Abstract
Background:
Complementary and alternative medicine (CAM) interventions are growing in popularity as possible treatments for long COVID symptoms. However, comprehensive analysis of current evidence in this setting is still lacking.
Objective:
This study aims to review existing published studies on the use of CAM interventions for patients experiencing long COVID through a systematic review.
Design:
Systematic review of randomized controlled trials (RCTs).
Methods:
A comprehensive electronic literature search was performed in multiple databases and clinical trial registries from September 2019 to January 2023. RCTs evaluating efficacy and safety of CAM for long COVID were included. Methodological quality of each included trial was appraised with the Cochrane ‘risk of bias’ tool. A qualitative analysis was conducted due to heterogeneity of included studies.
Results:
A total of 14 RCTs with 1195 participants were included in this review. Study findings demonstrated that CAM interventions could benefit patients with long COVID, especially those suffering from neuropsychiatric disorders, olfactory dysfunction, cognitive impairment, fatigue, breathlessness, and mild-to-moderate lung fibrosis. The main interventions reported were self-administered transcutaneous auricular vagus nerve stimulation, neuro-meditation, dietary supplements, olfactory training, aromatherapy, inspiratory muscle training, concurrent training, and an online breathing and well-being program.
Conclusion:
CAM interventions may be effective, safe, and acceptable to patients with symptoms of long COVID. However, the findings from this systematic review should be interpreted with caution due to various methodological limitations. More rigorous trials focused on CAM for long COVID are warranted in the future.
Introduction
Following its emergence in Wuhan, China in December 2019, COVID-19 has spread rapidly across the world, with 220 countries reporting positive cases. As of 21 February 2023, over 757 million confirmed cases of COVID-19, including 6.8 million deaths, were reported to the WHO, 1 and approximately 43% of cases globally continue to suffer a variety of symptoms after recovering from COVID-19. 2 Long COVID (LC), also termed as post-COVID conditions, refers to a wide range of new, returning, or ongoing health problems that people can experience four or more weeks after first being infected with COVID-19 and that cannot be explained by an alternative diagnosis. 3 Current understanding of LC pathogenesis is still in its infancy, with hypotheses including possible persistent severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, other viral reactivations, virus-induced autoimmunity, persistent tissue damage, immunity-triggered inflammation, and microvascular thrombosis. 4
LC is a multiorgan debilitating illness characterized by a wide range of prolonged symptoms and signs. The most prevalent symptoms of LC include, but are not limited to, fatigue, post-exertional malaise, fever, musculoskeletal pain, rash, menstrual cycle disorders, cough, olfactory and gustatory impairment as well as other multiorgan impairments involving the cardiopulmonary, neurological, and digestive systems. 5 It imposes a substantial impact on social function and quality of life.6,7 Therefore, enhancing knowledge of effective treatments for LC is especially crucial. 8
To date, optimal treatment regimens for LC patients have not been identified. Some of the early empiric treatment options recommended for LC recovery included topical, inhaled, or systemic steroid treatment for pulmonary LC patients. 9 Minor symptoms, such as cough, pain, and myalgia, have been treated symptomatically with acetaminophen and cough suppressants. Antibiotics have demonstrated limited effectiveness in early clinical trials. 10 Multidisciplinary treatments are increasingly needed to provide for the expanding population of LC patients. 11
Recent literature has noted a surge in requests by members of the public and patients for complementary and alternative medicine (CAM) information from pharmacists and other healthcare staff, regarding its role in prevention, symptomatic relief, or treatment of COVID-19. 12 However, the effectiveness of CAM interventions for LC still warrants a more thorough evaluation. In 2022, Kim et al.13,14 conducted a scoping review and bibliometric analysis to explore the existing evidence of CAM interventions for patients with LC. Through analyzing 16 observational studies and study protocols, the authors found that research evidence regarding the use of CAM for LC was very limited and more high-level evidence was required for this vulnerable population.
As more studies have been performed over time, there is a need for a comprehensive and up-to-date report on the efficacy and safety of CAM use in LC. Therefore, the aim of this review is to systematically synthesize the current literature evidence on the use of CAM modalities in a general LC population.
