Abstract
Background:
There has been an increasing use of transcatheter tricuspid valve repair (TTVR) recently. However, the periprocedural, short-term, and long-term outcomes of TTVR remain unclear.
Objectives:
To determine the clinical outcomes in patients with significant tricuspid regurgitation undergoing TTVR.
Design:
Systematic review and meta-analysis.
Data Source and Methods:
The systematic review and meta-analysis is reported according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. PubMed and EMBASE were searched for clinical trials and observational studies until March 2022. Studies reporting the incidence of clinical outcomes after TTVR were included. The clinical outcomes included periprocedural, short-term (in-hospital or within 30 days), and long-term (>6-month follow-up) outcomes. The primary outcome was all-cause mortality whereas the secondary outcomes included technical success, procedural success, cardiovascular mortality, rehospitalization for heart failure (HHF), major bleeding, and single leaflet device attachment. The incidence of these outcomes across studies was pooled by a random-effects model.
Results:
A total of 21 studies with 896 patients were included. A total of 729 (81.4%) patients underwent isolated TTVR while only 167 (18.6%) patients underwent combined mitral and tricuspid valve repair. Over 80% of the patients used coaptation devices while approximately 20% used annuloplasty devices. The median follow-up duration was 365 days. Technical and procedural success was high at 93.9% and 82.1%, respectively. The pooled perioperative, short-term, and long-term all-cause mortality for patients undergoing TTVR was 1.0%, 3.3%, and 14.1%, respectively. The long-term cardiovascular mortality rate was 5.3% while the HHF rate was 21.5%. Major bleeding and single leaflet device attachment were two major complications, accounting for 14.3% and 6.4%, respectively, during long-term follow-up.
Conclusion:
TTVR is associated with high procedural success and low procedural and short-term mortality. However, all-cause mortality, cardiovascular mortality, and HHF rates remain high during long-term follow-up.
Registration:
PROSPERO (CRD42022310020).
Introduction
Tricuspid regurgitation (TR) is highly prevalent in echocardiography, of which 14.6% are classified as primary and 85.4% are classified as functional. 1 The prevalence of moderate or greater TR was 0.55%, which was estimated to be approximately one-fourth of all left-sided valve diseases, similar to the prevalence of aortic stenosis. 1 Moderate or greater TR (significant TR) has been associated with increased risks of cardiac and all-cause mortality.2,3 Despite improvements in surgical techniques and optimal medical therapy, isolated tricuspid valve surgery remains a high-risk surgery with an in-hospital mortality rate approaching 10%. 4 Therefore, tricuspid valve surgery is recommended to be concomitantly performed with left-sided valve surgery according to current guidelines. 5
The high surgical risks and mortality paved the way for transcatheter interventions. The transcatheter tricuspid valve intervention (TTVI) includes transcatheter tricuspid valve repair (TTVR), orthotopic tricuspid valve replacement (NaviGate devices), and heterotopic caval valve implantation (CAVI). 6 Depending on the anatomic target, TTVR can be further categorized as follows: (1) coaptation devices, including direct coaptation devices [transcatheter edge-to-edge repair (MitraClip, TriClip, PASCAL)] and indirect coaptation devices (FORMA and Mistral) and (2) annuloplasty devices, including direct annuloplasty devices (TriCinch, Trialign) and ring devices (Cardioband).7,8 Previous systematic reviews concluded that TTVR was feasible and effective in the reduction of TR severity and improvement of symptoms and exercise capacity in patients with severe TR.9,10 However, these studies mainly focused on functional and echocardiographic improvements, and major adverse events were rarely reported in their analyses. Therefore, we conducted this systematic review and meta-analysis to assess the periprocedural, short-term, and long-term effectiveness and safety outcomes of TTVR in patients with significant TR.
Methods
We performed the search strategy, study selection, data extraction and data analysis in line with the Cochrane Collaboration and the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. 11 The systematic review and meta-analysis were registered in the International Prospective Register of Systematic Reviews (PROSPERO; CRD42022310020).
