Abstract
Objective
To develop an experimental porcine model of tricuspid regurgitation using two induction routes: the inferior vena cava and superior vena cava.
Methods
Tricuspid regurgitation was generated using the loop wire cutting method. The tricuspid regurgitation jet direction was controlled by accessing the valve through the inferior (n = 7) or superior (n = 6) vena cava. The occurrence, direction, and progression of tricuspid regurgitation were assessed postoperatively, and echocardiography was performed at 4 to 6 weeks. Right heart dilatation was assessed using computed tomography after 6 weeks.
Results
Moderate to severe or torrential tricuspid regurgitation occurred immediately after the procedure in 12 of 13 animals. The jet was directed toward the septum in five of seven animals in the inferior vena cava group and toward the posterolateral side in four of six animals in the superior vena cava group. The dimensions of the right heart (right atrium, ventricle, and tricuspid valve annulus) were significantly enlarged at the 4- to 6-week follow-up echocardiographic examination and confirmed to be enlarged by computed tomography, independent of the route used.
Conclusion
The loop wire cutting method successfully induced a disease model of tricuspid regurgitation while controlling the regurgitation jet direction via two procedural routes.
Keywords
Introduction
Functional tricuspid regurgitation (TR) is induced by dilation of the tricuspid annular structure, resulting in right ventricular volume overload and/or increased right ventricular pressure. Annular dilatation develops gradually and asymmetrically, with leaflet coaptation dysfunction occurring primarily in the anterior and posterior regions. 1 Traditionally, treatment for tricuspid valve disease has been considered less important than that for left-sided valvular heart disease, and severe TR has been thought to be well tolerated by patients. TR was historically managed with conservative treatment, with surgical correction considered only when performing mitral valve surgery. 2 However, research has demonstrated that moderate or severe functional TR is associated with progressive heart failure and early death,3,4 leading to increased interest in the use of surgery to treat TR. Considering the aging population and increase in the number of patients for whom conventional open heart surgery or reoperation is not indicated, minimally invasive techniques and novel percutaneous treatment options are required. Several experimental studies and clinical case reports have documented the feasibility of aortic, pulmonary, and mitral valve transcatheter surgery.5,6 Percutaneous tricuspid valve surgery has also been documented.7–9 However, animal disease models of TR are required for preclinical testing of percutaneous TR surgery and in vivo and ex vivo training. Preclinical testing using animal disease models is an important part of the regulatory process used to determine the safety of newly developed techniques prior to clinical trials. 10 Although many animal models have been developed to examine heart valve disease, 11 animal models of TR have only been developed in recent years, primarily using the percutaneous transcatheter technique.12–14 However, a standard animal model has not yet been established, and no method of effectively controlling the direction of the TR jet has been reported.
This study was conducted under the assumption that TR can be effectively induced by damaging the tricuspid structure using a percutaneous technique and that it can be induced in different directions when two different routes are used. We aimed to develop a simple and reproducible experimental model for TR in live swine using a novel approach. Moreover, we investigated the effect of the two approach routes on the remodeling of the right heart, the damage caused to the tricuspid valve structure, and the direction of regurgitation.
Materials and methods
Animal preparation
This study involved 13 healthy adult Yorkshire farm swine weighing 39 to 48 kg (mean, 42.69 ± 3.09 kg). The animals were placed under general anesthesia using a MultiPlus EVD Type anesthesia ventilator (Royal Medical Co., Ltd., Pyeongtaek, South Korea). Alfaxalone (5 mg/kg) and xylazine (2 mg/kg) were administered intramuscularly for paranesthesia, followed by 3% isoflurane for maintenance anesthesia. Before endotracheal intubation, atropine sulfate (1 mg) was administered intravenously (IV). Tramadol HCl (50 mg) and gentamicin sulfate (80 mg) were administered after intubation. The anesthetized animals were placed in the supine position. Heparin sodium (5000 U) was administered IV after performing a venous puncture, and the muscle relaxant vecuronium (0.1 mg/kg) was administered IV at the start of the procedure. At the end of the follow-up monitoring, the animals were anesthetized and then euthanized with KCl (3 g/20 mL) administered IV.
