Abstract
Background:
Human epididymis protein 4 (HE4, also known as WFDC-2) has been implicated in fibrotic disorders pathobiology. We tested the hypothesis that HE4 may be used as a candidate biomarker for systemic sclerosis (SSc)-related interstitial lung disease (SSc-ILD).
Methods:
A total of 169 consecutive SSc patients and 169 age-and sex-matched healthy controls were enrolled and blood samples were collected. Pulmonary function tests (PFTs) and paired lavage was performed on 169 patients and 37 healthy controls. All patients were classified as having SSc-no ILD or SSc-ILD, based on high-resolution computed tomography (CT) scans of the chest, and a semiquantitative grade of ILD extent was evaluated through CT scans (grade 1, 0–25%; grade 2, 26–50%; grade 3, 51–75%; grade 4, 76–100%). Serum and bronchoalveolar lavage fluid (BALF) HE4 levels were measured by enzyme-linked immunosorbent assay.
Results:
Serum HE4 levels were higher in SSc patients [median (interquartile range), 139.4 (85.9–181.8) pmol/l] compared with healthy controls [39.5 (24.3–54.2) pmol/l, p < 0.001] and were higher in patients with SSc-ILD [172.1 (94.8–263.3) pmol/l] than in those with SSc-no ILD [97.4 (85.5–156.5) pmol/l, p < 0.001]. This observation was replicated in the BALF samples. Corresponding values were 510.8 (144.6–1013.8) pmol/l for SSc cohort, 754.4 (299–1060) pmol/l for SSc-ILD, 555.1 (203.7–776.2) pmol/l for SSc-no ILD, and 238.7 (97.7–397.6) pmol/l for controls. The semiquantitative grade of ILD on CT scan was significantly proportional to the HE4 levels and the lung function parameter (i.e., FVC) had a negative correlation with the HE4 levels.
Conclusion:
This is the first study to demonstrate the potential clinical utility of blood and BALF HE4 as a biomarker for SSc-ILD. Future prospective validation studies are warranted.
Introduction
Systemic sclerosis (SSc, scleroderma) is a complex fibrotic disease in which autoimmunity, inflammation, and vascular lesions lead to fibrosis of the skin and several internal organs, most notably the lung. 1 Interstitial lung disease (ILD) characterized by inflammation and/or fibrosis of lung tissue is a common manifestation and a leading cause of mortality in SSc patients.2,3 High-resolution computed tomography (HRCT) gives evidence that about two-thirds of the cases with SSc develop interstitial lung involvement; postmortem examination shows nearly all patients shows pulmonary fibrosis. 4 However, it remains a great challenge to develop circulating blood biomarkers that could help improve the prospective evaluation of patients with SSc-related ILD. 5
Human epididymis protein 4 (HE4) was originally described as an epididymis specific protein but more recently has been suggested as a well-known biomarker in cancer.6–8 Notably, HE4 was described to be expressed specifically in activated fibroblasts and to act as a pan-protease inhibitor to mediate kidney fibrosis. 9 Although the exact function of HE4 is not well known, HE4 inhibited the activity of multiple matrix metalloproteinases and specifically inhibited their contribution to degrade collagen I, 10 which suggests a functional role of HE4 in the pathogenesis of fibrosis. Moreover, Yamamoto et al. 10 suggested that HE4 may be used as a fibrosis marker to predict cardiac remodeling in patients with dilated cardiomyopathy. In previous studies of lung disease, HE4 serum levels correlate positively with the overall severity of cystic fibrosis (CF), 11 and elevated expression of HE4 mRNA was also described in CF lung biopsy specimens compared with the non-CF control samples. 12 In addition, there was a correlation between increased serum HE4 levels with idiopathic pulmonary fibrosis (IPF), which warrants further investigation. 13 More recently, Nagy et al. reported that plasma HE4 levels were inversely associated with lung function improvement in CF patients with ivacaftor treatment. 14
Given this aforementioned background, we performed a cross-sectional study to evaluate the performance of HE4 as a biomarker for diagnosis of SSc-ILD, and examined the correlation between HE4 and lung fibrosis severity.
