Abstract
Sensitization of dorsal horn ganglion sensory neuron plays a crucial role in the maintenance of chronic pain disorder. Multiple interventions targeting dorsal horn ganglion can provide considerable relief of pain, including selective nerve root block, pulsed radiofrequency, and electrical nerve stimulation technique. It remains controversial about the superiority of neuromodulation mentioned above due to distinct pattern of analgesic mechanism. Here, we reported one 71-year-old male presenting at our pain clinic with severe, unilateral lower limb pain caused by postherpetic neuralgia. An implantable stimulator with dual neuromodulation mode, combining pulsed radiofrequency with electrical nerve stimulation, was then placed into the lateral epidural space adjacent to dorsal root ganglion at L4 level. Standard stimulation programing was performed with technicians to achieve satisfactory control of pain, with numerical rating scale decreasing from 8 to 2 postoperatively. This novel dual function neuromodulation strategy may provide an alternative option for pain management for those with intractable neuropathic pain.
Background
It has been well accepted that dorsal root ganglion (DRG) contributes a central role in normal processing of pain signal, as well as in the induction and development of pathological pain conditions. Therefore, multiple modes of interventional neuromodulation treatment targeting DRGs have been considered for chronic pain management.1,2 Pharmacological therapy remains the first choice to reverse the sensitization of DRG sensory neurons; unfortunately, anti-neuropathic drug therapy remains unsatisfactory despite more than 100 medications having been tested. 3 Alternatively, invasive procedures directly functioning with DRGs may improve therapeutic efficacy and clinical outcomes, including selective nerve root block, continuous and pulsed radiofrequency, and implantable electrical nerve stimulation. 1 In most cases, single mode of neuromodulation is applied to seek pain relief; recently, emerging evidence has demonstrated that combination of distinct pattern of modulatory strategy can provide satisfactory control of neuropathic pain.4–6
Here, we present a case of severe lower limb pain caused by postherpetic neuralgia. A new dual function DRG stimulator was implanted to provide DRG electrical stimulation and pulsed radiofrequency neuromodulation.
Case presentation
A 71-year-old male came to our pain clinics with severe unilateral lower limb pain in February, 2023. He had been infected with herpes zoster located in the right dermatomes of L3 and L4 3 months ago. The characteristic vesicular rash gradually diminished with anti-viral treatment within 2 weeks. However, this patient reported ongoing and aggressive suffering of pain ever since, accompanied with a transition of pain sensation from ‘intermittent and mild pain’ to ‘persistent and stabbing, burning pain’, mechanical allodynia, and hyperthesia. At first visit in our pain clinics, pain severity was ranked 8 of 10 numerical rating scale (NRS, 0 = total pain free; 10 = worst imaginable pain). 4 In addition to sensory impairment, this patient presented with functional deficiency in relation to reduced strength and motor ability, that he could not stand or walk by himself.
Limited relief of pain was achieved with multiple oral drugs, including pregabalin (75 mg, Bid), diclofenac (50 mg, Bid), and the patient still felt moderate to severe pain (NRS: 6–8) which required rescue opiates’ usage before surgery. Satisfactory control of symptom was achieved by the pulsed radiofrequency therapy at L3 and L4 nerve root, with pain reduction over 50% compared with baseline. However, the analgesic effect lasted less than 48 h, and the patient still felt severe pain which frequently disrupted his sleep. Given the effective but short-term therapeutic effect, this patient was recommended and consented to undertake an alternative neuromodulation treatment with potentially lasting function, by implanting a DRG stimulator to deliver electrical nerve stimulation and pulsed radiofrequency with a new dual function device set (RUISHENAN Medical Co., LTD, Changzhou, China). One 4-electrode stimulation lead (SCL302D-75, RUISHENAN Medical Co., LTD, Changzhou, China) was placed in the ipsilateral epidural space adjacent to the DRG at L4 level, and the location of DRG lead was confirmed with the anterior-posterior view of fluoroscopy during the surgery [Figure 1(a) and (b)]. Intraoperative sensory testing was conducted to ensure the proper coverage of stimulation in the painful region.
