Abstract
Background:
Sleep disturbance and trace elements imbalance are common features in patients with decompensated cirrhosis, partially sharing similar mechanistic contributors and linking to adverse outcomes. However, there is a paucity of data concerning their relationship.
Objectives:
To investigate the association between serum trace elements levels and sleep quality in the context of cirrhosis.
Design:
Cross-sectional study.
Methods:
We consecutively enrolled 160 patients with decompensated cirrhosis. The sleep disturbance was determined by the Pittsburgh Sleep Quality Index (PSQI > 5). Serum trace elements [magnesium, calcium, iron, copper (Cu), zinc (Zn), lead, and manganese] was measured by inductively coupled plasma mass spectrometry. Association of examined trace elements levels and sleep disturbance was analyzed by multiple linear (global PSQI scores) and multivariate logistic (dichotomized PSQI categories) regression models, respectively.
Results:
In total, 91 patients (56.88%) represented PSQI-defined sleep disturbance, characterized by female preponderance, lower body mass index levels, and higher serum Cu levels (all p < 0.05). Looking into its clinical relevance with debilitating conditions, we showed that Cu/Zn ratio (CZr) is significantly higher in cirrhosis with poor sleep quality (1.77 versus 1.48, p = 0.003). Diagnostic performance analysis indicated CZr > 1.62 to exhibit better discrimination relative to respective Cu. Both multiple linear (β = 0.355, p < 0.001) and multivariate logistic regression (odds ratio = 2.364, p = 0.019) identified higher CZr as an independent risk factor associated with sleep disturbance.
Conclusion:
Our findings implied an association between higher CZr and the presence of sleep disturbance in patients with decompensated cirrhosis.
Introduction
Sleep is well documented to be essential for both physical and psychological health, characterized as a highly active, regulator but metabolically distinct process. 1 The mechanistic basis of sleep remains enigmatic, while it is speculated that both homeostatic driver and circadian oscillation tightly and mutually govern this complex behavior. 2 Notably, imbalance and dysregulation concerning these interlinking systems may lead to sleep disorders, and in consequence, promoting the advent and development of various liver diseases. 3 On the other hand, progression to cirrhosis, known as a terminal stage of different etiologies, is accompanying with additionally predisposing factors and pathogenic stressors like inflammatory activities, oxidative stress and insulin resistance.4,5 Intriguingly, abovementioned contributors also partially account for the pathogenesis of sleep disturbance.6–8 Looking into these close connections, it is anticipated that there is a considerably high prevalence of sleep disturbance among patients with cirrhosis in the existing literature.9,10 Our previous results also implied that more than half of cirrhotics manifested with poor sleep quality assessed by the Pittsburgh Sleep Quality Index (PSQI). 11 On the contrary, there remains unmet clinical need and lack of effective therapeutic option to manage sleep disturbance in the context of cirrhosis, due to significant side-effect and mixed effectiveness pertinent to pharmacological administrations such as zolpidem, hydroxyzine, and exogeneous melatonin.12–14 Collectively, it is imperative to pursue and clarify other alternative therapies. Accordingly, dietary supplements including various trace elements have been reported to improve sleep disturbance with little adverse events. 15
In patients with cirrhosis, dysregulated and imbalanced trace elements are frequently encountered, since the livers plays an indispensible and pivotal role for the maintenance of their homeostasis.16,17 Accumulating evidence has demonstrated a deficiency or an excess of essential trace elements, both of which may negatively impact prognostication in cirrhosis, attributable to pathophysiological pertubation in relation to aggravated inflammatory actions, inflammatory cascades as well as fibrogenic changes in the hepatic tissues.18–20 To counter those deleterious processes, some researches sought to elucidate the therapeutic effects regarding the supplementation of trace elements or their dietary intake influence in different populations with encouraging preliminary findings.21–23 For instance, Baradari et al. 21 found that sufficient supplementation with zinc (Zn) significantly increased serum Zn levels aligning with improved subjective sleep quality scored by the PSQI among intensive care unit nurses. Another study revealed that dietary magnesium intake may be beneficial to reduce the possibilities of daytime falling asleep in females. 22 Although these reports have shed light on novel therapeutic strategy to restore trace elements abnormalities aiming at ameliorating sleep disorders, little evidence is available to target abnormal trace elements levels in patients with cirrhosis and sleep disturbance. Taken together, we hypothesized that imbalanced trace elements are closely linked to poor sleep quality among patients with cirrhosis. We firstly delineated the differences of several trace elements among groups stratified by PSQI-defined sleep quality. Next, the association between the most pronounced trace elements (or relevant parameter) and sleep disturbance in cirrhotics was investigated in this cross-sectional study.
