Abstract
Background:
The factors affecting cardiovascular risk associated with vascular calcification in patients with chronic kidney disease are less well addressed. Distinct risk factors may contribute synergistically to this elevated cardiovascular risk in this population.
Objectives:
We aimed to determine whether echocardiographic left ventricular hypertrophy (LVH) affects the risk of major adverse cardiac events (MACE) associated with vascular calcification in end-stage kidney disease (ESKD) patients.
Methods:
In this retrospective cohort study, ESKD patients underwent chest radiography and echocardiography to assess aortic arch calcification (AoAC) and LVH, respectively, and were classified into three groups accordingly: non-to-mild AoAC without LVH, non-to-mild AoAC with LVH, and moderate-to-severe AoAC. The risks of MACE, cardiovascular mortality, and overall mortality were assessed using Cox proportional hazard analysis.
Results:
Of the 283 enrolled ESKD patients, 44 (15.5%) had non-to-mild AoAC without LVH, 117 (41.3%) had non-to-mild AoAC with LVH, and 122 (43.1%) had moderate-to-severe AoAC. After 34.1 months, 107 (37.8%) participants developed MACE, including 6 (13.6%), 40 (34.2%), and 61 (50%) from each respective group. Those with moderate-to-severe AoAC (Hazard ratio, 3.72; 95% confidence interval, 1.58–8.73) had a significantly higher risk of MACE than did those with non-to-mild AoAC without LVH or with non-to-mild AoAC and LVH (Hazard ratio, 2.73; 95% confidence interval, 1.16–6.46). A similar trend was observed for cardiovascular and overall mortality.
Conclusion:
Echocardiographic LVH could modify the risk of adverse cardiovascular events associated with vascular calcification in ESKD patients. Interventions aiming to ameliorate both morbidities might be translated into a lower MACE risk in this population.
Keywords
Introduction
Chronic kidney disease (CKD) is common worldwide, affecting approximately 11% of the global population. 1 Patients with CKD and particularly end-stage kidney disease (ESKD) are at a significantly higher risk of cardiovascular disease compared to the general population. This elevated cardiovascular risk emerges early during microalbuminuria and increases as the glomerular filtration rate (GFR) declines. A systematic meta-analysis reports that the risk of stroke increases linearly with lower GFR and higher albuminuria, with both indices showing additive and independent risk-increasing influences. 2 The risk for coronary heart disease similarly rises significantly when the GFR drops below 60 mL/min/1.73 m2 and increases dose-dependently in patients with increasing albuminuria.3,4 The mechanism underlying the cardiovascular risk associated with CKD remains under active investigation. Traditional morbidities (e.g. hypertension, diabetes mellitus, dyslipidemia) and the influences associated with pervasive uremic toxin exposure are the most renowned causes of such risk, but studies have shown that unhealthy dietary preferences 5 and complications such as untreated metabolic acidosis 6 and secondary hyperparathyroidism 7 might also increase this risk. Of the CKD-associated complications, chronic kidney disease-mineral bone disorder (CKD-MBD) is undoubtedly an important contributor to the elevated risk of cardiovascular events in these patients. One study revealed that CKD-MBD may account for an important share of the residual cardiovascular risk in patients with and even without CKD. 8
Vascular calcification is a manifestation of ectopic calcification caused by CKD-MBD and is an integral part of the bone-vascular axis of derangement that occurs with uremia. A meta-analysis reports that the prevalence of vascular calcification is as high as 60% among CKD patients. 9 The pathogenesis of vascular calcification in CKD involves the adoption of an osteoblast-like phenotype by vascular smooth muscle cells, the main constituent of the vasa media. 10 Multiple predisposing factors, including hyperphosphatemia, chronic inflammation, and oxidative stress as well as the loss of anti-calcific defenses facilitates the initiation and propagation of vascular calcification during CKD.11,12 From a clinical perspective, vascular calcification in patients with any stage of CKD increases the risk of cardiovascular events and cardiovascular mortality by nearly 2- and 4-fold, respectively. 9 Severe vascular calcification in CKD increases the cardiovascular risk by as much as 20%. 13
Despite advancements in our understanding of the clinical importance of vascular calcification, few studies have addressed factors that modulate the cardiovascular risks exerted by vascular calcification in CKD patients. Furthermore, multiple cardiovascular risk factors, including vascular calcification, likely contribute synergistically to patient risk, as observed in a recent population-based study. 14 CKD frequently induces left ventricular mass growth due to excessive neurohormonal stimuli and other accompanying morbidities. Left ventricular hypertrophy (LVH) is a widely recognized predictor of adverse cardiovascular prognosis in CKD patients. These findings suggest that LVH acts as an occult modifier of cardiovascular risk associated with vascular calcification in these patients. This hypothesis has not been affirmed by others, especially in those with CKD. Moreover, existing studies frequently used non-echocardiographic methods to assess LVH, such as electrocardiogram, 15 whose results might not be accurate. We hypothesized that echocardiography-identified LVH modifies the cardiovascular risk posed by vascular calcification in patients with ESKD, the most severe form of CKD. We examined this hypothesis in a prospectively enrolled patient cohort.
