Abstract
Background:
The pathophysiology of idiopathic intracranial hypertension (IIH), a condition characterized by raised intracranial pressure, is not well understood.
Objectives:
We hypothesized that the hypothalamus might exhibit alterations in patients with IIH, based on its established association with obesity and the potential role of hormonal and metabolic factors in IIH.
Design:
Retrospective single-center cohort study.
Methods:
Thirty-three individuals with IIH and 40 matched healthy individuals were studied, including levels of the hormones and proteins leptin, adiponectin, ghrelin, insulin, growth/differentiation factor 15 (GDF15), somatostatin, and melatonin.
Results:
The analysis of laboratory values showed significantly increased insulin, leptin, and melatonin levels for IIH patients in comparison to controls, while adiponectin levels were decreased in IIH; however, only melatonin level differences could be confirmed in the analysis with BMI matching. There was no statistical difference in total hypothalamus volumes between IIH and controls, but the hypothalamic morphology was altered in IIH patients with a lower volume of the anterior part of the hypothalamus and a higher volume of the posterior part; these results were identical in the analysis of the BMI-matched groups. The correlation analyses between hormonal changes and hypothalamic morphology did not achieve significant results.
Conclusion:
In this exploratory study, morphological abnormalities of the hypothalamus were observed to be associated with the IIH complex, although the mechanism remains to be unraveled. These findings expand the metabolic phenotype of IIH, but further functional studies are necessary to corroborate these data.
Introduction
Idiopathic intracranial hypertension (IIH) is a condition characterized by raised intracranial pressure (without hydrocephalus or other causative factors), papilloedema with the potential risk of permanent visual loss, and progressive headache which profoundly reduces the quality of life; international consensus guidelines on diagnostic principles and management were formulated in 2018. 1 There is a rising incidence of this disease, and, given that the condition has an established association with obesity, the incidence appears related to country-specific prevalences of obesity, mirroring obesity trends. 2 Obese young women in reproductive years are predominantly affected. 3 IIH is a condition of unknown etiology; 3 multiple hypotheses regarding the pathophysiology of this disorder have been presented over the last decades, but there is no consensus. 4 Although the pathophysiology and biochemistry of IIH is poorly understood, a number of factors are currently thought to contribute, including dysregulation of cerebrospinal fluid (CSF) dynamics, raised venous sinus pressure and in particular, the striking association with obesity and female gender have implicated the potential role of hormonal and metabolic factors. 5 In addition, there is evidence that female IIH patients have a unique signature of androgen excess, in the light of an impact of androgens on surrogate markers in CSF secretion. 6 A recent study in 97 female IIH patients provided evidence that IIH can be regarded as a disease of systemic metabolic dysregulation with an insulin- and leptin-resistant phenotype in excess of that driven by obesity. 7
The hypothalamus plays a crucial role within the complex cortical-subcortical network for the homeostatic regulation of energy balance and metabolic control. 8 Leptin, produced by fat cells and in the hypothalamus, is involved in the regulation of food intake, and early data have shown that a dysregulation of leptin is present in female IIH patients, independent of the body mass index (BMI). 9 In addition, somatostatin, an inhibiting hormone of the hypothalamus within the cascade of growth hormones, could improve IIH symptoms when applied as a synthetic somatostatin-analogon. 10 In the light of this constellation and the strong association with obesity in the majority of patients, an involvement of the hypothalamus in the pathophysiological processes of IIH was hypothesized. There is currently no information available on hypothalamic morphology in patients with IIH. In order to assess the hypothalamic structure, we performed an exploratory study with a magnetic resonance imaging (MRI)-based analysis of hypothalamic volumes, separated for its anterior and posterior fractions, in a group of IIH patients and compared it to healthy controls.
Subjects and methods
In this retrospective single-center cohort study over 10 years, 33 IIH patients and 40 healthy controls were included in total. Because the disease is rare, the sample size is limited to patients who were admitted to our clinic during the study period and met inclusion criteria. IIH patients had to fulfill the standard diagnostic criteria according to Friedman
Controls were matched for age and gender. Since IIH is associated with obesity, two age-, gender-, and BMI-matched subgroups were selected from the patients and controls, respectively, and were analyzed separately (Table 1). None of the BMI-matched controls had any radiological signs of IIH.
Clinical characteristics of patients with idiopathic intracranial hypertension (IIH) and healthy control subjects.
