Abstract
Background:
Most people with coronavirus disease 2019 (COVID-19) experience resolution of symptoms within days to weeks following initial infection. In a subset of individuals, symptoms persist longer than 4 weeks, known as ‘Long COVID’. Many gastrointestinal (GI) symptoms persist as part of this syndrome; yet, an approach to treatment remains unclear. Prior studies have demonstrated the efficacy of complementary and alternative medicine (CAM) for the treatment of acute COVID-19 infections, but little data exist regarding the potential use of CAM in the treatment of Long COVID.
Objectives:
Identify CAM approaches useful in treating the GI symptoms of Long COVID.
Design:
A systematic review of studies reporting on the use of CAM for the treatment of GI symptoms of Long COVID was performed.
Data Sources and Methods:
Five electronic databases were searched from January 2019 to November 2022. Studies describing the use of CAM to treat GI symptoms of Long COVID were included and assessed by two independent reviewers. Studies not reporting on GI symptoms or using CAM were excluded. Studies chosen for final review underwent quality and bias assessment using predetermined criteria. The extracted data were synthesized utilizing a framework derived from the National Center of Complementary and Integrative Health categories.
Results:
The initial search yielded 396 articles. After applying the eligibility criteria, a total of four studies (three case reports and one case series) were included for final review. Two studies used nutritional supplements and two studies used traditional Chinese medicine. Reductions in nausea, loss of appetite, diarrhea, acid reflux, epigastric pain, and bloating were reported.
Conclusion:
This is the first systematic review to explore the role of CAM in treating GI manifestations of Long COVID. The review identified four studies, all reporting reductions in the GI symptoms of Long COVID. Despite the positive studies included in this review, the overall search yielded few results, all of which were non-experimental. As the post-infectious sequelae of COVID-19 become better recognized in the wake of the pandemic, higher-quality clinical studies are needed.
Introduction
SARS-CoV-2 is a novel coronavirus that emerged in 2019. 1 On 11 March 2020, infection with SARS-CoV-2, known as coronavirus disease 2019 (COVID-19), was declared a pandemic. 2 Acute infection with SARS-CoV-2 typically manifests as an acute respiratory syndrome, characterized by fever, cough, and shortness of breath. While most patients have a symptomatic resolution within 2–14 days following initial exposure, a subset of patients experience persistent symptoms. This syndrome has been named ‘Long COVID’ by the Centers for Disease Control and Prevention and describes a wide range of ongoing health problems lasting more than 4 weeks from the initial infection.3,4 Common symptoms include fatigue, shortness of breath, and difficulty concentrating. A significant subset of these patients develop gastrointestinal (GI) predominant illness, most commonly marked by abdominal pain, diarrhea, and loss of appetite. In addition, the development of post-COVID irritable bowel syndrome (IBS) has been well described.5–7
Currently, a clear strategy for managing the GI symptoms of Long COVID does not exist. 8 Given the global burden of this disease, there has been considerable interest in exploring potential therapeutic options to address these chronic disturbances. One area of focus is complementary and alternative medicine (CAM). As outlined by the National Center for Complementary and Integrative Health (NCCIH), CAM describes healthcare systems, practices, and medical products not normally considered part of conventional medical care. 9 Complementary therapies are used in addition to standard treatment, and alternative therapies are used in replacement of standard treatment. The NCCIH categorizes CAM approaches by how the therapy is delivered. These categories include nutritional (diet, dietary supplements, herbs, probiotics), psychological (mindfulness), physical (massage, spinal manipulation), and combinations (yoga, tai chi, acupuncture).
CAM therapies are effective in treating many symptoms of acute COVID-19. 10 Other studies have demonstrated the efficacy of CAM in reducing the duration and severity of acute and inflammatory infections, such as influenza, influenza-like illness, and the common cold. 11 The majority of these interventions fall within the NCCIH category of nutrition, and many have been shown to reduce the severity and/or duration of upper respiratory symptoms. These include zinc,12–14 elderberry extract,15–17 probiotics, 18 buckwheat honey, 19 and Pelargonium sidoides.20, 21 Qigong and acupressure have shown benefits in reducing pulmonary symptoms (dyspnea, cough), improving lung function, and reducing hospital stay in patients with severe COVID-19. 22 CAM is also commonly used for patients receiving care in outpatient GI clinics, particularly among those dissatisfied with conventional therapies. More than half of this population has reported using supplements or dietary modification, with 44% using both. 23
Despite prior studies demonstrating the efficacy of CAM in the treatment of acute COVID-19 and other viral infections, minimal data is available for evaluating the potential use of CAM in the treatment of the chronic GI symptoms of Long COVID. As the number of patients impacted by Long COVID increases, there has arisen a clinical need for safe and effective treatment options to improve GI symptoms in this syndrome. The primary objective of this study is to perform a systematic review assessing the role of CAM for this purpose, acknowledging the limited data currently available. This systematic review is timely and necessary, as it will help identify gaps in the existing literature and generate hypotheses for future studies.