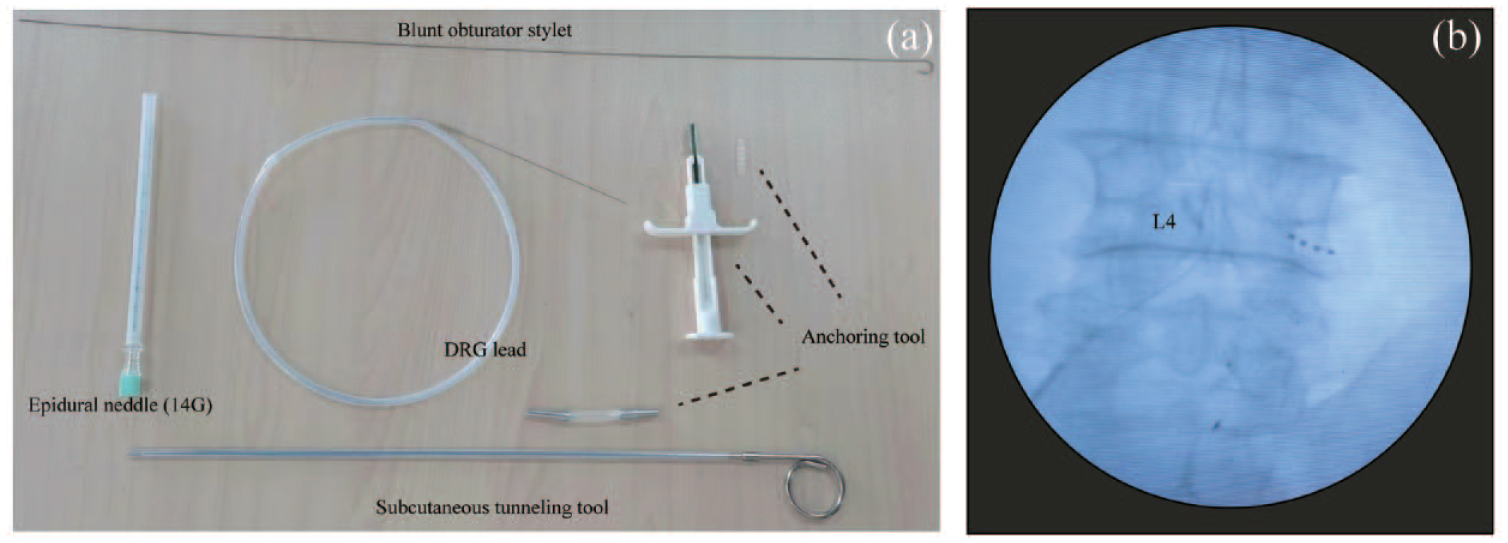
Detail of implantation procedure. (a) Surgical tools for DRG stimulator placement. (b) Location of implantable DRG lead was confirmed with intraoperative fluoroscopy in an anterior-posterior view.
A technician was responsible for the programing of stimulation parameter. To delivery electrical stimulation, an external stimulator was connected to the percutaneous stimulation lead. DRG electrical stimulation was set between the first two contacts (40 Hz, pulse width of 200 μs), and the current intensity was adjusted according to the sensation of patient. An alternative stimulation generator (Beiqi, R-2000BA1, Beijing, China) was connected to the stimulation lead to conduct pulsed radiofrequency. Stimulation protocol of pulsed radiofrequency was set as described previously. 4 Specifically, pulsed radiofrequency frequency was set at 2 Hz (20 ms pulse width) for 240 s and repeated for three cycles. Schematic of DRG stimulation is shown in Figure 2. The patient underwent one session of pulsed radiofrequency treatment following initial DRG stimulator implantation. The stimulator was removed after 10-day neuromodulation period; during this period the patient received another session of pulsed radiofrequency treatment.
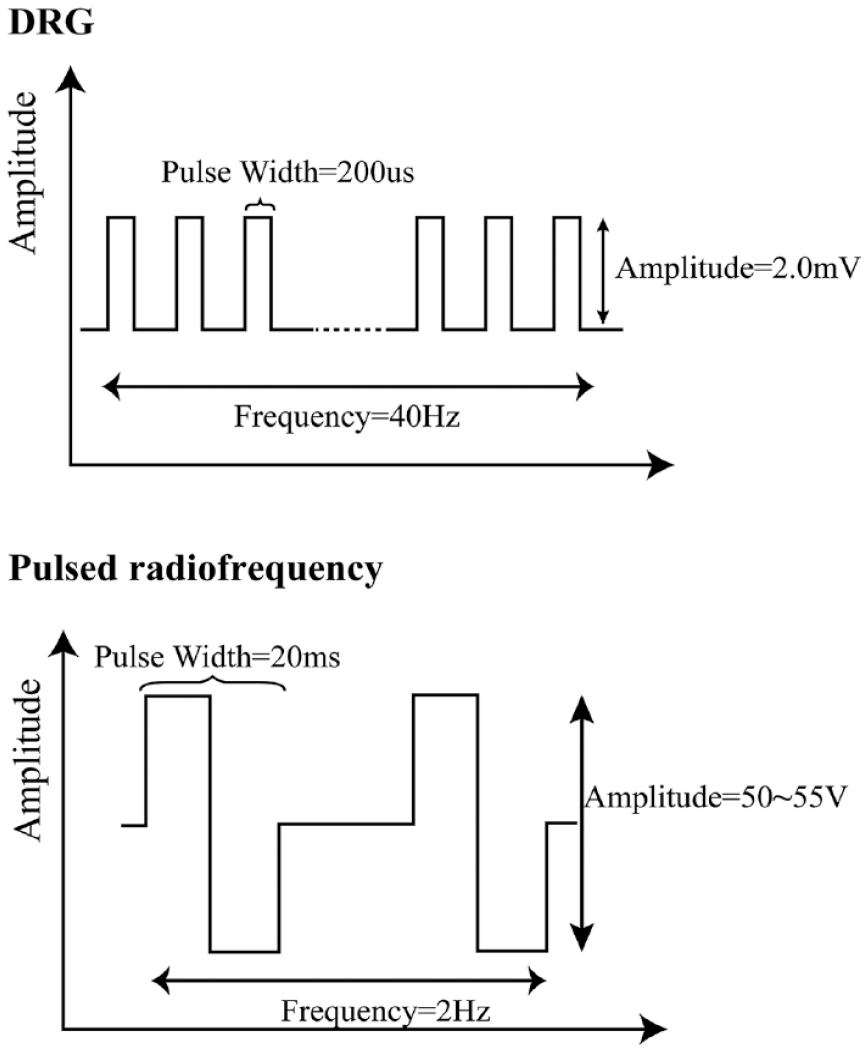
Schematic of stimulation protocol for DRG electrical stimulation (up panel) and pulsed radiofrequency (bottom panel) neuromodulation respectively.