Methods
Participant enrollment
This study was done based on a retrospective analysis of a prospective cohort, with details described previously. 16 Patients with ESKD receiving chronic hemodialysis, defined as an estimated GFR (eGFR) <15 mL/min/1.73 m receiving hemodialysis for more than 3 months, were identified from the hemodialysis unit of Shin Kong Wu Ho-Su Memorial Hospital, a medical center in Northern Taiwan, between 1 October 2018 and 31 December 2018 (Figure 1). Patients who received posteroanterior chest roentgenography and echocardiography examination on the mid-weekly non-dialysis day during the enrollment period were included, according to our prior study. 16 Inter-operator differences in echocardiography results were found to be low in this institute. ESKD patients were excluded if they received hemodialysis via tunneled cuff catheters, as left ventricular hypertrophy, which is influenced by the type of vascular access 17 and patients with tunneled cuff catheters, who were more likely to be incident patients with a short dialysis vintage and having heterogeneous mortality risk determinants, including infection. 18 In addition, we would like to enrich those with a high cardiovascular risk through increasing LVH prevalence, and thought that including only those without dialysis catheter might decrease the influences of catheter infection-related outcome events during subsequent analyses.
Participant selection process.
We subsequently divided participants into groups according to the severity of aortic arch calcification (AoAC) and the LVH status as determined by echocardiography. Using a severity classification scheme established and validated previously, 12 AoAC was categorized as follows: score range, 0–3; 0, none; 1, mild; 2, moderate; and 3, severe. Echocardiographic LVH (UCG-LVH) was defined as a left ventricular mass (standardized by body surface area) >95 g/m2 for females and >115 g/m2 for males, as recommended by the American Society of Echocardiography. 19 Participants were initially divided into four groups (AoAC <2 without UCG-LVH; AoAC <2 with UCG-LVH; AoAC ⩾2 without UCG-LVH; and AoAC ⩾2 with UCG-LVH) but later were re-assigned into three groups [none to mild AoAC (AoAC <2) without UCH-LVH; none to mild AoAC (AoAC <2) with UCH-LVH; and moderate-to-severe AoAC (AoAC ⩾2) regardless of UCG-LVH status] because very few patients (5.6%) had AoAC ⩾ 2 without UCG-LVH. We collected their demographic data, comorbidity status, dialysis profiles (duration and clearance efficacy), laboratory parameters, and concurrent medications after enrollment. Other echocardiographic parameters were documented during the examinations. Participants were followed up until the occurrence of designated outcomes or 31st December 2020, whichever came first.
Outcome definition
The primary endpoint of this study was the occurrence of a major adverse cardiac event (MACE), defined as any of the following: myocardial infarction, coronary revascularization, stroke, hospitalization for heart failure, or death from cardiovascular causes. Two secondary endpoints, cardiovascular mortality and all-cause mortality, were also examined. The cause of death and identity of cardiovascular events were determined by independent physicians unaware of the study design.
Statistical analysis
Continuous variables, are reported as the mean ± standard deviations. Categorical variables are reported as numbers with percentages. Continuous variables were compared between the groups using Student’s t-test for normal distribution or the Mann–Whitney U-test for skewed distributions. Categorical variables were compared between groups using the Chi-square or Fisher’s exact test. The one-way analysis of variance (ANOVA) or the Kruskal–Wallis test was used to compare more than two groups.
Demographic data, comorbidity profiles, laboratory parameters, and medication use were compared between the three groups. Univariate analysis then was used to compare baseline variables between participants with and without MACE (primary endpoint). Cox proportional hazard regression was then conducted to evaluate the association between AoAC/UCG-LVH severity and the risk of primary and secondary outcomes. Statistical analyses were conducted using SPSS version 19 (IBM Inc., Statacorp LLC, Texas, USA). In all analyses, p < 0.05 was deemed statistically significant.