Materials and methods
Study population
This study was conducted at the Shanxi Bethune Hospital between January 2018 and July 2019 and was approved by the local medical ethical committee (2018LL001). We enrolled 169 consecutive patients with a diagnosis of systemic sclerosis and 169 age-and sex-matched healthy controls. All patients met the 2013 European League against Rheumatism/American College of Rheumatology (EULAR/ACR) classification criteria for SSc. 15 Data on demographics, SSc subset, anti-centromere antibody, anti-topoisomerase I antibody, disease duration, and use of immunosuppressive drugs were collected. Immunosuppressive therapy was defined as treatment with any immunosuppressive drugs at enrollment. Disease onset was calculated from the first non-Raynaud phenomenon (RP) symptom, and disease duration was defined as the time between the first non-Raynaud symptom and blood sampling. SSc subsets were defined as a limited and diffuse subset according to LeRoy classification, 16 and skin involvement was determined by the modified Rodnan skin score (mRss). 17
The assessment of ILD was based on the findings of pulmonary function tests (PFTs) and HRCT scans at entry. Extensive lung disease was defined in a manner as previously described: >20% extent of fibrosis on HRCT or 10–30% extent of fibrosis and a forced vital capacity (FVC) of <70%. 18 Lung fibrosis was diagnosed using HRCT (ground glass opacification, interlobular septal thickening, fibrosis, and honeycombing) as judged by a pulmonologist or radiologist.
Key exclusion criteria were significant other organ co-morbidity, including cardiac, hepatic, or renal impairment; malignancy; pregnancy; radiologic results suggesting concurrent illness (i.e., pleural effusions or pneumonia) limiting the HRCT scan interpretation; and incapacity for providing written informed consent. In addition, subjects who did not have a HRCT, bronchoalveolar lavage (BAL), and PFTs performed within 3 months of each other were excluded. All healthy controls were asymptomatic, not taking any medication, and had normal chest HRCT. Written informed consent was obtained from all participants.
Chest HRCT scanning
HRCT was performed using a dual-source computed tomography (CT) scanner (Somatom definition flash, Siemens, Erlangen, Germany). All patients were scanned from lung apices to bases, supine, at full inspiration, with 1 mm section thicknesses using a peak voltage of 120 kV with tube current modulation (range 100–111 mA). All images were viewed on Syngo.via Oncology workstations using axial images with a window level of –600 Hounsfield units (HU) and a window width of 1500 HU. Each HRCT scan was evaluated independently by two radiologists with over 10 years thoracic imaging experience, respectively, blinded to all clinical and lung function information, as described previously. 18
Visual HRCT analysis
All HRCT scans were evaluated for the presence of ILD (1, no ILD; 2, ILD present) by two blinded radiologists in consensus. 19 The extent of ILD of HRCT scans was graded semi-quantitatively based on lung involvement, using a Likert scale (grade 1, 1–25%; grade 2, 26–50%; grade 3, 51–75%; grade 4, 76–100%) for extent of four categories of parenchymal abnormality (pure ground glass opacity, lung fibrosis, honeycombing, and emphysema). 20 The scoring was performed in each of three lung zones as indicated below: upper, extending from apex to aortic arch; middle, from aortic arch to inferior pulmonary veins; and lower, from inferior pulmonary veins to diaphragm). 21 If any definitive reason for exacerbation of HRCT findings other than ILD was present, such as pneumonia, those HRCT scans were excluded from the study. 22 Patients with indeterminate ILD were excluded from the primary analyses except where indicated. 23 Discrepancies were repeated and then resolved by consensus. The presence or absence of emphysema was also recorded.
