Abstract
Frailty exhibits diverse influences on health-related outcomes and represents a surrogate of increased susceptibility to harmful injuries. Patients with chronic kidney disease (CKD) are at a higher risk of accelerated biologic aging, and, in this population, the concept of frailty emerges as an instrumental measurement of physiologic reserves. However, a comprehensive description of known independent contributors to, and risk associates of, frailty in these patients remain unavailable. In the present review, original studies up to 28 February 2019 that assessed frailty in patients with all stages of CKD were retrieved and reviewed, with results extracted and summarized. By pooling 62 original investigations, 58.1% and 49.1% used cohort and cross-sectional designs, respectively. Dialysis-dependent end-stage renal disease patients (n = 39; 62.9%) were the most commonly examined population, followed by those with nondialysis CKD (n = 12; 19.4%) and those receiving renal transplantation (n = 11; 17.7%). Contributors to frailty in CKD patients included sociodemographic factors, smoking, CKD severity, organ-specific comorbidities, depression, hypoalbuminemia, and low testosterone levels. Conversely, the development of frailty was potentially associated with the emergence of cardiometabolic, musculoskeletal, and cerebral complications; mental distress; and a higher risk of subsequent functional and quality-of-life impairment. Moreover, frailty in CKD patients increased healthcare utilization and consistently elevated mortality among affected ones. Based on the multitude of contributors to frailty and its diverse health influences, a multifaceted approach to manage CKD patients with frailty is needed, and its potential influences on outcomes besides mortality need to be considered.
Keywords
Frailty: an ever-evolving concept with pleiotropic influences
Since the inception of the concept of frailty in the 1950s, frailty has been found to be prevalent among geriatric population and exhibits substantial influences on multiple health-related outcomes; furthermore, there has been an exponential increase in publications on frailty. 1 Originally conceived to characterize the extensive vulnerability to external or endogenous insults in the elderly, frailty has vague content and ambiguous meaning; not until the operational definition of frail phenotype structuralized by Fried and colleagues in 2001 did the measurement of frailty become standardized and subject to extensive investigation. 2 This status of vulnerability can stem from an individual’s demographic background, biologic illnesses with or without organ degeneration, psychologic competency, environmental features, social statuses, etc., with a cumulative and additive effect across different spectrums. 3 Physical performance such as frail phenotype is also commonly used to consolidate frailty, and serves as a robust surrogate of one’s biological age. 4
Regardless of the approaches used to assess frailty, the presence of frailty correlates with various detrimental outcomes among geriatric patients. A meta-analysis identified that frailty, whether measured by the frail index or frail phenotype, was associated with a higher risk of developing premature mortality, prolonged hospitalization, being institutionalized, having disability in basic or instrumental activities of daily living (ADLs), falls, fractures, cognitive impairment, and greater healthcare resource utilization. 5 Similar findings have been corroborated by other.6,7 Conversely, several factors, including, but not limited to, biologic aging, genetic background, lifestyle factors, cardiovascular morbidities, and dietary and nutritional balances, play a role in the pathogenesis of frailty in the geriatric population.8,9
Frailty in patients with chronic kidney disease
The importance of frailty has also been acknowledged in patients with other chronic disorders irrespective of age, including those with chronic kidney disease (CKD) and end-stage renal disease (ESRD). The presence of frailty increases the risk of mortality in these patients, and its adverse influences in other health-related outcomes are being discovered. A previous systematic review of 30 reports focused on the relationship between functional, cognitive impairment or frailty, and adverse outcomes in patients with predialysis CKD or dialysis-dependent ESRD. 10 The authors found that in these patients, functional impairment or frailty was consistently associated with a significantly higher risk of mortality or hospitalization. Another narrative review reached a similar conclusion regarding the negative effects of frailty on survival of ESRD patients. 11 However, accumulating evidence suggests other frailty-related adverse effects besides mortality and hospitalization of CKD patients, although this has not been confirmed to date. A comprehensive understanding of the biology of frailty in CKD patients, including its risk factors, accompanying features, and complications, is therefore needed to facilitate the design of intervention strategies in this disproportionately affected population. In this review, we summarize evidence from the literature to answer this gap in existing knowledge.