Results
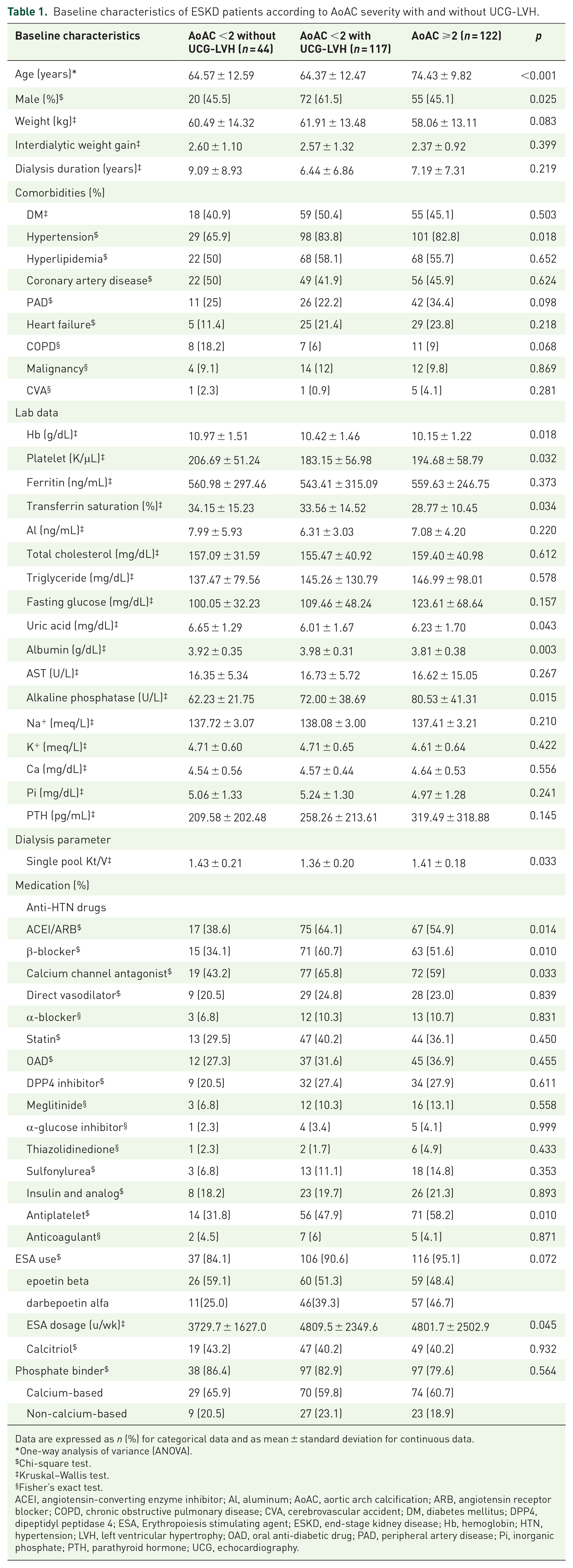
A total of 313 patients with ESKD receiving chronic hemodialysis was screened, and 30 (9.6%) were excluded based on the exclusion criteria (Figure 1). The final cohort included 44 patients (15.5%) with AoAC score <2 without UCG-LVH, 117 (41.3%) with AoAC score <2 with UCG-LVH, and 122 (43.1%) with AoAC score ⩾2 (Figure 1). ESKD patients with AoAC score ⩾2 were significantly older than those with AoAC score <2, regardless of UCG-LVH status (p < 0.001). The prevalence of males was highest among those with AoAC score <2 with UCG-LVH (p = 0.025; Table 1). The group with AoAC score <2 without UCG-LVH had the lowest prevalence of hypertension (p = 0.018). Regarding laboratory profiles, participants with AoAC score ⩾2 had the lowest hemoglobin (p = 0.018), transferrin saturation (p = 0.034), and albumin (p = 0.003) levels but the highest alkaline phosphatase levels (p = 0.015; Table 1). Participants with AoAC score <2 without UCG-LVH had significantly higher dialysis clearance (p = 0.033) than did those in the other two groups. Participants with AoAC score ⩾2 and those with AoAC score < 2 with UCG-LVH were more likely to use renin-angiotensin system inhibitors (RASi; p = 0.014), β-blockers (p = 0.01), calcium channel antagonists (p = 0.033), and antiplatelet agents (p = 0.01) than those with an AoAC score <2 without UCG-LVH (Table 1).
Baseline characteristics of ESKD patients according to AoAC severity with and without UCG-LVH.
