Abstract
Colon capsule endoscopy (CCE) has been available for nearly two decades but has grappled with being an equal diagnostic alternative to optical colonoscopy (OC). Due to the COVID-19 pandemic, CCE has gained more foothold in clinical practice. In this cutting-edge review, we aim to present the existing knowledge on the pros and cons of CCE and discuss whether the modality is ready for a larger roll-out in clinical settings. We have included clinical trials and reviews with the most significant impact on the current position of CCE in clinical practice and discuss the challenges that persist and how they could be addressed to make CCE a more sustainable imaging modality with an adenoma detection rate equal to OC and a low re-investigation rate by a proper preselection of suitable populations. CCE is embedded with a very low risk of severe complications and can be performed in the patient’s home as a pain-free procedure. The diagnostic accuracy is found to be equal to OC. However, a significant drawback is low completion rates eliciting a high re-investigation rate. Furthermore, the bowel preparation before CCE is extensive due to the high demand for clean mucosa. CCE is currently not suitable for large-scale implementation in clinical practice mainly due to high re-investigation rates. By a better preselection before CCE and the implantation of artificial intelligence for picture and video analysis, CCE could be the alternative to OC needed to move away from in-hospital services and relieve long-waiting lists for OC.
Keywords
Introduction
Focus on early detection and treatment of colorectal cancer (CRC) has improved symptom awareness and implementation of population-based CRC screening programmes. 1 Consequently, millions of optical colonoscopies (OCs) are performed annually in Europe. 2 OC is a sensitive and accurate diagnostic tool. There are, however, some concerns. The number needed to scope (NNS) in CRC screening programmes to diagnose one is increasing. In symptomatic patients referred by their general practitioner (GP), the NNS to diagnose one CRC is even higher. Moreover, as the current practice is to resect almost every polyp found during an OC, millions of time-demanding procedures are generated throughout Europe, and resource demands for highly specialised medical personnel, facilities and equipment are increasing. Waiting lists for OC are unacceptably high in a few countries; concerns have been expressed that the required resources negatively affect other activities within surgical and endoscopy departments. Even though OC’s complication rate is low, the sheer number of procedures performed leads to greater than 1000 deaths and greater than 100,000 significant complications in the EU each year. Furthermore, the rate of minor complications may be underreported. 3 One investigation reported a 6% sick leave after OC ranging from 1 to 9 days, significantly adding to society’s costs. 4 Finally, one colonoscopy produces more than 1.5 kg of nonrecyclable plastic and uses high energy and water levels. 5 Globally, healthcare sectors are responsible for 4.6% of CO2 emissions, whereas endoscopy activity is a significant contributor. 6
CCE has been available since 2006.7,8 Technical and clinical challenges have prevented CCE’s widespread adoption and use for years. However, despite improvements in imaging quality, pictures from CCE still fail to match the picture quality from OC (Figure 1). Consequently, due to lack of evidence, European guidelines recommend CCE only for a few indications. 9 However, with the pioneering ScotCap programme and, more recently, the English Camera Capsule Endoscopy programme, there seems to be a change of hearts.10,11 More than 10,000 CCEs have been used in routine clinical practice mainly to exclude colonic neoplasia. CCE’s obvious advantages (noncontact procedure, favourable safety profile), and the immense pressure generated by the disruption of colonoscopy services, facilitated a better pitch for CCE during the COVID-19 pandemic.12,13 We know patients will likely prefer CCE, especially if an OC does not follow the CCE test. 14 The advantage of providing the capsule camera test close to local healthcare centres or even in the patient’s residence is undoubtedly attractive. Data are accumulating that the sensitivity and quality of CCE can match OC incomplete investigations. 15 The environmental impact of nonrecyclable colon capsules still needs to be estimated and will of course increase the overall carbon footprint from the diagnostic pathway when a subsequent OC is warranted.