Pain severity was significantly attenuated before discharge, with NRS decreasing from 8 to 2. Mechanical allodynia and hyperthesia was restored after neuromodulation treatment. The patient took only pregabalin (75 mg Bid) to maintain satisfactory control of pain postoperatively, and did not require additional nonsteroidal anti-inflammatory or opiate agents. Additionally, functional improvement was provided with this short-term implantation of DRG stimulator. Before surgery, the patient could not stand or walk; he was able to walk with crutch 3 days post implantation. The muscle strength of herpetic lower limb was at 3–4 level at admission, and 5 level after treatment. Quantitative assessment was conducted with the Oswestry Disability Index (ODI) tool, 7 indicating a functional improvement of total ODI scores decreasing from 41 to 26. Enduring pain relief and functional improvement was reported by this case up at the last follow-up 3 months after treatment (Figure 3). In this case, we did not observe any obvious complication, including infection, lead migration, hematoma, cerebrospinal fluid leakage, or spinal nerve root injury. Patient felt very satisfactory after this novel therapy.

DRG stimulation therapy provided pain relief and functional improvement in this case. Comparison of NRS and (a) ODI scores. (b) Between pre- and post-treatment.
Discussion
DRG is a swelling nodule located in the proximal intervertebral foramen above the posterior root of the spinal nerve, gathering primary sensory neuron cells. It contains pseudo-unipolar neuron, glial cell, and fibrous connective tissue, which are responsible for receiving and transmitting peripheral impulses to central nerve system. Lacking of neurovascular barrier expression increases the permeability of circulating chemokines, eventually leading to a generalized hyperactivation of the DRG sensory neuron. 8 Thus, novel neuromodulation device such as DRG stimulation has been emergingly innovated in the last decade, for the purpose of treating a variety of chronic pain conditions, including complex regional pain syndrome and/or causalgia in the groin and lower extremity. 9 To date, only limited evidence from sparse case reports demonstrated its validation in pain management for postherpetic neuralgia.10,11 To our knowledge, it is for the first time we report a dual function DRG stimulator combining electrical nerve stimulation with pulsed radiofrequency neuromodulation in pain treatment. A common technical signature of this dual neuromodulation is that painful region should be relatively fixed and non-spread. In this case, painful area covered the L4 dermatome and the corresponding DRG level was selected for neuromodulation therapy.
Although mechanism underlying analgesic function of DRG stimulation remains unclear, it may differ between distinct pattern of neuromodulation approach. The fact that limited stimulation duration provided by pulsed radiofrequency treatment may explain its effective but short-term pain relief in this case. Likely, persistent electrical stimulation of trigeminal nerve branch combined with or without pulsed radiofrequency modulation demonstrated to achieve superior clinical efficacy than separate pulsed radiofrequency. 5 However, it remains controversial about therapeutic superiority of electrical nerve stimulation over radiofrequency, due to lack of high-quality data from well-designed clinical trial. In addition, radiofrequency alone may provide considerable relief of herpetic neuralgia compared with other conservative oral medication. 12 Thus, we propose that it is necessary to conduct comprehensive assessment of etiology, duration, intensity, location of pain and even costing to apply different neuromodulation approaches. It is necessary to conduct well-designed, case-controlled, prospective studies to further confirm the superiority of each neuromodulation. In future, pain treatment approaches perhaps will take full advantage of multimode neuromodulation strategy in terms of stimulation site, novel high frequency or bursting stimulation parameter, and as in this case, combination of dual or multiple neuromodulation devices to treat intractable pain.4–6,13,14
Conclusion
Dual function DRG stimulation may be a promising option in the future management of pain, to provide supplementary control of symptom via distinct mechanism. Therefore, prospective and well-controlled studies with larger sample size are necessarily needed in the future.
Supplemental Material
sj-pdf-1-taj-10.1177_20406223231206224 – Supplemental material for A new dual function dorsal root ganglion stimulation in pain management: a technical note and case report
Supplemental material, sj-pdf-1-taj-10.1177_20406223231206224 for A new dual function dorsal root ganglion stimulation in pain management: a technical note and case report by Qiao Wang, Rui Han, Rong Hu, Qianxi Liu, Dong Huang and Haocheng Zhou in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