Strategy of literature search
We used a systematic approach to identify relevant articles assessing frailty in patients with all stages of CKD in their titles or abstract using keywords, such as ‘frailty’ or ‘frail phenotype’, and ‘chronic kidney disease’, ‘renal insufficiency’, ‘chronic renal failure’, ‘end-stage renal disease’, or ‘chronic dialysis’, from databases, including PubMed, MEDLINE, and Google Scholar. Reports between 1980 and 28 February 2019 were retrieved. Inclusion criteria were original reports involving adult human subjects that examined the relationship between frailty and any types of clinical features or outcomes among the target population of CKD. Eligible studies were independently reviewed by two reviewers (P.Y.W. and C.T.C.). We excluded review articles, articles without abstract available, those that failed to measure the effects of frailty in CKD patients, or non-CKD target population (Figure 1). We further screened the abstracts and reference lists of the retrieved articles to identify additional studies that contained original data focusing on the same issue. Any discrepancy between the two reviewers was resolved by discussing with another senior author (D.C.C.). CKD (nondialysis) was mostly defined according to the estimated glomerular filtration rate based on the Modification of Diet in Renal Disease, while very few CKD cases were evaluated based on elevated serum creatinine levels. Staging of CKD, whichever available, was performed based on the Kidney Disease Improving Global Outcome criteria. 12

The algorithm of literature search and results retrieval.
We extracted the following parameters from the included studies: publication data, participants’ baseline CKD stages, method of frailty measurement, results from univariate analyses of clinical features between frail and nonfrail participants, and multivariate analyses of frail associates or complications, depending on the study design. We tabulated the study characteristics into the following categories: unadjusted risk associates of frailty, adjusted potential causes of frailty, and adjusted risks of health outcomes conferred by frailty according to the biologic relationship between frailty and clinical features that were extracted. Factors adjusted for in the multivariate analyses included at least age and gender in all studies and could further include study-specific parameters such as comorbidity, anthropometric data, and laboratory profiles.
Overview of studies addressing frailty influences in CKD patients
Our database search identified 537 articles addressing frailty and CKD in whole or in part. After an initial screening of the title and abstract, we excluded review articles, those without abstract available, those discussing frailty literally without direct measurement of frailty, and those that did not measure frailty in CKD patients (Figure 1). Overall, 62 original investigation articles with their full text (or abstract if published as conference proceedings) were finally reviewed with results extracted for summarization.13–74 We found that nearly half of these investigations were conducted in the United States (n = 28, 45.2%), followed by Taiwan (n = 7, 11.3%), Canada (n = 4, 6.5%), and Brazil (n = 4, 6.5%). Among the 62 articles, 58.1% used a cohort study design with follow up, while 41.9% had a cross-sectional design; more than half of the articles (n = 37; 59.7%) were based on single-center data, whereas others were analyzed using b multicenter registries. ESRD patients undergoing chronic hemodialysis (stage 5D) (n = 28; 45.2%) were the most common population being evaluated, followed by patients with nondialysis CKD (n = 12; 19.4%), those receiving renal transplantation (stage 5T) (n = 11; 17.7%), and ESRD patients receiving either hemodialysis or peritoneal dialysis (n = 8; 12.9%). Most retrieved studies used the Fried phenotype with or without modifications to measure frailty, whereas seven (11.3%) and four (6.5%) defined frailty according to the FRAIL scale and the Edmonton frail scale, respectively.
Among the 62 articles, 79% (n = 49) included univariate analyses of the relationship between frailty and risk features among CKD patients and 83.9% (n = 52) conducted multivariate analyses to account for influences from confounders. Most of the retrieved studies addressed frailty-related adverse complications in these patients (n = 45; 72.6%), whereas six (9.75%) evaluated potential contributors and complications in the same study; nine (14.5%) of the retrieved studies examined potential contributors to frailty only.