Materials and methods
Study cohort
This is a cross-sectional study by consecutively enrolling patients with cirrhosis hospitalized due to decompensating events at the Department of Gastroenterology and Hepatology, Tianjin Medical University General Hospital (TJMUGH). Study inclusion criteria referred to (1) age ⩾18 years, (2) capability to complete questionnaire pertinent to sleep quality, and (3) informed consent. The sample size was determined by the period of the study (from 2019 to 2021) and the number of admissions rather than calculation due to a paucity of valid prevalence regarding trace elements imbalance in the context of cirrhosis. This study conformed to the Declaration of Helsinki and was approved by Ethics Committee of TJMUGH (IRB-YX-004-01). The diagnosis of decompensated cirrhosis was built according to medical history along with a spectrum of workup including radiologic findings, biopsy results, elastography assessment, and endoscopic data. Furthermore, the decompensating events were recorded indicative of ascites, variceal hemorrhage, serious jaundice, and hepatic encephalopathy (HE). 24 We excluded from final analyses all participants having acute-on chronic liver failure, concomitant malignancies, uncontrolling Wilson’s diseases, consistently renal dysfunction with eGFR < 60 mL/min, using ongoing trace elements supplement and severe HE. In our department, HE was detected from the time to complete the number connection test performed upon hospitalization and categorized as presence if ⩾60 s needed to complete the test.25,26 Therefore, aforesaid exclusion criterion with respect to severe HE alluded to complete a number connection test of more than 120 s, equivalent to West Haven ⩾2. 27 Consistent with previous reports, we did not exclude patients with active alcohol since they represent significant subsets of cirrhotics.9,28 All baseline biochemical examinations were obtained within 24 h of index hospitalization. The written informed consent was provided by each participant.
Collection of data and measurement of serum trace elements
The following data were obtained at the time of inclusion: demographic features (age and sex), etiology of cirrhosis [chronic viral infection, alcoholic, autoimmune liver disease (AILD), cholestasis, non-alcoholic fatty liver disease (NAFLD), and cryptogenic cirrhosis], blood test results [total bilirubin, alanine aminotransferase (ALT), alkaline phosphatase (ALP), platelet, albumin, hemoglobin, electrolytes, prothrombin time-international normalized ratio (PT-INR), creatinine, and neutrophil-to-lymphocyte ratio (NLR)], and the presence of cirrhosis-related complications. The liver disease severity was classified by the model for end-stage liver disease (MELD) score and Child-Turcotte-Pugh (CTP) class.
Measurement of serum trace elements was in agreement with our previously applied methodology. 29 Taking into consideration the circadian variability of serum trace elements, we collected all samples at the early morning from fasting participants within 24 h upon hospitalization. In brief, collected peripheral blood samples in serum separating tubes were kept at room temperature for 30 min, and then set out for centrifugating and storing at −80°C in aliquots. In total, seven trace elements, including magnesium (Mg), calcium (Ca), iron (Fe), copper (Cu), Zn, lead (Pb), and manganese (Mn), were assessed using inductively coupled plasma mass spectrometry (ICP-MS, Agilent 7900, Agilent Technologies, Santa Clara, CA, USA) following the manufacturer’s instruction.
Assessment of sleep quality
Sleep quality was assessed via the PSQI of a Chinese version aimed at identifying poor sleepers (a global PSQI score of >5), which has been widely implemented and validated on the basis of population epidemiological studies. 30 It encompasses a range of 10 queries covering seven dimensions of sleep pattern: (i) subjective sleep quality, (ii) sleep latency, (iii) sleep duration, (iv) habitual sleep efficiency, (v) sleep disturbances, (vi) sleep medication use, and (vii) daytime dysfunction. Each dimension scores from 0 to 3 points. Accordingly, we calculate the total score by summing seven dimensions together, indexing from 0 to 21, where a higher value refers to poor sleep quality.