Literature retrieval
We systematically searched PubMed and Embase for clinical trials and observational studies until March 2022 using the following search terms: (1) percutaneous OR transcatheter and (2) tricuspid. The Boolean operator ‘and’ was used to combine the two search terms (Supplementary Table 1). We also checked the reference lists of the retrieved articles and previous meta-analyses and reviews for additional eligible studies. In addition, to avoid missing eligible articles, we also checked citations from the Transcatheter Cardiovascular Therapeutics (TCT) conference.
Inclusion and exclusion criteria
We included studies if they fulfilled the following criteria: (1) they included patients with greater or equal to moderate TR who underwent TTVR, regardless of whether isolated TTVR (ITTVR) or concomitant transcatheter mitral and tricuspid valve repair (TMTVR) was being performed; and (2) they reported at least one of the clinical outcomes of interest. Studies including fewer than five patients were excluded. In addition, studies that included patients undergoing transcatheter TV replacement and CAVI without reporting the specific outcomes of the TTVR subgroup were also excluded due to the heterogeneity of the interventions. Case reports, comments, editorials, and reviews were excluded due to insufficient data. For studies with overlapping populations, the study with the largest sample size was selected. To elucidate the clinical outcomes at different follow-up durations, studies reporting the same cohort at different stages were also included into the analysis of periprocedural, short-term, and long-term outcomes, respectively.
Study outcomes
The clinical outcomes included periprocedural, short-term (in-hospital or within 30 days), and long-term (>6-month follow-up) outcomes. The primary outcome was all-cause mortality. The secondary outcomes included technical success, procedural success, cardiovascular mortality, conversion to surgery or reintervention, rehospitalization for heart failure (HHF), major bleeding, access site complications, acute kidney injury, stroke, acute myocardial infarction, coronary complications, arrhythmia, cardiac tamponade, pulmonary embolism, device-related thrombosis and single leaflet device attachment (SLDA). Technical success, also referred to as device success, was defined as successful device implantation in the absence of major device- or procedure-related serious adverse events. 12 Procedural success was defined as the successful implantation of at least 1 device with a postprocedural TR grade ⩽2+, with no mortality or conversion to surgery. 13 Other definitions of the outcomes were consistent with the retrieved studies.
Data extraction
The data were extracted independently by two authors (Z.W., and W.Z.). Duplicates were removed first, and titles and abstracts were screened followed by full texts studies for potential eligibility.
From the retrieved studies, the following characteristics were extracted: first author, year of publication, inclusion period, study design, data source, device types, sample size, number of ITTVR and TMTVR patients, patient age and sex, European System for Cardiac Operative Risk Evaluation II (EuroSCORE II) score, proportion of functional TR, medical history of hypertension, diabetes mellitus, atrial fibrillation, permanent pacemaker (PPM)/implantable cardioverter defibrillator (ICD)/cardiac resynchronization therapy (CRT) implantation, follow-up time and number of aforementioned events.
Study quality assessment
The Newcastle-Ottawa Scale (NOS) was used to evaluate the quality of the included studies. 14 The NOS tool consists of three domains and a total score of 9 points, including cohort selection (4 points), cohort comparability (2 points), and outcome assessment (3 points). Studies with an NOS score of ⩾ 6 points were considered moderate-to-high quality studies, whereas studies with an NOS score of < 6 points were considered as low-quality studies.
Statistical analysis
We estimated the incidence of procedural, short-term, and long-term outcomes and reported them separately. To explore the incidence of the clinical outcomes, we used the exact binomial (Clopper-Pearson) method to calculate 95% confidence intervals (CIs). The Freeman-Tukey double arc-sine transformation was used for standard estimates.15,16 All the estimates were presented after back transformation. Consistency across the included studies was assessed by Cochran’s Q statistic and the I2 statistic. Heterogeneity was considered to be low for I2 < 25%, moderate for 25% <I2 < 75%, and high for I2 > 75%. 17 Due to the study design of the single-arm study and a tendency toward high heterogeneity, a random-effect model (DerSimonian and Laird method) was used to improve the reliability. 15
Subgroup analyses were performed based on the types of TTVR (coaptation devices versus annuloplasty devices). Sensitivity analysis was performed by omitting each study in turn. Visual inspection of funnel plots and the Egger linear regression test were used to determine publication bias when more than 10 effect estimates were available.