In the following section, we summarize findings from the 62 articles according to the role of each factor in CKD patients into four sections: unadjusted frailty associates, potential contributors to frailty (adjusted), potential modifiers of frailty course (adjusted), and health-related outcomes affected by frailty (adjusted).
Unadjusted associates of frailty in CKD patients
Existing literature examined a diverse spectrum of risk associates accompanying frailty in CKD patients (Supplementary Table), including demographic factors, anthropometric parameters, multiple types of comorbidity, psychological illnesses, physical examination parameters, nutrition, body composition details, bone mineral density, laboratory data, duration and clinical features of dialysis, residual renal function, ADL, quality of life (QoL), and functional and overall outcomes. Higher age; larger waist circumference; lower blood pressure; higher prevalence of comorbidities (heart failure, peripheral vascular disease, diabetes, and obesity); greater fat mass but less lean mass and bone mass; lower serum albumin, hemoglobin, and cholesterol levels but higher creatinine and C-reactive protein (CRP) levels; less residual renal function; worse cognitive function; lower frequency of physical activity and worse ADL; poorer nutrition and QoL; and a higher degree of healthcare utilization were consistently found in frail CKD patients compared with those in nonfrail CKD ones. However, these relationships were all unadjusted, and only some of them have been validated in multivariate analyses, as detailed in the following sections.
Potential contributors to frailty in CKD patients