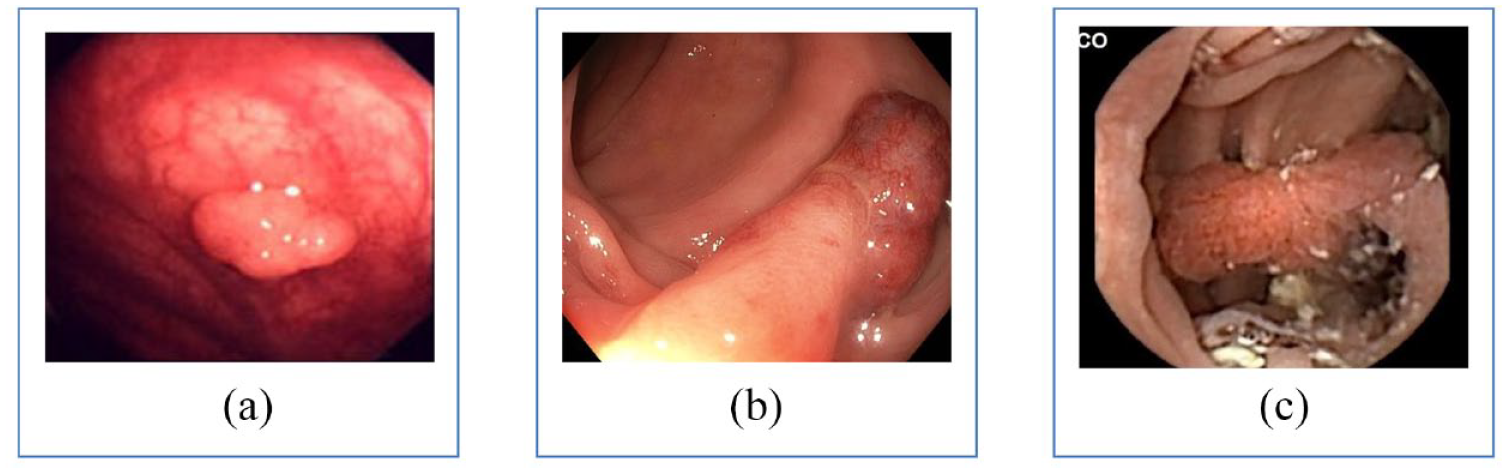
Presentation of colonic polyps depicting picture quality improvements over the last decades. (a) Picture of a polyp during colonoscopy from 1998. (b) Picture of a polyp during colonoscopy anno 2022. (c) Same polyp as depicted in b but obtained during colon capsule endoscopy.
Definitions
The literature contains many clinical trials on CCE performance.15–17 The definition of endpoints and terminology vary and can be confusing. The term completion rate means in some publications complete transit and others complete investigation, including adequate bowel preparation.
The terms used here are as follows:
Complete transit: Capsule passes to the anal cushions with active battery.
Bowel cleanliness: According to the Lexington-Rex scale. Acceptable cleanliness is fair or better.
Complete investigation: Complete transit with active battery and acceptable cleanliness without technical failures. It is conclusive even if no pathology is demonstrated.
Conclusive investigation: Complete investigation or incomplete investigation demonstrating pathology that needs therapy.
Re-investigation rate: Needs an ensuing investigation because of incomplete CCE or for therapy.
Requirements for widespread implementation
Patient preference: The CCE procedure is less uncomfortable for patients, causes less anxiety and intimidation and can be regularly performed on an out-of-hospital basis. Conversely, OC is a test that passed the test of time and continues to evolve. More than 80% of patients tend to gravitate towards the former when asked to choose between CCE and OC. 17 This represents only a ‘convenient’ representation of the whole truth. Patients do not tend to favour one or the other diagnostic test alone; instead, they state their preference based on the assumption that this test will be the one to give answers and lead to the next step in the management. Therefore, when presented with the possible consequences of undergoing an inclusive procedure tend to shift towards the test that will give the answers. In light of this, a recent meta-analysis shows a 50/50 distribution in patient preferences between CCE and OC diagnostic pathways. 14 Essentially, ‘champions’ for the CCE pathway are those with a complete (and negative) examination; hence, the patient group does not require a further endoscopic test or other interventional therapy. However, the ‘losers’ of the CCE diagnostic pathway are those needing two rounds of laxative preparation, either because the CCE was incomplete or inconclusive or because it showed pathology requiring more procedures. Therefore, a low re-investigation rate is a prerequisite for a clear CCE preference.
