Abstract
Background:
Previously, data mining methodology was used to identify 71 patented prescriptions in Chinese patent databases, indicating that Yin-nourishing therapy (YNT) may be an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome (pSS). The purpose of this study was to investigate effects of the addition of YNT, which includes tonifying liver and kidney therapy (TLKT) and replenishing Qi and nourishing Yin therapy (RQNYT), in the treatment of pSS.
Methods:
Fourteen databases (including Chinese, English, Japanese, Korean and Latin databases) were searched to identify randomised controlled trials (RCTs) of YNT plus hydroxychloroquine (YNTPH) versus hydroxychloroquine alone in patients with pSS. Relevant publications up to June 2021 were selected. A meta-analysis and trial sequential analysis (TSA) were performed using Review Manager 5.3, Stata 14.0 and TSA 0.9 software. The quality of identified studies was assessed based on the Cochrane risk of bias tool and GRADE (grading of recommendations, assessment, development and evaluation) criteria.
Results:
We included five RCTs with a total of 345 participants. Pooled results indicated significant differences in short-term outcomes, which were assessed via European League Against Rheumatism (EULAR) Sjögren’s Syndrome Patient Reported Index (ESSPRI), EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI), tear production, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and immunoglobulin G (IgG) results when YNTPH was compared with hydroxychloroquine use alone (p < 0.05). No significant difference in salivary flow rate was reported. The most common side effect reported for both groups was gastrointestinal reaction. Sensitivity analyses suggested that heterogeneity might be ascribed to clinical methodology. Subgroup analyses revealed heterogeneities regarding salivary flow rate were eliminated. TLKT appeared to be better than RQNYT for improving salivary flow rate. TSA only supported changes in ESSPRI, ESSDAI and ESR values. For all studies, the quality of evidence was low.
Conclusion:
YNTPH may be an effective complementary therapy. Current evidence, however, is insufficient for determining whether YNTPH is more effective than hydroxychloroquine alone. Well-designed RCTs are needed to determine the role of YNT in pSS treatment.
Introduction
Primary Sjögren’s syndrome is a type of chronic inflammatory rheumatism that affects mainly middle-aged women.1,2 The syndrome is caused by lymphocyte infiltration into exocrine glands, especially lacrimal and salivary glands. 3 In addition to sicca symptoms (e.g. xerophthalmia, xerostomia), most patients in late-stage pSS suffer systemic damage such as interstitial pneumonia, myositis or kidney damage, all of which can be life-threatening. 4 Recent pSS epidemiological studies2,5,6 have shown that its pooled prevalence rate is 60.82 per 100,000 residents [95% confidence interval (CI) = 43.69–77.94] and that pSS affects 0.01–0.72% of the global population. Currently, there is no cure for the disease; therefore, the main purpose of pSS treatment is to relieve local inflammation, delay disease progression and prolong survival. 7
At present, therapeutic drugs for pSS treatment 8 include both locally acting therapies and systemic medications. The application of local and symptomatic treatments (e.g. artificial tears, cyclopentadiene thioketone, anetin trisulfide, nonsteroidal anti-inflammatory drugs and potassium preparations) can relieve the dryness and pain caused by pSS to a certain extent; however, the extent to which symptoms are alleviated is limited among patients in whom important organs are affected. Systemic medications include immunosuppressive agents and glucocorticoid therapies. Among them, hydroxychloroquine (HCQ) is prescribed widely among pSS patients in China.9,10 A systematic review 11 based on 32 studies, however, showed that the efficacy of HCQ is limited and no safety advantage was observed in the treatment of SS. Moreover, long-term HCQ use may lead to adverse reactions such as liver and kidney damage, gastrointestinal reactions and bone marrow suppression; thus, many researchers have investigated other complementary and alternative therapies.
In China, traditional Chinese medicine (TCM) as an add-on to HCQ therapy is widely utilised in the treatment of pSS.12,13 It is worth noting that TCM clinical therapy research should be designed according to syndrome differentiation. Wu et al. 14 reported that Yin-nourishing herbs combined with HCQ could reduce Fas/FasL expression levels in the salivary glands of NOD mice with dry syndrome. Furthermore, it was reported that a combination of TCM (nourishing Yin) and HCQ reduced levels of fatigue, dryness and sleep disorders in pSS patients. 15 In this study, we searched national databases for studies on any of 71 patented prescriptions associated with nourishing Yin therapy for pSS treatment (Figure 1), including replenishing Qi and nourishing Yin therapy (RQNYT) and tonifying liver and kidney therapy (TLKT). Despite the large number of patented prescriptions found in the databases, there is a lack of solid evidence demonstrating the efficacy of Yin-nourishing therapy (YNT) as a supplement of HCQ in the treatment of pSS. For this reason, we performed a meta-analysis to verify the efficacy and safety of common Yin-nourishing prescriptions in combination with HCQ in the treatment of pSS. In addition, we used trial sequential analysis (TSA) to quantify the stability of meta-analysis results and limit conclusions based on false positives caused by random errors.

The flow chart of the study design is shown.