Statistical analysis
Descriptive statistical analysis was used to characterize baseline features of recruited population. Statistics were depicted as mean ± standard deviation, median [interquartile range (IQR)] and number (proportion) as appropriate. Continuous variables were compared by the Student’s t test or Mann–Whitney U test in cases with a skewed distribution. Categorical variables were compared by χ2 test or Fisher’s exact test. The optimal cutoff regarding serum trace elements levels was determined when the Youden index exhibited highest value according to area under the curve (AUC). The receiver operating characteristics (ROC) curve was used to calculate sensitivity and specificity of index parameter. Multiple linear regression and multivariate logistic regression models were used to determine independent risk factors, including serum trace elements and relevant parameter, for the presence of PSQI-defined sleep disturbance (poor sleep quality) as continuous values and dichotomized categories, respectively. The variables with a p < 0.1 were included in the subsequent multivariable analysis. The SPSS version 21.0 (IBM, New York, NY, USA) and Graphpad Prism version 8.0.1 (GraphPad software, La Jolla, CA, USA) were carried out for all statistical analyses. We considered p < 0.05 as statistically significant differences.
Results
Characteristics of the study population
As described in Table 1, we showed the baseline demographic and biochemical information of the study population. Overall, 160 patients with decompensated cirrhosis were included with a mean age of 62.41 ± 9.48 years, and 53.75% of them were male. The prominent etiologies of cirrhosis were AILD/cholestasis (30.62%) and alcoholism (29.38%), followed by chronic viral hepatitis and cryptogenic/NAFLD. Ninety-nine (61.88%) patients had ascites upon hospitalization, when this number was 15 with HE (9.38%). Within the entire cohort, 46 (28.75%) were classified as CTP class A, 83 (51.88%) as class B, and 31 (19.37%) as class C. The median MELD score was 9 (IQR: 6, 12) points. Ninety-one (56.88%) participants were classified as poor sleepers, when those with global PSQI scores of >5 were considered as having sleep disturbance. Compared with good sleepers, those with sleep disturbance were inclined to be female (53.85 versus 36.23%, p = 0.037) and present with lower body mass index (BMI) levels (22.07 versus 25.83 kg/m2, p < 0.001).
Characteristics of hospitalized cirrhotic patients classified by PSQI score.

Data were expressed as mean ± standard deviation, median (IQR), and number (proportion) as appropriate. We classify all cirrhotic patients into good sleepers (PSQI ⩽ 5) and poor sleepers (PSQI > 5).
AILD, autoimmune liver disease; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; BMI, body mass index; Ca, calcium; CTP, Child-Turcotte-Pugh; Cu, copper; CZr, Cu/Zn ratio; Fe, iron; HE, hepatic encephalopathy; IQR, interquartile range; K, potassium; MELD, model for end-stage liver disease; Mg, magnesium; Mn, manganese; Na, sodium; NAFLD, non-alcoholic fatty liver disease; NLR, neutrophil-to-lymphocyte ratio; Pb, lead; PSQI, Pittsburgh Sleep Quality Index; PT-INR, prothrombin time-international normalized ratio; TBIL, total bilirubin; Zn, zinc.
Comparison of serum trace elements levels according to sleep quality
No significant differences were observed with the exception of Cu between good and poor sleeper groups. Notably, patients with sleep disturbance had considerably higher levels of Cu than those without (108.37 versus 98.81 μg/dL, p = 0.012). Next, we considered synergistic and combined effects of Cu and Zn on sleep architecture and other health status;31,32 thus comparison pertaining to Cu/Zn ratio (CZr) was performed between groups. Intriguingly, our results indicated that serum CZr is significantly increased in poor sleepers compared to good sleepers (1.77 versus 1.48, p < 0.003).
Determination of cutoff to stratify sleep disturbance
Given the significant differences concerning serum Cu levels and CZr between patients with good relative to poor sleep quality, we further sought to identify sleep disturbance in cirrhotics by determining the optimal cutoff (Figure 1). In terms of ROC analysis, Cu > 81.87 μg/dL represented best discriminative capability with an AUC of 0.616 [95% confidence interval (CI): 0.526, 0.706, p = 0.012], sensitivity of 33.33%, and specificity of 92.31%. As expected, CZr exhibited a higher AUC value, that is 0.638 (95% CI: 0.552, 0.724, p = 0.003, sensitivity: 60.87%, specificity: 62.64%), for classifying participants into distinct groups. Taken together, we selected CZr as the surrogate in relation to sleep disturbance in the context of cirrhosis. Overall, no significant differences were found between groups regarding age, sex, presence of HE, and MELD scores. On the contrary, poor sleepers were more prevalent in the group with CZr > 1.62 than with CZr ⩽ 1.62 (67.06 versus 45.33%, p = 0.007). In addition, PSQI scores were significantly higher in patients with CZr > 1.62 compared to those with CZr ⩽ 1.62 (8 versus 5, p < 0.001).