The state of anesthesia was continuously monitored during the procedure using the heart rate (HR), blood oxygen saturation, and electrocardiography. Arterial blood pressure was also monitored by a percutaneously inserted pressure line in the left femoral artery.
Ethical statement
All animals were treated humanely following the guidelines established by the National Institutes of Health and the policies of the Animal Care and Use Committee of Pusan National Yangsan University Hospital, Korea. All experimental protocols were approved by the Institutional Animal Care and Use Committee of Pusan National Yangsan University Hospital (IRB no. 2017-045) and performed in accordance with the ARRIVE guidelines.
TR model induction
We induced TR using a wire and snare catheter-based procedure performed via the inferior vena cava (IVC) (n = 7) or superior vena cava (SVC) (n = 6) under fluoroscopic guidance using the Integris H5000F system (Philips, Amsterdam, Netherlands). All animals were closely monitored during the follow-up period.
Loop wire cutting technique via IVC
After puncturing the right femoral vein and inserting an 8-Fr sheath, a 5-Fr pigtail catheter (145°, 100 cm; Jung Sung Medical Co., Ltd., Seongnam, South Korea) or multipurpose angiographic catheter (100 cm; Cook Group, Bloomington, IN, USA) was introduced through the IVC along the narrow space between the tricuspid valve chordae tendineae from the right atrium (RA) to the right ventricle (RV). A 0.035-inch stiff wire (Cook Group) or a 0.014-inch guidewire was used to back up and track the catheter after confirming its correct position. A snare catheter (EN Snare; Merit Medical Systems, Inc., South Jordan, UT, USA or EV3 Goose Neck Snare; Medtronic, Minneapolis, MN, USA) was placed near the chordae tendineae and papillary muscle of the target tricuspid valve. The wire and catheter were passed between the tricuspid valve leaflet and chordae tendineae to form a loop that when pulled acted as a cutter, severing the chordae or papillary muscle passing through it (Figure 1(a)). All procedures were fluoroscopy-guided. Before the loop was completely removed, the movement of the heart in the pulling direction was monitored to ensure that the loop was positioned in the desired direction. Typically, this process was successfully completed in two or three attempts. Assuming that porcine and human hearts have similar anatomical structures, 15 the IVC route was used to damage the septal and anterior tricuspid leaflets (Figure 1(b)) because the inserted catheter and wire could be positioned near the septal side of the tricuspid valve.

Schematic illustration of the two routes and the damaged tricuspid valve structure. (a, b) IVC route. (a) A wire-guided catheter and a snare are introduced through the IVC and placed along the narrow space among the tricuspid valve chordae tendineae between the RA and RV. A loop cutter is then formed to inflict damage to the tricuspid valve structure. (b) Pulling the loop through the IVC causes damage to the septal side of the tricuspid valve structure. (c, d) SVC route. (c) A wire-guided catheter and a snare are introduced through the SVC and placed along the narrow space among the tricuspid valve chordae tendineae near the pulmonary artery. A loop cutter is then formed to inflict damage to the tricuspid valve structure and (d) pulling the loop through the SVC causes damage on the posterolateral side. IVC, inferior vena cava; RA, right atrium; RV, right ventricle; SVC, superior vena cava.
Loop wire cutting technique via SVC
The SVC route was used to damage the posterolateral side of the tricuspid valve. The wire and catheter were passed through the SVC junction with the pulmonary artery (PA) and positioned close to the chordae on the posterolateral side of the tricuspid valve. The operator inserted an 8-Fr sheath through the right jugular vein followed by a 5-Fr pigtail or multipurpose angiographic catheter, as described above. The catheter was advanced toward the PA along the SVC. Catheter backup and tracking were performed with a 0.035-inch stiff wire or a 0.014-inch guidewire as described above. The catheter was placed in the narrow space between the chordae tendineae of the tricuspid valve leaflet to form a loop with the wire (Figure 1(c)). The remainder of the procedure followed the steps described above; damage to the posterior tricuspid leaflet was induced via this route (Figure 1(d)).