Methods
Literature selection and data extraction
This study was performed following the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) guidelines. Multiple literature databases were searched from September 2019 to January 2023: Scopus, OVID. The standard definition of LC was aligned with the diagnosis developed by Centers for Disease Control and Prevention (CDC), requiring the presence of COVID-19 symptoms extending beyond 4 weeks after initial infection (https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects/index.html). Healthcare practices described as CAM were defined as per the National Center for Complementary and Alternative Health (https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name). All randomized clinical trials (RCTs) evaluating the effects of CAM interventions for LC were included in this study. Comments, reviews, nonrandomized prospective trials, case reports, case series, animal or cellular trials, and studies that were published in languages other than English were beyond the scope of this study. LC symptoms, study demographics, interventions, study results, and adverse events were extracted. Data screening and extraction were performed independently by two author reviewers with pre-specified Microsoft Excel sheets. Any disagreement was settled through discussions.
Quality assessment and risk of bias
Risk of bias of each study included in this review was performed with the Cochrane Collaboration assessment tool. 15 Each RCT was judged in the following domains: random sequence generation and allocation concealment, blindness of participants and personnel, blindness of the outcome assessment, incomplete outcome data, selective outcome reporting, and other biases. Each item was regarded as ‘low risk’, ‘uncertain risk’, or ‘high risk’. Review Manager version 5.4.1 (Cochrane, London, United Kingdom) was used for all risk of bias evaluation.
To evaluate the quality of included studies, the Mixed Methods Appraisal Tool (MMAT) (version 2018. Registration of Copyright (#1148552), Canadian Intellectual Property Office, Industry Canada) for quantitative RCTs was used. In the MMAT tool, five key methodological components, including appropriate randomization, comparable baseline, complete outcome data, outcome assessor blinding, and participants adherence were evaluated. Each component was scored with a Y (Yes: score 1), C (Cannot tell: score 0), or N (No: score 0). The overall quality score is the lowest score of the study components. 16 Two author reviewers evaluated the quality assessment and risk of bias independently. Any disagreements between the two author reviewers were settled by a third reviewer.
Data analysis
The Review Manager version 5.4.1 was used for data analysis. A qualitative analysis was performed due to heterogeneity among the included studies.
Results
Literature search
The study search strategy identified 2636 publications. After removing 4 duplicates, 2632 publications were screened via titles and abstracts. Based on the selection criteria, 2600 publications were excluded, and 32 full texts were evaluated for eligibility. Among them, 18 publications were excluded for various reasons. Finally, 14 records were included in the qualitative synthesis (Figure 1).

Flow chart of study selection.
Characteristics of included studies
The characteristics of the 14 included RCTs are summarized in Table 1. The sample sizes in these studies ranged from 12 to 281 participants, including 854 females, 338 males, and 3 other genders or prefer not to say. Studies were conducted in Brazil (n = 1), Denmark (n = 1), Egypt (n = 1), France (n = 1), Italy (n = 3), Spain (n = 2), Sweden (n = 1), the United Kingdom (n = 2), and the United States (n = 2). Of the trials included in this review, 2 were multicenter trials17,18 and the other 12 were single-center trials.19–30 Except for one four-armed trial, 19 one three-armed trial, 28 the other were two-armed trials.17,18,20–27,29,30 Interventions used in the 14 trials varied: 2 studies used olfactory training,17,26 1 used transcutaneous auricular vagus nerve stimulation, 30 2 used a dietary supplement,20,29 2 used olfactory training plus dietary supplement,18,27 1 used neuro-meditation, 28 1 used aromatherapy, 25 3 used inspiratory muscle training (IMT),22–24 1 used IMT and concurrent training, 19 and 1 used ENO Breathe (an online breathing and wellbeing program). 21 Study compliance rates ranged from 71% 24 to 100%.17,18,27,28 The general quality assessment for all the included studies was not less than 40% quality criteria met (Table 1).
Included study characteristics.