ROC analysis of serum CZr levels for classifying sleep disturbance in patients with decompensated cirrhosis.
Although the BMI, potassium, NLR, PT-INR, hemoglobin, ALT, and total bilirubin were similar between the groups, we showed the group with CZr > 1.62 has higher proportions of alcoholic and AILD/cholestatic cirrhosis, ascites, and CTP class C, higher serum platelet and ALP levels than with CZr ⩽ 1.62 (Table 2). However, the group with CZr > 1.62 exhibited lower serum sodium and albumin levels than with CZr ⩽ 1.62.
Characteristics of hospitalized cirrhotic patients classified by CZr.

Data were expressed as mean ± standard deviation, median (IQR) and number (proportion) as appropriate. We classify all cirrhotic patients into good sleepers (PSQI ⩽ 5) and poor sleepers (PSQI > 5).
AILD, autoimmune liver disease; ALB, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; ; BMI, body mass index; CTP, Child-Turcotte-Pugh; CZr, Cu/Zn ratio; HE, hepatic encephalopathy; IQR, interquartile range; K, potassium; MELD, model for end-stage liver disease; Na, sodium; NAFLD, non-alcoholic fatty liver disease; NLR, neutrophil-to-lymphocyte ratio; PSQI, Pittsburgh Sleep Quality Index; PT-INR, prothrombin time-international normalized ratio; TBIL, total bilirubin.
Identification of risk factor independently related to sleep disturbance
Next, we investigated whether higher CZr is an independent risk factor for the presence of sleep disturbance. The overall population were dichotomized into two distinct groups referring to good and poor sleepers judged by global PSQI scores of >5. As depicted in Table 3, the univariate analysis indicated that females [odds ratio (OR): 2.053, 95% CI: 1.082, 3.898, p = 0.028], BMI (OR: 0.862, 95% CI: 0.792, 0.938, p = 0.001), and CZr >1.62 (OR: 2.455, 95% CI: 1.293, 4.662, p = 0.006) are associated with sleep disturbance. Multivariate logistic regression analysis further verified that CZr > 1.62 (OR: 2.364, 95% CI: 1.149, 4.864, p = 0.019) is independently associated with sleep disturbance adjusting for sex and BMI.
Univariate and multivariate logistic regression analyses to determine risk factors associated with sleep disturbance according to dichotomized PSQI categories.

Bold font indicated statistical significance.
Adjusting for sex, BMI, and CZr.
ALT, alanine aminotransferase; BMI, body mass index; CI, confidence interval; CTP, Child-Turcotte-Pugh; CZr, Cu/Zn ratio; HE, hepatic encephalopathy; Na, sodium; MELD, model for end-stage liver disease; OR, odds ratio; PSQI, Pittsburgh Sleep Quality Index; TBIL, total bilirubin.
Another sensitivity analysis was carried out by employing continuous PSQI scores to confirm the validity of our findings (Table 4). In simple regression analysis, PSQI scores were associated with female (β coefficient = 0.168, 95% CI: 0.013, 0.323, p = 0.034), BMI (β coefficient = −0.259, 95% CI: −0.421, −0.096, p = 0.002), and CZr (β coefficient = 0.278, 95% CI: 0.127, 0.429, p < 0.001). Similar result was found incorporating CZr in multiple linear regression analysis, rendering it an independent risk factor for poor sleep quality assessed by PSQI.
Simple and multiple linear regression analyses to determine risk factors associated with sleep disturbance according to global PSQI scores.