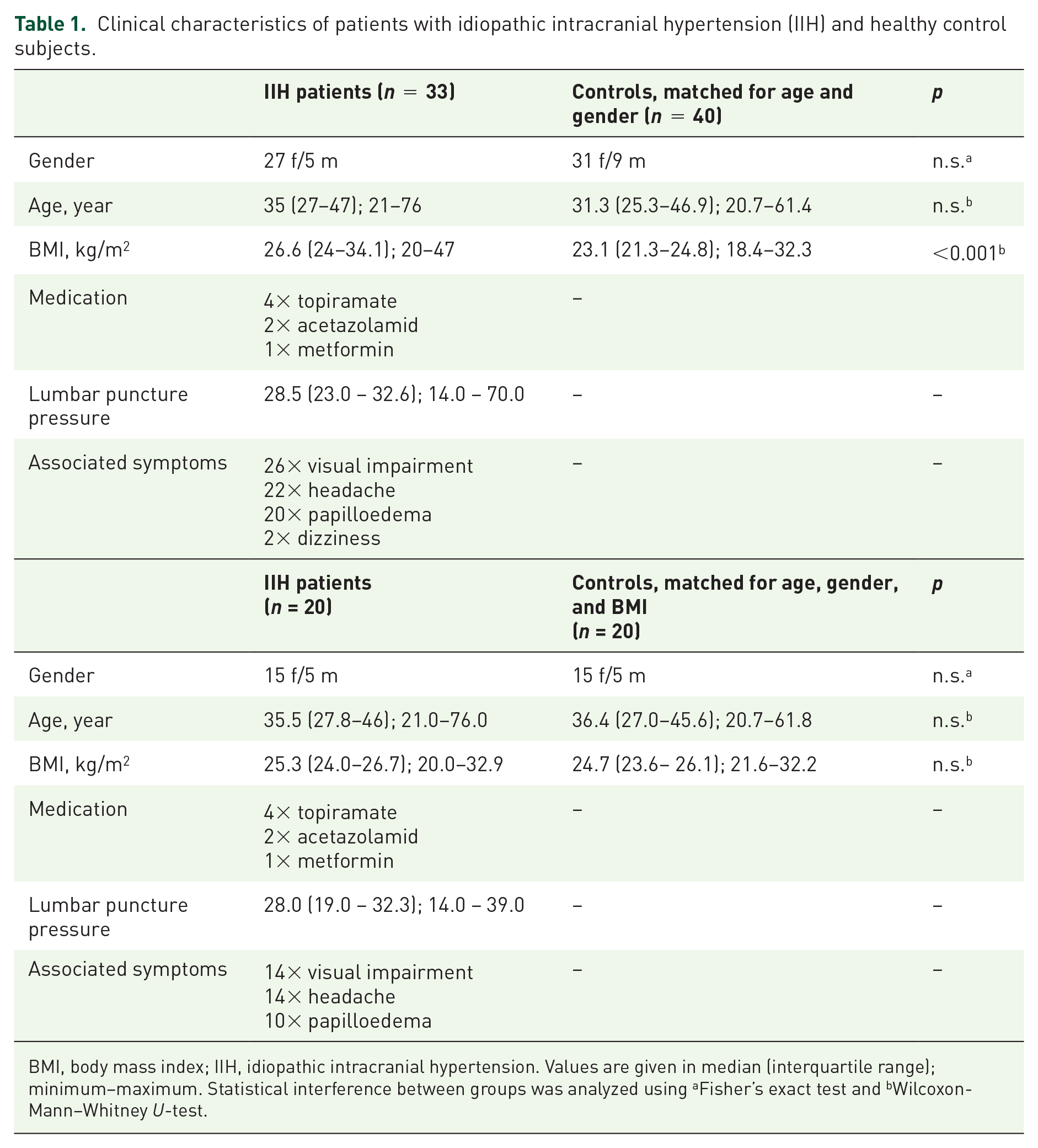
BMI, body mass index; IIH, idiopathic intracranial hypertension. Values are given in median (interquartile range); minimum–maximum. Statistical interference between groups was analyzed using aFisher’s exact test and bWilcoxon-Mann–Whitney
All participants provided written informed consent for the study protocol according to institutional guidelines which had been approved by the Ethics Committee of Ulm University, Germany (reference #159/17).