Data are expressed as n (%) for categorical data and as mean ± standard deviation for continuous data.
One-way analysis of variance (ANOVA).
Chi-square test.
Kruskal–Wallis test.
Fisher’s exact test.
ACEI, angiotensin-converting enzyme inhibitor; Al, aluminum; AoAC, aortic arch calcification; ARB, angiotensin receptor blocker; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; DM, diabetes mellitus; DPP4, dipeptidyl peptidase 4; ESA, Erythropoiesis stimulating agent; ESKD, end-stage kidney disease; Hb, hemoglobin; HTN, hypertension; LVH, left ventricular hypertrophy; OAD, oral anti-diabetic drug; PAD, peripheral artery disease; Pi, inorganic phosphate; PTH, parathyroid hormone; UCG, echocardiography.
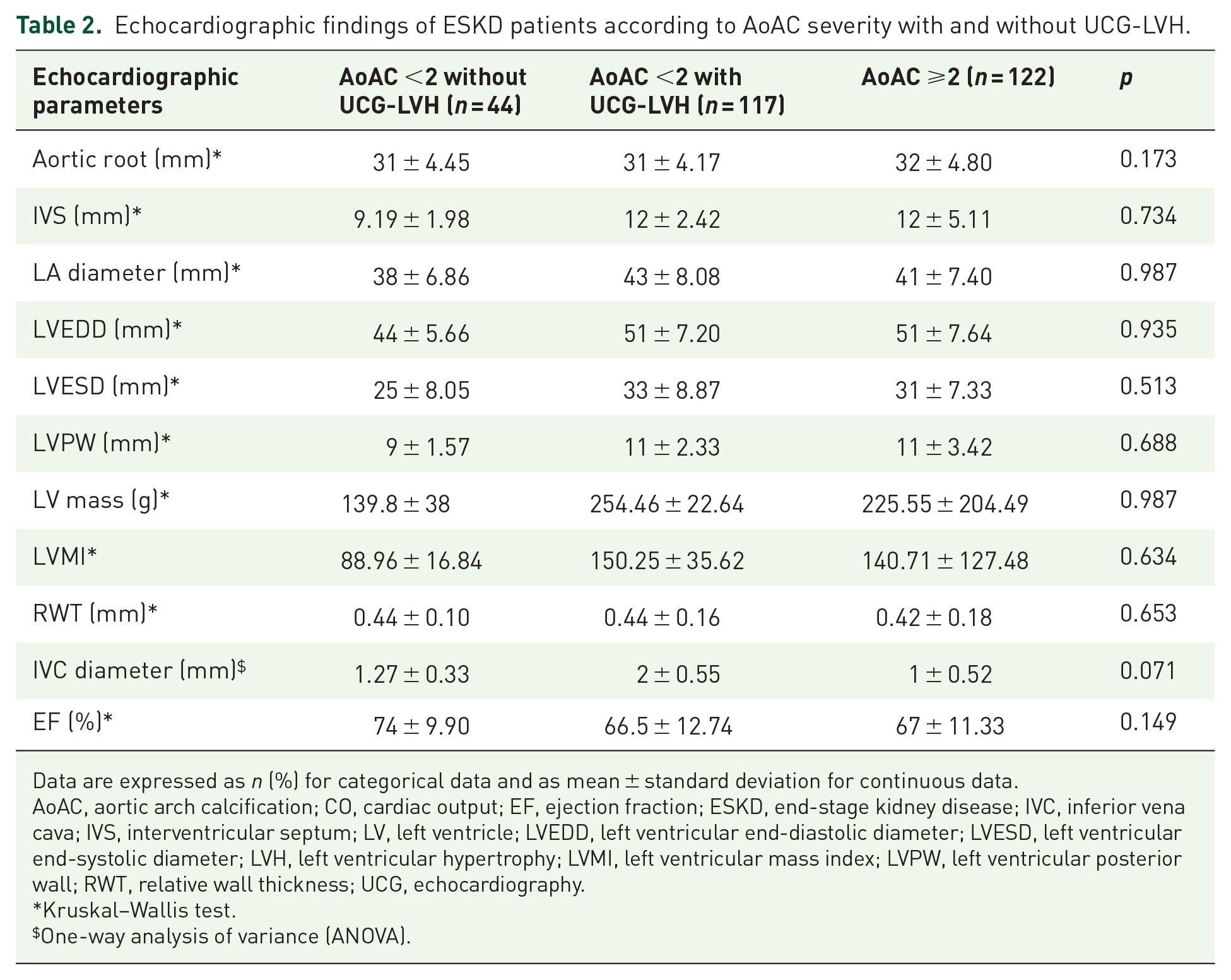
Regarding echocardiographic findings, no significant difference was observed between the three groups in most structural parameters, including the left ventricular chamber size and wall thickness and aortic root/vena cava diameters (Table 2). The ejection fraction also did not differ between the groups (Table 2).
