Abstract
Background:
The relationship between short-term cardiac function changes and long-term outcomes in heart failure (HF) patients undergoing cardiac resynchronization therapy (CRT) remains uncertain, especially when stratified by diabetes status.
Objectives:
This study aims to assess the association between short-term cardiac function changes and outcomes such as all-cause mortality and HF hospitalization in patients undergoing CRT, stratified by diabetes status.
Design:
This is a cohort longitudinal retrospective study.
Methods:
A total of 666 HF patients, treated with CRT between March 2007 and March 2019, were included in this study. Among them, 166 patients (24.9%) were diagnosed with diabetes. Cardiac function was assessed at baseline and again at 6 months, incorporating evaluations of left ventricular ejection fraction (LVEF), left ventricular end-diastolic diameter (LVEDD), left atrial diameter (LAD), N-terminal prohormone of brain natriuretic peptide (NT-proBNP), and QRS duration. The QRS duration represents the time required for a stimulus to spread through the ventricles (ventricular depolarization). The primary endpoints of the study were all-cause mortality and HF-related hospitalization.
Results:
During a median follow-up of 2.51 years, 172 (25.8%) patients died and 197 (29.6%) were hospitalized for HF. Changes in LVEF, LVEDD, and LAD within 6 months had similar effects on adverse outcomes in both diabetic and nondiabetic patients. However, the presence of diabetes significantly modified the association between changes in NT-proBNP and QRS duration and adverse outcomes. Short-term changes in NT-proBNP and QRS duration were positively associated with all-cause mortality and HF hospitalization in patients without diabetes. However, the relationship between short-term changes in NT-proBNP and QRS duration and adverse outcomes was non-linear in diabetic patients.
Conclusion:
Improvement of cardiac function after CRT implantation can reduce long-term risk of all-cause mortality and HF hospitalization in HF patients. However, the presence of diabetes may affect the association between short-term changes in NT-proBNP and QRS duration and adverse outcomes.
Introduction
Heart failure (HF) is a significant growing global public health concern that affects approximately 26 million people worldwide. 1 Cardiac resynchronization therapy (CRT) is recommended for HF patients with reduced left ventricular ejection fraction (LVEF) and wide QRS who remain symptomatic despite optimal medical therapy. 2 Although CRT has been shown to improve HF symptoms, exercise capacity, and left ventricular function, up to one-third of patients are considered non-responders, showing no significant improvement in LVEF following CRT.3,4
Non-response to CRT is associated with higher risks of adverse outcomes in HF patients, and identifying predictors of non-response to CRT is essential for optimizing treatment selection and improving the outcomes of HF patients.5,6 Short-term changes in cardiac function parameters are useful indicators for assessing the efficacy of CRT, and adverse changes are strongly correlated with long-term adverse outcomes. 7 For example, early improvement in left ventricular function, as measured by changes in LVEF, predicts better outcomes following CRT. 8 From this perspective, changes in other cardiac function parameters, such as left ventricular end-diastolic diameter (LVEDD) and left atrial dimension (LAD), N-terminal prohormone of brain natriuretic peptide (NT-proBNP), and QRS duration, also have prognostic values.9–12
Diabetes and cardiovascular disease are highly prevalent comorbidities, and the presence of diabetes may influence CRT response in HF patients.13,14 While some studies suggest that diabetes is significantly related to poorer outcomes in HF patients receiving CRT, including increased risks of all-cause mortality and HF hospitalization,15,16 others report no significant difference in the effectiveness of CRT in reducing mortality and HF hospitalization between patients with and without diabetes.17,18 The limited and inconsistent evidence hinders a clear understanding of the impact of diabetes on short-term changes in cardiac function and long-term clinical outcomes in HF patients undergoing CRT.
To resolve this issue, our study aims to investigate the relationship between short-term changes in cardiac function parameters and long-term outcomes in HF patients receiving CRT, with or without diabetes. These findings could aid in the early detection of cardiac dysfunction and implement appropriate interventions to mitigate the adverse impact of diabetes on cardiac function in HF patients treated with CRT.
Methods
Study design and population
We conducted a retrospective observational cohort analysis at the Arrhythmia Center of Fuwai Hospital, including 686 consecutive HF patients who received CRT implantation between March 2007 and March 2019, meeting class I or II guideline recommendations.19,20 Study inclusion criteria: (1) patients with HF aged 18 years and above; (2) adherence to the 2021 ESC guidelines for CRT, encompassing both class I and class II recommendations; (3) cardiac function assessments conducted post-CRT and at the 6-month mark; (4) provision of written informed consent for participation in the study. The exclusion criteria are as follows: (1) absence of diabetes history documentation; (2) lack of recorded study outcomes; (3) loss to follow-up within 6 months post-CRT implantation; (4) incomplete cardiac function data records. As a result of the above criteria, after excluding 5 patients with missing diabetes history and 15 cases with missing study outcome records, we analyzed data from a final sample of 666 patients. The flow of study selection is illustrated in Figure 1.