High accuracy: CCE has excellent sensitivity. A recent meta-analysis showed that CCE’s polyp and adenoma detection rate (ADR) is between 90% and 99%. 15 A back-to-back comparison between OC and CCE, where a second OC solved any disagreement, revealed that most of the so-called CCE ‘false positives’ were detectable. In other words, they were OC’s false negatives. 18 The impressive performance of AI-supported solutions in detecting colon pathology missed by ‘unsupported’ human assessment corroborates this. 19 Another facet of the problem is that although CCE has higher sensitivity, especially for flat adenomas, it delivers severe errors in size estimation. The size estimation tool integrated with Rapid Reader (which, despite the company’s disclaimer, is regularly used in clinical care) provides a 30% polyp size overestimation compared with OC and pathology measurements for matched polyps. 18 It may lead to over-reporting of large and under-reporting of diminutive/small polyps.
Double counting during CCE can sometimes lead to upstaged polyp burden. Dual reporting can be a significant problem as the camera capsule often passes back and forth several times in colon segments. This often confuses the following therapeutic OC and leads to prolonged OC procedures, looking for polyps that do not exist. It has been shown that the polyp detection rate (PDR), corrected for double reporting, is higher for CCE than OC, which increases re-investigation rates. 18 Cancer miss rate and interval cancer rate in CCE have not yet been established.
Quality reporting
Each CCE produces greater than 25,000 images, and reading is time-consuming and costly. 20 Furthermore, CCE reporting is burdened by remarkable intra- and interobserver variations. Unpublished data from our team show that although polyp detection has an acceptable interobserver agreement, polyp size reporting and bowel cleanliness grading carry unacceptable kappa values. Artificial intelligence (AI) algorithms in capsule endoscopy have shown promising results but developing trustworthy AI is complicated. 21 The results of AI grading on cleanliness, based on the total number of CCE images of each video, might be quite different from human assessment and indeed, the ‘truth’; capsule might spend much more time in the caecum than in the transverse colon. Furthermore, the amount of information on a particular frame on cleanliness depends on the proximity of the capsule dome to the colon wall. Landmarks such as colonic flexures are used when reporting the localisation of lesions and segmental bowel cleanliness. Still, as the capsule might pass one flexure several times, research is needed to investigate the reliability hereof.
Re-investigation rate: Essentially, CCE is a glorified screen-out test with two possible outcomes: One is a complete investigation with no findings. Patients can be reassured, and their care is taken forward. The other outcome comprises cases with an incomplete examination (inadequate bowel cleanliness/incomplete transit) or the need for biopsy or polyp removal. The three ways to reduce re-investigations (Figure 2) are as follows:
To improve the rate of complete transits and bowel cleanliness.
To select patients for CCE who have a low risk of positive findings.
To constrain therapeutic re-investigation to those individuals who will benefit from the procedure.

Venn diagram presenting factors to consider to diminish the re-investigations rate after colon capsule endoscopy.
Published data from the more extensive clinical trials designed on an intention-to-treat basis show that the incomplete investigation rates are high and remain a concern regarding patients’ experience and economy. A recent French study of CCE in clinical practice reported a complete investigation rate of 48.9%. 22 ScotCap, using CCE in symptomatic and surveillance patients, reported a re-investigation rate of 63% and 70%, respectively. 11 There is a consensus that to improve patient experience and economy, the ideal investigation should be equivalent to OC, that is, a completion rate of greater than 90% and good visualisation of greater than 90%. A recent study introducing a new booster to reduce transit times showed promising results. 23 With the use of prucalopride, complete transit can be achieved in greater than 95% with the caveat that a few investigations will be too fast for proper colon inspection. The bowel preparations in CCE vary, but a high dose of polyethylene glycol (PEG) is the norm. 24
The regimen and timing of bowel preparation are of importance. A significant improvement can be achieved by careful timing between the cleansing doses and the boosters. Successful bowel cleansing is more likely when the capsule is ingested in the morning before 10 o’clock. Patient instructions and motivation are being investigated, but no hard evidence exists. At this time, it is necessary to improve the completion rate by optimising the bowel preparations and boosters, patient instructions and motivation and very likely by a pretest selection of patients for CCE based upon patient demographics, medical history, medication and indication for the CCE. The use of blood samples and biomarkers might be necessary for the future. Improved CCE in clinical practice will be significant if these causes of too many and too long re-investigations can be solved.