The loops formed during TR induction are shown in Figure 2(a) (IVC route) and Figure 2(b) (SVC route). When the loop was pulled back, the destroyed tendon and papillary muscle were sometimes released from the sheath with the looped wire and catheter (Figure 2(c)). Although this was not observed in all cases, it did occur in both approach routes; therefore, we assumed that the loop wire cutting method exerted a force strong enough to affect the tricuspid valve structure.

Tricuspid regurgitation induction and specimen of ruptured tricuspid valve structure. The guidewire and snare catheter form a loop around the tricuspid valve structure following entry through (a) the right femoral vein in the inferior vena cava route and (b) the right internal jugular vein in the superior vena cava route and (c) chordae tendineae and papillary muscles are ruptured by pulling the loop and released from the sheath.
Echocardiographic assessment
All pigs underwent echocardiography by an expert before initiation of the experiment to confirm the absence of structural or valvular heart disease. We confirmed the presence and severity of TR and the regurgitation jet direction during the procedure using transthoracic echocardiography. A Vivid Q portable electrocardiogram (GE HealthCare, Chicago, IL, USA) was used to generate images that were analyzed by experts. The results were visually confirmed by quantifying the TR jet grade and determining its direction using color Doppler. TR was graded as mild, moderate, severe, or torrential by visually estimating the color flow jet area according to clinical guidelines. 16 The best views of the tricuspid valve structure and TR were obtained using the modified four-chamber view or the right heart inflow view with the swine in the supine position. The dimensions of the RA, the mid-portion of the RV, and the tricuspid valve annulus (TVA) were measured immediately after the procedure and 4 to 6 weeks after the induction of TR, depending on the condition of the animals. The RA diameter was assessed from the edge of the aortic wall to the edge of the anterior wall of the RA at end-systole. The diameter of the mid-portion of the RV (Mid-RV) was evaluated from the inner edge of the right ventricular lateral wall to the edge of the ventricular septum wall at end-diastole. The size of the TVA was evaluated at end-diastole.
Computed tomography
To assess the effect of TR, we evaluated the dilatation of the RA and RV by computed tomography (CT) 6 weeks after TR induction, selecting one animal in which severe or torrential TR had been induced by each route. Axial CT images were used to demonstrate the best approximated four-chamber view. Calipers were placed from inner wall to inner wall at the maximal diameter to measure the RV/left ventricle ratio (R/L ratio).
Statistical analysis
Categorical variables are expressed as frequency. Continuous variables are expressed as mean ± standard deviation and were analyzed using the Wilcoxon signed-rank test of nonparametric statistics. A P value of <0.05 was considered statistically significant. All statistical analyses were performed using SPSS software version 28.0 (IBM Corp., Armonk, NY, USA).
Results
From July 2017 to July 2021, 13 Yorkshire farm swine were treated using the wire and snare catheter-based percutaneous technique to generate a survival model of TR via two approach routes. All procedures were completed with no signs of unintended damage, such as cardiac arrest. Arterial blood pressure and HR monitoring did not change significantly after TR induction during the procedure via the IVC route (n = 7). Systolic blood pressure (SBP) was 87.20 ± 4.49 mmHg before and 88.79 ± 2.72 mmHg after TR induction. Diastolic blood pressure (DBP) was 58.45 ± 1.93 mmHg before and 58.85 ± 1.48 mmHg after TR induction. HR was 78.87 ± 1.87 bpm before and 78.97 ± 1.86 bpm after TR induction. Similarly, no significant changes were observed in these parameters when the SVC route was used (n = 6). SBP was 87.46 ± 4.80 mmHg before and 88.84 ± 2.94 mmHg after TR induction. DBP was 58.13 ± 1.91 mmHg before and 58.79 ± 1.59 mmHg after TR induction. HR was 78.53 ± 1.81 bpm before and 78.59 ± 1.75 bpm after TR induction. No significant changes were observed at the 4- to 6-week follow-up. In animals with TR induced via the IVC route (n = 7), SBP was 89.48 ± 2.17 mmHg, DBP was 59.92 ± 1.35 mmHg (P = 0.06), and HR was 79.37 ± 1.65 bpm. In animals with TR induced via the SVC route (n = 6) SBP was 89.40 ± 2.34 mmHg, DBP was 59.67 ± 1.21 mmHg, and HR was 78.60 ± 1.78 bpm.
