Abstract
Introduction:
Although recent advances in chemotherapy for lung cancer are remarkable, most clinical trials have excluded patients with interstitial lung disease (ILD) due to the concern of developing acute exacerbation (AE) of ILD. Hence, accumulating original evidence of cancer treatment for this population is important.
Methods:
Between 2016 and 2020, a prospective observational study was conducted across 11 Japanese hospitals. Patients with chemotherapy-naïve, inoperable, advanced lung cancer with ILD were included. The primary outcome was the frequency of AE-ILD after registration; the secondary outcomes were the risk factor of AE-ILD and the efficacy of chemotherapy.
Results:
Among 124 patients enrolled, 109 patients who received chemotherapy were analyzed. The median age was 72 years, and the majority showed usual interstitial pneumonia (UIP)/probable UIP pattern upon chest computed tomography. The median percent-predicted forced vital capacity (%FVC) was 81% (interquartile range: 66–95%). After registration, 23 patients (21.1%; 95% confidence interval [CI]: 14.4–29.7%) developed AE-ILD. The logistic analysis revealed that lower %FVC slightly but significantly increased the risk of AE-ILD (odds ratio per 10% decrease: 1.27; 95% CI: > 1.00–1.62). Overall response rates/median overall survival times in non-small-cell lung cancer and small-cell lung cancer for the first-line chemotherapy were 41% (95% CI: 31–53)/8.9 months (95% CI: 7.6–11.8) and 91% (95% CI: 76–98)/12.2 months (95% CI: 9.2–14.5), respectively.
Conclusion:
AE-ILD during chemotherapy is a frequent complication among patients with lung cancer with ILD, particularly those with lower %FVC. Conversely, even in this population, passable treatment response can be expected.
Introduction
Lung cancer is one of the major complications in patients with interstitial lung disease (ILD), particularly those with idiopathic pulmonary fibrosis (IPF) and/or those with heavy smoking history.1–5 For patients with advanced lung cancer, systemic chemotherapy may be the only treatment option, but if the patients originally have ILD in their lungs, the treatment can trigger acute exacerbation of preexisting ILD (AE-ILD) with an incidence rate of 13.3–30.6%.4,6–9 Although recent advances in chemotherapy for advanced lung cancer are remarkable, including molecular targeted drugs and immune checkpoint inhibitors,10,11 most clinical trials have excluded patients with ILD due to the concern of developing AE-ILD or severe drug-induced pneumonitis, which precludes such patients from evidence-based treatment. Hence, so far, whether chemotherapy outweighs the risk of AE-ILD is empirically judged in each clinical setting; this indicates the need to accumulate original evidence for this population regarding the safety, particularly for the risk of AE-ILD, and efficacy of systemic chemotherapy.
To date, several researchers have identified the risk factors for chemotherapy-associated AE-ILD among patients with lung cancer with ILD, including the usual interstitial pneumonia (UIP) pattern on chest computed tomography (CT), 6 younger age (<70 years), 6 higher serum C-reactive protein (CRP), 7 lower percent-predicted forced vital capacity (%FVC), 8 non-small-cell lung cancer (NSCLC), 8 and higher GAP index, 12 although results were inconsistent. Unfortunately, all the candidate factors reported were derived from retrospective cohort analyses, suggesting that prospective evaluation would be valuable to increase data reliability.
Based on these backgrounds, we planned a prospective observational study for patients with advanced lung cancer with preexisting ILD. Through the observation of our cohort, we aimed to discuss about the appropriate treatment approach for this population.
Materials and methods
Study design and participants
This prospective study included patients with chemotherapy-naïve, inoperable, advanced lung cancer with preexisting ILD. Patients were recruited from 11 Japanese hospitals since 1 November 2015, until 30 September 2019. The data cut-off was 31 October 2020. The study protocol was registered under number UMIN000019370 and was also approved by the review board of each participating institution (Hamamatsu University School of Medicine: E15-126). Written informed consents were obtained from all the participants at the registration.
Inclusion criteria were as follows: (1) diagnosis of primary lung cancer with histopathological evidence; (2) chemotherapy-naïve, advanced clinical stage without indication of curative surgery and radiotherapy; and (3) presence of preexisting ILD. ILD diagnosis was defined as bilateral reticular shadows and/or ground-glass attenuations on chest CT, which was confirmed in each institution at registration. Patients who had postradiation fibrosis alone were excluded in advance. The included patients were managed without any treatment suggestion from the executive secretariat of the study.
