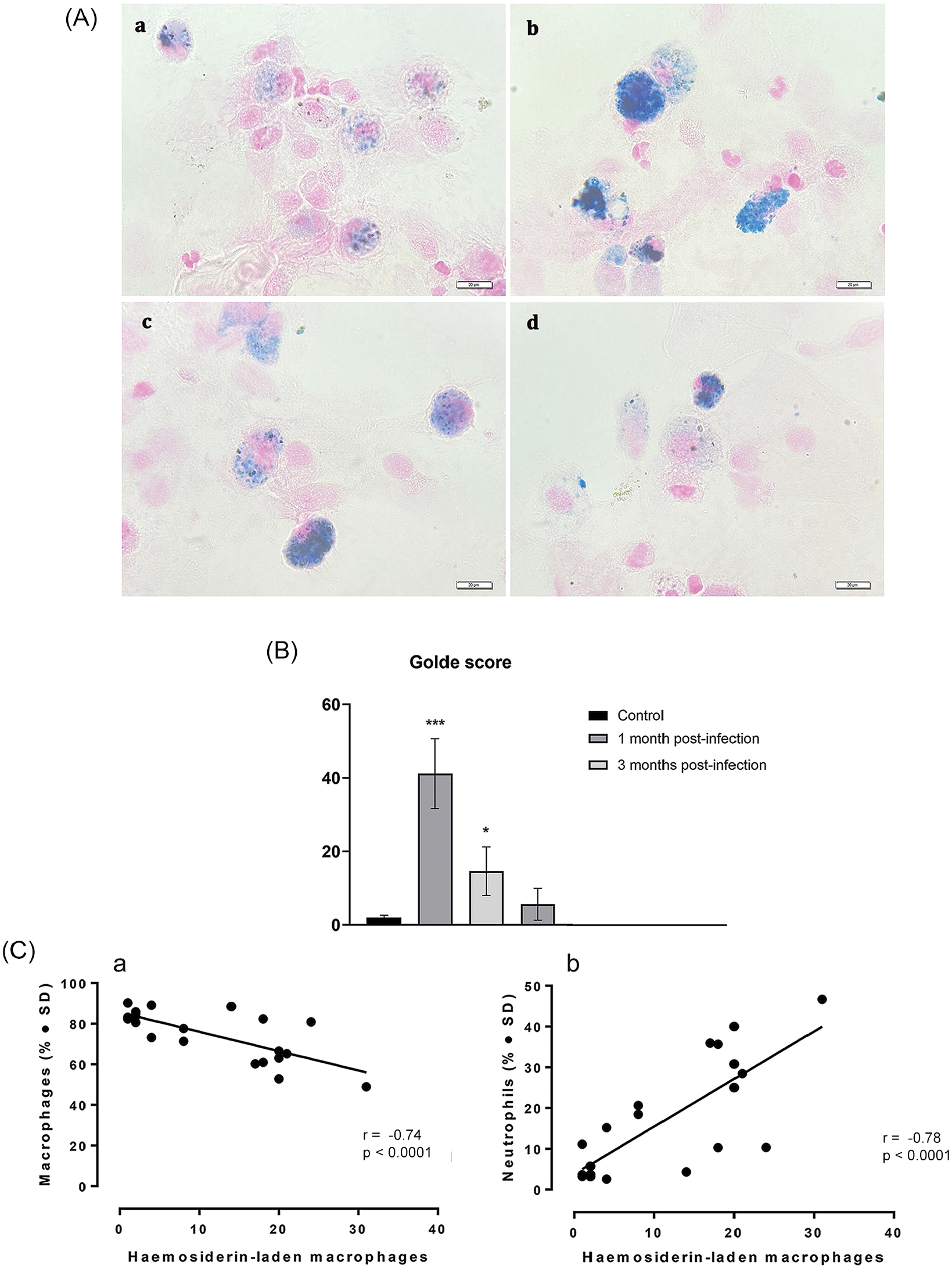
Perls’ Prussian blue stains the iron-laden macrophages. (A) Overview of iron-laden macrophages cells found in a BAL sample from (a) Control; (b) COVID-19 patient with interstitial lung disease at 1 month post-infection; (c) COVID-19 patient with interstitial lung disease at 3 months post-infection; (d) COVID-19 patient with interstitial lung disease at 6 months post-infection, (B) Golde Score, (C) positive Pearson’s correlations between percentage of neutrophils and percentage of hemosiderin-loaded macrophages in BAL, and (D) negative Pearson’s correlations between total percentage of macrophages and percentage of hemosiderin-loaded macrophages in BAL; The r coefficient of correlation (from Pearson’s correlation of determination) and the respective p values are shown.