Flow chart of study participants.
Assessment of changes in cardiac function
In this study, patients underwent cardiac function assessment at CRT implantation and at 6 months using various measures including LVEF, LVEDD, LAD, NT-proBNP, and QRS duration. The LVEF, LVEDD, and LAD values were obtained using transthoracic two-dimensional guided M-mode echocardiography (EPIC7, iE33; Philips Medical Systems, Amsterdam, Netherlands) as per current guidelines by a qualified ultrasound physician. 21 The LVEDD value was calculated by measuring the left ventricular (LV) internal diameter in the parasternal LV long-axis view, while the LA diameter was measured by assessing the LA anterior–posterior linear dimension in the parasternal long-axis view. 22 The LVEF was calculated using the Simpson method and derived from LV end-diastolic volume and LV end-systolic volume. The NT-proBNP concentration was analyzed by electrochemiluminescence immunoassay using blood samples collected into Ethylene Diamine Tetraacetic Acid (EDTA) anticoagulant tubes, which were then evaluated using the Roche Elecsys 2010 Immunology Analyzer, Roche, Basel, Switzerland. The QRS duration was measured by calculating the duration from the start point to the end point of the QRS wave group on a standard 12-lead electrocardiogram. All these cardiac function parameters required re-examination at 6 months after CRT. Change in cardiac function was defined as the difference between the cardiac function measurement at 6 months after CRT and the baseline value.
Study outcomes and follow-up
The study assessed all-cause mortality and HF hospitalization as the primary outcomes. The follow-up period spanned from CRT implantation until the earliest occurrence of HF rehospitalization or mortality. The researchers gathered outcome data by regularly contacting the patients via telephone, and the follow-up period ended in January 2021. The study’s total follow-up period extended up to 12.36 years, with a median of 2.51 years.
Assessment of covariates
This study included a variety of factors, including demographic information, disease history, medication history, and cardiac function parameter. Demographic data, smoking, and drinking habits were collected through questionnaires. Body mass index was calculated using weight and height measurements. Clinicians used the New York Heart Association classification to determine cardiac function based on clinical tests and patient symptoms. Medical and disease histories were obtained from electronic medical records, including atrial fibrillation, left bundle branch block, coronary artery disease, stroke, ischemic cardiomyopathy, hypertension, diabetes, and ventricular tachycardia/ventricular fibrillation (VT/VF). The medication history included both inpatient and post-discharge medications, such as β-blockers, angiotensin-converting enzyme inhibitors/angiotensin receptor blockers, diuretics, amiodarone, statins, anticoagulants, and antiplatelets. Estimated glomerular filtration rate (eGFR, ml/min/1.73 m2) was calculated using the simplified of the Modification of Diet in Renal Disease equation (MDRD) formula. 23 Detailed definitions of disease history are provided in Supplemental Table 1.
Post hoc power analysis
A post hoc power analysis was conducted to justify the sample size selected for this study. Based on the mortality rates observed in previous studies for patients treated with CRT, which are 26% for diabetic patients and 20% for non-diabetic patients, 24 the calculated sample size to achieve 80% power at a 5% significance level was approximately 322 patients per group. Our study, with a total of 666 patients (166 diabetic and 500 non-diabetic), is adequately powered to detect significant differences in mortality rates between diabetic and non-diabetic patients treated with CRT.
Statistical analysis
The study’s baseline characteristics were presented based on diabetes status (diabetes and non-diabetes). Continuous variables were expressed as mean ± standard deviation (SD), and categorical variables were expressed as percentages (%). Student’s t test and Chi-square test were used to compare continuous and categorical variables, respectively. The distribution of NT-proBNP was skewed, and therefore the log transformation was applied for statistical analyses (Supplemental Figures 1 and 2). Multivariate Cox proportional hazards regression models were used to estimate the hazard ratio (HR) and 95% confidence interval (CI) for the association of cardiac function changes with all-cause mortality and HF hospitalization in the overall population and stratified by diabetes. The adjusted variables were selected based on clinical relevance or univariate association with outcomes, and a final regression model was determined based on the number of available events. To detect nonlinear associations, restricted cubic smoothing curve fitting was used, and the likelihood ratio test was used to determine the inflection point. If a non-linear relationship was found, the best-fitting model with the maximum log likelihood was identified using a recursive algorithm to find the inflection point. The HRs and their corresponding 95% CI for the two-piece-wise regression model were calculated using Bootstrap resampling.