Between November 2020 and June 2021, clinical information about the enrolled patients was collected. During the collection period, the baseline ILD patterns (UIP compatibility) upon chest CT and the ILD diagnosis (primarily IPF or not) were determined in each institution based on the most recent criteria. 13 In addition, the semiquantitative extents of emphysema and normal lung area on chest CT were also evaluated in each institution. The lung cancer stage was classified according to the eighth edition of the UICC TNM classification. AE-ILD was diagnosed using the following criteria: (1) acute worsening or development of dyspnea, (2) CT findings showing newly developed bilateral ground-glass attenuations and/or nonsegmental consolidation superimposed on preexisting ILD shadows, (3) exclusion of pulmonary infection by negative respiratory culture or ineffectiveness of wide-spectrum antibiotics, and (4) deterioration not fully explained by cardiac failure or fluid overload. AE-ILD (or pneumonitis) severity was graded according to the Common Terminology Criteria for Adverse Events version 5.0.
Outcomes
The primary outcome was the frequency of AE-ILD after registration. Because AE-ILD can spontaneously occur in the clinical course of ILD even without chemotherapy, 14 rigorously distinguishing chemotherapy-associated ‘triggered’ AE-ILD from ‘idiopathic’ AE-ILD is difficult. In this study, therefore, all the event of AE-ILD after registration was included in the primary outcome. Conversely, AE-ILD frequency in each chemotherapy line was determined according to the onset timing; that is, AE-ILD during ‘X’th-line chemotherapy was defined as the event between the initiation of ‘X’th-line treatment and that of ‘(X + 1)’th-line treatment or death/censoring. Variables for risk factor analysis included the following: sex; age; smoking dose (pack-years); performance status defined by Eastern Cooperative Oncology Group (ECOG); type of tumor histology; lung cancer stage; UIP compatibility on chest CT (UIP/probable UIP pattern or not); ILD type (IPF or not); semiquantitative extents of emphysema and normal lung area on chest CT; AE-ILD history before registration; pulmonary function parameters including %FVC and percent-predicted diffusing capacity for carbon monoxide (%DLco); GAP index calculated from age, sex, %FVC, and %DLco; 15 and clinical laboratory parameters including CRP, lactic acid dehydrogenase, Krebs von den Lungen-6, and surfactant protein-D. The secondary outcomes were the risk factor of AE-ILD and the efficacy of chemotherapy including treatment response and overall survival (OS) time after treatment initiation.
Sample size
Assuming that a point estimate of the frequency of AE-ILD, the primary endpoint, could be obtained around the 13.3–30.6% previously reported,4,6–8 it was calculated that 300 cases would be required to achieve an estimation precision (95% confidence interval [CI]) of ±5%, which was set as the target sample size in this study. However, since the case collection period was actually completed with 124 cases, post hoc power analysis yielded a point estimate accuracy of ±8%.
Statistical analysis
Data were presented as numbers with percentages or medians with interquartile ranges (IQRs). For group comparison, Fisher’s exact test or Mann–Whitney U test was used as appropriate. Among baseline variables, logistic regression models were used to evaluate the risk factors for AE-ILD. The Kaplan–Meier survival method was used to estimate median OS time. Cox proportional hazard model was used to evaluate the risk of death. All p values < 0.05 were considered statistically significant.
Results
Baseline patient characteristics
Over the study period, a total of 124 patients were enrolled, but five patients were excluded due to not meeting inclusion criteria. Among the remaining 119 patients, 109 patients were started with systemic chemotherapy and were analyzed for our primary/secondary outcomes. Table 1 shows the baseline characteristics of the 109 patients. The median age was 72 years (IQR: 65–77), and the majority were men (90%) with a heavy smoking history of more than 40 pack-years. Emphysema was detected in as many as 86% of the total cohort. NSCLC was more frequent than small-cell lung cancer (SCLC). Regarding ILD, UIP or probable UIP pattern on CT was observed in 69%, and the diagnosis of IPF was confirmed in 53%. Other non-IPF patients included those with non-IPF idiopathic interstitial pneumonias (n = 41), collagen vascular disease-associated ILD (n = 6), pneumoconiosis (n = 3), and IgG4-related disease-associated ILD (n = 1). The median %FVC and %DLco in the total cohort were 81% (IQR 66–95) and 73% (IQR 58–83), respectively. At registration, six patients (6%) had been pharmaceutically treated for their ILD, which included nintedanib (n = 2), pirfenidone (n = 1), and corticosteroids with/without immunosuppressive drugs (n = 3). Ten patients (9%) had a history of prior ‘idiopathic’ AE-ILD.