The successful induction of moderate to severe or torrential TR was confirmed by echocardiography immediately after the procedure in 12 of the 13 animals. TR was moderate and moderate to severe in two pigs in the IVC route group, mild to moderate in one pig in the SVC route group, and severe or torrential in the other five pigs in each group. We confirmed that mild to moderate TR could be induced via the SVC in one animal, increasing the regurgitation volume over time, and the generated TR jet was maintained at the moderate grade 4 weeks later.
In most cases, postoperative echocardiography confirmed that the TR jet was successfully generated on the septal or posterolateral side, depending on where the damage was inflicted. Echocardiography confirmed that the TR jet direction was septal in five of the seven pigs with TR induced via the IVC route (Figure 3(a)). Eccentricity was not observed in the remaining two pigs, making it impossible to determine the direction of the regurgitation flow. One pig had severe TR accompanied by chordae rupture, and the other had torrential TR. A posterolaterally directed TR jet was confirmed in four of the six pigs treated via the SVC (Figure 3(b)). The remaining two pigs had torrential TR, and this was accompanied by chordae rupture in one animal (Table 1).

Echocardiographic assessment at 4-week follow-up and dimensions of right heart. (a) Tricuspid regurgitation on the septal side through the TV (white arrow) after performing the procedure through the inferior vena cava. (b) Tricuspid regurgitation from the posterolateral side was observed after performing the procedure through the superior vena cava and (c) the modified four-chamber view with the swine in the supine position. The dimensions of the RA, Mid-RV, and TV annulus were measured as shown (dotted arrows). LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle; TV, tricuspid valve; Mid-RV, mid-portion of right ventricle.