Statistical analyses were performed using MetaXL version 5.3 (EpiGear International, Sunrise Beach, Queensland, Australia) and R software (version 4.0.3). A P value < 0.05 indicated statistical significance.
Results
Study identification and selection
The literature retrieval process is presented in Supplementary Figure 1. A total of 5247 studies were screened through the PubMed and Embase databases. After removing the duplicates and screening the title and abstracts, 66 studies were selected for the full-text assessment. We further excluded 45 studies due to overlapping populations (n = 32), no available outcome data (n = 5), patients undergoing tricuspid valve replacement or CAVI (n = 7), and sample size < 5 (n = 1) (Supplementary Table 2). Ultimately, a total of 21 studies (9 prospective single-arm studies, 6 prospective cohort studies, and 6 retrospective cohort studies) were included in this meta-analysis. The studies of Kodali et al., 18 Nickenig et al., 12 Fam 2019 13 with short-term follow-up were included for analyses of perioperative or short-term outcomes only, while the corresponding studies with long-term follow-up (Kodali et al., 19 Nickenig et al., 20 and Kitamura et al. 21 ) were included for long-term outcome analyses. All of these included studies had a moderate-to-high quality with an NOS score of ⩾ 6 points (Supplementary Table 3).
Baseline patient characteristics
The baseline characteristics of the included studies are shown in Table 1. A total of 896 patients were included in this meta-analysis (studies sharing the same population but with different follow-up durations were counted only once). The surgical risks were high with a mean EuroSCORE II ranging from 3.76% to 10.4%. A total of 729 (81.4%) patients underwent ITTVR while 167 (18.6%) patients underwent TMTVR. Functional TR accounted for over 90% of the patients and 190 of 835 (22.8%) patients had previous PPM/ICD/CRT implantation. The median follow-up duration for all included studies excluding overlapping populations (excluding the studies of Kodali et al., 18 Nickenig et al. 12 and Fam et al. 13 ) was 313 days, and the median follow-up duration for studies included in the long-term analysis was 365 days.