After adjustment for confounders, multiple factors emerged as independent contributors to the development of frailty in CKD patients (Table 1). Sociodemographic factors, including advanced age, female gender, certain ethnicity (non-White), unemployment, lower education, and smoking, particularly age and being female, are associated with a significantly higher risk of frailty among CKD patients than among nonfrail ones. Increasing CKD severity correlates with a higher frailty risk; however, a dose-response relationship has not been consistently observed. Comorbidities such as the cardiovascular, pulmonary, and central nervous system disorders, metabolic disturbance, and musculoskeletal disorders were all significant risk factors for developing frailty in CKD patients. Among these comorbidities, endothelial dysfunction, chronic obstructive pulmonary disease, obesity, and arthritis were associated with more than two-fold risk elevation. Psychiatric impairment and disability were associated with an even higher risk of frailty (more than three-fold) relative to other contributors. Among patients undergoing chronic hemodialysis, laboratory data such as hypocreatininemia, hypoalbuminemia, and low testosterone levels, with a similar degree of risk elevation, were predictors of developing frailty in CKD patients. A summary of potential causes of frailty in CKD patients is illustrated in Figure 2.
Potential causes of frailty in patients with CKD reported in the literature.
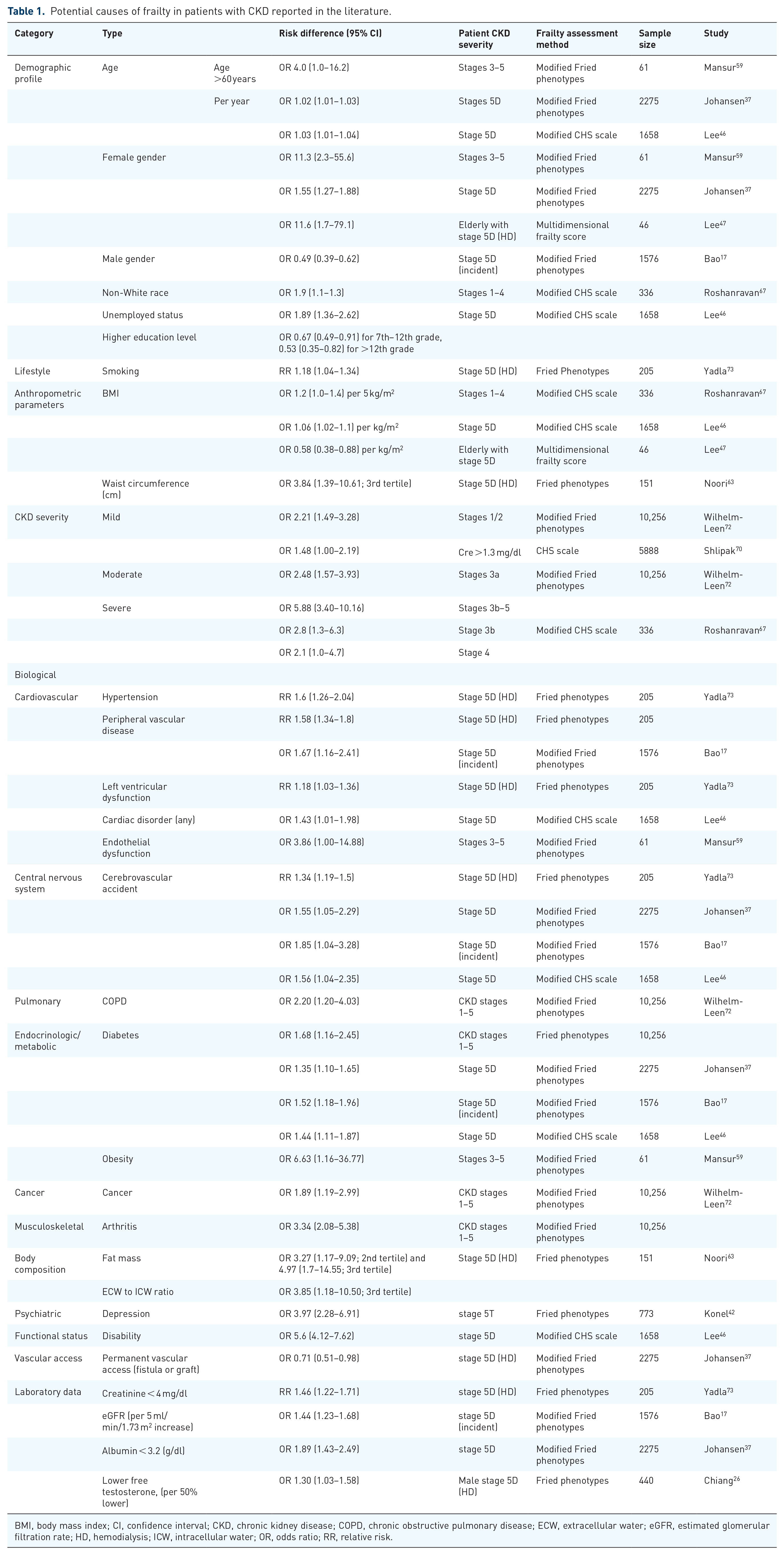
BMI, body mass index; CI, confidence interval; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; ECW, extracellular water; eGFR, estimated glomerular filtration rate; HD, hemodialysis; ICW, intracellular water; OR, odds ratio; RR, relative risk.