Realistic medicine: The current guidelines for resection of polyps have been created in the light of colonoscopy where you are at the lesion site, and resection is just a tiny addition in procedure length, cost and risk. 25 The diagnosis of polyps requiring resection in CCE has high consequences regarding a new bowel cleansing procedure and OC. The guidelines have to be tested in light of implications for the patients. Recently, Campbell et al. 26 suggest leaving diminutive polyps untreated or allocating those to other activities such as participation in the screening system or repeated FIT testing. The savings introduced by these realistic medicine approaches might leave room and economy for lowering the fecal immunochemical test (FIT) threshold value. The net effect is likely to be positive for the long-term patient outcome.
AI algorithms
Deep learning algorithms in convolutional neural networks (CCN) are tailored for image analysis. They have demonstrated high diagnostic performance levels in small-bowel capsule endoscopy. 27 However, few studies have reported the yield of AI in CCE. 28 Ribeiro et al. developed an AI algorithm for detecting blood in CCE images. 29 They retrospectively extracted greater than 5,000 CCE pictures used to train and validate the CNN, achieving a sensitivity of 99.8% and specificity of 93.2% for the detection of blood. In another study, the same authors found a deep learning algorithm capable of detecting protruding lesions in CCE images with a sensitivity of 90.7% and specificity of 92.6%. 30 Implementing AI for picture analysis in CCE seems to be the apparent solution to cope with a large-scale CCE roll-out. That said, we are faced with critical ethical considerations here. Are we ready and willing to rely on AI to diagnose colorectal lesions, including malignancies? Probably not. We have to await studies on large datasets from different centres and applied to varying populations before we can loosen the reins in a stepwise course.
Preselection
CCE will not be a realistic alternative to all colonoscopy procedures. CCE makes sense only in a limited population where the share of patients avoiding re-investigation after CCE is high. In our minds, CCE should be offered to patients only with a priory risk of re-investigation of less than 25%. To achieve this, we need efficient procedures for bowel preparation and transit times and, indeed, carefully selected patient groups. Therefore, the steps here should be defined as
Identify low-risk population cohorts for colorectal neoplasia;
Identify factors which favour high completion rates;
Apply a realistic medicine approach;
Sum up 1–3 and decide whether the patient is a good candidate for CCE.
Low-risk populations
The frequency of positive findings varies with indication. The ScotCap data showed that symptomatic patients referred by GPs have a low risk for pathology. 11 A FIT triage offering CCE only to those with a low FIT value can significantly reduce this risk. Essentially, around 80–90% of the patients are referred for endoscopy. Other populations included in the Scottish and English CCE pathways are screenees with low positive FIT values and patients requiring follow-up after a previous polypectomy. Other populations should also be considered. These include children, young surveillance patients with familial adenomatous polyposis (FAP), patients needing anaesthesia for a colonoscopy and patients refusing colonoscopy.