MRI acquisition and analysis
MRI data were acquired at a clinical 1.5 T scanner (Siemens Magnetom Symphony) with a standard headcoil. Morphological data were obtained using high-resolution T1-weighted magnetization-prepared gradient echo image (MPRAGE) sequence (192 sagittal slices, no gap, 1 mm3 isovoxel,
To divide the hypothalamus into anterior and posterior part, the criteria reported by Piguet
Statistics
Statistical group comparisons were performed using Fisher’s exact test or Wilcoxon–Mann–Whitney
Results
Clinically, the IIH patient group represented a classical phenotype with a preponderance of women and a significantly higher BMI than controls. Nineteen patients (68.5%) had an empty sella in MRI. The analysis of laboratory values showed significantly increased insulin, leptin, and melatonin levels for IIH patients in comparison to healthy controls (Table 2), while adiponectin levels were decreased in IIH patients. However, the comparison of age, gender, and BMI-matched subgroups only showed a statistically significantly decreased level of melatonin in those patients with IIH (Table 2), while the other hormone levels did not significantly differ between the age, gender, and BMI-matched subgroups.
Biochemical characteristics and results of hypothalamic volumetric analysis of patients with idiopathic intracranial hypertension (IIH) and healthy control subjects.
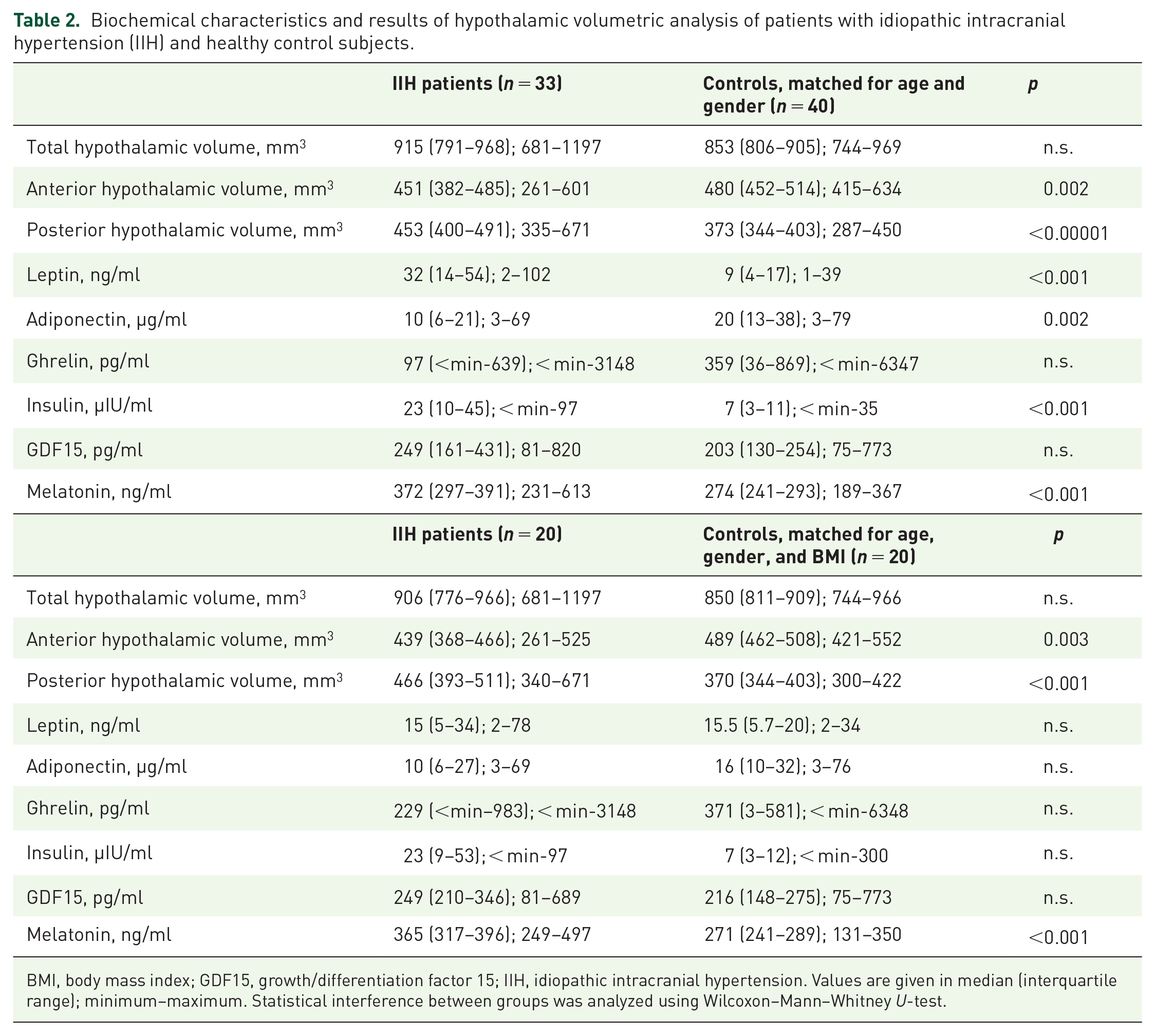