Characteristics and results of experimental model of tricuspid regurgitation.
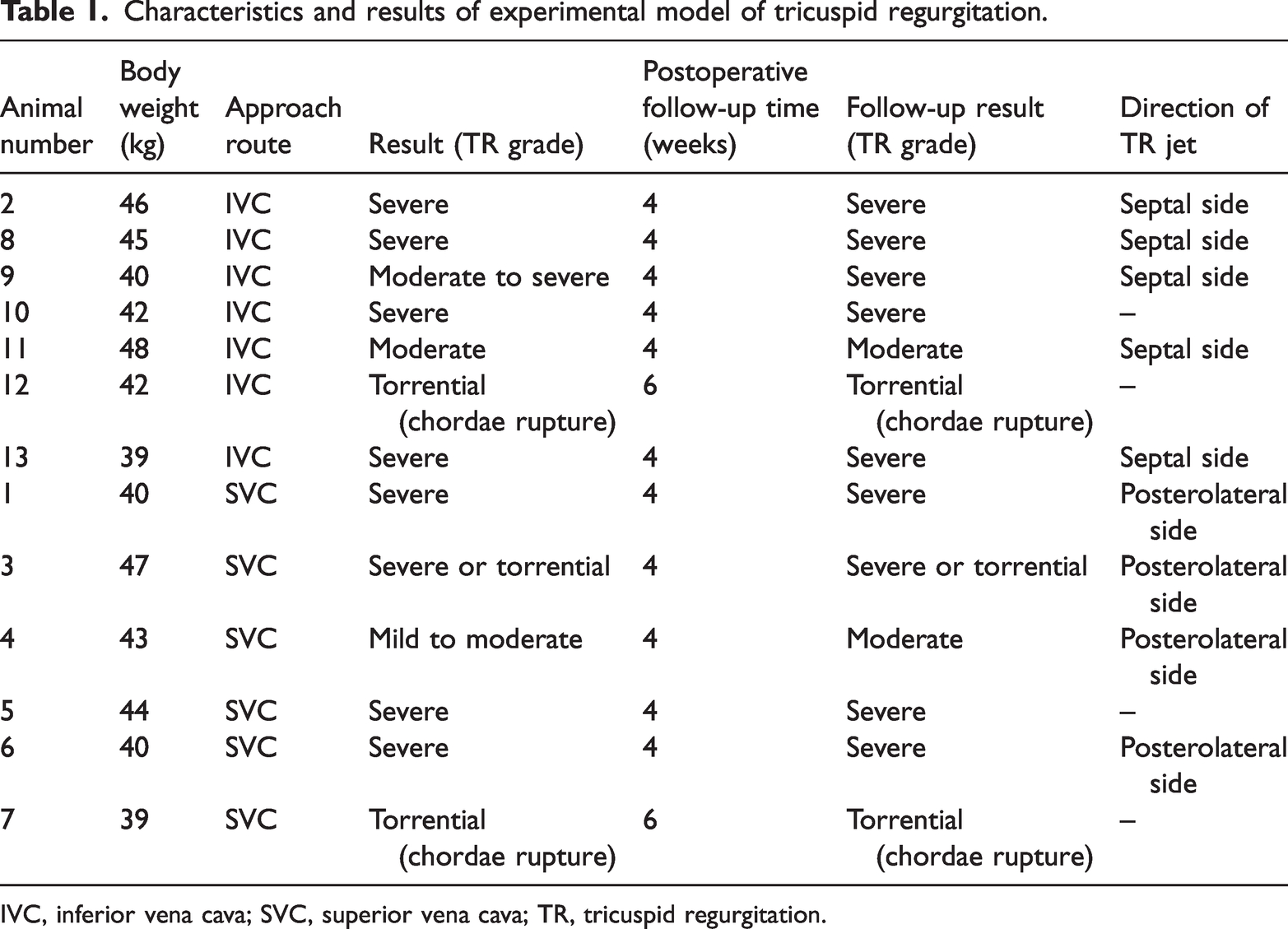
IVC, inferior vena cava; SVC, superior vena cava; TR, tricuspid regurgitation.
Compared with baseline, there was a significant increase in the size of the right side of the heart as measured by the RA, Mid-RV, and TVA diameters 4 weeks after TR induction. The measurements taken are shown in Figure 3(c). In the IVC route group (n = 7), the RA diameter was 3.06 ± 0.11 cm before and 3.58 ± 0.18 cm after TR induction (P = 0.018), the Mid-RV diameter was 3.16 ± 0.15 cm before and 3.70 ± 0.10 cm after TR induction (P = 0.018), and the TVA diameter was 2.79 ± 0.11 cm before and 3.33 ± 0.18 cm after TR induction (P = 0.018). In the SVC route group (n = 6), the RA diameter was 3.03 ± 0.08 cm before and 3.49 ± 0.09 cm after TR induction (P = 0.028), the Mid-RV diameter was 3.13 ± 0.11 cm before and 3.72 ± 0.10 cm after TR induction (P = 0.027), and the TVA diameter was 2.77 ± 0.11 cm before and 3.30 ± 0.17 cm after TR induction (P = 0.028) (Table 2). The dimensions of the right side of the heart were also measured 6 weeks after TR induction in the one animal per group that maintained torrential TR accompanied by chordae rupture. In these animals, the heart remained significantly larger than that at 4 weeks. Pig No. 12, in which TR was induced via the IVC route, had 4-week diameters of 3.41, 3.65, and 3.23 cm and 6-week diameters of 3.78, 3.91, and 3.57 cm for the RA, Mid-RV, and TVA, respectively. Pig No. 7, in which TR was induced via the SVC route, had 4-week diameters of 3.45, 3.65, and 3.20 cm and 6-week diameters of 3.67, 3.89, and 3.54 cm for the RA, Mid-RV, and TVA, respectively.
Changes in right heart dimensions via both approach routes.

*Baseline vs. 4-week follow-up.
Data are expressed as mean ± standard deviation.
IVC, inferior vena cava; Mid-RV, mid-portion of right ventricle; RA, right atrium; SVC, superior vena cava; TVA, tricuspid valve annulus.