Baseline characteristics of 109 patients who received chemotherapy.

GAP index is calculated according to a previous study. 15 AE, acute exacerbation; CRP, C-reactive protein; CT, computed tomography; DLco, diffusing capacity for carbon monoxide; FVC, forced vital capacity; ILD, interstitial lung disease; IPF, idiopathic pulmonary fibrosis; IQR, interquartile range; KL-6, Krebs von den Lungen-6; LDH, lactic acid dehydrogenase; NSCLC, non-small-cell lung cancer; PS, performance status; SCLC, small-cell lung cancer; SP-D, surfactant protein-D.
AE-ILD after registration
The total cohort has a median follow-up period of 292 days (range: 19–1548 days). Over this period, 23 patients (21.1%; 95% CI: 14.4–29.7%) developed AE-ILD. The flowchart is shown in Figure 1. During the first-line chemotherapy period, AE-ILD was observed in 11.9% of the patients, and the event was frequent during chemotherapy cycles of three or more. Conversely, during the second-line chemotherapy period, AE-ILD was observed in 14.1% of the patients, mainly in the cycles of two or less. The representative CT images of AE-ILD are shown in Figure 2.
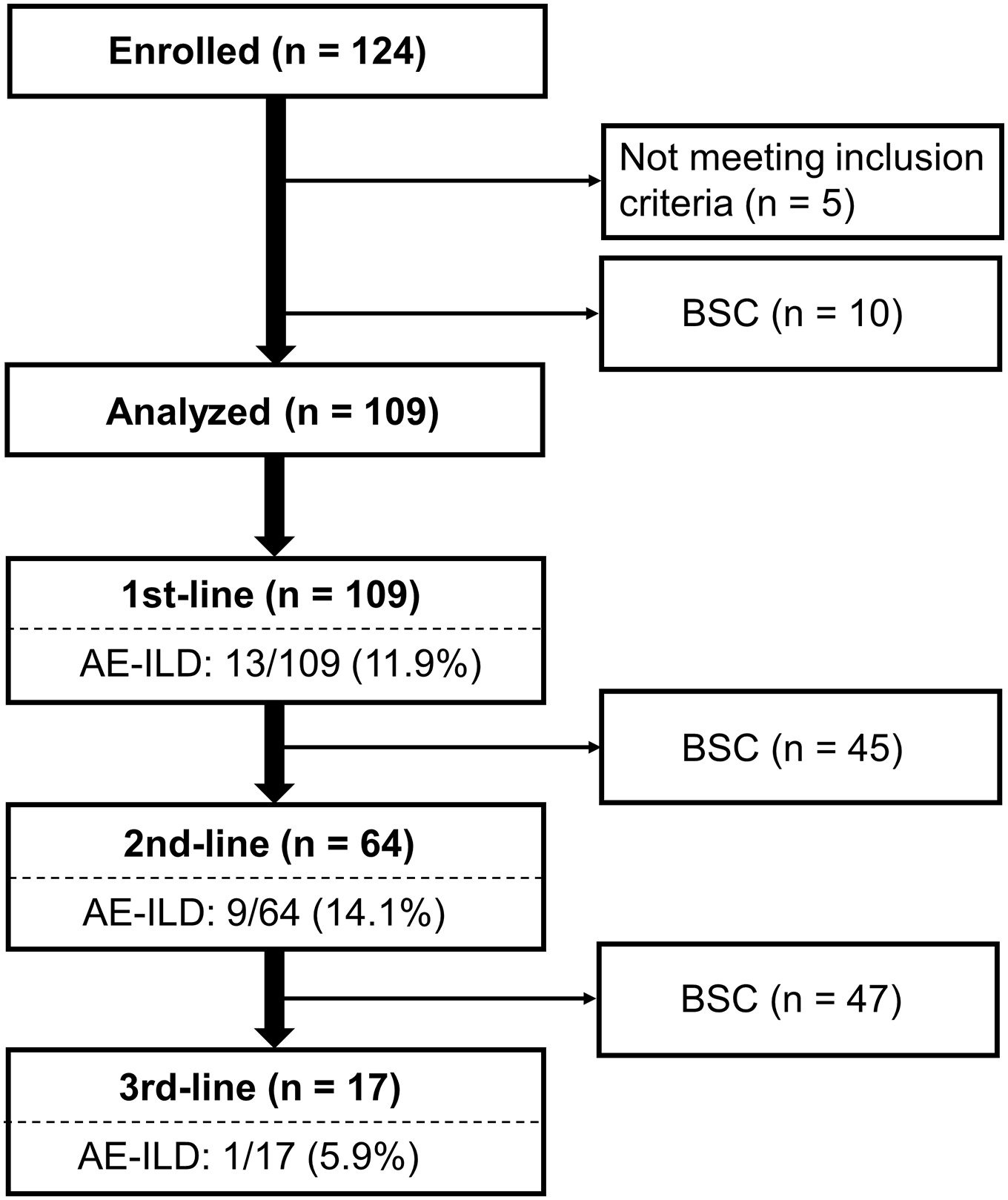
Flowchart of patients.

Representative chest computed tomography images of acute exacerbation of preexisting interstitial lung disease (AE-ILD). Case1 (left): a 69-year-old male patient with lung adenocarcinoma with rheumatoid arthritis-associated interstitial lung disease who received carboplatin plus nab-paclitaxel. Case2 (right): a 65-year-old male patient with lung squamous cell carcinoma with idiopathic pulmonary fibrosis who received carboplatin plus nab-paclitaxel.
Table 2 and Supplementary Table 1 list the frequency and the severity of AE-ILD in each administered regimen. The most common severity of AE-ILD (pneumonitis) events was grade 3, regardless of drug selected. Three patients died of AE-ILD (mortality rate: 13%), and the remaining patients survived mostly after high-dose corticosteroid therapy with an oxygen supplement.
Frequency and severity of acute exacerbation of preexisting interstitial lung disease in each treatment regimen.