BMI, body mass index; GDF15, growth/differentiation factor 15; IIH, idiopathic intracranial hypertension. Values are given in median (interquartile range); minimum–maximum. Statistical interference between groups was analyzed using Wilcoxon–Mann–Whitney
The total hypothalamic volume was similar in IIH patients and controls as shown in Figure 1(a). Although there was no significant difference between those two groups, there was a tendency toward enlarged hypothalamus volumes in IIH patients. The anterior hypothalamus was reduced in volume in IIH patients (vs controls: –10.9%) with a large effect size of 0.81. In contrast to this lower volume, the volume of the posterior hypothalamus was significantly higher in IIH patients (vs controls: +23.5%) with a very large effect size of 1.40. The comparison of the age, gender, and BMI-matched subgroups of patients and healthy controls [Table 2 and Figure 1(b)] confirmed these findings by also showing a reduction of the anterior hypothalamus (with a large effect size of 1.1) and an enlarged volume of the posterior hypothalamus in patients with IIH (with a very large effect size of 1.41), while the volume of the total hypothalamus did not differ.
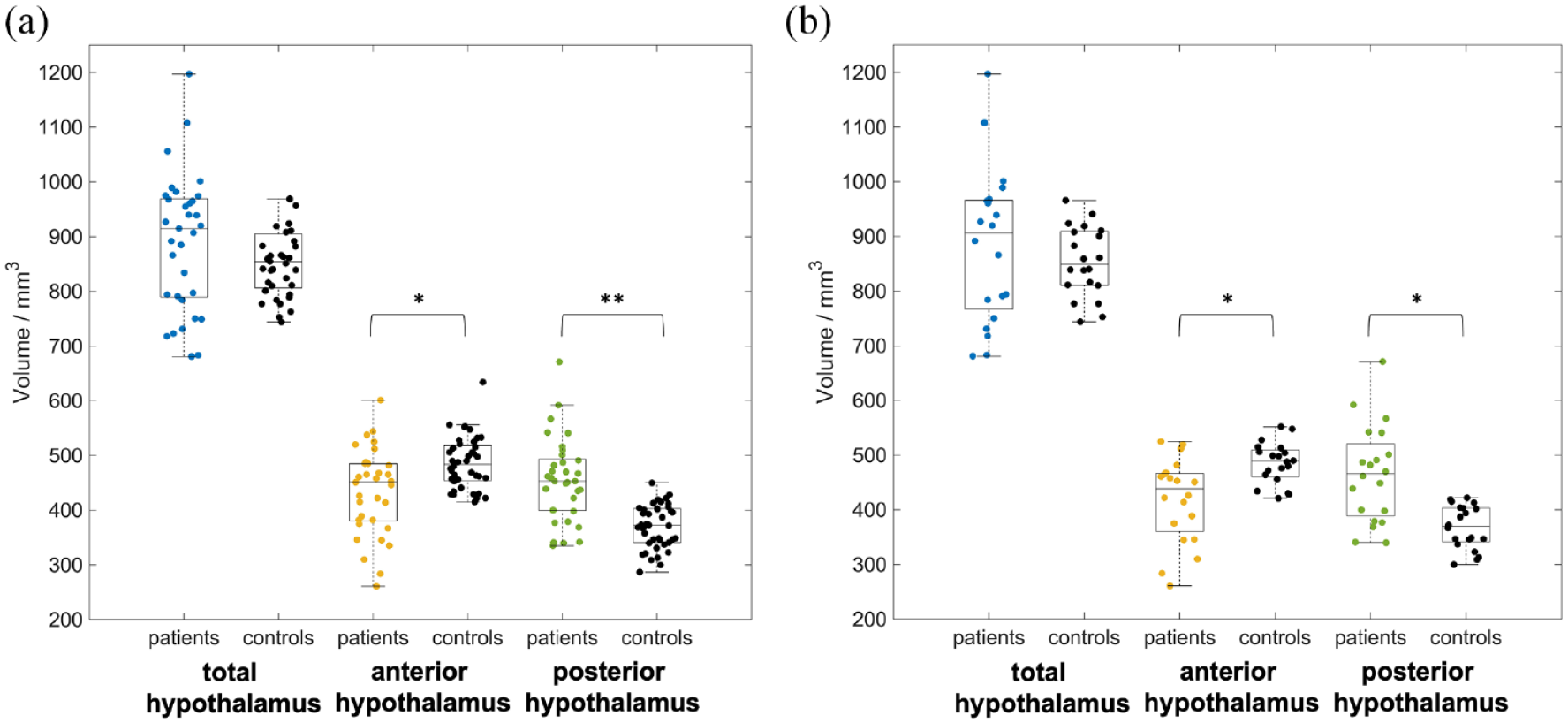
Alterations in patients with idiopathic intracranial hypertension (IIH) in anterior and posterior hypothalamus volume: (a) comparison of age- and gender-matched patient and control groups and (b) comparison of age-, gender-, and body mass index-matched patient and control groups.