Multiplanar reformatted CT was used to assess the effect of sustained severe or torrential TR on the right side of the heart. The R/L ratio was 1.4 in Pig No. 7 with torrential TR after chordae rupture through the SVC route and 1.3 in Pig No. 12 with torrential TR through the IVC route. We considered this to be indicative of enlargement of the RA and RV chambers 6 weeks after the procedure because the R/L ratio measured by CT was >1.0 in both animals (Figure 4).

Multiplanar reformatted computed tomography 6 weeks after procedure. The RV/LV (R/L) ratio was (a) 1.4 in Pig No. 7 from the superior vena cava group with torrential tricuspid regurgitation following chordae rupture and (b) 1.3 in Pig No. 12 from the inferior vena cava group with torrential tricuspid regurgitation following chordae rupture. L, left; LA, left atrium; LV, left ventricle; R, right; RA, right atrium; RV, right ventricle.
Discussion
Among the tricuspid valve diseases, functional TR due to tricuspid valve complex elongation following RA or RV dilatation and remodeling is the most common. 1 Surgical and transcatheter tricuspid valve repair or replacement procedures have recently been attempted. 17 A preclinical TR animal model would be useful to test surgical techniques prior to translation to the clinical setting. The valve mechanism is analogous across mammalian species; therefore, animal models can be used to investigate valve injury, its underlying mechanisms, and potential treatments for human valvular diseases. 18
We investigated various methods in our quest for a simple, reproducible, and efficient experimental model of TR. Initial experiments involved tricuspid valve damage inflicted by pulling and fixing the valve during open chest surgery. While this approach had the potential to create an annular expansion effect resembling functional TR, it carried significant risks, resulting in fatal bleeding and cardiac arrest in some cases. In the method involving rupture of the chordae tendineae, severe TR was induced by direct structural damage during open chest surgery, leading to immediate regurgitation. However, this method required blood vessel incision and purse-string suturing to control bleeding. Additionally, the lack of predictability in the direction of surgical tools mandated image guidance through echocardiography, and confirming long-term complications of TR proved challenging. We subsequently explored a catheter-based procedure in which a balloon was inflated with a guidewire from the tricuspid valve side to damage the tricuspid structure. However, this method was sometimes ineffective in damaging the tricuspid valve structure depending on the balloon size.
As a result, we explored the use of a catheter-based wire and snare interventional procedure to induce TR in swine by causing structural damage. TR was successfully and simply induced to at least a moderate grade in 12 of the 13 animals. Reproducibility was confirmed by repeating the same procedure in all 13 animals, achieving various levels of TR. Echocardiography confirmed that the RA, Mid-RV, and TVA diameters increased significantly over the 4- to 6-week follow-up period. The generated TR jet was effectively maintained throughout the follow-up period, and this was sufficient to cause remodeling of the right side of the heart without adverse effects on survival.
Based on these results, we expect that this animal model can be used to study the effects of TR, such as pulmonary hypertension and RA-RV remodeling. Previous reports have also described experimental animal models of TR. Hoppe et al. 12 induced TR in sheep by damaging the papillary muscle of the tricuspid valve using a guidewire loop, causing an immediate significant increase in the mean RA pressure and HR. Although this approach produced TR simply and effectively, it was an acute study involving only seven animals, and the survival of the study animals was not guaranteed. Bai et al. 13 induced TR using specially designed forceps in seven healthy sheep to disrupt the tricuspid valve structure through the catheter. In their method, the RA pressure increased immediately after TR was induced, but there was no increase in the RV dimension on the 6-month echocardiographic follow-up examination. Yan et al. 14 also induced TR using specially designed grasping forceps with percutaneous access in 11 healthy pigs. The diameters of the RA, RV, and TVA were significantly larger 1 month after TR induction. However, a limitation of their study was that the forceps could not be controlled during the procedure; thus, the amount of tissue removed could not be accurately predicted. In our study, all 13 animals survived the 4- to 6-week follow-up period, and the TR jets generated through both routes were maintained in their respective orientations. The two animals with torrential TR had persistent torrential TR and enlarged RV dimensions for 6 weeks after TR induction as assessed by CT and echocardiography. We suggest that this animal model has the potential to be used for the development and testing of devices or technologies to treat TR by inducing TR through two easy-to-access routes.