Pulmonary function testing
PFT was performed with a Master Screen pulmonary function instrument (CareFusion, Bagheria, Germany) in accordance with American Thoracic Society/European Respiratory Society recommendations.24–26 The following indices were recorded: the median forced expiratory volume in 1 s (FEV1), forced vital capacity (FVC), and diffusing capacity of the lung for carbon monoxide (DLco). These values were described as a percentage of the predicted values for the patient’s age, sex, and height. 27
BAL procedure
BAL was performed using a CV-260SL flexible optic bronchoscope (Olympus, Tokyo, Japan). Briefly, a total volume of 60–120 ml (divided into 3–4 aliquots) of sterile buffered saline was instilled into the right middle lobe with immediate aspiration after each aliquot. The recovery rate was about 47%. The recovered BALF was filtered through a two-layer sterile gauze and centrifuged at 1800 × g for 20 min. 28 The supernatant was collected and stored at –80°C until further procedures. BAL fluid (BALF) specimens were obtained from 169 SSc patients and 37 healthy individuals.
Measurements of HE4
Blood collection occurred within 3 months before PFTs, BAL, and HRCT. Blood samples obtained were centrifuged at room temperature within 30 min, and serum aliquots were stored at –80°C until analysis. HE4 levels from serum and BALF was measured by enzyme-linked immunosorbent assay (EIA, Fujirebio Diagnostics, Gothenburg, Sweden) according to the manufacturer’s instructions, as previously described. 29 In brief, the sample was incubated with Biotin Anti-HE4 (2H5). Horseradish peroxidase (HRP) Anti-HE4 (3D8) antibody was added as a second antibody following a phosphate-buffered saline (PBS) wash. After the second washing, the TMB (3,3′,5,5′-tetramethylbenzidine) HRP-substrate was added to each well. Finally, the absorbance was read at 405 nm in a micro-plate spectrophotometer. Samples with HE4 concentrations higher than 900 pmol/l were re-measured following dilution according to the manufacturer’s handbook. To adjust for dilution of BALF in estimating the HE4 concentrations in the respiratory epithelial lining fluid, we analyzed urea in BALF and concomitant serum samples with a SYNCHRON®SYSTEMS Urea Assay Kit (Beckman Coulter, Brea, CA, USA) according to the protocol provided by the manufacturer. The ratio [(urea in serum)/(urea in BALF)] were used as a coefficient for dilution to adjust HE4 levels as described by Aspelund et al. and Pocino et al.30,31
Statistical analysis
Statistical significance was defined as p < 0.05. Descriptive statistics were given as mean ± standard deviation (SD) or median [interquartile range (IQR)]. Pearson coefficients, Spearman coefficients, chi-square, Wilcoxon rank-sum tests and Mann–Whitney U test were applied as appropriate. Normality of continuous variables was assessed by the Kolmogorov-Smirnov test. The diagnostic performance of HE4 was determined using receiver operating characteristic (ROC) analyses. Optimal cut-off values for HE4 were based on the Youden index. Statistical differences between AUCs were assessed using the DeLong test. In all models, patients with indeterminate-ILDs were excluded. Data analyses were conducted using SPSS Version 18.0 (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 7 (GraphPad Software Inc., San Diego, CA, USA).
Results
Cohort characteristics
Demographic and clinical data of patients and healthy controls are summarized as shown in Table 1. There was a female predominance, and the mean age for all patients was 58.0 years (SD = 14.7 years); 70.4% of the SSc cohort had never smoked and median disease duration was 5.1 years (range: 0.3–9.9 years). Out of 169 patients, 53 (31.4%) were treated with immunosuppressive drugs at the evaluation: 39.6% (21/53) received Cyclophosphamide, 32.1% (17/53) received Mycophenolate mofetil, 20.8% (11/53) received Methotrexate, 7.5% (4/53) received Prednisone. Laboratory findings are shown in Table 2.
Demographic and clinical data in the study population.

Values are mean ± standard deviations, median (range) or n (%).
Pulmonary hypertension was defined as estimated right ventricular pressure greater than 40 mmHg on echocardiogram and none of the patients had confirmatory right heart catheterization.
Use of immunosuppressive agents includes cyclophosphamide, mycophenolate, methotrexate, or prednisone.
p < 0.05, **p < 0.01, ***p < 0.001 compared with SSc -no ILD using the Mann–Whitney U test or Fisher’s exact test.