Echocardiographic findings of ESKD patients according to AoAC severity with and without UCG-LVH.
Data are expressed as n (%) for categorical data and as mean ± standard deviation for continuous data.
AoAC, aortic arch calcification; CO, cardiac output; EF, ejection fraction; ESKD, end-stage kidney disease; IVC, inferior vena cava; IVS, interventricular septum; LV, left ventricle; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; LVH, left ventricular hypertrophy; LVMI, left ventricular mass index; LVPW, left ventricular posterior wall; RWT, relative wall thickness; UCG, echocardiography.
Kruskal–Wallis test.
One-way analysis of variance (ANOVA).
Factors influencing the risk of developing pre-specified outcomes
After nearly 3 years (34.1 ± 10 months) of follow up, 107 participants (37.8%) developed MACE: 6 (13.6%) of those with AoAC score <2 without UCG-LVH, 40 (34.2%) with AoAC score <2 and UCG-LVH, and 61 (50%) with a AoAC score ⩾2. Participants with an AoAC score <2 without UCG-LVH had the lowest risk of MACE, followed by those with an AoAC score <2 with UCG-LVH and those with an AoAC score >2 [p < 0.001; Figure 2(a)]. Regarding secondary outcomes, 59 (20.8%) participants died from cardiovascular causes and 76 (26.9%) from any cause. Three (6.8%), 20 (17.1%), and 36 (29.5%) died from cardiovascular causes, while 6 (13.6%), 24 (20.5%), and 46 (37.7%) died from any cause in those with an AoAC score <2 without UCG-LVH, with an AoAC score <2 with UCG-LVH, and with an AoAC score ⩾2, respectively. The same trend of rising cardiovascular mortality [p = 0.002; Figure 2(b)] and overall mortality [p < 0.001; Figure 2(c)] with greater AoAC severity and LVH was noted.
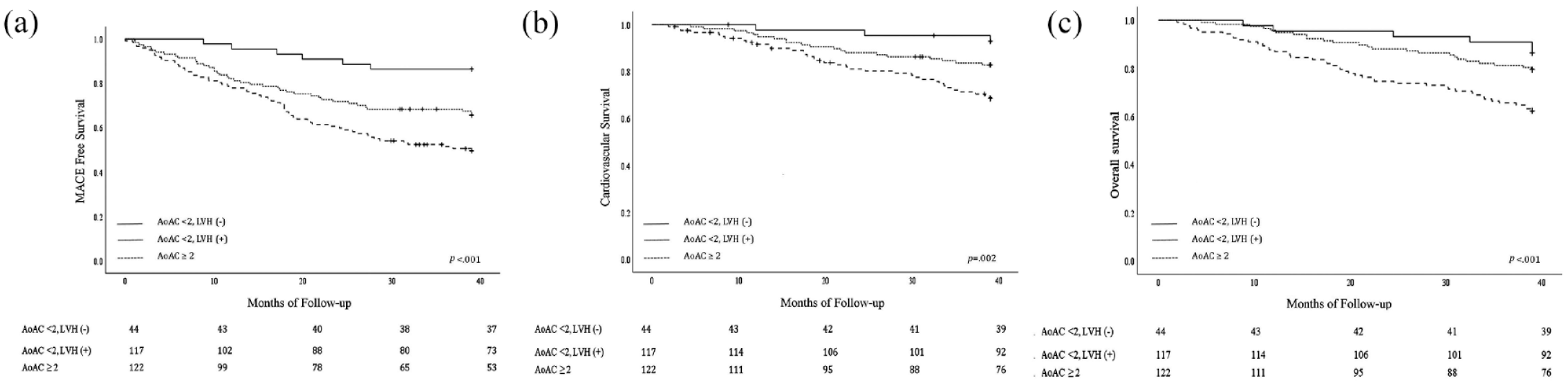
Event-free curves according to aortic arch calcification severity and left ventricular hypertrophy status (a) MACE-free curves (b) Cardiovascular survival curves (c) Overall survival curves.