The statistical analyses in this study were conducted using SPSS statistical software (version 24.0), IBM, New York, USA and R statistical software is an open source software (version 4.0.2). A two-sided p value of less than 0.05 was considered statistically significant.
Results
Baseline characteristics
From March 2007 to March 2019, 666 HF patients treated with CRT were included in the final analysis. Among them, 166 (24.9%) had diabetes, with a mean age of 59.1 ± 11.5 years, and 214 (32.1%) were female. At baseline, the mean value of LVEF was 30.3 ± 7.9%, LVEDD was 69.7 ± 9.8 mm, LAD was 44.7 ± 7.6 mm, QRS was 164.2 ± 23.9 ms, and the median NT-proBNP was 1371 (741.5–2611.2) pg/ml. Compared to non-diabetic patients, diabetic patients were older, more obese, had a worse cardiac function, and had a higher incidence of co-morbidities such as coronary artery disease, cardiomyopathy, and hypertension, but a lower incidence of VT/VF (Table 1).
Baseline characteristics of the total population and stratified by diabetes.

Data are presented as n (%) and mean ± standard deviation, if not otherwise indicated.
ACEI/ARB, angiotensin converting enzyme inhibitor/angiotensin receptor blocker; AF, atrial fibrillation; BMI, body mass index; CAD, coronary artery disease; CM, cardiomyopathy; eGFR, estimated glomerular filtration rate; LAD, left atrial diameter; LBBB, left bundle branch block; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; NYHA, New York Heart Association; VT/VF, ventricular tachycardia/ventricular fibrillation.
Trajectories of changes in cardiac function within 6 months
Figure 2 depicts the trajectories of cardiac function changes over 6 months in HF patients who underwent CRT, classified into three groups based on overall population and diabetes status. After 6 months of CRT treatment, all patient groups (general population, diabetic, and non-diabetic) exhibited improved cardiac function, including an increase in LVEF and a decrease in LVEDD, LAD, NT-proBNP, and QRS duration. Compared to non-diabetic patients, diabetic patients had smaller increases in LVEF [Figure 2(a)] and smaller decreases in NT-proBNP [Figure 2(d)] following 6 months of CRT treatment. However, the presence of diabetes did not significantly affect changes in LVEDD, LAD, or QRS duration over the same treatment period.