Several patient-related factors have been linked to an increased risk of colorectal neoplasia. Johnson et al. 31 showed by a meta-analysis that patients with inflammatory bowel disease and a history of first-degree relatives with CRC had a much higher risk of developing CRC. In the same study, increased body mass index (BMI), red meat intake, cigarette smoking, low physical activity, and low fruit and vegetable consumption were associated with a moderately increased risk of CRC. In another study, a higher vitamin D and marine omega-3 fat intake were associated with a lower risk for the development of colorectal adenoma. 32 Chemoprevention has also gained increased attention, but medications’ protective effect on polyp and cancer development must be balanced against adverse events. Aspirin, nonaspirin nonsteroidal anti-inflammatory drug (NSAIDs) and metformin have been associated with a lower adenoma and CRC incidence, but the recommended routine chemoprevention is scarce. 33 Big data-based algorithms might facilitate the selection of patients who should be advised a CCE and who should not.
Factors associated with a low completion rate
Several risk factors for incomplete OC have been identified. In a Japanese study, female sex, age ⩾60 years, history of prior pelvic or abdominal surgery, inflammatory bowel disease and poor bowel cleansing were risk factors for incomplete OC. 34 In another study in a Danish bowel cancer screening population, lower income quartiles were associated with increased odds of incomplete OC due to poor bowel preparation. 35 Higher BMI has also been reported as a risk factor for incomplete OC.36,37 For small-bowel capsule endoscopy, gastric transit time longer than 45 min, previously small-bowel surgery, hospitalisation and poor bowel preparation have been identified as risk factors for an incomplete examination. 38 To date, risk factors for incomplete CCE have not been investigated. Future studies need to identify independent factors for incomplete CCE to optimise patient preselection and increase the odds of a successful CCE.
Safety: As the colon capsule is moved forward by the bowel peristalsis without counteracting mechanical forces, the risk of major complications is very low. A possible severe complication is capsule retention, defined as the capsule remaining in the gastrointestinal tract for a minimum of 2 weeks, confirmed by abdominal imaging or the need for surgery, endoscopic or medical intervention to remove or aid in the capsule passage. It has been reported in 0.26% of cases. 13 However, in most instances, the capsule will be egested spontaneously after 2 weeks.
Accessibility: CCE seems well suited for servicing remote areas and societies with no setup for OC. General practitioners and nurses can be trained to deliver capsules and upload videos to a remote facility for picture analysis. To implement CCE with remote analysis, all that is needed is some receiver belts, computers and safe electronic connections.
Cost-effectiveness
National health service (NHS) highlands presented a cost-efficiency analysis on the use of CCE compared with a colonoscopy-first diagnostic pathway, indicating a 20–30% higher cost with the CCE pathway. The calculations are very sensitive to the re-investigation rates, which, in both UK pathways, are high. We believe we can reach an economic break even by reducing the re-investigation rate to approximately 25%. According to calculations based upon the Danish Care-for-Colon trial, including 6000 FIT-positive screening individuals in a randomised trial between CCE and colonoscopy, it is indicated that a possible prize reduction in the procedure of about 20% by adding AI reading to the pathway and reducing manual reading from 60 to 75 minutes to approximately 10 minutes is achievable. The breakeven will likely be reached within 1–2 years. The economy will likely further be facilitated as more commercial colon capsule providers enter the market within 2022.
AI-supported Image Analysis in Large Bowel Camera Capsule Endoscopy (AICE)
AICE is the Horizon Europe programme awarded in 2022 to support the international project: ‘AICE’ led by the centre for clinical implementation of capsule endoscopy (CICA) in Svendborg, Denmark (Figure 3). The aim of this programme, including 13 international centres, is to develop a complete, evidence-based cost-efficient diagnostic pathway for CCE analysis. It includes sky-based analytical facilities that hopefully will provide global availability even without onsite facilities other than trained nurses for capsule delivery and a safe connection to the sky-based analytical and report generation. Recognising multiple ethical challenges, the programme has allocated a large proportion of the grant to this topic.

AICE pathway showing the patients’ journey from referral to large bowel diagnostics until completed CCE diagnostics.
Conclusion
CCE has some apparent advantages compared with OC, including less discomfort, a low risk of adverse events, and the fact that it can be performed out of the hospital. However, the re-investigating rate needs improvements through developing algorithms for patient preselection and implementation of AI in picture analysis before a broader implementation in clinical practice is favourable for patients and a cost-effective approach.