Cytokine and chemokine profiles
The gene expression levels of pro-inflammatory cytokines such as IL-6, IL-8, tumor necrosis factor alpha (TNF-α), and interferon-gamma (INF-γ) significantly diminished at the 6-month time interval compared to 1 month after COVID-19 infection (Figure 3). However, the expression of chemokines CCL2 and CCL3 displayed a rising trend over the same period (Figure 3). Similarly, protein levels of IL-6 and IL-8, which are known to be involved in the inflammation process, showed a significant reduction over time. By contrast, anti-inflammatory cytokines/chemokines, including IL-13, CCL2, CCL3, CCL13, and CCL18, exhibited an increasing trend over the same time period (Figure 4). This indicates a potential shift from a pro-inflammatory to an anti-inflammatory state in the lung tissue of post-COVID patients as recovery progresses.

RT-PCR analysis of (a) IL-6, (b) IL-8, (c) TNF-α, (d) INF-γ, (e) IL-10, (f) IL-13, (g) CCL2, and (h) CCL3 gene expression. BAL samples were collected from 16 females diagnosed with COVID at 1, 3, and 6 months post-infection (N = 16 for each time point, for a total of 48 samples). Control BAL samples from 10 healthy females.

Analysis of (a) IL-6 (pg/mL), (b) IL-8 (pg/mL), (c) IL-13 (pg/mL), (d) CCl2 (pg/mL), (e) CCL3 (pg/mL), (f) IL-13 (pg/mL), and (g) CCL2 (ng/mL) protein levels. BAL samples were collected from 16 females diagnosed with COVID at 1, 3, and 6 months post-infection (N = 16 for each time point, for a total of 48 samples). Control BAL samples from 10 healthy females.
Investigating the interrelationships between the expression levels of various pro-inflammatory and anti-inflammatory cytokines and chemokines, a positive Pearson’s correlation coefficient within each group was found (Figures 5 and 6). Interestingly, a negative correlation observed between INF-γ (a pro-inflammatory cytokine) and IL-10 (an anti-inflammatory cytokine) in terms of their mRNA expression (r = −0.32; p < 0.05) was evident [Figure 6(g)].
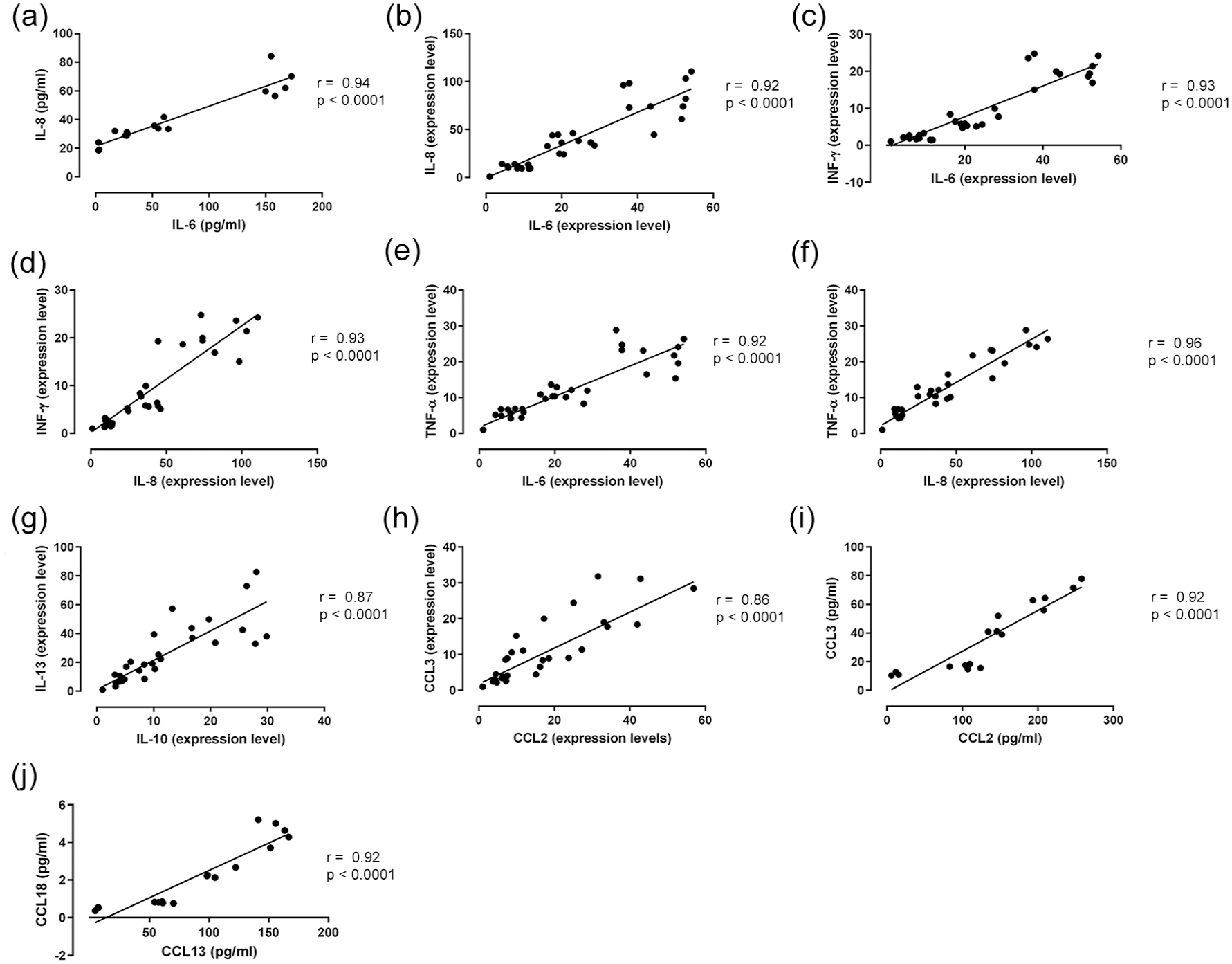
Pearson’s correlation analysis of (a) IL-6 and IL-8 (pg/mL), (b) IL-6 and IL-8 (expression level), (c) INF-γ and IL-6 (expression level), (d) INF-γ and IL-8 (expression level), (e) TNF-α and IL-6 (expression level), (f) TNF-α and IL-8 (expression level), (g) IL-10 and IL-13 (expression level), (h) CCL3 and CCL2 (expression level), (i) CCL3 and CCL2 (pg/mL), and (j) CCL18 and CCL13 (expression level).
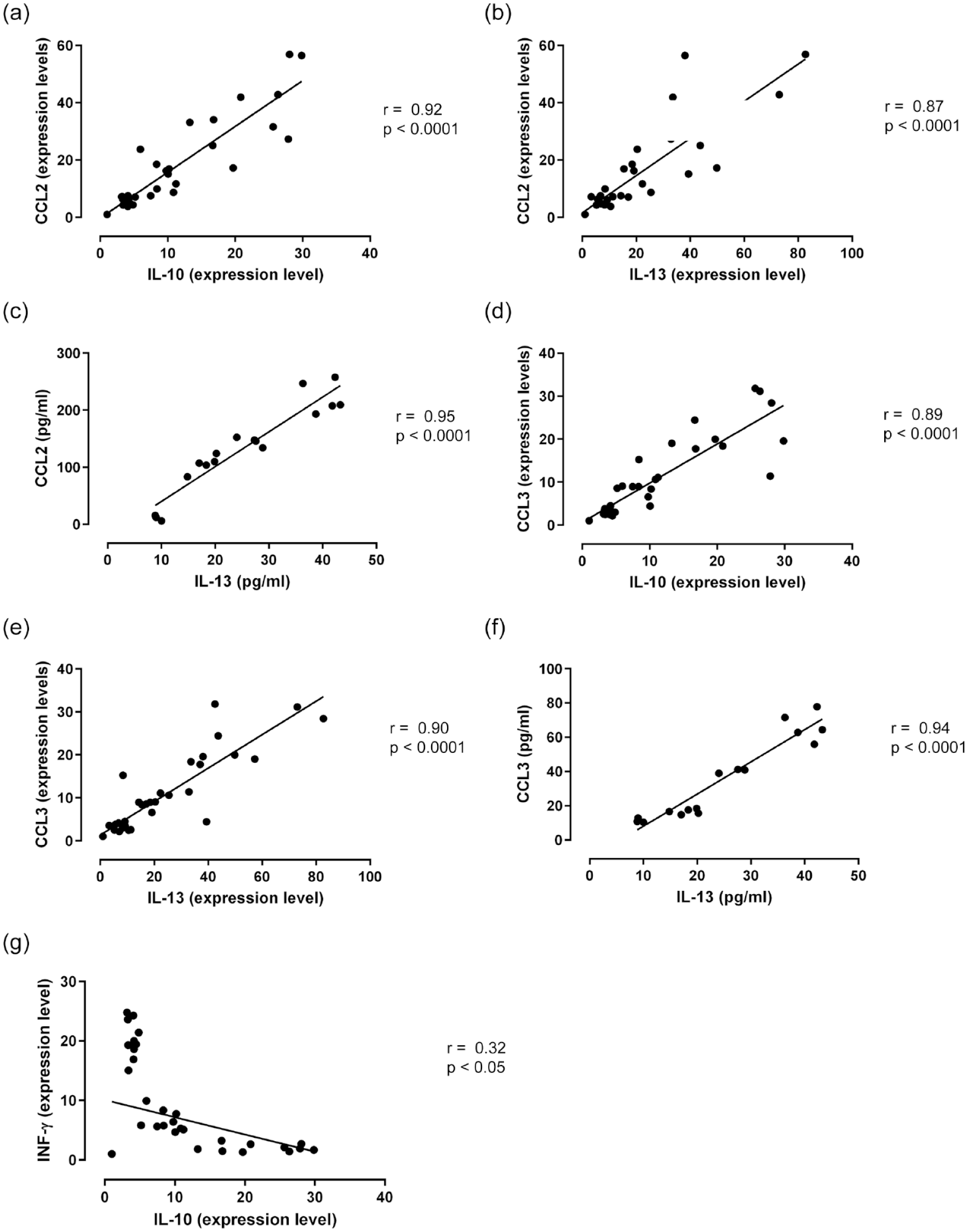
Pearson’s correlation analysis of (a) CCL2 and IL-10 (expression level), (b) CCL2 and IL-13 (expression level), (c) CCL2 and IL-13 (pg/mL), (d) CCL3 and IL-10 (expression level), (e) CCL3 and IL13 (expression level), (f) CCL3 and IL-13 (pg/mL), and (g) INF-γ and IL-10 (expression level).
Prognostic marker of disease evolution
CD28, a poor outcome predictor of interstitial pulmonary fibrosis, showed a notable upregulation in BAL 1 month after infection, subsequently dropping and being significantly downregulated at the 6-month point (Figure 7).

Time-dependent mRNA CD28 profile of the moderate to severe COVID-19 patients with interstitial lung disease.
Discussion
This pilot study aimed to evaluate the time-based alterations in the profiles of gene and protein levels of pro-inflammatory and anti-inflammatory cytokines/chemokines in the BAL of patients with interstitial lung disease, particularly those who had experienced moderate to severe COVID-19. By examining three specific time points (1, 3, and 6 months after viral exposure) and comparing the results to those obtained from healthy volunteers, the study aimed to uncover insights into the immune mechanisms underlying post-COVID lung complications.