The correlation analyses between hormonal serum levels and hypothalamic morphology did not achieve any significant results. There was also no significant correlation between all of the analyzed hormone levels and global, anterior, and posterior hypothalamic volumes. Regarding clinical parameters, there was also no association of any morphological or hormonal parameter with BMI, lumbar puncture pressure, age, or gender. We found no association between medication with topiramate or metformin (which was taken in a subsample of the patients) or presence of an empty sella and hypothalamic volumes as well as between either BMI, age, and gender. Regarding disease duration, there were no differences in hypothalamic volumes between patients with a short duration of disease and patients with a longer duration of disease.
Discussion
Since in IIH, increase of BMI often leads to a substantial disease progression, and on the contrary, weight loss is therapeutic, adipose tissue might be one of the major players in IIH.18,19 However, the role of the hypothalamus, as a structure which is crucial in the regulation of the energy balance, has not been investigated in detail in the pathogenesis of IIH. In this pilot study, we hypothesized that, in association with IIH, the morphology of the hypothalamus might exhibit changes in the assessment by
As a novel extension of the metabolic IIH phenotype, this study demonstrated the existence of structural hypothalamic abnormalities. There was no statistical difference of hypothalamus size between IIH and non-IIH control individuals with respect to global volumes; however, we observed a lower volume of the anterior part of the hypothalamus of about 11% in IIH patients with a higher volume of the posterior part of about 24%. In order to control for body weight, a comparison with controls matching not only for age and gender but also for BMI was performed, and confirmed these findings by demonstrating a reduction of the anterior hypothalamus and an enlarged volume of the posterior hypothalamus in patients with IIH. It may be safe to conclude that these morphological alterations of the hypothalamus structure are a specific finding associated with IIH, although not presenting as gross volume changes, and might be regarded as a cerebral imaging correlate of pathophysiological processes. Hypothalamic abnormalities have not been reported yet in the spectrum of IIH-associated findings: neuroimaging is required to demonstrate normal brain parenchyma with no evidence of structural lesions or cerebral venous sinus thrombosis for the diagnosis, while a number of neuroimaging features suggestive of IIH have been reported, including empty sella and alterations of the optic nerve.1,5 With respect to the hypothalamus, no consistent structural hypothalamic abnormalities have been established in association with obesity in general, although its involvement in the functional networks of food intake and energy metabolism is beyond all doubt. 20 A multiparametric MRI study demonstrated a relation between BMI and higher proton diffusivity in the hypothalamus but also not with hypothalamic total volume. 21 On the contrary, in systemic diseases which are associated with substantial metabolic alterations, body weight loss due to hypermetabolism, and/or reduced food intake has been demonstrated to be associated with hypothalamic atrophy, for example, in neurodegenerative diseases like amyotrophic lateral sclerosis 13 or Huntington’s disease 22 and in anorexia nervosa. 23 In dementia of the frontotemporal lobar degeneration type, a neurodegenerative disease with symptoms of eating disorder, it could be demonstrated by structural neuroimaging that this disease was associated with atrophy of the hypothalamus which was related to eating behavior; this atrophy was more pronounced posteriorly, that is, in a hypothalamic region containing nuclei that play a critical role in regulating food intake behavior. 16 Other (diffusion-based) neuroimaging studies in healthy controls demonstrated abnormalities in the anterior–superior hypothalamic subregion which contains the paraventricular nucleus which is suggested to be involved in inhibitory control of food intake and energy expenditure. 24 In the light of these studies, the current result of abnormal hypothalamic morphology with significantly higher volumes of the posterior hypothalamus than controls might indicate to be a correlate of the involvement in the metabolic control dysregulation. Given that the hypothalamus is anatomically a small, but functionally differentiated area, 25 the smaller volume of the anterior part which is involved in inhibitory control of food intake and the larger posterior part which is involved in regulating food intake behavior16,24 might mirror the altered functional state with respect to structure.