Methods
Eligibility criteria
The methodology guiding this systematic review closely adhered to that proposed by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist. 24 The study was registered in PROSPERO with the identifier CRD42022372333. All case reports, case series, observational studies, and experimental studies exploring the use of CAM therapies to treat the GI symptoms experienced as part of Long COVID were included and assessed by two independent reviewers. Only human studies were included. Studies not reporting on GI symptoms as a part of Long COVID or not using CAM were excluded. Studies not written in English were excluded. All review articles, opinion pieces, protocol reviews, and letters were excluded.
Search strategy
Five electronic databases (APA PsycINFO, EBM Reviews –Cochrane Central Register of Controlled Trials, Embase, Ovid MEDLINE, and Scopus) were searched from May 2019 to November 2022. The search strategy was designed and conducted by an experienced librarian with input from the study’s principal investigator. Controlled vocabulary supplemented with keywords was used to search for the use of CAM in the management of GI manifestations of COVID-19. The search was limited to English and human subjects. The actual strategy listing all search terms used and how they are combined is available in Supplemental Appendix 1. Results obtained from database searches were combined and duplicate studies were removed. Two independent reviewers screened studies for inclusion and reviewed these studies for data extraction.
Data extraction and synthesis
Studies meeting eligibility criteria underwent analysis by two independent reviewers. Extracted data included study information (author and year of publication), location of study, intervention used, study design, number of participants, duration of intervention, and primary and secondary outcome(s). Only primary and secondary outcomes describing GI symptoms were extracted. Loss of smell was not considered to be a GI symptom. Measures of outcome including measures of central tendency and association were extracted as available. No statistical models were fitted given this study’s focus on qualitatively assessing the available data. CAM approaches were categorized by those suggested by the NCCIH, including nutritional, psychological, physical, and combination.
Quality assessment
Studies selected for final review were assessed for quality using predefined criteria by two independent reviewers. All case reports and case series were evaluated using the Consensus-based Clinical Case Reporting Guideline Development (CARE) guidelines. The CARE guidelines are a framework designed to improve the accuracy, transparency, and usefulness of case reports by providing a systematic approach to their presentation and structure. The guideline’s 13-item checklist was developed by experts in clinical medicine, epidemiology, and research methodology, and has been widely embraced by medical journals as a standard for case report publication.25,26
Results
Study selection
An initial search of the five databases yielded 396 articles. Following the removal of 258 duplicate records, 138 articles were screened by title and abstract. Ultimately, 29 articles were sought for full-text retrieval. Of the 29 articles, only four met the eligibility criteria and were included in the final review. Excluded studies with the reason for exclusion are shown in Supplemental Table 1. Full details of the study selection are found in the PRISMA flowchart depicted in Figure 1.