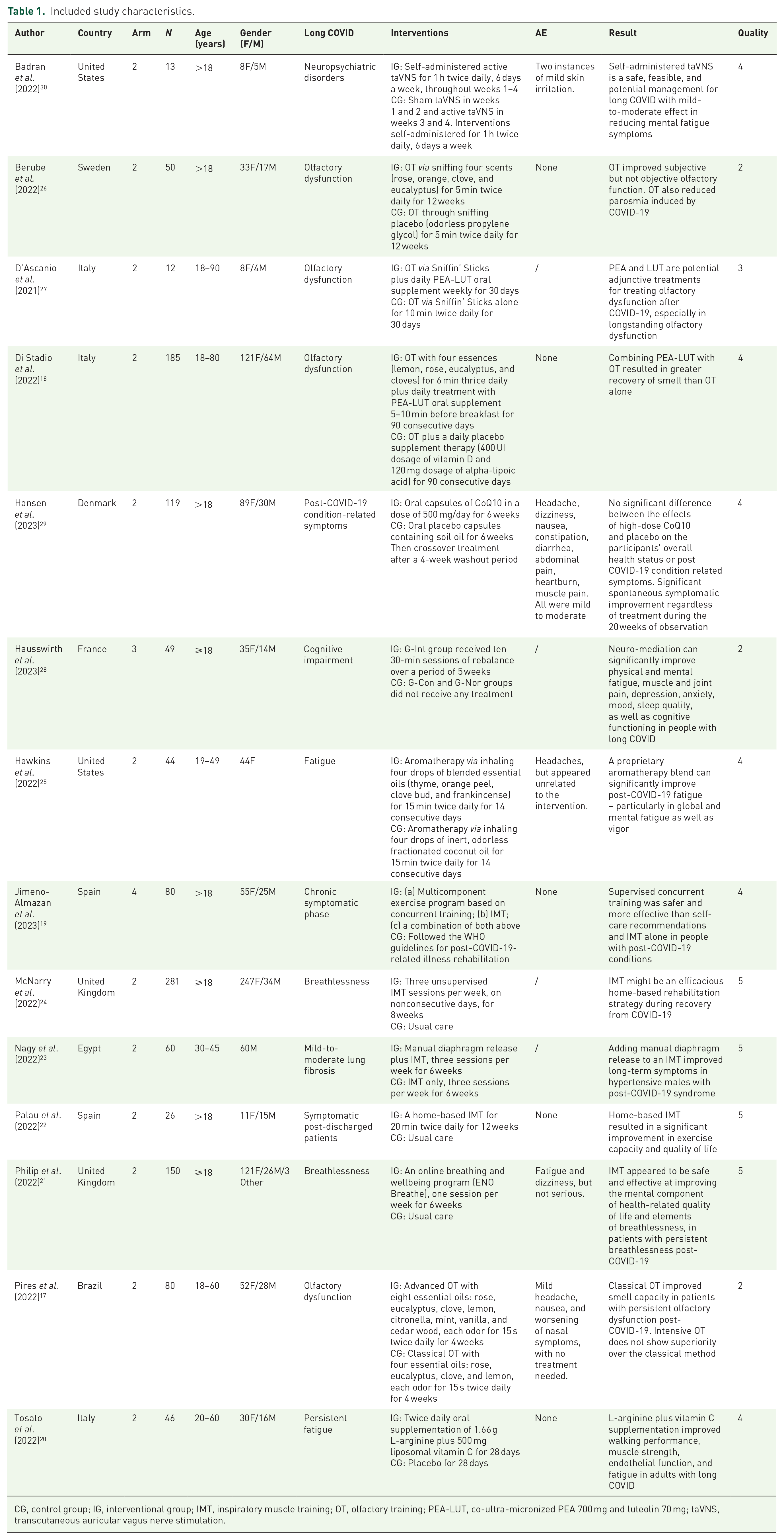
CG, control group; IG, interventional group; IMT, inspiratory muscle training; OT, olfactory training; PEA-LUT, co-ultra-micronized PEA 700 mg and luteolin 70 mg; taVNS, transcutaneous auricular vagus nerve stimulation.