We next performed univariate analysis to identify potential factors influencing the risk of MACE during follow-up in these ESKD patients (Table 3). Those who developed MACE during follow-up had significantly higher age (p = 0.002), a higher prevalence of diabetes mellitus (p = 0.001), hypertension (p = 0.006), coronary artery disease (p = 0.014), peripheral artery disease (p = 0.026), and heart failure (p = 0.043) than did those without. Those developing MACE had significantly lower total cholesterol (p = 0.003) and fasting glucose (p = 0.032) but higher serum albumin (p < 0.001) than those who did not. Dialysis clearance was somewhat lower in those with MACE than those without (Table 3). The medications taken also did not differ significantly between those with and without MACE except for analogs (p < 0.001) and antiplatelet drugs (p < 0.001).
Clinical characteristics of ESKD patients according to primary end-point status.
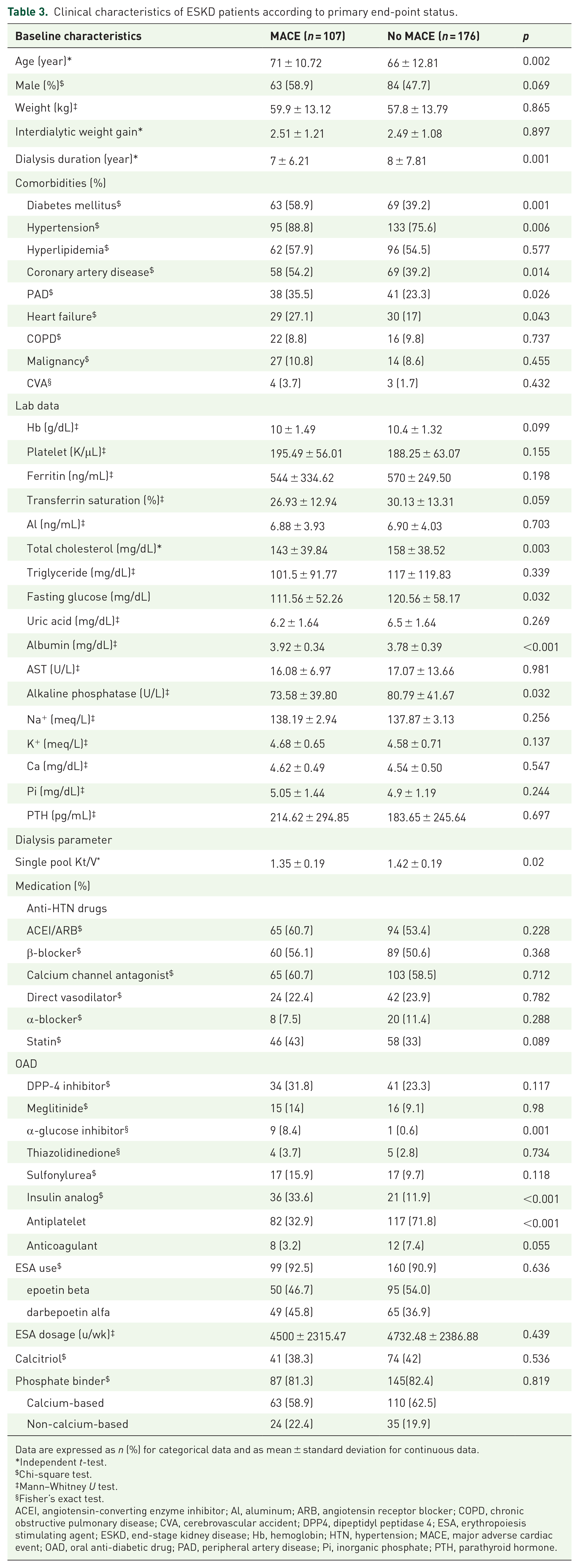
Data are expressed as n (%) for categorical data and as mean ± standard deviation for continuous data.
Independent t-test.
Chi-square test.
Mann–Whitney U test.
Fisher’s exact test.
ACEI, angiotensin-converting enzyme inhibitor; Al, aluminum; ARB, angiotensin receptor blocker; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; DPP4, dipeptidyl peptidase 4; ESA, erythropoiesis stimulating agent; ESKD, end-stage kidney disease; Hb, hemoglobin; HTN, hypertension; MACE, major adverse cardiac event; OAD, oral anti-diabetic drug; PAD, peripheral artery disease; Pi, inorganic phosphate; PTH, parathyroid hormone.