“–”, not available.
BALF, bronchoalveolar lavage fluid; BMI, body mass index; CT, computed tomography; ILD, interstitial lung disease; IST, immunosuppressive therapy; mRSS modified Rodnan skin score; SSc systemic sclerosis.
Laboratory findings in patients and controls.

Values are presented as mean ± SD or median (interquartile range) unless otherwise stated.
169 healthy individuals underwent PFTs and venous blood collection.
37 healthy individuals underwent BAL.
p < 0.05 versus SSc-ILD; **p < 0.01 versus SSc-ILD; ***p < 0.001 versus SSc-ILD; Other abbreviations as in Table 1.
BALF, bronchoalveolar lavage fluid; DLCO %, diffusing capacity of carbon monoxide % predicted; EV1, forced expiratory volume in 1 s; FVC%, forced vital capacity % predicted; HE4, human epididymis protein 4; PFTs, pulmonary function tests.
Serum and BALF HE4 levels
As shown in Figure 1A, median serum HE4 level was significantly elevated in patients with SSc [median (IQR) 139.4 (85.9–181.8) pmol/l] compared with healthy control subjects [39.5 (24.3–54.2) pmol/l, p < 0.001]. BALF HE4 level was 510.8 (144.6–1013.8) pmol/l in SSc patients and 238.7 (97.7–397.6) pmol/l in healthy control subjects (Figure 1B). Moreover, when we pooled the data from all cohorts, a significant relationship was observed between the serum and the BALF HE4 levels (r = 0.798, p < 0.001, Figure 2).