Baseline characteristics of the included studies of this meta-analysis.
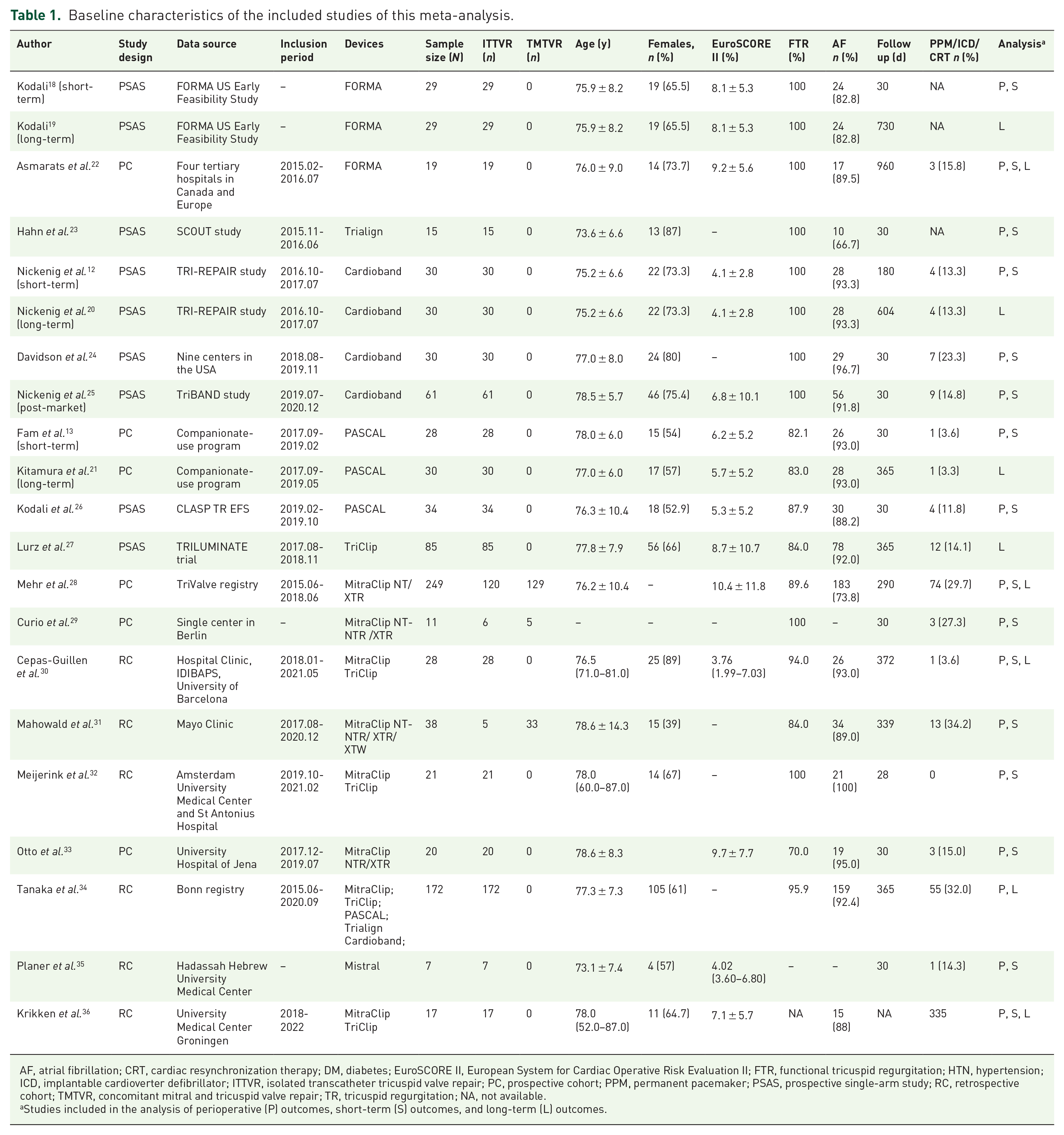
AF, atrial fibrillation; CRT, cardiac resynchronization therapy; DM, diabetes; EuroSCORE II, European System for Cardiac Operative Risk Evaluation II; FTR, functional tricuspid regurgitation; HTN, hypertension; ICD, implantable cardioverter defibrillator; ITTVR, isolated transcatheter tricuspid valve repair; PC, prospective cohort; PPM, permanent pacemaker; PSAS, prospective single-arm study; RC, retrospective cohort; TMTVR, concomitant mitral and tricuspid valve repair; TR, tricuspid regurgitation; NA, not available.
Studies included in the analysis of perioperative (P) outcomes, short-term (S) outcomes, and long-term (L) outcomes.
The baseline functional and echocardiographic characteristics are presented in Supplementary Table 4. A total of 87.4% of the patients were in the New York Heart Association (NYHA) Class III or IV groups, and 96.3% of the patients had severe or greater TR.
Distributions of the devices used in TTVR
The devices used across the included studies are shown in Figure 1 and Supplementary Table 5. By omitting the overlapping cohorts, coaptation devices and annuloplasty were used in 728 (81.3%) and 167 (18.6%) patients, respectively. Only one patient underwent combined therapy of coaptation (PASCAL) and annuloplasty device (Cardioband). Transcatheter edge-to-edge (TEER), as the most widely used coaptation leaflets method, accounted for 673 (75.1%) patients, while FORMA accounting for 48 (5.4%) patients and Mistral for only 7 (0.8%) patients. Cardioband and Trialign were the two major devices for annuloplasty, used for 151 (16.9%) and 18 (2.0%) patients, respectively.

Different devices included in the present analysis. (a) Devices used in this meta-analysis. (b) Proportions of coaptation and annuloplasty devices. (c) Proportions of the devices used in this meta-analysis.