In unadjusted Cox proportional hazard regression analyses, ESKD patients with an AoAC score <2 with UCG-LVH [hazard ratio (HR), 2.94; 95% confidence interval (CI), 1.25–6.93] and those with an AoAC score ⩾2 (HR, 4.76; 95% CI, 2.06–11.02) had an elevated risk of developing MACE during follow-up compared to those with an AoAC score <2 without UCG-LVH (Table 4). After adjusting for age and sex, those with an AoAC score <2 and UCG-LVH (HR, 2.73; 95% CI, 1.16–6.46) and those with an AoAC score ⩾2 (HR, 3.72; 95% CI, 1.58–8.73) still had a significantly higher risk of developing MACE compared to those with an AoAC score <2 without UCG-LVH (model 1; Table 4). The results remained essentially the same after additional adjustment for relevant medications and laboratory data (model 2; Table 4).
Cox proportional hazard analysis of factors determining the risk of primary and secondary outcomes.

ACEI, angiotensin-converting enzyme inhibitor; AoAC, aortic arch calcification; ARB, angiotensin receptor blocker; CI, confidence interval; CV, cardiovascular; ESA, erythropoiesis stimulating agent; HR, hazard ratio; LVH, left ventricular hypertrophy; MACE, major adverse cardiac event; UCG, echocardiography.
Adjusted for age, sex.
Adjusted for age, sex, hypertension, Hb, platelet, transferrin saturation, uric acid, albumin, alkaline phosphate, single pool Kt/V, ACEI/ARB, ß -blocker, calcium channel antagonist, antiplatelet use, ESA dosage.
Cox proportional hazard regression analysis of secondary outcomes showed that ESKD patients with an AoAC score ⩾2 (HR, 5.19; 95% CI, 1.59–16.86) had a significantly higher risk of cardiovascular mortality compared to those with an AoAC score <2 without UCG-LVH (Table 4). These associations remained after accounting for age and sex [CV mortality, model 2 (Table 4)]. ESKD patients with an AoAC score ⩾2 (HR, 3.32; 95% CI, 1.42–7.77) also had a significantly higher risk of all-cause mortality compared to those with an AoAC score <2 without UCG-LVH (Table 4), although the association did not persist after adjusting for other factors.
Discussion
In this study examining the influence of vascular calcification on the risk of MACE and mortality in a group of ESKD patients, we found that those with more severe vascular calcification together with UCG-LVH exhibited the highest risk of MACE, followed by those with less severe vascular calcification with UCG-LVH. Similar trends were observed for cardiovascular mortality and overall mortality, although the relationship was less prominent, likely due to lower event numbers during the 3 years of follow-up. Based on these findings, we believe it prudent to consider the adverse cardiovascular influences jointly exerted by vascular calcification and LVH in ESKD patients to offer appropriately targeted management strategies to mitigate these effects.
Previous studies have addressed the combinatorial effect of vascular calcification and LVH on patient outcomes in the general population and older adults. In a study investigating the effect of thoracic aortic calcification in a group of older adults with UCG-LVH on outcomes, Cho et al. 20 found those with LVH and a thoracic aortic calcification score above the median had a 4.51-fold greater risk of MACE after nearly 5 years of follow-up. Similarly, Grossman et al. 21 reported that hypertensive patients with UCG-LVH and coronary calcification had a 4.4-fold greater risk of cardiovascular events than did those with neither condition, while those with UCG-LVH alone were not at risk. A larger population-based study showed that individuals with AoAC as assessed by chest radiography and electrocardiogram-assessed LVH had a 1.8-fold higher risk of cardiovascular events after >10 years of follow-up. 14 This study also reports that LVH alone did not correlate with elevated cardiovascular risk, while AoAC did. Similar studies have been performed in cohorts of ESKD patients under hemodialysis, but echocardiography was rarely used to assess LVH. In a small cohort of ESKD patients, Kamiura et al. 22 reported that thoracic aortic calcification but not LVH predicted cardiovascular events. However, another study suggested that only the concurrent presence of aortic calcification and LVH predicted a 1.8- to 2-fold higher risk of cardiovascular events and mortality, while each feature alone did not. 23 Interestingly, vascular calcification, but not LVH, consistently predicts a higher cardiovascular risk in the non-CKD general population.14,20,21 In contrast, the correlation between vascular calcification and cardiovascular risk was not so consistent among ESKD patients.22,23 From this perspective, we add to the existing literature reporting that vascular calcification is a strong predictor of cardiovascular risk in ESKD patients and that LVH likely plays a modulatory role in this risk elevation (Table 4). Moreover, we observed that within a prolonged follow-up period, vascular calcification and LVH in combination conferred a 3- to 4-fold higher cardiovascular risk compared to ESKD patients without either of these conditions. Our results are closer to those derived from a population-based study, 14 although the clinical setting differed.