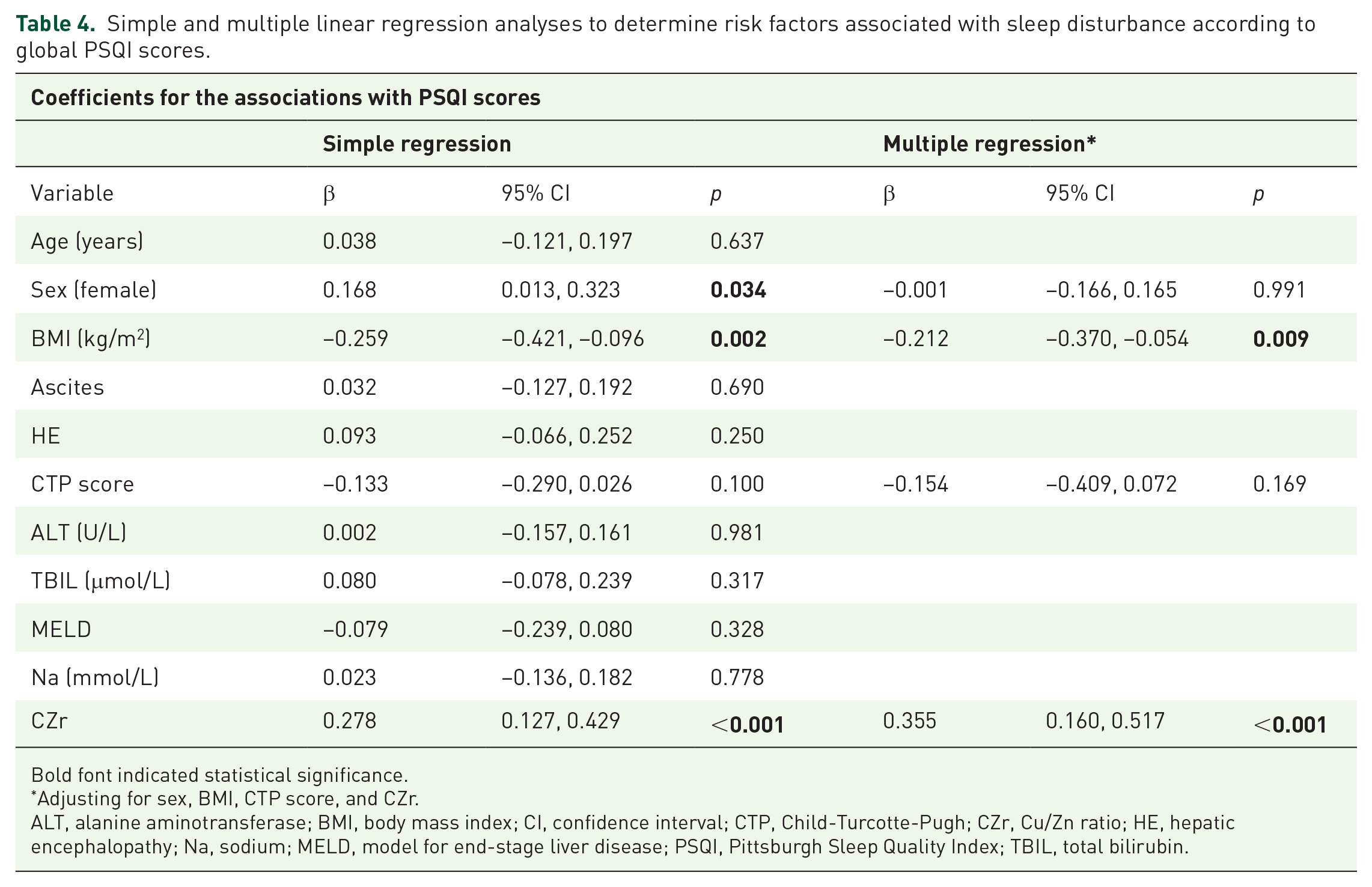
Bold font indicated statistical significance.
Adjusting for sex, BMI, CTP score, and CZr.
ALT, alanine aminotransferase; BMI, body mass index; CI, confidence interval; CTP, Child-Turcotte-Pugh; CZr, Cu/Zn ratio; HE, hepatic encephalopathy; Na, sodium; MELD, model for end-stage liver disease; PSQI, Pittsburgh Sleep Quality Index; TBIL, total bilirubin.
Correlation between serum Cu levels/CZr and PSQI
In the overall population, we observed a positive correlation between serum Cu levels (rs = 0.272, p < 0.001)/CZr (rs = 0.310, p < 0.001) and PSQI scores (Figure 2). When classified by sleep disturbance, a positive correlation between serum Cu levels (rs = 0.256, p = 0.014)/CZr (rs = 0.235, p = 0.025) with PSQI scores was evident in poor sleepers with decompensated cirrhosis. On the contrary, no significant correlation was found between serum Cu levels (rs = 0.094, p = 0.444)/CZr (rs = 0.166, p = 0.172) in good sleepers.

Correlation analysis between serum Cu levels and PSQI scores in the overall population (a), poor sleepers (b), and good sleepers (c). In addition, correlation analysis between CZr and PSQI scores in the overall population (d), poor sleepers (e), and good sleepers (f).