Procedural outcomes
A total of 17 studies12,13,18,22–26,28–36 were included in the analysis of procedural outcomes (Table 2 and Supplementary Table 6). The pooled technical and procedural success rates were 93.9% (95% confidence interval (CI): 90.7%–96.4%) and 82.1% (95% CI: 74.0%–89.0%), respectively. The perioperative all-cause mortality rate was 1.0% (95% CI: 0.3%–1.9%) (I2 = 0.0) (Figure 2). The pooled incidence of the secondary outcomes was also estimated: 1.1% for reintervention or conversion to surgery, 5.5% for major bleeding, 3.5% for access site complications, and 2.6% for acute kidney injury. The pooled incidence for cardiovascular complications was relatively low, with a rate of 0.6% for acute myocardial infarction, 2.0% for arrhythmia, 1.4% for cardiac tamponade, and 1.0% for stroke. For patients undergoing the TEER method, the incidence of perioperative SLDA was 5.6%.
Incidence of clinical outcomes of patients undergoing transcatheter tricuspid valve repair using random-effects meta-analysis.

CI, confidence interval.
The Egger’s test was not performed for those with study numbers <10.
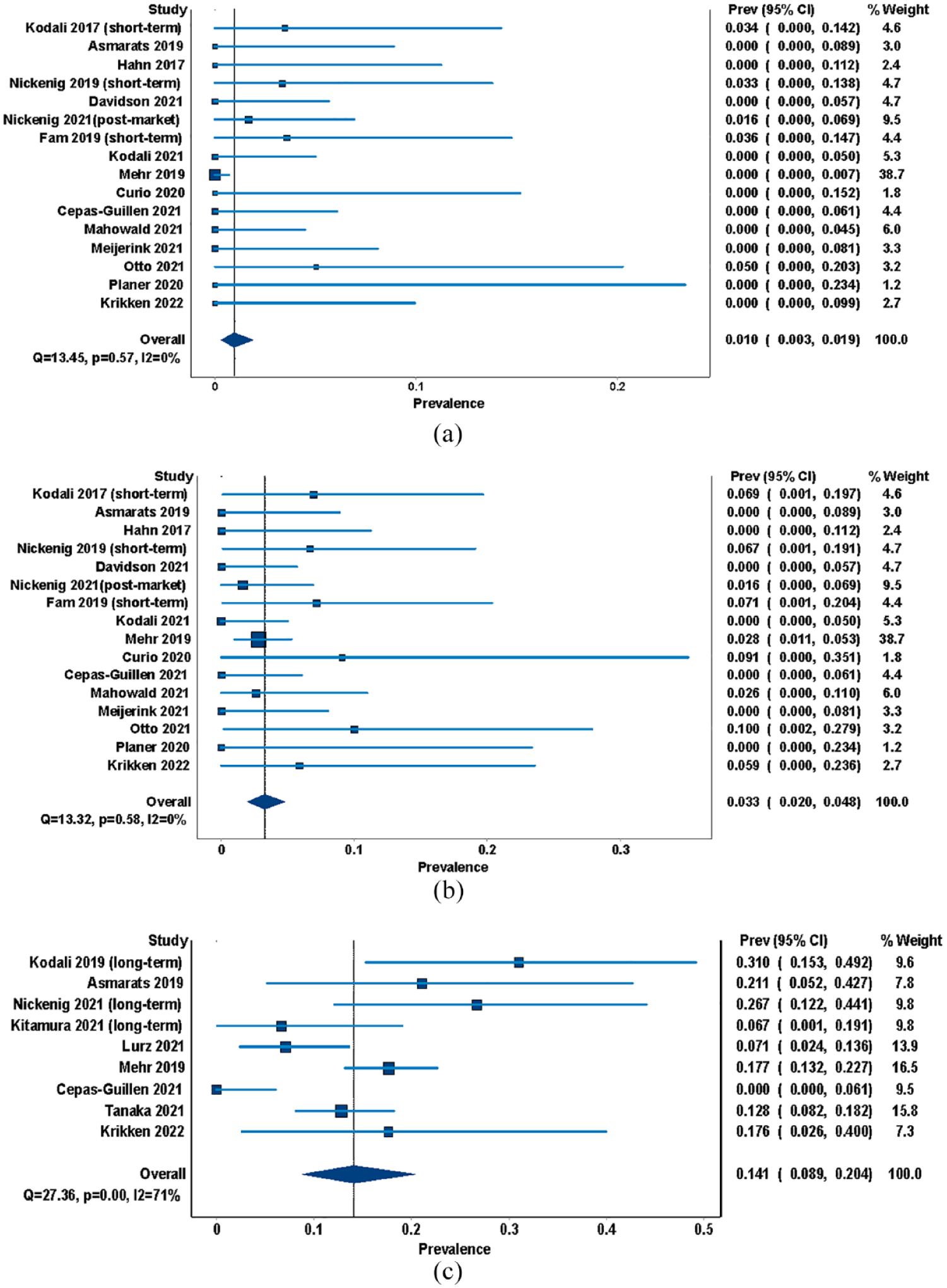
Pooled incidence of procedural, short-term, and long-term all-cause mortality of patients with severe TR undergoing TTVR. (a) Procedural all-cause mortality. (b) Short-term all-cause mortality. (c) Long-term all-cause mortality. TTVR, transcatheter tricuspid valve repair; TR, tricuspid regurgitation.
Short-term outcomes
A total of 16 studies12,13,18,22–26,28–33,35,36 were included for analyzing short-term outcomes (Supplementary Table 7). The short-term all-cause mortality rate was 3.3% (95% CI: 2.0%–4.8%) (I2 = 0.0) (Table 2 and Figure 2). Procedural success declined to 74.5% (95% CI: 66.7%–81.5%) within a 30-day follow-up post-TTVR. The pooled incidence rate of major bleeding was 8.0% while the rate of access site complications was 4.9%. Coronary complications were reported for patients who received FORMA or annuloplasty devices, with a pooled incidence rate of 2.2%. In addition, the rate of SLDA was estimated to be 8.6%. Otherwise, the incidence of acute kidney injury and cardiovascular complications was comparable with the procedural incidence.