CAM interventions for LC
The included RCTs regarding the use of CAM modalities to alleviate LC could be divided into the following areas: self-administered transcutaneous auricular vagus nerve stimulation, olfactory training, use of dietary supplements, neuro-meditation, aromatherapy, IMT, supervised concurrent training, as well as an online breathing and well-being program (ENO Breathe). The search returned one RCT, which evaluated self-administered transcutaneous auricular vagus nerve stimulation for neuropsychiatric disorders in LC management. 30 Four RCTs used olfactory training alone17,26 or olfactory training plus dietary supplements18,27 as a treatment for post-COVID-19 olfactory dysfunction. One RCT assessed oral high-dose CoQ10 for post-COVID-19 condition-related symptoms. 29 One study used a neuro-meditation program for cognitive impairment post-COVID-19 infection. 28 Two studies apsssssssplied aromatherapy 25 or dietary supplement 20 for relieving fatigue after COVID-19. Three studies evaluated IMT22–24 for persistent breathlessness and mild-to-moderate lung fibrosis. One trial reported on supervised concurrent training to improve the chronic symptomatic phase in outpatients with post-COVID-19 symptoms. 19 Lastly, one trial conducted an online breathing and well-being program for persistent breathlessness post-COVID-19. 21
Self-administered transcutaneous auricular vagus nerve stimulation
Aim: Improvement of neuropsychiatric disorders
In a pilot double-blind RCT, 13 patients diagnosed with neuropsychiatric disorders associated with LC were randomized to transcutaneous auricular vagus nerve stimulation or sham group. Study findings implied that self-administered transcutaneous auricular vagus nerve stimulation intervention demonstrated a mild improvement in self-reported LC symptoms as well as mental fatigue. 30 This supervised self-administered brain stimulation could be a safe, feasible, and remote technology to manage LC symptoms in the home setting.
Olfactory training, olfactory training plus dietary supplement
Aim: Improvement of olfactory dysfunction
Of the 14 trials, 4 trials investigated CAM interventions for olfactory dysfunction post-COVID-19. Berube et al. 26 assessed the effect of olfactory training on post-COVID-19 olfactory dysfunction, such as anosmia, hyposmia, parosmia, and phantosmia for 2 months or more. Fifty participants were randomized to olfactory training or placebo groups equally. Participants were required to sniff four scents (rose, clove, orange, and eucalyptus) or placebo (odorless propylene glycol) over 5 min twice daily, 12 weeks in total. Olfactory function assessments were performed with the University of Pennsylvania Smell Identification Test (UPSIT), Visual Analog Scale (VAS), and the presence of parosmia before and after the training. Compared to the placebo group, subjective olfactory function was observed to have significantly improved in the olfactory training group. However, objective olfactory function testing did not differ. Parosmia rates also reduced with olfactory training. Another multicenter RCT by Pires et al. 17 investigated optimizing the number of fragrances in intensive olfactory training targeted at persistent olfactory dysfunction, which has lasted for at least a month. Eighty participants were randomized to receive either four (rose, clove, eucalyptus, and lemon) or eight (rose, clove, citronella, eucalyptus, lemon, mint, cedar wood, and vanilla) essences over 4 weeks. UPSIT and VAS were evaluated pre- and post-olfactory treatment. There was no significant improvement using intensive olfactory training compared to the classical method. In a randomized controlled pilot study, D’Ascanio et al. 27 randomized 12 participants with hyposmia or anosmia after COVID-19 to olfactory training alone twice daily or olfactory training weekly plus the daily dietary supplements palmitoylethanolamide and luteolin (PEA-LUT, with PEA 700 mg and luteolin 70 mg), over 30 days. Sniff scores for assessing olfactory function change including detection threshold, odor discrimination, and odor identification, were evaluated. The intervention combining olfactory training with dietary supplements was associated with superior improvement of olfactory function, especially in patients with longstanding olfactory dysfunction. Since the quality of this study was limited due to a small sample size, another multicenter double-blinded RCT was performed by the authors to further investigate the efficacy of PEA-LUT supplement plus olfactory training for the recovery of post-COVID-19 olfactory impairment. One hundred thirty individuals were treated with PEA-LUT plus olfactory training in the intervention group and 55 with olfactory training plus placebo in the control group. This study by Di Stadio et al. 18 found that combining PEA-LUT supplement with olfactory training was superior in recovery of smell in post-COVID-19 olfactory impairment patients compared to their counterparts treated with olfactory training alone, over 90 days.