The cytological examination of BAL samples from the enrolled patients highlighted significant neutrophilia (>3% neutrophils) 8 even at the 6-month mark post-infection, which can be a sign of sustained inflammation and lung injury. Notably, an inverse correlation was observed between both macrophages and lymphocytes with neutrophils, hinting at the simultaneous occurrence of ongoing inflammation and the initiation of pro-healing processes. In addition, an intriguing pattern was observed with hemosiderin-laden macrophages: there was a negative correlation with the overall percentage of macrophages in the BAL and a positive correlation with the percentage of neutrophils. This could be indicative of a shift in the lung’s cellular environment following the viral infection, characterized by a prolonged inflammatory response potentially leading to tissue damage and subsequent hemorrhage. 8 The observed variation in the macrophage populations might also reflect alterations in immune response dynamics during the recovery phase. Notably, 1-month post-infection, we documented efferocytosis of neutrophils, a process that helps expedite the resolution of pulmonary inflammation and promotes healing. 11
In the immunological investigation, one of the primary objectives was to analyze BAL samples from study participants, with a particular focus on the gene expression of IL-6 and IL-8. Prior research has underscored the critical role of IL-6 concentration in the bloodstream of patients in intensive care, rendering it a significant parameter. 12 Monitoring systemic levels of IL-6 may serve as a parameter for monitoring the cellular response to SARS-CoV-2. 13 Similarly, serum IL-8 has been identified as a biomarker for predicting the prognosis of COVID-19 patients, with IL-8 levels showing a better correlation than IL-6 levels with overall clinical disease scores at different stages of the disease. 14 The findings demonstrated that the mRNA expression of both IL-6 and IL-8 in BAL cells samples continued to be significantly upregulated 1 month post-SARS-CoV-2 infection in comparison to control. However, their expression gradually downregulated during the subsequent post-infection stages. Intriguingly, the comparison of mRNA expressions of these cytokines in post-COVID-19 patients and controls revealed a marked upregulation of both IL-6 and IL-8 at the 6-month post-infection mark – a 4-fold and 11-fold increase, respectively – indicating an enduring pro-inflammatory cytokine profile. 15 This pattern was echoed in the protein profile of these cytokines. This persistent inflammatory response is likely driven, at least in part, by the activities of M1 macrophages and virus-activated T cells, which are recognized as the chief sources of IL-6 secretion. 16 In addition, we noted upregulation of CCL3 in BAL samples from patients at 6 months post-infection, which displayed higher expression levels than samples from 3 months post-infection and controls. This suggests that the recruitment and activation of monocytes/macrophages could be contributing to the perpetuation of the inflammatory state. 17
To gain more insight into the inflammatory response, a thorough correlation statistical analysis was conducted. The results showed a robust positive correlation between the gene and protein expression of IL-6 and IL-8. A negative correlation between these pro-inflammatory cytokines and the anti-inflammatory cytokines IL-10 and IL-13 was not observed, nor with the chemokines associated with the M2 macrophage phenotype (data not shown), which are known to play a crucial role in long-healing processes. Such observations suggest that even 6 months post-infection, the inflammatory mechanisms continue to be considerably active. The signals that trigger and facilitate healing mechanisms do not seem to exceed the inflammatory intensity, which implies a persistent state of inflammation within the lungs. This enduring inflammation could potentially foster continuous pro-fibrogenic signaling and drive the advancement of post-inflammatory pulmonary fibrosis.18,19
T-cell immunity plays a vital role in the host’s defense against SARS-CoV-2. Specifically, CD4+ T-cell responses targeting the Spike protein of the virus have been observed in 100% of post-COVID patients. 20 Autopsies of severe COVID-19 patients have revealed impaired formation of follicular germinal centers due to defective T follicular helper cell responses. 21 Moreover, memory T-cell responses can persist in patients exposed to the closely related SARS-CoV-1 for up to 4 years after the initial infection. 22 In the context of post-COVID interstitial lung disease, changes in the cellular and molecular lung environment play a crucial role in fibrogenesis. Specific subtypes of T cells may either promote or reverse the progression of these mechanisms. 23 Th1 cells, Th2 cells, Th17 cells, and regulatory T cells have been detected in BAL samples from idiopathic pulmonary fibrosis patients, and they have been shown to have an impact on the pathogenesis of the disease.24,25 In this study, we noted an increase in IFN-γ and TNF-α levels at the 3-month interval following COVID, relative to the control group. Although direct profiling of T cells was not conducted, this increase may imply a significant presence of the Th1 profile in BAL during this period. This upregulation gradually decreased during the 6 months, but remained twofold, respectively, fourfold higher than in healthy individuals. This observation may be correlated with a decrease in the pro-inflammatory Th1 population and M1 macrophages, as well as the levels of IFN-γ in the BAL fluid or circulation of patients with interstitial lung disease. 26 A notable negative correlation between IFN-γ and the anti-inflammatory cytokine IL-10 was noticed. The decrease in IFN-γ mRNA expression after the resolution of COVID-19 infection indicates the cessation of its activation as a viral response 27 and may be suggestive of the activation of the healing mechanisms.
Persistent activation of macrophages during tissue repair after infection can lead to fibrosis, characterized by excessive accumulation of extracellular matrix components.19,28 In this context, M2 macrophages, which are involved in wound healing, can acquire a pro-fibrotic phenotype and play a crucial role in inducing and regulating fibrosis. Chemokines produced by M2 macrophages, such as CCL18/PARC, promote collagen production in lung fibroblasts, establishing a feedback loop with alveolar macrophages. 29 Here it is shown a progressive increase in CCL18 levels from 3 to 6 months after COVID-19, indicating a potential association with fibrosis progression. This finding aligns with previous correlations observed between CCL18 levels and the severity of fibrosis in idiopathic pulmonary fibrosis and systemic sclerosis. 30 Thus, CCL18 has the potential to serve as an early marker for identifying progressive post-COVID interstitial lung disease.