Only regimens with n > 5 are included in this table and all of the raw data are shown in Supplementary Table 1. NSCLC, non-small-cell lung cancer; SCLC, small-cell lung cancer.
Table 3 compares the baseline characteristics of patients who developed AE-ILD or those who did not. Among the factors, patients who developed AE-ILD showed significantly lower %FVC than those who did not (median: 72.4% versus 82.8%, p = 0.04). Particularly, when the %FVC value was less than 70%, the overall incidence of AE-ILD was as high as 32% (95% CI: 17.8–50.8%). No significant association was observed between other factors, including age, sex, NSCLC, CRP, UIP/probable UIP pattern on CT, IPF diagnosis, %DLco, GAP index, and the extent of normal lung area on CT, and the development of AE-ILD. Consistent with that, logistic analyses revealed that only lower %FVC was associated with the increased risk of AE-ILD (odds ratio per 10% decrease: 1.27; 95% confidence interval [CI]: >1.00–1.62, p = 0.047).
Comparison of baseline factors between patients who developed acute exacerbation of preexisting interstitial lung disease and those who did not.

AE-ILD, acute exacerbation of preexisting interstitial lung disease; CRP, C-reactive protein; CT, computed tomography; DLco, diffusing capacity for carbon monoxide; FVC, forced vital capacity; IPF, idiopathic pulmonary fibrosis; IQR, interquartile range; KL-6, Krebs von den Lungen-6; LDH, lactic acid dehydrogenase; NSCLC, non-small-cell lung cancer; PS, performance status; SCLC, small-cell lung cancer; SP-D, surfactant protein-D.
Evaluated by Fisher’s exact test or Mann–Whitney U test as appropriate.
Efficacy of chemotherapy
Table 4 summarizes the efficacy profile of chemotherapy. For the first-line chemotherapy, the overall response rate (ORR) in NSCLC patients was 41.3% (95% CI: 30.9–52.6). Remarkably, the rate in SCLC patients was as high as 91.1% (95% CI: 76.3–97.7). For the second-line chemotherapy, the ORRs were 13.6% (95% CI: 6.0–27.1) and 30.0% (95% CI: 14.3–52.1) in NSCLC and SCLC patients, respectively.