In addition, we attempted to control the direction of the TR jet generated by inducing TR via the IVC and SVC; to the best of our knowledge, this has not been previously attempted. The advantage of this method is that the two catheter approach routes could control the direction of TR without thoracotomy, allowing survival of the animals. Xie et al. 19 reported a method of controlling the degree of TR by excising as many tricuspid leaflets as desired through right thoracotomy; however, their study involved a limited number of animals, some of which developed complications such as bleeding and infection following thoracotomy. The leaflet and chordae along the septal side can rupture when approached through the IVC, presumably because of the angle of the wire when passing to the PA through the tricuspid valve and its proximity to the septal side of the valve. The use of a loop wire cutting approach through the SVC can induce TR by rupturing the anterior and/or posterior leaflets, given that the wire and snare are near the posterolateral side. Functional TR leads to RV dilatation and remodeling, which changes the tricuspid annular structure, particularly the posterior and anterior leaflets. 1 Because eccentricity of the TR jet was confirmed in five of seven animals in the IVC group and four of six animals in the SVC group, our intended control of the TR jet direction was not successful in all animals in each group. In two animals of each group, in which neither the size nor the pulling force of the loop wire cutter had been adjusted to the desired level, the chordae and papillary muscle of the tricuspid valve were almost completely torn by the loop cutter. This resulted in rapid TR with an almost torrential jet. However, we confirmed that the direction of the TR jet was generated differently according to the two access routes. The posterolateral TR jet generated through the SVC route was thought to be in a direction similar to the regurgitant jet seen in functional TR. This suggests the possibility of using the model to study various TR-induced diseases of the right side of the heart.
Limitations
This model has several limitations. First, when approaching the SVC and progressing toward the lateral target, the TR jet was generated in a direction similar to that of functional TR. However, we induced TR by valvular structural injury, an entirely different mechanism from that involved in functional TR. Our method generated severe (torrential) TR that would not be clinically observed in functional TR. Nonetheless, our study showed that the direction of the jet can be altered by using different approach routes.
Second, the extent of damage to the chordae and papillary muscle caused by the snare and wire loop could not be precisely controlled. The loop could not be accurately sized, and its location and pulling force may have differed between the experiments. The chordae and papillary muscle of the tricuspid valve were almost completely torn off by the loop cutter in two pigs, resulting in torrential TR with a nearly central jet. Repeated experimentation and increased proficiency are expected to minimize these issues as indicated by our ongoing experiments.
Third, this was a small animal study in which we attempted to induce TR in a porcine model. Limited data were generated, and CT was performed on only two pigs to assess the remodeling of the RA and RV. A continuous follow-up study is needed to confirm the effect of TR induced through this technique on RV remodeling.
Conclusions
In this study, we used a loop wire cutting approach to successfully generate a long-term animal model of TR, which showed evidence of chamber dilatation 6 weeks after TR induction. The direction of TR could be controlled by using different approach routes and damaging different parts of the tricuspid valve structure. This novel method of TR induction is simple and may facilitate TR research and the development and testing of drugs or devices for TR treatment.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241233524 - Supplemental material for Novel percutaneous technique for creation of porcine model of tricuspid regurgitation via two routes
Supplemental material, sj-pdf-1-imr-10.1177_03000605241233524 for Novel percutaneous technique for creation of porcine model of tricuspid regurgitation via two routes by Ji Soo Oh, Ga Yun Kim, Seok Hyun Kim, Sang Hyun Lee, Yong Hyun Park, June Hong Kim and Min Ku Chon in Journal of International Medical Research
Footnotes
Author contributions
All authors contributed to the study conception and design. JS Oh and MK Chon wrote the first draft of the manuscript. GY Kim, SH Kim, SH Lee, YH Park, and JH Kim performed the data collection and analysis. All authors actively participated in this study, contributed to the writing of the manuscript, and approved the final mauscript.
Data availability
The data that support the findings of this study are available upon request from the corresponding author.
Declaration of conflicting interest
The authors have no conflicts of interest to declare.
Funding
This study was supported by the Research Institute for Convergence of Biomedical Science and Technology (30-2017-003, Pusan National University Yangsan Hospital).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