The analysis of the cytology of the BAL fluid revealed an intriguing phenomenon known as efferocytosis, where macrophages were observed engulfing apoptotic neutrophils. This indicated a notable shift in the macrophage population toward the M2 phenotype, which is notoriously associated with several beneficial effects. The M2 macrophages display a reduced production of pro-inflammatory cytokines like TNF-α and IL-6 while increasing the release of anti-inflammatory mediators such as IL-10, IL-13, and TGF-β. In addition, they release pro-resolving molecules that aid in the resolution of inflammation, mainly by efferocytosis as a regulator of macrophage polarization.31 –33 Furthermore, regulatory T cells play a crucial role in the immune system by interacting with various components of both the innate and adaptive immune responses. These specialized cells possess potent immunosuppressive abilities. They achieve this by inhibiting T-cell function, promoting the transition of macrophages to the M2 phenotype, releasing anti-inflammatory mediators, enhancing immune tolerance, and facilitating the resolution of inflammation. 34 Interestingly, regulatory T cells secrete IL-13, which, in turn, stimulates the production of IL-10 in macrophages. The presence of IL-10 then initiates a cascade of events, leading to macrophage efferocytosis. Specifically, IL-10 triggers the accumulation of Rac1-associated actin in the phagosome, facilitating the internalization of apoptotic cells by macrophages, 35 and promoting tissue recovery and healing.
CD28 expression, a major activator of naive T cells and crucial for T-cell survival, was monitored here. 36 There was a significant time-dependent downregulation of the CD28 in the patient’s BAL, suggesting a worrying evolution of the post-COVID interstitial lung disease for those patients. This is especially when correlated with other findings in which downregulation of checkpoint CD28 and ICOS on the CD4 T-cell surface was associated with pulmonary function decline and impacted all-cause mortality for patients with idiopathic pulmonary fibrosis. 37 Consistent with our findings, a recent study also reported changes in markers of lymphocyte activation and exhaustion, including reduced CD28 expression in CD8+ T cells, in patients with mild to moderate post-COVID-19 conditions. 38
Consistently, cytokine profiles of the post-COVID-19 patients group were enriched for anti-inflammatory cytokines. The literature reported an increased expression of TH2 cytokines (especially IL-13) in idiopathic pulmonary fibrosis. 39 In accordance with this, a significant upregulation of the IL-13 in the BAL of the post-COVID patients is shown here. Particularly sevenfold 1 month after infection, 20-fold at month 3, and approximately 40-fold higher at month 6 compared to healthy volunteers. To date, this may be the first argument in demonstrating the interstitial lung disease from the post-COVID syndrome. In addition, to promote the pro-fibrotic response despite the anti-inflammatory one, it is necessary to induce the polarization of macrophages to an M2-like phenotype, which may be promoted by IL-10. 40 Indeed, gene expression analysis revealed an IL-10 overexpression in the same manner as IL-13 but with half the value compared to this one.
Overall, these results provide insights into how innate immune responses are modulated by pro- and anti-inflammatory cytokines secreted by T cells and macrophages, likely contributing to the initiation of the pro-fibrotic responses observed in evolution in post-COVID interstitial lung disease. It has been done by analyzing the same patients at three time intervals after viral infection.
The study, however, is limited in its small sample size, single-center experience, and used just several representative pro-and anti-inflammatory cytokines expression panels, due to the restriction related to BAL fluid samples from the patients.
In future directions, it would be interesting to confirm the results, especially in relevant cytokines by a classic ELISA analysis for all cytokines. In addition, further multicentric corroboration of the findings would be of interest.
Conclusion
In conclusion, our findings suggest that the immune response in post-COVID interstitial lung disease is marked by a dual process of sustained inflammation and attempts at resolution, which may collectively drive the pathogenesis of pulmonary complications. These insights can pave the way for targeted therapeutic strategies aimed at modulating these immune mechanisms to prevent or mitigate long-term lung damage in affected patients. Further research is warranted to validate these mechanisms and to explore the potential for intervention strategies that can balance immune response and promote healing in the lungs.