Treatment response of 1st-line and 2nd-line chemotherapy.

CI, confidence interval; CR, complete response; NE, not evaluable; NSCLC, non-small-cell lung cancer; PD, progressive disease; PR, partial response; SCLC, small-cell lung cancer; SD, stable disease.
Among the total 109 patients, 103 died during the observation period. Median OS times from the initiation of the first-line chemotherapy were 8.9 months (95% CI: 7.6–11.8) and 12.2 months (95% CI: 9.2–14.5) in NSCLC and SCLC patients, respectively. For the second-line chemotherapy, median OS times were 3.9 months (95% CI: 2.7–5.9) and 7.4 months (95% CI: 3.0–12.8) in NSCLC and SCLC patients, respectively. Survival curves are presented in Figure 3. According to Cox proportional hazard model, AE-ILD development after registration was not associated with an increased risk of death (hazard ratio: 0.79; 95% CI: 0.49–1.29; p = 0.35).

Survival curves after first-line chemotherapy initiation: (a) patients with non-small-cell lung cancer (NSCLC) and (b) patients with small-cell lung cancer (SCLC). Dotted lines indicate 95% confidence intervals.
Discussion
This was a multicenter prospective study reporting a series of patients with inoperable advanced lung cancer and preexisting ILD who received systemic chemotherapy. Most patients were elderly men with a heavy smoking history and had UIP/probable UIP pattern on CT. AE-ILD occurred in 23/109 patients (21.1%; 95% CI: 14.4–29.7%) after registration, which was a comparable frequency to that in previous retrospective reports for this population.4,6–9 In contrast, the efficacy profiles seemed relatively favorable, suggesting the importance of appropriate clinical management for this population.
AE-ILD has been an active research topic particularly in IPF, which is a major form of ILD, and several researchers have proposed the risk factors. In ‘idiopathic’ AE of IPF, so far, lower FVC seems to be the most evidence-rich risk factor.14,16–18 Conversely, other factors have also been reported, including lower DLco,16,18 shorter 6-min-walk distance, 16 poorer oxygenation,16,19 increased dyspnea, 16 and a prior history of AE. 17 As a point in common, importantly, the event of AE frequently occurred in patients with deteriorated IPF. In this study, although all of these parameters have not been evaluated, our finding that lower %FVC is a risk factor for AE-ILD during chemotherapy is consistent with the perception obtained by these past clinical studies. FVC can be measured noninvasively using spirometry, and the procedure is not so difficult if the patient’s general condition is expected to withstand systemic chemotherapy. Even after developing inoperable advanced lung cancer, FVC measurement would be useful not only in evaluating the ILD severity but also in estimating the risk of AE-ILD during chemotherapy.
When focusing on ‘chemotherapy-associated’ AE-ILD, several retrospective studies have reported on the candidates of risk factors, including UIP pattern on CT, 6 younger age, 6 higher CRP, 7 lower %FVC, 8 NSCLC, 8 and higher GAP index. 12 In the present prospective study, all of these parameters were evaluated for their potential as risk factors. As a result, at least in our cohort, only lower %FVC was again verified as the risk factor for AE-ILD during chemotherapy. However, unexpectedly, the odds ratio of %FVC was as small as 1.27. This suggests the limitation of this parameter probably because %FVC value can be affected by the local cancer conditions such as the presence or absence of pleural effusion, surgical resection, and atelectasis. Another parameter, if present, that can monitor the severity of ILD and is not subject to the cancer conditions would be promising to more precisely evaluate the risk of AE-ILD in patients with lung cancer.
The efficacy of chemotherapy was evaluated as a secondary outcome in this study. Because this was an observational study recruiting heterogeneous patients, each patient received different treatment regimens, which means that the efficacy data should be regarded just as reference. However, according to recent clinical trials for advanced lung cancer patients, mostly without preexisting ILD, the ORR/median OS data in the first-line chemotherapy (using cytotoxic agents with/without bevacizumab) were reported as 20.4–35%/6.0–17.1 months and 73–84.4%/9.6–12.8 months in NSCLC20–25 and SCLC patients,26,27 respectively; these are comparable with our data, and they suggest the benefit of systemic chemotherapy even in ILD patients. Consistently, the treatment response for the second-line chemotherapy in our cohort also seems comparable with historical data,28–30 although the number of our patients who received second-line chemotherapy was small. By contrast, our data indicate that AE-ILD development during chemotherapy did not significantly affect the prognosis. Collectively, this study proposes that, even for patients with preexisting ILD, systemic chemotherapy would be worth trying.