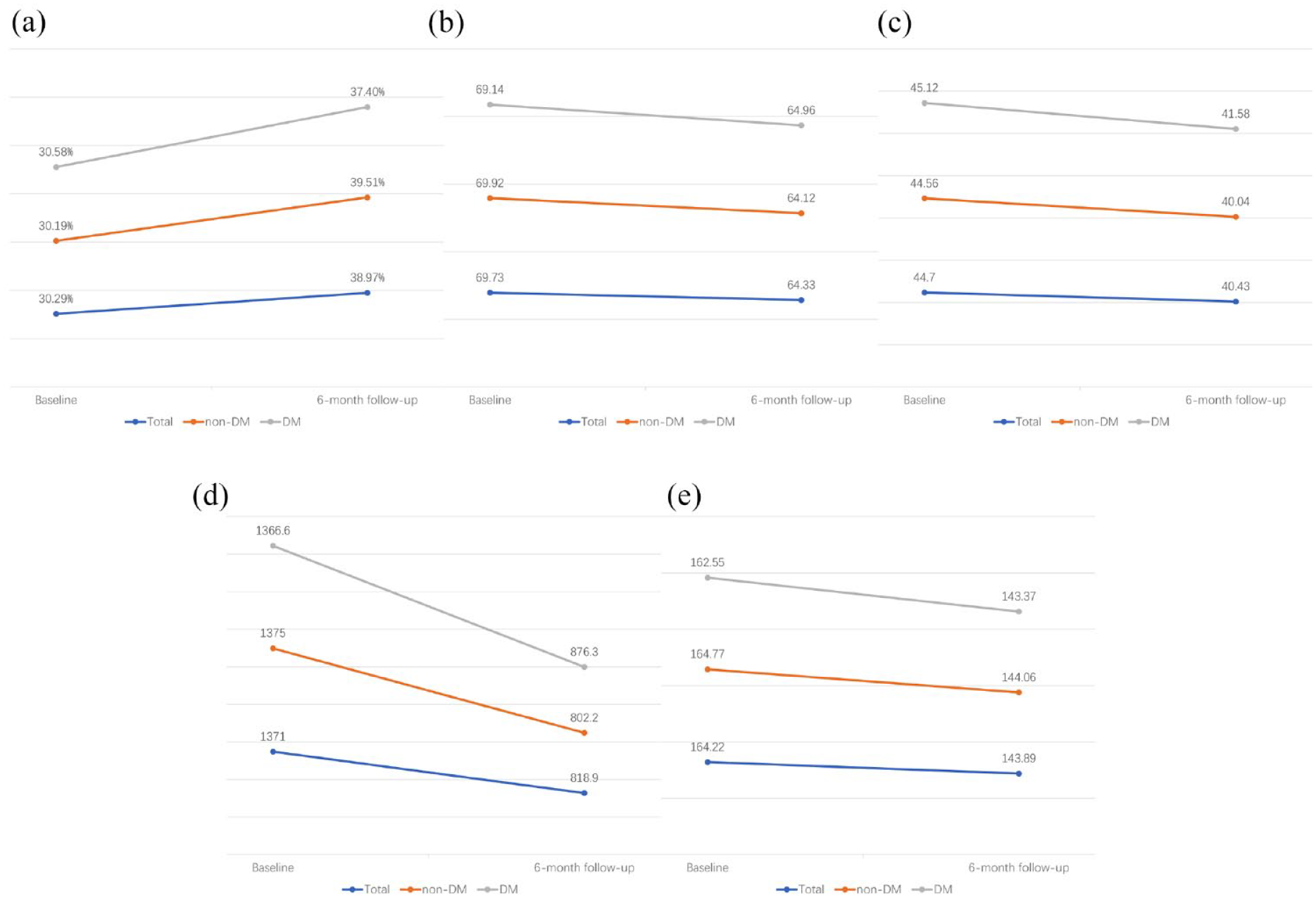
Trajectory of changes in cardiac function within 6 months. (a) Trajectory of LVEF (%) within 6 months. (b) Trajectory of LVEDD (mm) within 6 months. (c) Trajectory of LAD (mm) within 6 months. (d) Trajectory of NT-proBNP (pg/ml) within 6 months. (e) Trajectory of QRS duration (ms) within 6 months. Adjusted for age, sex, BMI, NYHA class, AF, LBBB, hypertension, VT/VF, ACEI/ARB, and eGFR.
Association between changes in cardiac function within 6 months and all-cause mortality
During a median follow-up of 2.51 years (range up to 12.36 years), 172 (25.8%) deaths occurred. In the overall population, there was a negative association between ΔLVEF and all-cause mortality (HR, 0.67; 95% CI, 0.60–0.75), while ΔLVEDD (HR, 1.74; 95% CI, 1.47–2.06), ΔLAD (HR, 1.19; 95% CI, 1.04–1.37), and ΔNT-proBNP (HR, 1.25; 95% CI, 1.07–1.45) were positively associated with all-cause mortality. However, no correlation was observed between ΔQRS duration and all-cause mortality (p > 0.05) (Table 2). In Figure 3, the association between ΔLVEF, ΔLVEDD, and ΔLAD with all-cause mortality was similar in both diabetic and nondiabetic patients. However, Figure 3(d) and (e) showed inconsistent associations between ΔNT-proBNP and ΔQRS with mortality among diabetic and nondiabetic patients. In nondiabetic patients, ΔNT-proBNP and ΔQRS were positively associated with all-cause mortality (ΔNT-proBNP: HR, 1.31; 95% CI, 1.10–1.57; ΔQRS: HR, 1.08; 95% CI, 1.00–1.17) (Table 2). However, in diabetic patients, a non-linear relationship was observed between ΔNT-proBNP and ΔQRS and all-cause mortality with an inflection point at 0. Below the inflection point, a positive correlation was observed between ΔNT-proBNP (HR, 2.48; 95% CI, 1.27–4.83) and ΔQRS (HR, 1.32; 95% CI, 1.01–1.73) with all-cause mortality; whereas above the inflection point, a negative correlation was observed between ΔNT-proBNP (HR, 0.14; 95% CI, 0.02–0.94) and ΔQRS (HR, 0.24; 95% CI, 0.08–0.79) with all-cause mortality, as shown in Table 4.
Association between cardiac function changes and all-cause mortality.