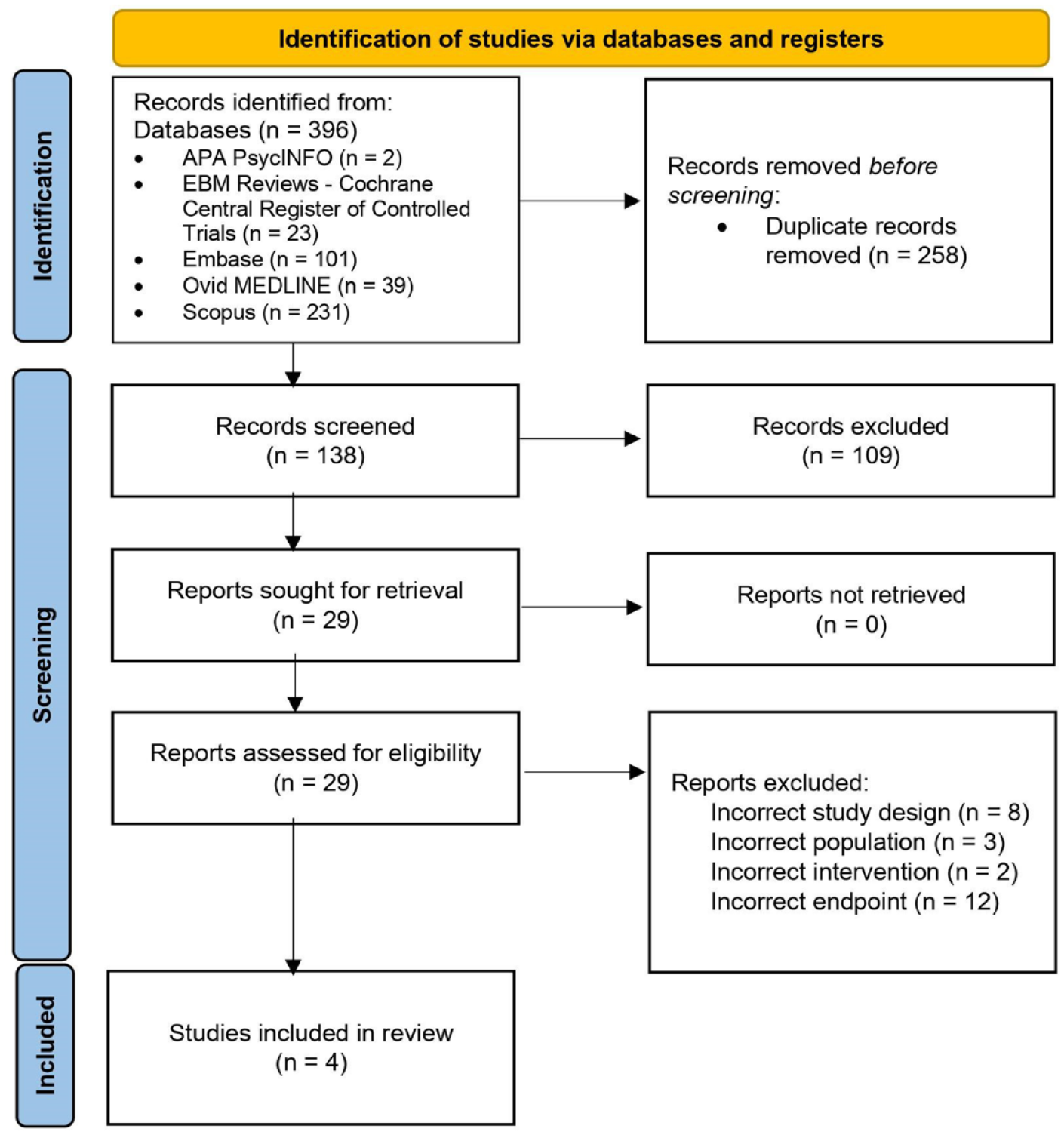
Preferred reporting items for systematic reviews and meta-analyses flowchart demonstrating the selection process for included studies.
Study characteristics
After applying the inclusion and exclusion criteria, a total of three case reports and one case series involving a total of five individuals were included in the final review (Figure 1).27–30 All four studies were found to adhere to the CARE guidelines per assessment by study reviewers. Further characteristics of these studies are described in Table 1. NCCIH categories included nutritional (n = 2) and combination (n = 2). Specifically, both studies in the combination category used traditional Chinese medicine (TCM). All four studies reported on the effect of the intervention on GI manifestations of Long COVID. Studies reported a wide range of GI symptoms including nausea, loss of appetite, diarrhea, bloating, abdominal discomfort, epigastric pain, and acid reflux.27–30 All patients were treated in the outpatient setting and with multiple follow-up visits after initiation of the CAM intervention. In the studies using nutritional approaches, the study investigators administered the intervention to the patients, whereas the studies using TCM had the intervention administered by licensed practitioners.
Characteristics and extracted data from included studies.

GI, gastrointestinal; mg, milligrams; NCCIH, National Center for Complementary and Integrative Health; r, Spearman’s rank correlation coefficient; TCM, traditional Chinese medicine.