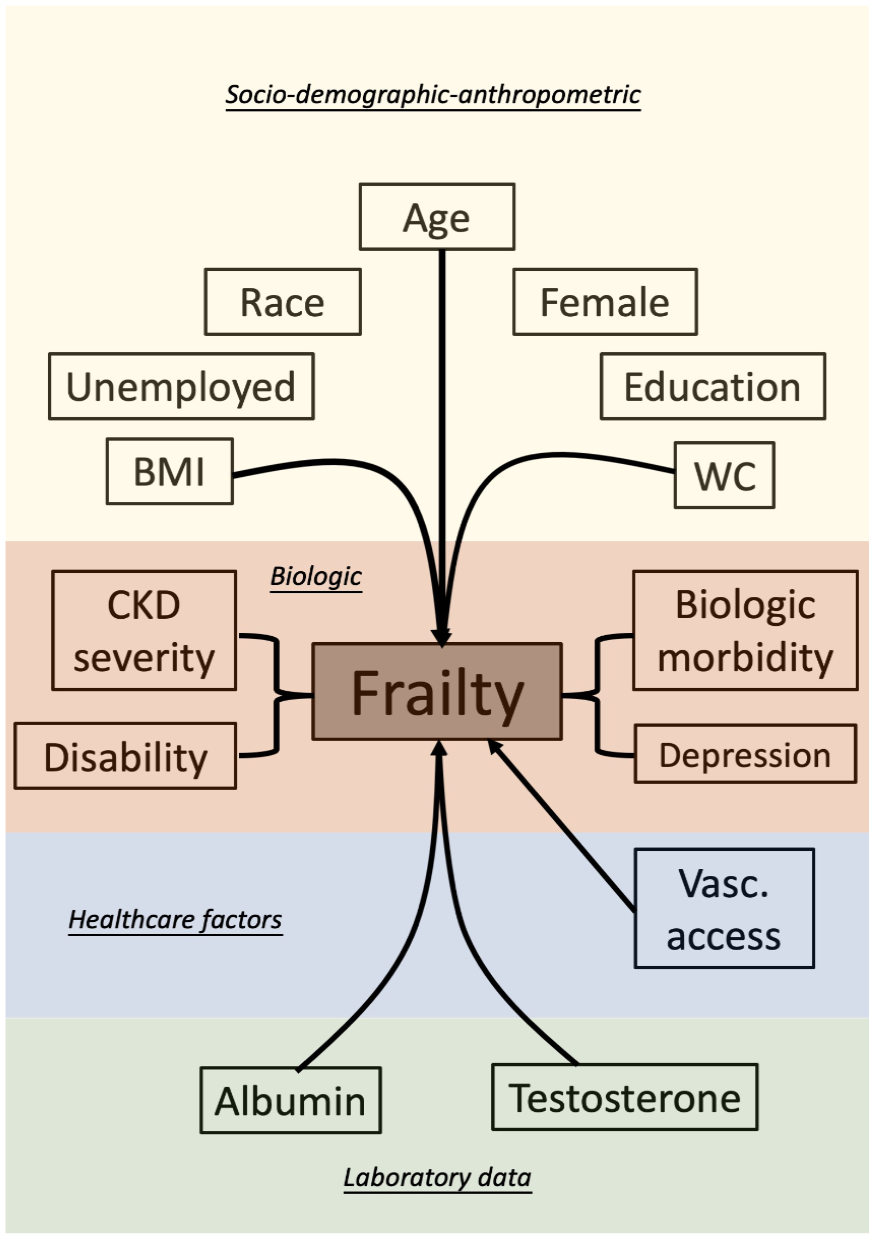
An illustrative diagram showing potential contributors to frailty in CKD patients.
Potential modifiers of frailty courses in CKD patients
Three of the retrieved studies examined factors that modified the course of frailty in CKD patients (Table 2).26,28,40 Johansen and colleagues revealed that diabetes mellitus, certain ethnicity, and higher interleuin-6 (IL-6) levels were associated with worsening frailty over a 2-year follow-up period among chronic dialysis patients, whereas higher serum albumin levels were associated with improving frailty. Chiang and colleagues reported that a baseline lower free testosterone level predicted the risk of developing frailty over 1 year among male dialysis patients. In contrast, in renal transplant patients, Chu and colleagues found that an African-American origin was associated with improved frailty after transplantation, whereas diabetes and longer dialysis period predicted having persistent frailty despite transplantation.
Potential modifiers of frailty trajectories in patients with CKD reported in the literature.

CI, confidence interval; CKD, chronic kidney disease; HD, hemodialysis; IL-6, interleukin-6; OR, odds ratio; RRR, relative risk reduction.