Model I: adjusted for age, sex. Model II: adjusted for age, sex, BMI, NYHA class, AF, LBBB, hypertension, VT/VF, ACEI/ARB, and eGFR.
Each unit changed in LVEF is 5%.
Each unit changed in LVEDD is 5 mm.
Each unit changed in LAD is 5 mm.
NT-proBNP value is log2-transformed.
Each unit changed in QRS is 10 ms.
ACEI/ARB, angiotensin converting enzyme inhibitor/angiotensin receptor blocker; AF, atrial fibrillation; BMI, body mass index; CI, confidence interval; eGFR, estimated glomerular filtration rate; LAD, left atrial diameter; LBBB, left bundle branch block; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; NYHA, New York Heart Association; VT/VF, ventricular tachycardia/ventricular fibrillation.

Dose–response association of cardiac function changes with all-cause mortality and HF hospitalization in HF patients treated with CRT. (a–e) The relationship between change in LVEF, LVEDD, LAD, NT-proBNP, QRS duration, and all-cause mortality in patients with and without diabetes. (f–j) The relationship between change in LVEF, LVEDD, LAD, NT-proBNP, QRS duration, and HF hospitalization in patients with and without diabetes, respectively.
Association between changes in cardiac function within 6 months and HF hospitalization
During a mean follow-up of 3.3 years, 197 (29.6%) hospitalizations for HF occurred. In the general population, ΔLVEF was inversely associated with HF hospitalization (HR, 0.70; 95% CI, 0.62–0.78), while ΔLVEDD (HR, 1.62; 95% CI, 1.39–1.88), ΔLAD (HR, 1.20; 95% CI, 1.06–1.35), and ΔNT-proBNP (HR, 1.51; 95% CI, 1.30–1.75) were positively associated with HF hospitalization. However, there was no significant association between ΔQRS and HF hospitalization (p > 0.05). The negative association between ΔLVEF and HF hospitalization, and the positive associations between ΔLVEDD, ΔLAD, and HF hospitalization were consistent regardless of diabetes status [Table 3 and Figure 3(f) and (h)]. However, positive associations between ΔNT-proBNP and ΔQRS and HF hospitalization were only present in non-diabetic patients, not in diabetic patients (Table 3). In diabetic patients, the association between ΔNT-proBNP and HF hospitalization was non-linear, as shown in Figure 3(i), with a positive association observed when ΔNT-proBNP was less than the saturation point (HR, 1.81; 95% CI, 1.13–2.89) (Table 4). Similarly, a non-linear relationship was observed between ΔQRS and the risk of HF hospitalization in diabetic patients, with an HR (95% CI) of 1.41 (1.10–1.81) when ΔQRS was less than 0, and an HR (95% CI) of 0.36 (0.21–0.64) when ΔQRS was greater than or equal to 0 [Table 4 and Figure 3(j)].
Association between cardiac function changes and HF hospitalization.

Model I: adjusted for age, sex. Model II: adjusted for age, sex, BMI, NYHA class, AF, LBBB, hypertension, VT/VF, ACEI/ARB, and eGFR.
Each unit changed in LVEF is 5%.
Each unit changed in LVEDD is 5 mm.
Each unit changed in LAD is 5 mm.
NT-proBNP value is log2-transformed.
Each unit changed in QRS is 10 ms.
ACEI/ARB, angiotensin converting enzyme inhibitor/angiotensin receptor blocker; AF, atrial fibrillation; BMI, body mass index; CI, confidence interval; eGFR, estimated glomerular filtration rate; HF, heart failure; LAD, left atrial diameter; LBBB, left bundle branch block; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; NYHA, New York Heart Association; VT/VF, ventricular tachycardia/ventricular fibrillation.
Threshold effect analysis of changes of NT-proBNP and QRS duration on all-cause mortality and HF hospitalization in patients with diabetes.