Effects of CAM on GI symptoms of Long COVID by NCCIH category
Nutritional
Two studies utilized CAM approaches in the NCCIH category of nutrition to treat the GI symptoms of Long COVID. One study used a botanical-based, high-fiber formula for 60 days in a patient experiencing loss of appetite and nausea for 1 year. This intervention resulted in a significant reduction in nausea (r = −0.328, p = 0.011) and loss of appetite (r = −0.451, p < 0.001) as formula intake increased. 27 In the other study, two pediatric patients experiencing diarrhea-predominant Long COVID received 400–600 mg of lactoferrin via oral formulation every day for 90 days. In both cases, oral lactoferrin led to the resolution of diarrhea within 2 weeks. 28
Traditional Chinese medicine
The two case reports in the NCCIH category of combination used TCM. One of the case reports described a 32-year-old female with epigastric pain, bloating, and diarrhea as part of Long COVID. She was treated with an individualized herbal decoction daily and weekly acupuncture for a total of 6 weeks. The patient reported complete resolution of her GI symptoms following treatment. 29 The other case described a 42-year-old female experiencing nausea, abdominal discomfort, and acid reflux as part of Long COVID. She was treated with an individualized herbal decoction for an unclear duration. At the end of the treatment period, the patient had a complete resolution of symptoms. 30
Discussion
While the acute impacts of COVID-19 have been well described, the capacity for COVID-19 to induce chronic disease in previously healthy patients is becoming increasingly evident. The syndrome of persistent symptoms following an acute COVID-19 infection has been collectively named ‘Long COVID’. Distressing GI symptoms are common in patients with Long COVID. 5 Given the novelty of this syndrome and the lack of available therapeutics, attention has turned to CAM to address these chronic symptoms following acute COVID-19. To the authors’ knowledge, this is the first systematic review to comprehensively assess the role of CAM in treating the GI manifestations of Long COVID. The present study identified three different CAM modalities effective in treating the GI symptoms of Long COVID in limited patient samples.
Two of the therapeutic modalities identified were in the NCCIH category of nutrition. One approach utilized a botanical-based, high-fiber formula, while the other utilized an oral formulation of lactoferrin, both with resolution of symptoms.27,28 While the pathophysiology of Long COVID has yet to be fully elucidated, the therapeutic hypothesis guiding these studies posited that disruption to the gut microbiome was responsible for driving the GI symptoms of Long COVID. In the study utilizing the high-fiber formula, the patient had measured ecologic shifts in the gut microbiota that correlated with the resolution of the patient’s symptoms. 27 Notably, the patient had an enrichment of short-chain fatty-acid-producing bacteria following treatment, which has been shown to inversely correlate with the development of Long COVID. 31 Importantly, studies have shown that COVID-19 can cause direct disruption of the gut microbiome. 32 With better characterization of the gut microbiota in patients with Long COVID, notable changes are becoming better recognized and could offer a reasonable explanation for why previously healthy patients develop a persistence of GI symptoms following acute COVID-19.33–35 Thus, nutritional supplementation highlights an important area for future research given its potential to shift the underlying microbiome and treat the persistent GI symptoms of Long COVID.
Lactoferrin is a glycoprotein contained in exocrine secretions, including intestinal secretions. Lactoferrin has been implicated in modulating the gut microbiota.36–38 While microbiota data were not collected in this study, the treatment was effective at alleviating GI symptoms. 28 Lactoferrin also has anti-inflammatory and anti-viral properties. SARS-CoV-2 elicits host damage by binding to ACE2 receptors throughout the body, many of which are found in the GI tract. When bound to this receptor, SARS-CoV-2 induces the GI inflammatory response, leading to tissue damage, which can elicit GI symptoms such as abdominal pain and diarrhea. 39 Given SARS-CoV-2 can persist in the GI tract for months following initial infection, ongoing viral shedding could serve as a nidus for the persistent GI symptoms in Long COVID.40,41 Thus, a gut-directed anti-inflammatory compound, such as lactoferrin, could have a therapeutic effect by curbing the ongoing GI inflammatory cascade following COVID-19, allowing for healing of the GI tract.
The other studies included in this systematic review used TCM, which falls in the NCCIH category of combination. TCM is a health system developed thousands of years ago that characterizes disease by six main syndromes, which guide diagnosis and development of personalized treatment. TCM combines modalities such as acupuncture, tai chi, and herbal medicine. One study used herbal medicine alone, while the other used this in combination with acupuncture. Both studies reported a complete resolution of GI symptoms, including nausea, abdominal discomfort, bloating, acid reflux, loss of appetite, and diarrhea by the end of the treatment period.29,30
The CAM therapies included in this review belong to the NCCIH categories of nutrition and combination. While these studies primarily targeted the gut microbiome and the inflammatory response of the GI tract, no interventions targeted the gut–vagal–brain axis (GVBA). Neuro-modulatory techniques, such as gut hypnotherapy, have been effective in treating functional GI disorders of the GVBA, such as IBS.42,43 Yoga and meditation have also shown benefits in treating symptoms of IBS. 44 Post-COVID IBS has become increasingly recognized, and Long COVID frequently has neuropsychiatric manifestations along with GI symptoms.5–7,45 Thus, future studies should target the therapeutic potential of this area of intervention.