Discussion
Accumulating evidence implies that disrupted sleep may contribute to the development and progression of various liver diseases and vice versa, in particular, their confluence is substantially related to impaired quality of life and poor physical/psychological health.33–36 Of note, the proportion of decompensated cirrhotics with sleep disturbance in the present study was 56.88%, compatible with data in the existing literature. 37 Our preliminary findings revealed that CZr and serum Cu levels are significantly higher in patients manifested with poor sleep quality relative to good sleepers. Additionally, CZr > 1.62 exhibited better discriminative performance than serum Cu levels in separation to identify poor sleepers, and was independently associated with sleep disturbance after adjusting for sex and BMI. A higher CZr was independently associated with a higher PSQI score. Finally, CZr was positively correlated with global PSQI score, especially among the sleep disturbance setting in the context of cirrhosis.
As a matter of fact, it is a common manifestation that patients with chronic liver diseases experiencing dysregulation to trace elements metabolism, as evidenced by increased levels of Cu and Fe along with decreased levels of Zn, selenium, Ca, and Mg. 38 More recently, the clinical relevance concerning serum trace elements imbalance has leveraged on a spectrum of pathological entities, debilitating conditions, and patient populations. For instance, Asker et al. 39 showed that serum trace minerals levels (Mg, Cu, Zn, Fe, Mn, and Pb) are increased among patients with obstructive sleep apnea, known as a sleep breathing disorder, and speculated these changes may serve as favorable biomarkers pertinent to oxidative stress, inflammatory response, and disease status. Furthermore, a cross-sectional study based on the National Health and Nutrition Examination Survey recruiting 5076 subjects indicated that serum Zn levels and the Zn/Cu ratio are inversely related to long sleep duration in adults. 31 Xu et al. 40 also reported a connection between lower blood selenium contents and severe sleep disturbance in MHD populations, whose pathogenesis can be attributed to anti-oxidative and anti-inflammatory activities of selenium. Taken together, our results fill a significant gap by addressing higher CZr as a potential surrogate to identify patients with decompensated cirrhosis and concurrent sleep disturbance.
A major finding of the present study characterized CZr > 1.62 to be an independent risk factor for PSQI-defined sleep disturbance, exhibiting better discriminative performance relative to serum Cu levels judged by AUC analysis. Likewise, the standardized coefficient was demonstrated with the strongest value (β = 0.355, p < 0.001) in comparison with other variables. Therefore, it is tempting and practical to manage Cu and Zn imbalance with the purpose of restoring abnormal sleep parameters. As a matter of fact, Himoto et al. administered polaprezinc (a complex of Zn and L-carnosine) on 14 patients with HCV-related chronic liver diseases for 6 months in addition to their regular therapeutics. 41 Notably, a considerable increase in serum Zn levels was observed following medicinal supplementation, being in alignment with attenuated hepatic dysfunction and liver inflammation. Last but not least, the serum Cu levels were not influenced at this dosage of Zn supplementation (225 mg/day orally). Collectively, it is speculated to deliberately manage CZr on the basis of selective drugs and appropriate dose, since excessive Zn administration may partially account for the Cu deficiency by inhibiting its absorption. 42
Multiple converging pathways are responsible for serum trace elements dysregulation and sleep disturbance among different patient populations and diseases settings. As we herein addressed a novel knowledge with respect to a pathogenic link between higher CZr and the presence of sleep disturbance, several potential interpretations should be stated as following. First, the serum Cu and Zn levels are strictly relevant to their respective carrier proteins, that is, ceruloplasmin and albumin in plasma. These proteins are mainly synthesized and regulated at the hepatic levels. Although there is a paucity of data regarding ceruloplasmin in the study population, significantly lower albumin levels were found in patients with CZr > 1.62 (26 versus 30 g/L, p < 0.001). Therefore among the contributory factors for increased CZr, it is indispensible to consider inadequate nutrition fostering increased synthesis and secretion of Cu-ceruloplasmin as well as reduced synthesis and secretion of Zn-albumin. Actually, malnutrition is suggested to be one of the most common complications in cirrhosis. 43 It was noted that poor sleepers have significantly lower BMI values constituting a major criterion to evaluate nutritional status among Asian populations, which has been advocated by the Global Leadership Initiative on Malnutrition aiming at standardizing the diagnosis of malnutrition and facilitating data comparison across national borders and disease settings. 