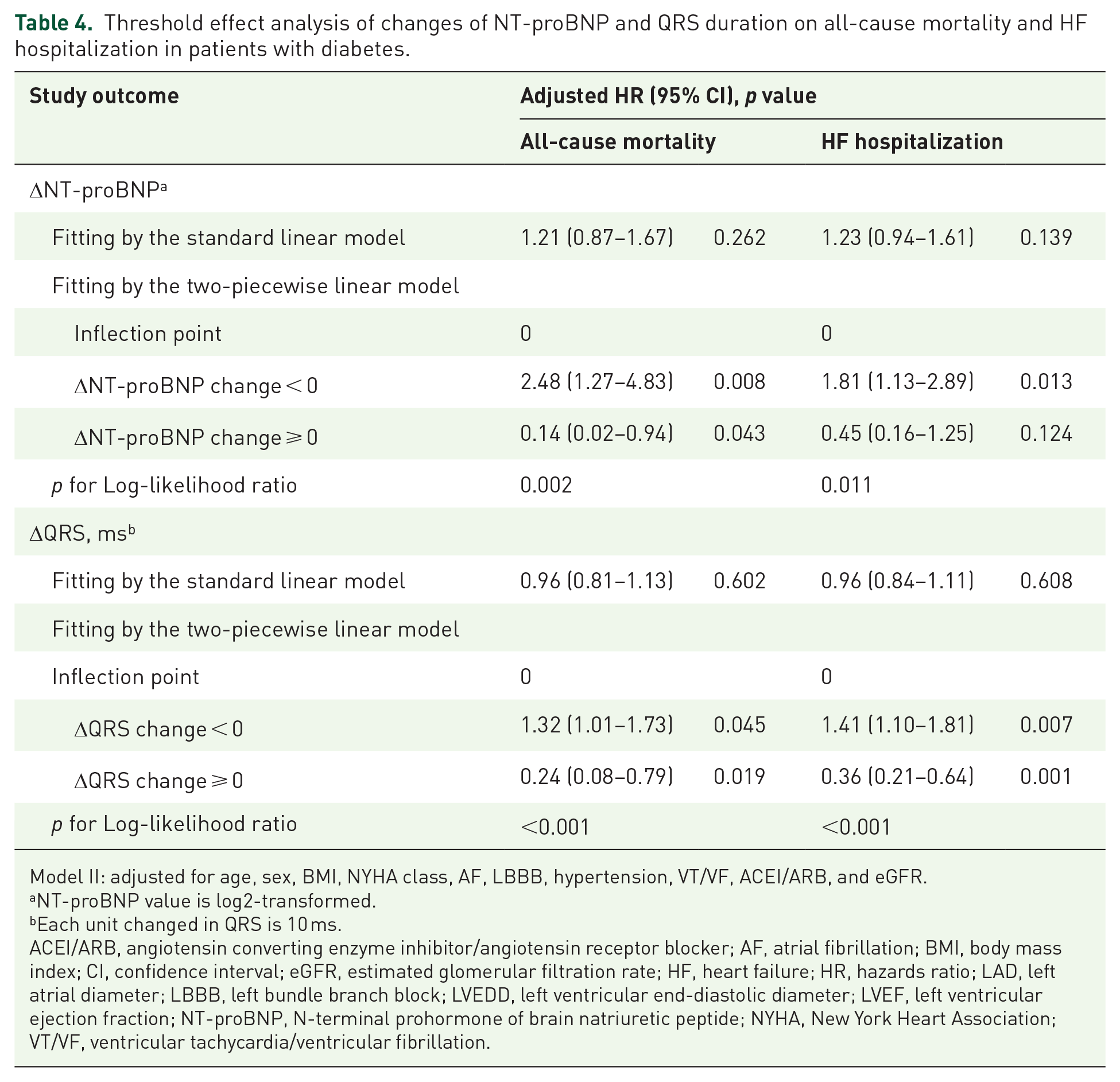
Model II: adjusted for age, sex, BMI, NYHA class, AF, LBBB, hypertension, VT/VF, ACEI/ARB, and eGFR.
NT-proBNP value is log2-transformed.
Each unit changed in QRS is 10 ms.
ACEI/ARB, angiotensin converting enzyme inhibitor/angiotensin receptor blocker; AF, atrial fibrillation; BMI, body mass index; CI, confidence interval; eGFR, estimated glomerular filtration rate; HF, heart failure; HR, hazards ratio; LAD, left atrial diameter; LBBB, left bundle branch block; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; NYHA, New York Heart Association; VT/VF, ventricular tachycardia/ventricular fibrillation.
Discussion
To our knowledge, this is the first study to investigate the effect of diabetes on association between short-term changes in cardiac function parameters and long-term adverse outcomes. Specifically, we observed that ΔLVEF within 6 months was negatively associated with poor prognosis, while ΔLVEDD, ΔLAD, and ΔNT-proBNP were positively associated with an increased risk of death and HF hospitalization in HF patients treated with CRT. However, we did not observe a significant association between ΔQRS and prognosis. The presence of diabetes had little impact on the prognostic value of ΔLVEF, ΔLVEDD, and ΔLAD, but the association between ΔNT-proBNP and ΔQRS and adverse outcomes differed between diabetic and nondiabetic patients.