The question of how LVH influences cardiovascular risk can be approached from several perspectives. LVH is a cardiac adaptation to rising pressure or volume workload and is associated with anemia, hypertension, and hypervolemia in CKD patients. LVH predisposes these patients to subsequent heart failure via demand/supply mismatch and myoischemia, which underlie the elevated risk of coronary occlusion in CKD patients. 24 One possibility is that LVH introduces independent cardiovascular risks that are independent of the conventional risk factors in these patients. Indeed, a Japanese study showed that the addition of electrocardiographic LVH to cardiovascular prediction significantly improved outcome stratification according to the Framingham 10-year risk score in CKD patients. 25 Alternatively, LVH may be an indicator of other occult risk factors in these patients, including but not limited to the abovementioned LVH causal factors. Indeed, the prevalence of hypertension and hemoglobin concentrations differed significantly between participants with an AoAC score <2 with and without LVH (Table 1). The latter possibility is also supported in part by the reported lack of association between LVH and the risk of cardiovascular events in ESKD patients22,23 and a recent meta-analysis concluding that interventions to ameliorate LVH did not consistently yield cardiovascular benefits. 26 Regardless of the underlying mechanism, LVH is a strong predictor of cardiovascular risk in CKD patients, and we believe that LVH may be better viewed as a cardiovascular-risk subdivider instead of a main-outcome–stratifying variable.
Previous studies have identified factors that influence the risk of cardiovascular events posed by vascular calcification. Watanabe et al. 27 showed that the inclusion of radiographic pericardiac fat as an indicator in addition to vascular calcification improved cardiovascular risk prediction in patients with at least 1 risk factor. They argued that pericardial fat served as a marker for visceral fat, metabolic syndrome, or dyslipidemia; a greater amount of visceral fat or a greater degree of metabolic syndrome might be indicative of more severe inflammation that precipitates future cardiovascular events. 28 In this study, we observed that the presence of LVH increased the cardiovascular risk posed by vascular calcification (Table 4). Vascular calcification is proposed to cause LVH by chronically increasing vascular stiffness and cardiac afterload. 29 The emergence of LVH, therefore, may represent a maladaptive cardiac response to the downstream pathophysiology of vascular calcification, culminating in a potential vicious cycle. Consequently, LVH may indicate a more aberrant response to vascular calcification, further increasing the future risk of cardiovascular events.
Our findings may have clinical importance. Therapeutic strategies that aim to reduce LVH severity include lifestyle modification (more exercise and weight reduction), dietary salt reduction, and anti-hypertensive medication use (especially RASi). These strategies may carry additional cardiovascular benefits in patients with CKD and more severe vascular calcification by lowering the residual risk associated with vascular calcification. Further, clinically available therapies for vascular calcification remain scant and the existing candidate compound may not be widely accepted. 11
Our study has strengths and limitations. We used echocardiography to identify LVH in patients with ESKD, a more sensitive modality that reveals more subtle degrees of LVH compared to electrocardiography. 30 Our findings fill the knowledge gap in the existing literature, which does not adequately clarify the outcome-modifying role of LVH in cardiovascular risk estimation in patients with ESKD. An extensive array of variables was collected and analyzed, lending support to the validity of our findings. However, several limitations still remain. A larger sample size is needed to subdivide participants into subgroups based on additional features in future studies. We mainly focused on patients with dialysis-dependent ESKD, and whether the results are applicable to those with non-dialysis CKD is unclear. We identified only ESKD patients who did not use dialysis catheter for treatment in this study, and this enrichment approach might risk losing a proportion of candidates with very poor vasculature and elevated vascular risk. Nonetheless, the potentially increased risk of infection-related mortality might be lessened, somewhat justifying our strategy. Vascular calcification was assessed using chest radiography, but computed tomography may provide more precise measurements than plain radiography. More studies are needed to validate and expand our findings.
Conclusion
In a cohort of ESKD patients, we found that LVH increased the risk of adverse cardiovascular events associated with vascular calcification but not to the degree exerted by vascular calcification. A similar trend in increasing risk was observed for cardiovascular mortality and overall mortality, although our estimates might be compromised by fewer events. Our findings indicate that we should place emphasis on the proper management of vascular calcification and LVH in this population to reduce their future risk of adverse cardiovascular events.