Neuro-meditation program
Aim: Improvement of cognitive impairment
A study randomized 34 adult patients with cognitive impairment post-COVID-19 infection to either a neuro-meditation program intervention group (n = 17) or a control group (n = 17), whereas an additional 15 healthy volunteers were placed in a normative group. Various cognitive tasks and self-administered questionnaires were evaluated before and after the interventions. Study results noted that ten 30-min neuro-meditation sessions could significantly improve all the subjective and cognitive performances in LC. 28 The neuro-meditation program appeared to be a promising non-pharmacological treatment for cognitive impairment after COVID-19 infection.
High-dose coenzyme Q10 therapy
Aim: Reducing number and/or severity of post-COVID-19 symptoms
Hansen et al. 29 hypothesized that the constellation of post-COVID-19 symptoms was caused by acquired chronic mitochondrial dysfunction. They conducted a double-blinded crossover RCT to determine whether high-dose CoQ10 could benefit patients with post-COVID-19 symptoms. One hundred twenty-one participants were randomly assigned to the observational group receiving oral capsules of CoQ10, 500 mg/day or control group receiving placebo containing soy oil for 6 weeks; this was followed by a washout period of 4 weeks, after which participants crossed over to the alternative treatment. They found that high-dose CoQ10 intervention did not significantly improve post-COVID-19 symptoms when compared to placebo. However, this study was solely observed based on self-reported data with questionnaires, which could have led to recall bias.
Aromatherapy, dietary supplement
Aim: Improvement of fatigue
Two studies assessed aromatherapy or supplements in patients with fatigue after COVID-19 infection. In a double-blinded RCT by Hawkins et al., 25 44 female participants who suffered from fatigue for more than 5 months after COVID-19 infection were randomized to inhale essential oils (clove bud, orange peel, frankincense, and thyme) or placebo (inert, odorless fractionated coconut oil) for 2 weeks. Results demonstrated that aromatherapy significantly improved energy levels among women recovering from COVID-19.
In the other single-blind RCT by Tosato et al., 20 46 participants with persistent fatigue were divided to receive either oral supplementation (1.66 g L-arginine plus 500 mg liposomal vitamin C) or a placebo twice daily for 28 days. Study findings indicated that L-arginine plus vitamin C appeared to restore physical performance and improve persistent fatigue in LC population.
Inspiratory muscle training
Aim: Improvement of breathlessness and mild-to-moderate lung fibrosis
Four studies were identified evaluating IMT for breathlessness post-COVID-19 infection, including studies using IMT for breathlessness, home-based IMT for post-discharged patients, manual diaphragm release plus IMT for mild-to-moderate lung fibrosis, and an online breathing and well-being program (ENO Breathe) for breathlessness. McNarry et al. 24 investigated 281 adults who suffered from breathlessness and received either home-based IMT thrice weekly, or usual care, over 8 weeks. They found that home-based IMT group had significant improvement in breathlessness recovery, despite a high withdrawal rate. In another trial by Palau et al. 22 assessing the efficacy of home-based IMT on maximal functional capacity in post-discharged patients after COVID-19 infection, 26 patients were randomized to receive either home IMT twice daily for 12 weeks or usual care (no intervention). Results showed that home-based IMT resulted in a significant improvement in physical performance and quality of life among symptomatic post-COVID-19 discharged patients. Another study by Nagy et al. 23 assessed whether combining manual diaphragm release with IMT therapy was more effective than IMT alone in patients with mild-to-moderate lung fibrosis post-COVID-19 infection. Study results indicated that the addition of manual diaphragm release might enhance the effect of IMT in rehabilitation of symptomatic male patients with LC.