Recent clinical studies have attempted to make evidence of suitable chemotherapy regimens for patients with lung cancer with ILD. According to these studies, carboplatin plus (nab-)paclitaxel with/without bevacizumab or carboplatin plus S-1 combination chemotherapies for NSCLC31–33 and carboplatin plus etoposide combination chemotherapy for SCLC 34 seem to be acceptable regimens with low incidences of chemotherapy-associated AE-ILD, which is mostly reproduced in our cohort. To more appropriately evaluate the suitable regimens for patients with ILD, future clinical trials for this population are necessary to consider the pretreatment ILD severity including %FVC, as already adopted in recent trials,32–36 which leads to avoiding over- or underestimation of the incidence of chemotherapy-associated AE-ILD. Moreover, to decrease the overall risk of AE-ILD, prophylactic administration of anti-fibrosis agents, including pirfenidone and nintedanib, is now under discussion and may be promising.37,38 These challenges would further optimize the treatment regimens for ILD patients.
This study has several limitations. First, unfortunately, we could not recruit a planned sample number during the predetermined study period. Due to the small number of subjects, the risk for AE-ILD between regimens, particularly cytotoxic agents versus immune check point inhibitors, between chemotherapy lines, or between tumor histologies could not be compared. Second, a central review of CT findings by identical radiologists could not be performed, which might affect the reproducibility of ILD diagnosis and the extent of shadows, although all the evaluation was conducted by experienced respiratory physicians in each institution. Finally, because of the shortage of data, progression-free survival times for chemotherapy and chronological changes in clinical parameters including %FVC could not be evaluated, although both of them had been originally planned to be assessed at the protocol registration. A decline in FVC has been suggested as another risk factor for ‘idiopathic’ AE of IPF, 19 which should be validated in future studies even for the setting of chemotherapy-associated ‘triggered’ AE-ILD.
Conclusion
With a prospective design, we identified the overall incidence of chemotherapy-associated AE-ILD as 21.1% (95% CI: 14.4–29.7%), which was frequently observed in patients with lower %FVC. In contrast, as a positive result, the efficacy of chemotherapy was almost comparable with that in patients without ILD. Our results could be helpful in considering the treatment strategy for this population.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221108395 – Supplemental material for Chemotherapy for patients with advanced lung cancer with interstitial lung disease: a prospective observational study
Supplemental material, sj-docx-1-taj-10.1177_20406223221108395 for Chemotherapy for patients with advanced lung cancer with interstitial lung disease: a prospective observational study by Keigo Koda, Yasunori Enomoto, Yoichiro Aoshima, Yusuke Amano, Shinpei Kato, Hirotsugu Hasegawa, Takashi Matsui, Koshi Yokomura, Eisuke Mochizuki, Shun Matsuura, Naoki Koshimizu, Meiko Morita, Suguru Kojima, Ayano Watanabe, Yoshiyuki Oyama, Masaki Ikeda, Hideki Kusagaya, Tomohiro Uto, Jun Sato, Shiro Imokawa, Masato Kono, Dai Hashimoto, Yosuke Kamiya, Mikio Toyoshima, Kazuhiro Asada, Masako Morita, Masashi Mikamo, Hideki Yasui, Hironao Hozumi, Masato Karayama, Yuzo Suzuki, Kazuki Furuhashi, Tomoyuki Fujisawa, Noriyuki Enomoto, Yutaro Nakamura, Naoki Inui and Takafumi Suda in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
Guarantor statement: Y.E. had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
We thank Dr. Masaru Tsukui and all investigators for their kind support.
Ethics approval and consent to participate
The study protocol has been registered under number UMIN000019370 and approved by the review board of each participating institution (Hamamatsu University School of Medicine: E15-126).
Consent for publication
Written informed consents, including publication of the results, were obtained from all the participants at the registration.
Author contributions
Availability of data and material
The data sets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