44 Furthermore, we previously corroborated a close link between PSQI-defined sleep disturbance and high malnutrition risk. 45 These results in combination reveal that lower BMI is a phenotypic representation of malnutrition in the context of cirrhosis. Second, patients with cirrhosis are susceptible to debilitating condition designated as cirrhosis-associated immune dysfunction, characterized by combined immunodeficiency and systemic inflammation. 46 Plenty of studies have unraveled that inflammatory activities can impact serum Cu and Zn levels by upregulating the former and downregulating the latter via a hepatic organized mechanism.47,48 Notably, pro-inflammatory cytokines were capable of enhancing the Zip-14 expression, that is a Zn transporter, and eliminating serum Zn in the liver during inflammation. 49 Last, both infection and inflammatory cascade induce oxidative stress, and in consequence raise serum Cu-ceruloplasmin levels. Likewise, many physiological and pathological pertubations pertaining to the redox state of human systemic albumin substantially affect its binding properties. 50 Overwhelming oxidative stress is responsible for Zn ions displaced from albumin along with circulatory decreased albumin bound fraction of Zn.51,52 Collectively, it is argued that CZr may serve as a solid surrogate clinically with predictive and prognostic role in a wide range of pathological entities. 32 In addition, its utility appears to be comparable with or outperform other routine inflammation markers (e.g., erythrocyte sedimentation rate, C-reactive protein).
Concerning the combined effect of Cu and Zn on sleep disturbance, we speculate the positive correlation between CZr and PSQI score additionally provides therapeutic avenue. More recently, Japan’s Practical Guideline has recommended sufficient Zn administration for patients with low serum Zn levels, even if they are devoid of Zn deficiency symptoms. 53 Intriguingly, our results demonstrated that decompensated cirrhotics with concomitant sleep disturbance exhibit relatively lower Zn levels in comparison with good sleep counterparts, although this disparity did not reach statistical significance. The values referring to 60.35 μg/dL of serum Zn in poor sleepers indeed corresponded to marginal diagnostic criterion according to proposed guideline (i.e., 60–80 μg/dL) as well as our previously established cutoff (i.e., <64 μg/dL).29,53 Therefore, a low Cu diet with monitored Zn replacement is presumed to restore CZr for patients with decompensated cirrhosis and sleep disturbance, given these populations may already have high/normal Cu levels. Moreover, elevated CZr has also been linked to increased hepatocellular carcinoma-related mortality. 54 Notably, the positive connection was only found in poor sleepers with cirrhosis (Figure 2), indicating more intensively therapeutic scale and feasible administration of Zn for this group to avoid possible side effects. Because a challenging issue exists regarding the optimal dose and duration of Zn supplementation, practitioners should be aware of acute/chronic Zn toxicity such as gastric discomfort, impaired immune function, and decreased high density lipoprotein.55,56
The limitations of the current study merit discussion. First, this was a cross-sectional study on the basis of a relatively small sample size; therefore, an inevitable bias existed given the study design. Second, we only applied PSQI to determine sleep disturbance. Although this metric has been validated with acceptable sensitivity and specificity, further studies using objective sleep measurements (e.g., wearable devices, polysomnography) are needed. Third, we did not collect information on the dietary intake of cirrhotics considering its pivotal role on maintaining circulatory and organic trace elements levels. Of note, patients with cirrhosis may experience insufficient oral intake attributable to early satiety, anorexia, dysgeusia, consciousness impairment, and unpalatable prescribed diets. 57 Furthermore, malabsorption, altered protein metabolism, and hepatic dysfunction all result in disturbances to trace elements metabolism and impairment of trace elements homeostasis. 17 In this regard, further studies should take account distinct patterns of dietary intake as confounding variables to delineate relationship between any trace elements level and sleep disorders in this population. Fourth, we excluded patients with severe HE which may give rise to selection bias. Last, there was a lack of information with respect to some essential trace elements reported in other studies due to limited resource, such as selenium and cobalt. However, our preliminary findings are likely to pave the way for further multi-center investigation, taking into account the clinical importance of both trace elements imbalance and sleep disturbance on cirrhosis progression.
Conclusion
In summary, this is the first study to uncover an association between higher CZr and PSQI-defined sleep disturbance in patients with decompensated cirrhosis. To improve sleep quality in this population, we recommend that CZr is actively managed via the appropriate prescription of therapeutic agents.