Long-term outcomes
A total of nine studies19–22,27,28,30,34,36 were included for analyzing long-term outcomes (Supplementary Table 8). The median follow-up for the nine studies was 365 days. The pooled all-cause mortality rate was 14.1% (95% CI: 8.9%–20.4%) (I2 = 70.8%) (Table 2 and Figure 2). Cardiovascular mortality was available in six studies involving 364 patients with a pooled rate of 5.3% (95% CI: 2.1%–9.7%). In addition, the rate of HHF was 21.5% (95% CI: 16.3%–27.2%). The pooled incidences of major bleeding and SLDA were 14.3% and 6.4%, respectively. However, the incidence of stroke (1.6%), pulmonary embolism (1.8%), and device-related thrombosis (1.2%) was relatively low.
Sensitivity analysis and subgroup analysis
The sensitivity analyses showed that after removing a single study at one time, the pooled incidences of TTVR perioperative mortality varied from 0.8% to 2.0%, short-term all-cause mortality varied from 3.1% to 3.5%, and long-term all-cause mortality varied from 12.6% to 16.1% (Supplementary Table 9). Significant heterogeneity was indicated in the long-term group (I2 58.3%–74.1%).
Regarding the significant differences between coaptation and annuloplasty devices, subgroup analysis based on the device type was performed. The long-term all-cause mortality was estimated to be 13.0% (95% CI: 6.2%–21.2%) for coaptation devices, 26.7% (95% CI: 12.2%–44.1%) for annuloplasty devices, and 12.8% (95% CI: 8.2%–18.2%) for combined coaptation and annuloplasty therapy (Figure 3). The perioperative procedural success rate was 83.4% for coaptation devices and 78.8% for annuloplasty devices (Supplementary Figure 2). The rate of HHF was also estimated to be 20.1% and 43.3% for coaptation and annuloplasty devices, respectively (Supplementary Figure 3). Regarding the safety outcomes, the procedural and short-term incidences of major bleeding were 5.2% and 6.0%, respectively, for the coaptation device group; however, the incidence was 6.9% perioperatively and increased to 12.5% during short-term follow-up for the annuloplasty device group (Supplementary Figures 4 and 5).