The use of CAM in Long COVID holds significant potential in addressing a wide array of symptoms. For example, one study reported the benefit of TCM in patients with convalescent stage COVID-19 by improving radiographic signs of lung inflammation. 46 A case report documented improvement in Long COVID symptoms and decreased radiographic pulmonary exudates and fibrotic lesions. 47 Another study found significant improvement in cough, fatigue, and subjective well-being scores among patients with Long COVID being treated with a prebiotic and probiotic capsule for 30 days. 48 Other research has suggested the positive impact of acupuncture on Long COVID recovery. 49 While the current literature on CAM in Long COVID-19 remains limited, particularly regarding GI manifestations, it is essential to expand the understanding of these therapies and their potential role in managing this debilitating condition.
While this review revealed several positive studies, the overall search yielded few results, and all resulting studies were non-experimental. Although the included studies adhere to the CARE guidelines, the observational nature of case reports and case series limits the ability to draw definitive conclusions about the efficacy of CAM interventions in this context. Given the protracted disease course of Long COVID, patients may have improved spontaneously over time without the CAM intervention; however, the lack of a control group in the studies makes it impossible to determine this. In addition, no variant-specific data were available in these studies, thus limiting the certainty of using these approaches in treating different SARS-CoV-2 variants. Due to the limited number of patients included in these studies, there is minimal generalizability of the findings. Therefore, while the adherence to CARE guidelines supports the quality of the included studies, their overall trustworthiness and applicability to a broader population should be interpreted with caution.
This is the first study to comprehensively evaluate CAM approaches for treating the GI symptoms of Long COVID. Given the global impact of the pandemic, CAM provides an attractive area for future investigation, as this review suggests some therapeutic benefits and allows a multicultural approach to care. 50
As the sequelae of COVID-19 infections become better recognized, this review brings attention to the need for higher-quality, randomized controlled trials to explore the safety and efficacy of CAM in treating these distressing symptoms. In addition, future studies should obtain microbiome data as a means of further characterizing therapeutic response, which could provide additional mechanistic insight into the underlying drivers of the Long COVID phenomenon.
Conclusion
With the global persistence of SARS-CoV-2, an increasing number of individuals are at risk for developing chronic symptoms following initial infection, which results in a particularly debilitating condition. This systematic review identified several studies reporting the benefit of CAM approaches in treating the GI manifestations of Long COVID, emphasizing the potential role of CAM in optimizing the care of these patients. However, there is limited data regarding the use of CAM in treating Long COVID and only four case-based studies are ultimately included in this review. While the number of included studies is insufficient to draw definitive conclusions, this review serves as an exploratory investigation, providing preliminary information for future clinical trials in this area. Importantly, this study highlights the need for higher-quality experimental outcome studies to rigorously assess the safety and efficacy of CAM in treating these distressing symptoms.
In conclusion, this review offers an exploratory and timely perspective on the role of CAM in addressing GI symptoms associated with Long COVID. By presenting initial insights into the utilization of CAM therapies for Long COVID, this review aims to stimulate further scientific inquiry and encourage the development of more effective treatment strategies. As our understanding of Long COVID continues to evolve, CAM therapies may increasingly contribute to the holistic management of this complex condition.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231190548 – Supplemental material for The use of complementary and alternative medicine for the treatment of gastrointestinal symptoms in Long COVID: a systematic review
Supplemental material, sj-docx-1-taj-10.1177_20406223231190548 for The use of complementary and alternative medicine for the treatment of gastrointestinal symptoms in Long COVID: a systematic review by Brent Gawey, Juan Yang, Brent Bauer, Joanne Song and Xiao Jing Wang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223231190548 – Supplemental material for The use of complementary and alternative medicine for the treatment of gastrointestinal symptoms in Long COVID: a systematic review
Supplemental material, sj-docx-2-taj-10.1177_20406223231190548 for The use of complementary and alternative medicine for the treatment of gastrointestinal symptoms in Long COVID: a systematic review by Brent Gawey, Juan Yang, Brent Bauer, Joanne Song and Xiao Jing Wang in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We thank Mr. Larry Prokop for his expertise in assisting with the literature search. We also thank The HEAD Foundation, Singapore for their support of Dr. Yang and Dr. Bauer.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