Established health-related complications owing to frailty in CKD patients
After confounder adjustment, frailty remained associated with multiple adverse complications in CKD patients, including disorders involving the cardiac, musculoskeletal, metabolic, and central nervous system; mental distress; impaired functional status; increased fall risk; poorer QoL; greater utilization of healthcare resources (hospitalization, emergency visits, re-admission, longer length of stay, and total medical visits); and a higher mortality than nonfrail CKD one patient (Table 3). Specifically, frailty correlated independently with abnormal cardiac conduction, lower lean and bone mass but higher adiposity, increased fracture risk, and worsened cognitive function. Interestingly, in patients undergoing chronic dialysis, frailty conferred a 2.6-fold higher risk of vascular access failure compared with nonfrail patients. 23 In addition, among renal transplant recipients, frailty significantly increased the risk of subsequent graft loss; those with frailty were more likely to have immunosuppressive dose reduction than nonfrail ones.42,54 Among the spectrum of frailty-related complications in CKD patients, the risk for having sarcopenia was the highest [odds ratio (OR) 12.2], 41 followed by any ADL impairment (OR 11.3) 43 and renal allograft failure (OR 6.2). 42 The risk for fall in frail CKD patients was consistent among existing studies (differences in risk, 1.6 to 3),32,44,50,73 and a similar degree of risk increase was noted with regard to the endpoint of hospitalization-related events.17,37,46,51,73
Confounder-adjusted risk of complications resulting from frailty in CKD patients.
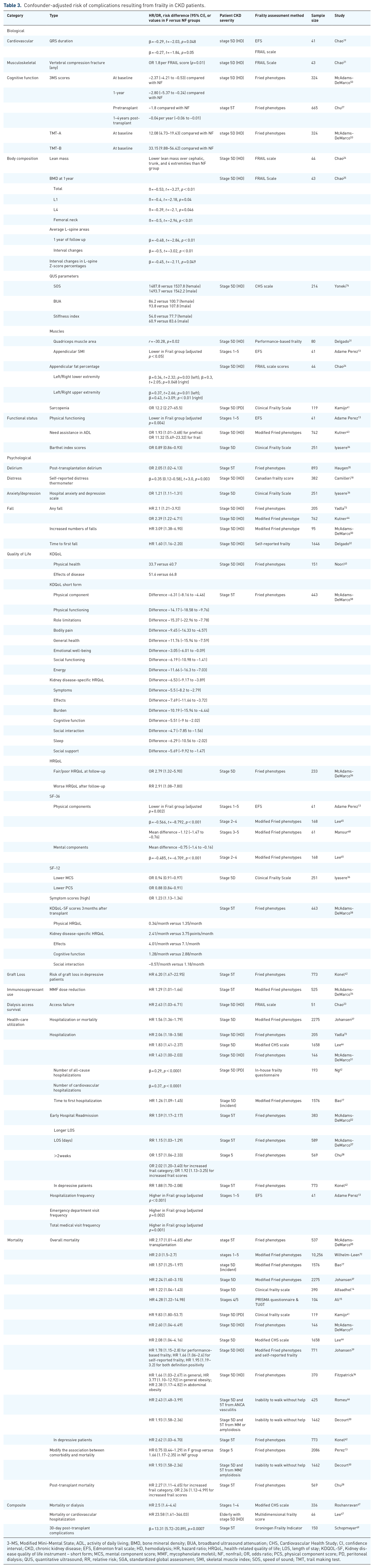
3-MS, Modified Mini-Mental State; ADL, activity of daily living; BMD, bone mineral density; BUA, broadband ultrasound attenuation; CHS, Cardiovascular Health Study; CI, confidence interval; CKD, chronic kidney disease; EFS, Edmonton frail scale; HD, hemodialysis; HR, hazard ratio; HRQoL, health-related quality of life; LOS, length of stay; KDQOL-SF, Kidney disease quality of life instrument – short form; MCS, mental component score; MMF, mycophenolate mofetil; NF, nonfrail; OR, odds ratio; PCS, physical component score; PD, peritoneal dialysis; QUS, quantitative ultrasound; RR, relative risk; SGA, standardized global assessment; SMI, skeletal muscle index; SOS, speed of sound; TMT, trail making test.