Pooled long-term all-cause mortality of patients with severe TR undergoing TTVR with coaptation devices or annuloplasty devices. TTVR, transcatheter tricuspid valve repair; TR, tricuspid regurgitation.

Periprocedural, short-term, and long-term outcomes of transcatheter tricuspid valve repair in this study.
Publication bias
As shown by the funnel plots (Supplementary Figure 6), there were seemingly no obvious publication biases regarding the primary outcomes except procedural all-cause mortality. Egger’s tests also suggested potential publication biases for the outcome of procedural mortality (P = 0.024). Notably, due to the small study numbers, Egger’s test was not conducted for the long-term outcomes.
Discussion
To our knowledge, this was the largest and latest meta-analysis assessing the clinical outcomes and safety of TTVR in patients with severe TR. A total of 21 studies involving 896 patients with significant TR were included in this meta-analysis. The main findings of our study were as follows: (1) 81.4% of the patients underwent ITTVR while only 18.6% of the patients underwent TMTVR. Over 80% of the patients received coaptation devices for TTVR, while approximately 20% used annuloplasty. Technical and procedural success rates were high at 93.9% and 82.1%, respectively. (2) The pooled perioperative, short-term, and long-term all-cause mortality rates for patients undergoing TTVR were 1.0%, 3.3%, and 14.1%, respectively. The long-term cardiovascular mortality rate was 5.3% while the rate of HHF was 21.5%; (3) Major bleeding and SLDA were the two major complications, accounting for 14.3% and 6.4% of the patients undergoing TTVR, respectively.
According to a systematic review and pooled analysis including 454 patients with severe TR undergoing TTVR, TTVR was feasible with a low all-cause mortality rate of 9% at a weighted mean follow-up duration of 265 days. 9 Another meta-analysis with a total of 771 patients undergoing ITTVR for moderate or greater TR, indicated the all-cause mortality rate to be 11% at a weighted mean of 212 days of follow-up. 10 However, the perioperative and short-term outcomes were not available in these meta-analyses. In our study, the pooled perioperative and short-term (within 30 days) all-cause mortality rates for patients undergoing TTVR were low at 1.0% and 3.3%, which were significantly reduced compared with those of patients undergoing tricuspid valve surgery. 4 Nevertheless, the long-term all-cause mortality and unplanned HHF rates remained high at 14.1% and 21.5%, respectively, during the 365-day follow-up, possibly due to the high fragile condition and advanced heart failure.
To be noted, in our study, 22.8% of patients with pre-procedural cardiac implantable electronic device (CIED) implantation, which was similar to that of a recent study derived from the TriValve Registry. 37 It was indicated that patients with or without CIED who underwent TTVI had comparable procedure success and short-term clinical outcomes in terms of TR reduction, NYHA functional class, and mortality irrespective of the devices used. 37 Indeed, it was the procedural success that significantly associated with improved survival, reinforced that need to obtain best procedural results and reduce residual TR. 37
Notwithstanding, the complications associated with the TTVR procedure are still worth mentioning. The incidence rate of major bleeding during the procedure, short-term, and long-term follow-up were 5.5%, 8.0%, and 14.3%, respectively. Furthermore, the annuloplasty group had a higher incidence of major bleeding than the coaptation group (6.9% versus 5.2% perioperatively, and 12.5% versus 6.0% during the short-term follow-up, Supplementary Figures 4 and 5). This tendency was consistent with a recent meta-analysis that reported that bleeding complications were higher in the annuloplasty group than in the TEER group (13.3% versus 2.8%) 38 ; however, when the bleeding occurred was not mentioned in this analysis. According to a real-world study including the first 60 patients treated with the Cardioband annuloplasty system, major bleeding was one of the most common postprocedural adverse events, accounting for 11.7% of patients. 39 Notably, right coronary artery complications were also common in the annuloplasty groups and occurred in 15% of the patients. 39 In our study, coronary complications were reported only in the annuloplasty group involving 9 out of 136 patients (6.62%) undergoing annuloplasty intervention. For patients who received the TEER therapy in our meta-analysis, the rates of SLDA during the procedure, and the short-term and long-term follow-ups were 5.6%, 8.6%, and 6.4%, respectively. According to a study including 20 patients with severe symptomatic TR treated with MitraClip, the rate of SLDA was 15%, and the median time to SLDA was 37 days. 33 In addition, larger coaptation gaps (>7.2 mm) and noncentral/nonanteroseptal location of the TR jet have both been identified as risk factors for procedural success in patients undergoing TEER. 40 Therefore, the use of devices with larger clip arms (MitraClip XTR system or the PASCAL system), annuloplasty devices, or transcatheter valve replacement may be a better alternative for patients with large tricuspid annuli, wide leaflet coaptation gaps and tethered leaflets.