Concurrent exercise
Aim: Improvement of cardiopulmonary fitness, strength, and symptom severity
To determine the effects of concurrent exercise combined with IMT in patients with LC, Jimeno-Almazan et al. 19 randomly assigned 80 non-hospitalized adults into one of four 8-week parallel interventions: IMT (n = 17), concurrent training (n = 20), concurrent training plus IMT (n = 23), or a control group (n = 20) following the WHO guidelines for post-COVID-19 related illness rehabilitation. Study findings indicated that concurrent training, either with or without IMT, seemed more effective than IMT alone or self-management, according to the WHO recovery recommendation guidelines alone.
Online breathing and well-being program of English National Opera (ENO Breathe)
Aim: Improvement of health-related quality of life
Philip et al. 21 compared the efficacy of an online breathing and well-being program of English National Opera (ENO Breathe) to usual care in adults with persistent breathlessness post-COVID-19. ENO improved breathlessness as well as mental health measures in patients with persistent symptoms after COVID-19.
Safety of CAM interventions
While four studies did not provide sufficient information about safety of CAM treatments for LC,23,24,27,28 10 studies reported relevant information about adverse effects from the interventions.17–22,25,26,29,30 Five studies noted no adverse effects during the study period.18–20,22,26 Five reported adverse effects in detail.17,21,25,29,30 Badran et al. 30 concluded that a 4-week self-administered taVNS was safe and could be a potential treatment for LC symptoms, particularly mental fatigue; they reported two incidences of mild skin irritation due to prolonged stimulation, which resolved without any medical treatment. Hansen et al. 29 applied 500 mg CoQ10 or placebo daily for 6 weeks to individuals with post-COVID-19 condition and found that only 6.4% of adverse events were related to the CoQ10 intervention, all of which were mild to moderate. Hawkins et al. 25 reported that breathing the scent of a proprietary essential oil blend twice daily for 14 days could significantly improve fatigue recovery from COVID-19. One participant in this study reported recurring headaches after breathing the essential oil and withdrew from the study on day 13. This patient had experienced headaches prior to participating in the study. No other adverse events were reported in this study. Philip et al. 21 found that the online ENO Breathe program could improve breathlessness and boost mental health measures in individuals with persisting breathlessness post-COVID-19. One participant in this study withdrew due to dizziness that was thought to be induced by a prolonged computer screen time during the sessions for the interventional group. Seven other participants withdrew due to excessive fatigue. There were other reports of fatigue or mild dizziness during the sessions, though they were not serious enough to warrant a withdrawal from the study. Pires et al. 17 reported that olfactory training intensification by increasing the number of essences over 4 weeks was not advantageous as compared to the classical method of four essences. Side effects comprising of headaches, nausea and worsening of nasal symptoms were self-limiting and did not require medical attention. In summary, the adverse effects reported in this review were minor, suggesting that CAM therapies served as safe interventions.
Bias risk in the trials
Four trials were considered to have unknown bias risk for generating the random sequence due to insufficient information about the specific method of randomization used,17,26,28,30 whereas the remaining nine trials were regarded as low bias risk.18–25,27,29 Five trials provided appropriate random allocation concealment and were classified as low bias risk,21–24,29 whereas the remaining nine provided insufficient data and were classified as unknown risk.17–20,25–28,30 Five trials were blinded to both participants and personnel and were regarded as low risk of bias,19,21,22,29,30 whereas six trials did not and were considered as high risk of bias.20–23,27,28 The remaining three trials were rated as unknown risk of bias since insufficient information was provided.17,19,24 Ten trials performed outcome assessment blinding and were judged to be at low risk of bias,18,20–23,25–27,29,30 whereas three trials did not and were judged as unknown risk of bias.17,19,24 The remaining trial did not perform outcome assessment blinding and was thus judged to be at high risk of bias. 28 One trial did not report detailed information about drop-out cases and was defined as high risk of bias. 26 Three study protocols were unavailable and had relevant outcomes that were not reported in the pre-specified way,17,27,28 whereas the other eleven did and were judged as low risk of bias.18–26,29,30 Six studies seemed to have no other sources of bias and were regarded as low risk,18,21–23,25,29 one study had was regarded as high risk, 28 whereas the remaining seven did not and were considered as unknown risk of bias due to other potential source of bias17,19,20,24,26,27,30 (Figure 2).