Several studies have investigated the impact of short-term changes in cardiac function on long-term prognosis of HF patients treated with CRT. Solomon et al. 25 reported that a 5% increase in LVEF at 12 months significantly lowered the risk of death or hospitalization for HF. Stassen et al. 26 observed that atrial reverse remodeling at 6 months after CRT reduced the risk of death. Similarly, Yu et al. 27 found that a significant reduction in NT-proBNP at 3 months after CRT lowered the risk of all-cause death and HF hospitalization. However, Gold et al. 28 did not find a significant association between changes in QRS duration during follow-up and adverse outcomes in CRT patients. Although these studies provide important insights, their findings may be limited by small sample sizes, different follow-up periods, incomplete cardiac function parameters, and heterogeneity across studies. To address these limitations, we investigated the impact of changes in multiple cardiac parameters at 6 months on the long-term prognosis of HF patients treated with CRT. Our study demonstrated that a decrease in LVEF was inversely associated with poor outcomes, while increases in LVEDD, LAD, and NT-proBNP were positively associated with poor outcomes. Monitoring changes in multiple cardiac parameters could provide a more comprehensive assessment of the response to CRT and help clinicians identify patients who may require additional interventions or adjustments in treatment plans to improve their prognosis.
Given the high prevalence and negative impact of diabetes, examining its impact on short-term changes in cardiac function parameters is crucial for understanding diabetes-related therapeutic management in HF patients treated with CRT. 29 However, previous studies have reported conflicting results regarding the effect of diabetes on short-term cardiac function changes in HF patients treated with CRT. Kahr et al. 30 found no significant impact of diabetes on LVEF improvement in 418 CRT patients (25.1% with diabetes) followed up for 4.8 years, but noted a significant difference in left ventricular remodeling between those with and without diabetes. However, Sardu et al. 31 found that diabetes status failed to affect the change trajectory of LVEF and LVEDD in 72 elderly CRT patients (44.4% with diabetes) followed up for 1 year. To address the inconsistent findings among previous studies, we conducted a larger study (24.9% with diabetes) examining the effect of diabetic status on changes in multiple cardiac function parameters. Our study suggests that diabetes may negatively impact the short-term changes in LVEF and NT-proBNP in HF patients treated with CRT. Therefore, clinicians should consider effects of diabetes when evaluating the response to CRT and devising treatment strategies.
As diabetes is a chronic disease, it can significantly impact the long-term prognosis of HF patients treated with CRT. However, prior studies have yielded inconsistent findings on the effect of diabetes on the long-term outcomes of HF patients receiving CRT, possibly due to differences in study populations, follow-up duration, and clinical response evaluation.14,32,33 To optimize treatment strategies, it is crucial to continue investigating the impact of diabetes on the long-term prognosis of HF patients treated with CRT using various cardiac function parameters. Our study found no significant difference between diabetic and nondiabetic patients in the relationship between ΔLVEF, ΔLVEDD, ΔLAD, and the risk of all-cause mortality and HF hospitalization. However, the presence of diabetes significantly modified the association between ΔNT-proBNP and ΔQRS and poor prognosis. Among nondiabetic patients, ΔNT-proBNP and ΔQRS were positively associated with adverse outcomes, while in diabetic patients, a non-linear relationship with a saturation value was observed. Clinicians should consider the impact of diabetes in their clinical decision-making when using ΔNT-proBNP and ΔQRS to predict the long-term prognosis of CRT patients. Further investigation is needed to confirm these findings and better understand the non-linear relationship between ΔNT-proBNP, ΔQRS, and outcomes in diabetic patients.
In our cohort, the observed higher eGFR may reflect the general population of patients receiving CRT at our center. However, it’s important to note that the inclusion of patients in our study was not based on their renal function, and CRT implantation was performed according to the prevailing guidelines and individual clinical assessments. 19 After adjusting for eGFR, the associations between short-term cardiac function changes post-CRT and long-term prognosis remained consistent. This suggests that the observed associations in our study are robust and not significantly affected by renal function. Furthermore, previous studies have also demonstrated that CRT can improve renal function, which in turn, may contribute to the observed improvements in cardiac function and long-term prognosis post-CRT. 34 Our findings align with these observations and provide further evidence on the beneficial effects of CRT in HF patients, irrespective of their renal function at baseline. Unfortunately, we did not collect data on glucose levels, which is a limitation of our study. The diagnosis of diabetes was based on the patients’ medical records from the hospital electronic system. Previous studies have highlighted the adverse effects of elevated glucose levels on cardiac function and outcomes in HF patients.35,36 Hyperglycemia, a hallmark of uncontrolled diabetes, can induce oxidative stress, inflammation, and endothelial dysfunction, which may exacerbate cardiac dysfunction and contribute to adverse outcomes. 37 The relationship between glucose control, diabetes status, and CRT outcomes warrants further investigation in future studies to provide a more comprehensive understanding of the potential benefits of CRT in this population.