Coaptation devices approximate the leaflets of the tricuspid valve to attenuate regurgitation, while annuloplasty devices mimic surgical sutures or ring annuloplasty to reduce the TR regurgitant orifice area. In our study, patients who received annuloplasty devices presented with lower procedural success, higher all-cause mortality, and a higher incidence of major bleeding than those who received coaptation devices. The reasons may be due to the small sample size and the heterogeneity among patients in the annuloplasty group. Notwithstanding, according to previous studies, the improvements in functional status and activity capacity were consistent with the correction of TR severity regardless of the use of coaptation devices or annuloplasty devices. 10 The reduction to moderate or less TR was associated with a 3-fold reduction in mortality and unplanned HHF at 1 year compared with those with severe TR or greater postprocedure (8.8% versus 24.5%; HR: 0.31; p = 0.041). 27 In addition, patients whose TR was corrected to moderate or less severity were free of death or HHF within 6-month follow-up postprocedure. 27 Further randomized clincal trials (RCTs) are warranted to explore the differences in complications and outcomes among different devices.
Study limitations
Several limitations of this study are worth mentioning. First, as TTVR was not carried out until recent years, this meta-analysis mainly included small single-arm feasibility studies without control groups. Future RCTs are warranted to demonstrate the benefits of TTVR over medical treatment in patients with severe TR. Currently, there are several ongoing pivotal clinical trials to compare the safety and effectiveness of tricuspid valve transcatheter repair system with optimal medical therapy including Clip for the tricuspid valve (TRI-FR trial, NCT04646811), Tricuspid Valve TriClip system (TRILUMINATE Pivotal; NCT03904147) and Edwards PASCAL System (CLASP II TR; NCT04097145). Second, the sample size of the studies included was small and different devices feature different mechanisms, resulting in overall significant heterogeneity. Nevertheless, individually omitting each study did not significantly change the primary outcomes. Third, the clinical outcomes of the ITTVR and TMTVR groups were not reported in every study, therefore, subgroup analyses between the two groups were not available. Fourth, most of these devices were still in the early clinical stage, and future device iterations and more robust RCTs with longer follow-up are warranted to evaluate the effects of TTVR over medical therapy before promoting the widespread use of TTVR instead of conventional surgery.
Conclusion
TTVR was associated with high procedural success and low procedural and short-term mortality. However, all-cause mortality, cardiovascular mortality, and the rate of HHF remained high during long-term follow-up.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231158607 – Supplemental material for Periprocedural, short-term, and long-term outcomes following transcatheter tricuspid valve repair: a systemic review and meta-analysis
Supplemental material, sj-docx-1-taj-10.1177_20406223231158607 for Periprocedural, short-term, and long-term outcomes following transcatheter tricuspid valve repair: a systemic review and meta-analysis by Zexuan Wu, Wengen Zhu, Wulamiding Kaisaier, Miriding Kadier, Runkai Li, Gulpari Tursun, Yugang Dong, Chen Liu and Yili Chen in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