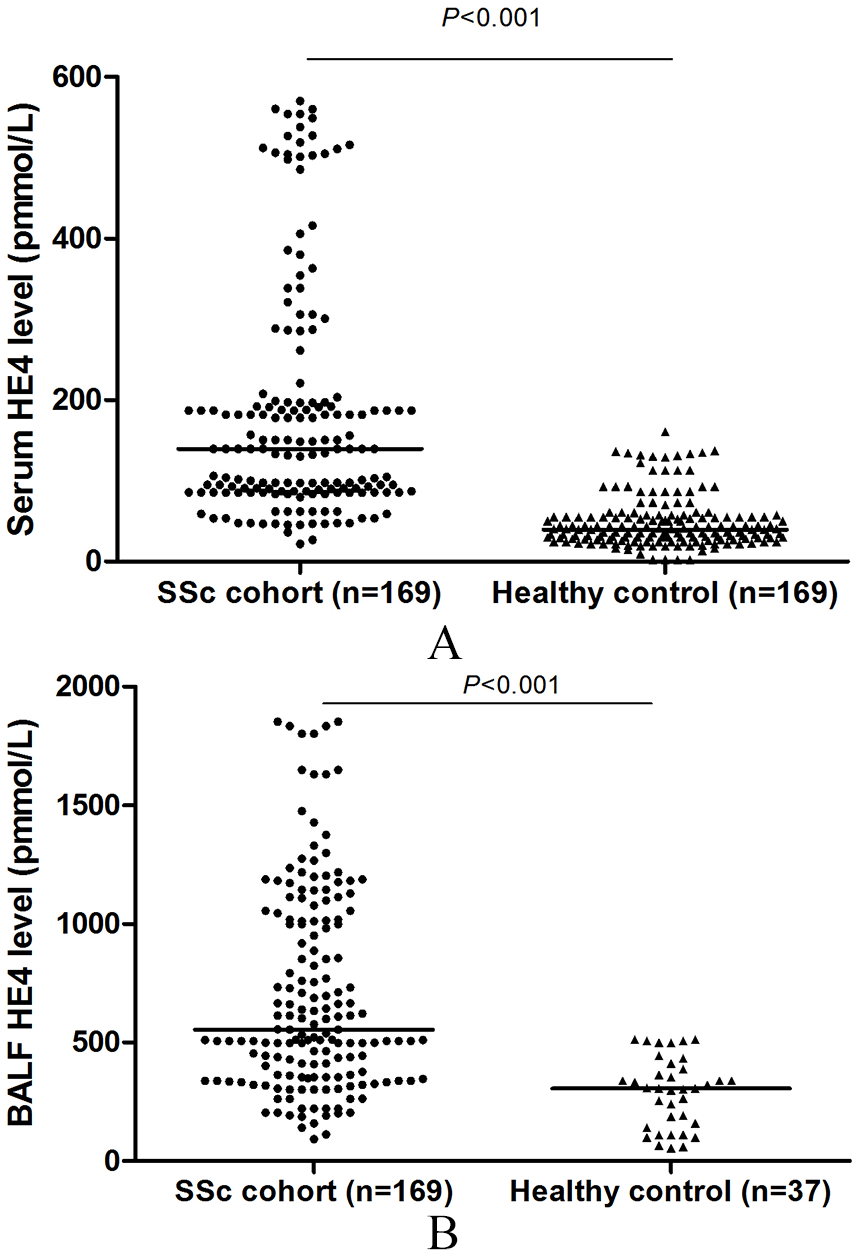
Scatter plots of (A) serum HE4 and (B) BALF HE4 levels in SSc patients and healthy controls. Horizontal bars indicate the median values. p values were determined by Mann–Whitney U test.
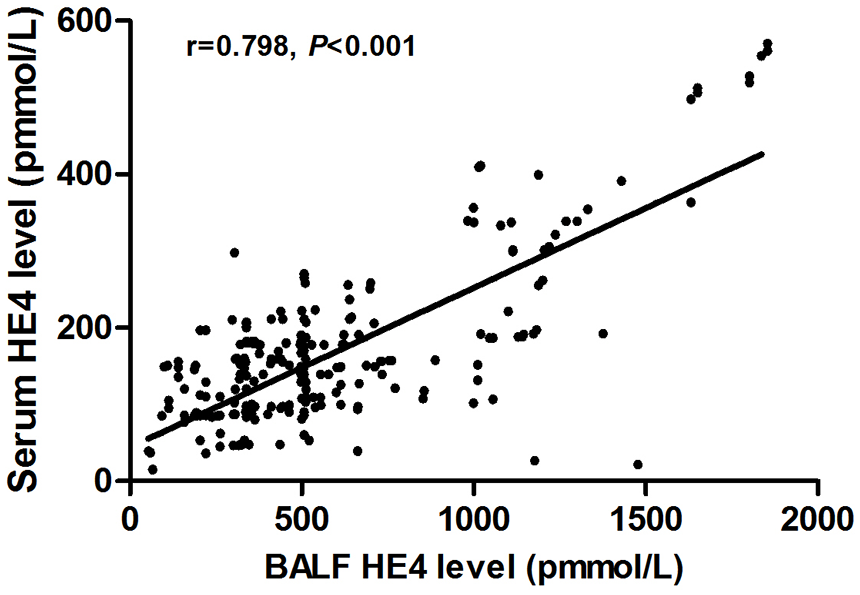
Associations between HE4 in serum and BALF. Correlation analyses were assessed using Spearman’s rank correlation.
With regard to SSc subgroups, SSc-ILD patients had markedly higher HE4 concentrations in both serum and BALF than SSc-no ILD patients [serum: 172.1 (94.8–263.3) pmol/l, 97.4 (85.5–156.5) pmol/l, respectively; BALF: 754.4 (299–1060) pmol/l, 555.1 (203.7–776.2) pmol/l respectively; Figure 3A, B and Table 2]. There was also a significant difference between HE4 levels in patients with SSc-no ILD and healthy controls (p < 0.001, Figure 3 and Table 2).

Scatter plots of (A) serum HE4 and (B) BALF HE4 levels in SSc-ILD and SSc-no ILD patients. Horizontal bars indicate median values. p values were determined by Mann–Whitney U test.