Our findings may provide a possible explanation for previously inconsistent conclusions regarding the relationship between ΔQRS changes and long-term prognosis, as the presence of diabetes may attenuate the effect of ΔQRS on prognosis. Given the complexity of cardiac function assessment, multiple indexes should be considered since their predictive value for long-term prognosis may differ, particularly in the presence of diabetes. Additionally, while several studies have delved into the relationship between cardiac function parameters and outcomes in HF patients receiving CRT, our research uniquely highlights the differential impact of diabetes on these associations.38,39 This novel insight underscores the need for personalized therapeutic strategies and monitoring protocols for diabetic HF patients undergoing CRT. Furthermore, our findings pave the way for future research to elucidate the underlying mechanisms driving the non-linear relationship between ΔNT-proBNP, ΔQRS, and outcomes in the diabetic population.
The mechanisms linking changes in cardiac function parameters to poor outcomes are complex and multifactorial. A decrease in LVEF indicates a decline in systolic function, increasing the risk of mortality and HF-related hospitalization. 40 Conversely, an increase in LVEDD and LAD suggests left atrial and ventricular remodeling and elevated filling pressure, respectively, contributing to HF progression and an elevated risk of adverse outcomes. 41 The impact of diabetes on the relationship between ΔNT-proBNP and ΔQRS and poor prognosis may be related to diabetes-induced cardiac dysfunction and remodeling. 42 Diabetes has been linked to increased oxidative stress, cardiomyocyte hypertrophy, inflammation, and fibrosis, all of which can contribute to HF progression and adverse outcomes. 43 These factors significantly impact the synthesis and secretion of NT-proBNP by ventricular myocytes, as it is induced by ventricular overload or dilation. 44 Additionally, long-term diabetes may result in diabetic cardiomyopathy, which may manifest as cardiac autonomic remodeling and abnormal cardiac conduction, leading to significant QRS duration widening. 45
Our study’s clear strength is investigating the trajectories of multiple cardiac function parameters and their relationship with long-term outcomes. Furthermore, we were the first to demonstrate differences in the association between NT-proBNP and QRS changes and long-term outcomes in diabetic and non-diabetic patients. However, our study has several limitations. First, our study is retrospective in nature, limiting the extent to which we can establish causality between the observed short-term cardiac function changes and long-term outcomes. Additionally, the retrospective design also poses the risk of confounding variables that may have influenced the results. Second, we acknowledge that the study included a small proportion of diabetic patients relative to non-diabetic patients undergoing CRT. This imbalance could potentially limit the generalizability of our findings to the broader diabetic population with HF undergoing CRT. Third, we did not record the use of hypoglycemic drugs, which may have impacted our results. Additionally, potential imbalances in baseline characteristics could exist due to the non-randomized controlled trial design. Last, the generalizability of our findings is limited as the study was conducted in a single medical center. Therefore, future multicenter prospective studies are recommended to confirm our findings.
Conclusion
The study emphasizes the significance of short-term changes in cardiac function as predictors of long-term prognosis in HF patients following CRT. Changes in ΔLVEF, ΔLVEDD, and ΔLAD within 6 months were equally effective predictors of long-term outcomes in both diabetic and non-diabetic patients. However, diabetes may disrupt the linear association between ΔNT-proBNP and ΔQRS duration, resulting in unfavorable outcomes with saturation values in diabetic patients. Further investigation is needed to confirm these findings and better understand the non-linear relationship between ΔNT-proBNP, ΔQRS, and outcomes in diabetic patients.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231223285 – Supplemental material for Effect of short-term cardiac function changes after cardiac resynchronization therapy on long-term prognosis in heart failure patients with and without diabetes
Supplemental material, sj-docx-1-taj-10.1177_20406223231223285 for Effect of short-term cardiac function changes after cardiac resynchronization therapy on long-term prognosis in heart failure patients with and without diabetes by Yu Yu, Ligang Ding, Hao Huang, Sijing Cheng, Yu Deng, Chi Cai, Min Gu, Xuhua Chen, Hongxia Niu and Wei Hua in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