Plot of the risk of bias of included studies.
Discussion
This study aimed to explore the emerging literature regarding CAM management of LC. The studies included in this review were heterogenous regarding diseases, CAM interventions, and outcome measurements. Study findings do not suggest a singular effective CAM modality for all symptoms of LC. The empirical results suggest that neuropsychiatric disorders, olfactory dysfunction, cognitive impairment, fatigue, breathlessness, and mild-to-moderate lung fibrosis were the most common conditions treated with CAM interventions in LC. All the interventions adopted were non-pharmacological therapies, including dietary supplements, olfactory training, aromatherapy, IMT, and concurrent training.
The COVID-19 pandemic has triggered an unmet need for safe and effective recovery modalities for the long-term sequelae of COVID-19 infection. However, treatments with robust efficacy for post-viral syndromes and related conditions have not been fully developed. This could be attributed in part to different definitions of post-COVID-19 sequelae by WHO, 31 CDC, 3 NIH, 32 and NICE. 33 This systematic review assessed eligibility using a double independent screening and extraction based on CDC guidelines. We synthesized studies if they sampled people currently suffering from a wide range of ongoing health problems at least 4 weeks after the COVID-19 infection. LC care in general practice can be improved through better defining the sequelae of the disease and their severity. Standardized definitions of the sequelae of COVID-19 infections and their severity, as well as enhanced access to multidisciplinary specialist services for patients should be established to monitor individuals with long-term COVID-19.
The implementation of interventions for LC requires multidisciplinary and multidimensional approaches to deal with the high prevalence of sequelae associated with COVID-19 infection. The integration of CAM therapies into conventional care not only enhances the communication between patients and their caregivers, but it also increases therapeutic options and empowers patients. About 71.4% of our included studies provided detailed information regarding the safety aspects of CAM interventions in the management of LC. Most reported side effects were minor, and no severe adverse events were observed. The reported CAM therapies seem safe and effective in improving various symptoms associated with LC.
Strengths and limitations
To our knowledge, this is the first study to synthesize the current evidence of CAM therapy recommendations across RCTs in populations experiencing LC. This review provides an up-to-date comprehensive review of clinical research in terms of CAM management of LC symptoms. Our findings may also inform patients about feasible alternative modalities to conventional medicine in producing better health outcomes. In addition, we highlighted various CAM modalities for LC management, which could be used to identify additional research objectives for future LC research.
Nonetheless, the findings presented in this study should be interpreted with caution. One of the limitations of this systematic review is that it was not registered with databases. The genesis of this study occurred during the initial stages of understanding LC, a time of rapid progression in both symptom recognition and disease characterization. This made it challenging to establish a stable protocol that could meet the registration criteria. The limitation might reduce the transparency and accountability of our research process. As our understanding of LC continues to evolve, we commit to ensuring that future studies adhere to relevant protocol registration practices, thereby minimizing this limitation in subsequent research. While case reports and other type of trials have been reported, only RCTs were included in our study, potentially overlooking some therapeutic modalities. More CAM interventions should be investigated for the treatment of LC in the future. Since the trials included in this review demonstrated considerable methodological and intervention heterogeneity, we were unable to provide more objective findings of included trials by quantitative analysis. Lastly, the majority of the included studies were of low quality due to poor methodological designs, particularly in terms of random allocation and blinding. Consequently, there is still no definite evidence supporting the efficacy of CAM in LC management. More rigorous and well-designed studies in this setting are warranted.
Conclusion
This systematic review provides insight into the efficacy and safety of various CAM interventions for LC. Study findings suggest that olfactory dysfunction, fatigue, breathlessness, and mild-to-moderate lung fibrosis were the main reasons LC patients sought CAM treatments. CAM interventions specifically self-administered transcutaneous auricular vagus nerve stimulation, neuro-meditation, diet supplements, olfactory training, aromatherapy, IMT, and concurrent training have shown some promising results for people experiencing LC. No serious adverse events were noted. However, methodological limitations of the included trials imply that these results should be interpreted with caution. More rigorous and methodologically sound studies in this setting should be evaluated in the future.