Of the 92 SSc-ILD and 77 SSc-no ILD patients, 52 and 1, respectively, received immunosuppressive drugs. Therefore, we limited the analysis to SSc-ILD patients to investigate differences between HE4 levels in patients with and without immunosuppressive therapy. The results showed that no significant differences were observed between patients with and without treatment [serum: 170.7 (90.1–315.2) pmol/l, 176.3 (99.7–291.6) pmol/l, respectively; BALF: 778.9 (232.6–1011) pmol/l, 746.1 (249.4–1192.4) pmol/l, respectively].
Correlation between ILD and HE4 levels in patients with SSc
We first correlated the HE4 levels and severity of SSc-ILD as measured by semiquantitative grades of ILD. The HE4 levels were significantly proportional to the grades of ILD on the CT scans. Serum and BALF HE4 levels differentiated grades 1 and 2 (p = 0.027, p < 0.01, respectively), grades 2 and 3 (both p < 0.01), and grades 3 and 4 (p < 0.001, p = 0.013, respectively) in patients with SSc -ILD (Figure 4). Therefore, our findings indicate that HE4 level may be a good biomarker of severity of ILD in patients with SSc.

Box plots of (A) serum HE4 and (B) BALF HE4 levels in SSc-ILD patients according to semiquantitative CT grades: grade 1, 0–25% involvement of ILD on chest CT; grade 2, 26–50%; grade 3, 51–75%; grade 4, 76–100%.
We further evaluated the relationship between HE4 levels and PFTs, using Spearman’s correlation analysis. Within the SSc-ILD cohort, serum and BALF HE4 showed both a significant inverse correlation with FVC (r = −0.85, p < 0.001, Figure 5A; r = −0.89, p < 0.001, Figure 5B, respectively). Furthermore, HE4 levels were also significantly correlated with FVC in all SSc patients, although the correlation was smaller (r = −0.70, p < 0.01, Figure 5C; r = −0.76, p < 0.01, Figure 5D, respectively). When the relationship between HE4 and the values of the FEV1 and DLco was analyzed, an inverse tendency was not found (data not shown).

Associations between (A) serum HE4 and FVC in the SSc-ILD cohort, (B) BALF HE4 and FVC in the SSc cohort, (C) serum HE4 and FVC in the SSc-ILD cohort, and serum HE4 and FVC in the SSc cohort. Correlation analyses were assessed using Spearman’s rank correlation.
HE4 levels in association with skin involvement in patients with SSc
In addition, we evaluated the relationship between HE4 levels and extent of skin disease. Contrary to our expectations, we found that mRSS tended to be inversely correlated with BALF HE4 levels in patients with SSc-ILD; however, the relationship did not reach statistical significance (r = 0.51, p = 0.076, Spearman’s correlation). Also, no significant association was observed between serum HE4 and mRSS in SSc-ILD (data not shown).
Diagnostic characteristics of HE4 in SSc
Receiver operating curve (ROC) analysis revealed strong performance characteristics of both serum and BALF HE4 for detecting the presence of ILD in SSc patients (Table 3). The optimal cut-offs (according to the Youden index) for serum and BALF HE4 to distinguish SSc-ILD from SSc-no ILD were 237.6 and 501.7 pmol/l, respectively. The corresponding areas under the curve (AUCs) were 0.83 (95% CI 0.67–0.91) and 0.91 (95% CI 0.87–1.0).
Diagnostic accuracy of serum and BALF HE4.

AUC, area under the curve; BALF, bronchoalveolar lavage fluid; CI, confidence interval; CT, computed tomography; HE4, human epididymis protein 4; ILD, interstitial lung disease; SSc systemic sclerosis.
When differentiating grade 1 versus 2, 3, and 4, the AUC of serum HE4 (0.77; 95% CI 0.51–0.89) was lower than that of BALF HE4 (p = 0.029), while the AUC of serum HE4 for grades1, 2, 3, and 4 (0.91; 95% CI 0.79–1.0) was similar to that of BALF HE4 (p = 0.99).