In our study, the metabolic phenotype of IIH could be confirmed with respect to altered hormone levels of leptin, adiponectin, melatonin, and insulin, in accordance with a recent study on metabolic determinants of the IIH phenotype. 7 Hyperinsulinemia, hyperleptinemia, and low levels of adiponectin suggest general metabolic dysbalance as part of the adipose tissue–insulin resistance circle. 26 However, in the comparison of subgroups of IIH and controls also matched for BMI, this result did not hold, and the hormone levels did not differ any more except from melatonin levels in IIH. As a consequence, these correlations seem to be based on the BMI differences only and cannot be considered to be an IIH-specific effect. For melatonin, which (among other functions) is suggested to normalize the expression and secretion of leptin and adiponectin, and therefore is thought to have a beneficial role in insulin resistance and maybe even obesity, 27 we observed higher levels in the IIH cohort including the comparison with BMI matching, but its role in IIH needs to be further defined.
Taken together, we found morphological alterations of the hypothalamus in IIH. The observed hypothalamic atrophy could result from tissue remodeling in response or in compensation to the important hormonal changes associated to IIH. Within the metabolic concepts of IIH, it is discussed that IIH represents a distinct neurological manifestation of a phenotype with female androgen excess and male androgen deficiency. 18 Indeed, it is possible that altered androgen metabolism, as observed in IIH, contributes directly to hypothalamic atrophy or indirectly through altered CSF dynamics. In that context, hypogonadism in men is associated with structural hypothalamic defects,28,29 and it is possible that the observed altered androgen metabolism in women with IIH 6 contributes to the observed hypothalamic atrophy. Future studies in IIH should include hormones of androgen metabolism which was, as a limitation, not done in our study. Our results cannot directly point to a functional role of the hypothalamus in the IIH-obesity complex, all the more since they do not indicate if the altered hypothalamic morphology is a primary or a secondary phenomenon within the development of IIH. The morphological changes could be secondary to raised intracranial pressure or a consequence of changes occurring in the pituitary gland, as it is well understood that patients with IIH frequently have non-specific intracranial findings associated with raised intracranial pressure. It seems possible that the metabolic changes are the consequences of a defect in the central nervous system (CNS), involving remodeling of hypothalamic networks, similar to what occurs in neurodegenerative diseases, but further studies are required.
If a stronger consideration of hypothalamic involvement in the therapeutic concepts of IIH, beyond the approaches with the somatostatin analogon octreotide, 30 might be helpful remains speculative at this stage. It has, however, to be considered that – although the therapeutic impact of weight loss in IIH is clear – the optimal strategy necessary to induce weight loss is not established and may be patient specific, as a combination of the existing approaches (lifestyle and behavioral interventions, bariatric surgery, and novel therapies such as glucagon-like peptide 1 receptor agonists) 31 and further components to be developed in the future.
The findings should be considered in the context of several limitations. First, the sample size was relatively small, but IIH is still regarded as a rare disorder, although it might be underdiagnosed. 32 Second, our imaging approach with T1w structural data and volumetric analysis of the hypothalamus might only be a first indication of abnormalities, but cannot address its functional integration in the pathophysiological concepts. Third, there were no clinical signs of raised intracranial pressure among the controls according to medical history and clinical impression and MRI, but no ophthalmoscopy or lumbar puncture was performed in the control subjects. Finally, future studies will have to use correlational analyses with fluid hormonal markers like leptin, somatostatin, and hormones of androgen metabolism together with advanced structural and functional neuroimaging techniques which allow for more detailed differentiation of hypothalamic subunits and target the hypothalamic connectivity.20,21,23,33,34
In summary, this volumetric analysis of the hypothalamus in IIH patients demonstrated structural abnormalities associated with the IIH-obesity complex, as an extension of the IIH metabolic phenotype. The mechanism of hypothalamic changes remains to be explored. Further studies with higher sample sizes and longitudinal design are necessary to corroborate these pilot data, but it might be useful to integrate the hypothalamus into human research activities and preclinical research in IIH 35 to improve our understanding of the underlying etiology and the mechanistic concepts in driving disease pathology in this burdening disease with the target of effective research programs and well-designed trials in IIH.