The relationship between frailty and mortality in CKD patients has been repeatedly examined in the literature (Table 3). Frailty is predictive of a higher risk of mortality in CKD patients across stages from early CKD to chronic dialysis or stage 5T patients, and the hazard ratios (HRs) ranged between 1.22 and 9.83 compared with nonfrail CKD patients, with most studies deriving a HR between 2 and 3. One study reported an exceptionally higher risk of mortality related to frailty (OR 9.83) 41 ; however, this likely resulted from the modest case number, the frailty measurement approach (clinical frailty scale), and the population they examined (peritoneal dialysis). We also noted that the mortality risk conferred by frailty did not increase linearly with higher CKD severity based on the literature search results; however, mortality risk increased substantially among elderly compared with others. 47 This suggests that chronologic aging substantially enhances the adverse influence of frailty in CKD patients who already have accelerated biologic aging.
A brief summary of frailty-related adverse health-related outcomes is illustrated in Figure 3.

An illustrative diagram showing potential complications of frailty in CKD patients.
Reciprocal relationship between frailty and clinical features in CKD patients
Several features have been examined both as contributors to and complications of frailty in CKD patients, with potential biologic plausibility. These risk features associated with frailty included hypoalbuminemia, higher fat mass, depression, and having a disability (Tables 1 and 3). In addition, it is interesting to note that having permanent vascular access (fistula or graft) is predictive of a lower frailty risk, whereas frail patents were at a higher risk of access failure among chronic dialysis patients.23,37 Similarly, musculoskeletal disorders such as arthritis were independent causes of frailty in CKD patients, whereas frailty in CKD patients might contribute to a higher risk of fractures.21,72
Serum albumin level has long been considered a composite indicator for nutritional status, inflammatory status, and possibly beyond, exhibiting a strong outcome-predictive ability in diverse clinical settings. 75 It is plausible that nutritional impairment contributes to an increased risk of frailty; conversely, the physical limitation imposed by frailty may further compromise nutrient-seeking ability and cause protein-energy malnutrition in affected individuals with CKD. Alternatively, it can be that subclinical inflammation or cytokine interplay stays at the core of this albumin-frailty connection. 76 Dysfunctional muscle and fat tissues with resultant metabolic defects, such as insulin resistance, are potential contributors to frailty and sarcopenia, and frailty can adversely affect eating behavior and lean mass building. 77 This vicious cycle is expected to perpetuate itself in CKD patients who are already at risk of deranged homeostasis with negative body composition alterations. Psychiatric disorders, particularly depression, suppress one’s appetite and decrease oral intake; moreover, frail individuals have poorer QoL and an increased risk of mood disorders. This bidirectional relationship between depression and frailty has been affirmed in older adults, 78 and likely still holds true in CKD patients. Disability and frailty frequently overlap in older adults, and crosstalk between these two adverse phenotypes exists and both independently contribute and act synergistically to an increased risk of mortality among elderly and possibly CKD patients as well. 79
Factors that exhibit an opposite relationship with frailty in CKD patients
Among the retrieved reports, body mass index (BMI) exhibited an inverse relationship with the risk of frailty depending on the population being examined. Greater body BMI increases the probability of frailty in CKD patients regardless of stages,46,67 but decreases the risk in one study involving elderly patients undergoing dialysis 47 (Table 1). A similar scenario has been reported by other studies involving the elderly,80,81 and may be explained partially by the close association between better nutritional status and higher BMI in geriatric patients but not in the general population. It may be worthwhile to note that interventions directed toward reducing BMI can have differential influences in general CKD patients and in older ones.