Discussion
This work is the first to describe the potential for HE4 to be used as a novel biomarker of SSc-ILD. Our results indicated for the first time that serum HE4 was significantly elevated in SSc-ILD patients compared with SSc-no ILD patients and control subjects, and HE4 is detectable in BALF.
Moreover, we showed that HE4 was associated with SSc-ILD severity, and that this association was independent of mRSS, which is another established risk factor for disease severity in SS. Also, HE4 exhibited an inverse correlation with lung function (i.e., FVC) in SSc patients. These results emphasize the potential for HE4 to be used as a diagnostic tool for SSc-ILD.
It has been reported previously that HE4 is able to bind and inhibit the activities of several proteases (e.g., Prss23, Prss35, trypsin, bacterial collagenase, MMP2, and MMP9),9,32,33 and HE4 has been characterized with respect to fibrosis. Accordingly, in view of its putative roles in fibrosis, we sought to investigate HE4 expression in systemic sclerosis.
As a first approach, we decided to study BALF based on a previous study showing that WFDC2 protein is readily detectable in respiratory tract tissues. 34 With this strategy, we were able to identity that HE4 was expressed in BALF from patients with SSc, and that elevated levels of BALF HE4 are associated with severity of ILD. BALF HE4 correlated positively with serum HE4 and was about 5-fold higher than the latter. Hence, we speculated that HE4 secreted at high levels into BALF is transferred into the circulation across the air–blood barrier.
HRCT is currently considered the gold-standard imaging method for ILD detection.35,36 However, considering cost-effectiveness and radiation hazard, serum biomarkers can be obtained easily and quickly and measured longitudinally. SP-D, KL-6, and CCL18 has been investigated as important circulating biomarkers associated with the pathogenic process of ILD, reflecting the progress of lung involvement.5,37 The most important finding in the present study was that serum HE4 was able to act as a predictive marker in SSc-ILD when scleroderma patients were categorized according to variable degree of severity by HRCT.
This study has some weaknesses. Firstly, it was a single-centre study. Therefore, the number of patients in the SSc-advanced group (n = 21) was rather small. However, it might be an advantage to be consistent in evaluation of subjects’ characteristics such as diagnostic process of SSc, interpretation of HRCT, and treatment strategies. Secondly, the cross-sectional nature of the study meant that determination of the relationship between HE4 and progression of SSc-ILD was not performed. Further studies using longitudinal data are required to establish exact and definite causal relationships. Thirdly, this study lacks a validation cohort to confirm the results. Furthermore, most of our patients were recruited from the rheumatology department in a tertiary hospital of Shan xi Province, raising the possibility of a referral bias toward more serious disease. Fourthly, mean disease duration was 5.1 years. Therefore, the results may not apply to other SSc patients with longer disease duration. Fifthly, in view of the fact that traditional Chinese medicine is relatively common in China, that is to say, our patients may take a small amount of traditional Chinese medicine irregularly (although there are no patients taking it in the questionnaire), which may affect the results. A final limitation is the lack of histopathologic examination. Direct serum/BALF-pathology correlation will be more convincing for HE4 as a biomarker for SSc-ILD.
In conclusion, this study showed that HE4 is markedly elevated both in BALF and in serum of patients with SSc-ILD, and associated with extent of ILD in scleroderma patients. We thus suggest that HE4 could be involved in lung fibrosis pathogenesis and be a biomarker for SSc-ILD. It would be interesting to study further the relevance of this pan-protease inhibitor in other connective tissue disease-associated lung disease.
Conclusion
We report that HE4 levels in serum and BALF were markedly increased in patients with SSc-ILD and had a positive correlation with ILD severity as measured using HRCT. Moreover, HE4 levels had a negative correlation with PFT parameter (i.e., FVC). HE4 could be a clinically useful biomarker in screening and evaluating SSc-ILD.
Footnotes
Acknowledgements
We gratefully thank Shanghai Bioprofile Technol-ogy Company Ltd. for providing technical support.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shanxi Bethune Hospital Scientific Research Fund Project (N0.2019YJ10).