Nonindependent risk features for frailty
The prevalence and values of many clinical features differed significantly between CKD patients with and without frailty (Supplementary Table); however, their relationship with frailty disappears after confounder adjustment. These factors include multimorbidity, blood pressure, individual morbidities such as osteoporosis and viral infection, and many laboratory parameters ranging from electrolytes (phosphate), hemogram (hemoglobin), lipid profile, and hormonal panel (parathyroid hormone or vitamin D). In addition, care modality, dialysis modality or duration, dialysis clearance, or several nutritional measurement parameters (standard global assessment, mini-nutritional assessment, and malnutrition-inflammation scores) were similarly neutral regarding their relationship to frailty after accounting for other variables in CKD and ESRD patients. It is possible that these factors are surrogates of other vital pathogenic players of frailty, such as serum albumin, cardiovascular morbidities, CKD severities, and residual renal function (Table 1). It will be more appropriate for researchers to account for these instrumental variables that contribute deeply to the development of frailty in subsequent studies aiming to examine frailty risk factors.
Implications for subsequent studies involving frailty in CKD patients
Understanding the risk factors and complications of frailty can be of importance in CKD population from both clinical and public health perspectives. Previous reviews and meta-analyses placed much emphasis on the adverse influences on survival conferred by frailty in CKD patients10,11; however, emerging studies hint at the diverse organ and functional influences posed by frailty. In addition, there are reports suggesting that frailty significantly modifies the association between other risk features and mortality. 82 Researchers are in the process of devising strategies to combat frailty in CKD patients, especially those with advanced CKD and dialysis-dependent ESRD. 83 With the information summarized in this review, we can gain more insight into the beneficial influences of frailty-targeted interventions besides mortality or hospitalization alone. Moreover, by targeting independent risk associates of frailty before or near its onset in CKD patients, we can more efficiently identify upstream etiologies amenable for reducing frailty, paving the way toward outcome improvement in the future. However, we should still remember that only some of the relationships that we described are causal because 41.9% of studies were cross-sectional in nature, precluding overinferences. More than half are single-center studies, and there may be center-specific frailty features that are not generalizable to other populations. Nonetheless, we believe that this comprehensive summarization of existing literature can facilitate the design of subsequent frailty studies in CKD patients.
Summary and conclusion
We conducted an extensive literature search and retrieved 62 reports that addressed the risk associates or complications of frailty in CKD patients. We found that more than half of these studies focused on dialysis-dependent ESRD patients, while only one-fifth of these studies examined those with nondialysis CKD or renal transplantation. Fried phenotype with or without modifications was the most common approach for measuring frailty in CKD patients, followed by FRAIL scale and Edmonton frail scale. Contributors to frailty in CKD patients include sociodemographic factors, smoking, higher CKD severity, several organ-specific comorbidities, depression, disability, hypoalbuminemia, and low testosterone levels. The development of frailty is independently associated with subsequent complications in CKD patients, including cardiometabolic, musculoskeletal, and cerebral disorders; mental distress; functional and QoL impairment; excessive healthcare consumption; and higher risk of mortality. Considering these wide array of frailty-related detrimental influences, frailty-reducing therapies are expected to produce a plethora of benefits in CKD patients. Further intervention studies are awaited to answer this unmet clinical need.
Supplemental Material
Supplementary_Table_3 – Supplemental material for Contributors, risk associates, and complications of frailty in patients with chronic kidney disease: a scoping review
Supplemental material, Supplementary_Table_3 for Contributors, risk associates, and complications of frailty in patients with chronic kidney disease: a scoping review by Patrick Yihong Wu, Chia-Ter Chao, Ding-Cheng Chan, Jenq-Wen Huang and Kuan-Yu Hung in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
Study design: CTC, PYH; Data analysis: PYH, CTC, DCC; Article drafting: PYH, CTC, JWH, KYH; All authors approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: The study is financially sponsored by National Taiwan University Hospital, National Taiwan University Hospital Beihu branch, and Ministry of Science and Technology, Taiwan (MOST 108-2314-B-002-055-).
Sponsor’s role
The sponsors have no role in the study design, data collection, analysis, and result interpretation of this study.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