Methods
The Preferred Reporting Item for Systematic Reviews and Meta-Analyses (PRISMA) Statement was used in the literature review process (Supplemental Table S1). 16 The registration of protocols related to this study was identified in PROSPERO, with the registration number CRD42021257118.
Search strategies
The following types of electronic databases were searched through 1 June 2021: four Chinese [China National Knowledge Infrastructure (CNKI), Chinese Bio-Medical Literature Database (CBM), China Science and Technology Journal Database (CQVIP) and Wanfang Database]; four English (PubMed, Web of Science, EMBASE and Cochrane Library); three Korean [Revised International Staging System (RISS), Korean Medical Article Database (KMbase) and DBpia Scholarly Database]; two Japanese [Citation Information by National Institute of Informatics (CiNii) and Japan Medical Abstracts Society (Igaku Chuo Zasshi, ICHUSHI-Web)] and one Latin [Latin American and Caribbean Health Science Literature Database (LILACS)] to identify trials eligible for inclusion in the meta-analysis. No language restrictions (including all Chinese, English, Japanese and Korean searches) were applied. Additional details regarding search strategies used to identify literature from above electronic databases are supplied in Supplemental Table S2.
Inclusion criteria
Participants
Participants were diagnosed according to the international classification of primary Sjögren’s syndrome, revised in 2002. 17 TCM syndrome differentiation results included Qi and Yin deficiency, or Liver and Kidney Yin deficiency.
Interventions
In the meta-analysis, the experimental group included patients who received a combination of Yin-nourishing formula and HCQ. The control group consisted of participants who received HCQ alone. All treatment was orally administered. Yin-nourishing treatments include TLKT and RQNYT.
Outcomes
Outcomes measured included European League Against Rheumatism (EULAR) Sjögren’s Syndrome Patient Reported Index (ESSPRI), EULAR Sjögren’s Syndrome Disease Activity Index (ESSDAI), Schirmer’s test, salivary flow rate, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) level, immunoglobulin G (IgG) level and adverse events (AEs).
Types of studies
Randomised controlled trials (RCTs) were considered eligible for inclusion in the assessment.
Exclusion criteria
Studies with the following characteristics were excluded from this study:
Those that included patients with overlapping secondary SS or other connective tissue diseases such as rheumatoid arthritis;
Nonclinical RCTs such as case reports, literature reviews and protocols;
Repeated published studies;
Animal experiments or cell research;
Studies for which data are not available.
Data extraction, quality assessment and evidence certainty
Data were extracted and summarised independently by two of the authors of this study in accordance with inclusion and exclusion criteria. Differences regarding the application of criteria among the two authors were addressed by a third author. The following key data were extracted from articles: first author, publication year, country, treatment groups, control groups, outcomes and further relevant information. A thorough investigation of herbal treatments was conducted using the Royal Botanic Gardens Medicinal Plant Names Service (available at: https://mpns.science.kew.org/ or http://www.theplantlist.org).
The methodological quality of selected studies was assessed independently by two authors (SL and DL) using the Cochrane Handbook for Systematic Reviews of Interventions 5.1.0 (available at: www.cochrane.de). The following items were taken into consideration: (1) random sequence generation (selection bias); (2) allocation hiding (performance bias); (3) implementer and participant blinding (attrition bias); (4) result evaluation blinding (detection bias); (5) incomplete result data (reporting bias); (6) selective reporting of results (publication bias) and (7) other bias. Bias risk associated with each item was recorded, as follows: low, unclear or high. The overall quality of each study was judged, as follows: weak, fair or good based on the number of domains considered to be low, unclear or high risk (<3, 3 or ⩾4 domains), respectively. The third reviewer (RO) appraised addressed disputes to make a final decision.
Based on the results of the meta-analysis, two independent reviewers (RO and DL) used the GRADE (grading of recommendations, assessment, development and evaluation) methodology 18 to evaluate the certainty of evidence. The third reviewer (SL) resolved disputes regarding the downgrading or upgrading of the assessment. In combination with clinical practice guidelines, evidence quality was evaluated as high, moderate, low or very low.
Strategy for data synthesis
Review Manager (version 5.3.0) and Stata (version 14.0) were used to perform the meta-analysis. After changes in continuous data pertaining to ESSPRI, ESSDAI, Schirmer’s test, salivary flow rate, ESR, CRP and IgG were evaluated and combined, the variables of values were expressed as mean difference (MD) with a confidence interval (CI) of 95%. Heterogeneity between results of selected studies was analysed using the chi-square (χ2) test (significance level set at α = 0.1). The magnitude of heterogeneity was measured along with a quantitative determination of heterogeneity, which was calculated using the I-Squared (I2) test. With I2 = 25% and 50% as cut-off margins, 0% ⩽ I2 ⩽ 25% indicated a lack of heterogeneity, 25% < I2 ⩽ 50% indicated mild heterogeneity, and I2 ⩾ 50% or p < 0.1 indicated substantial heterogeneity. Given that selected studies differed in terms of participant characteristics, random-effects models were used to address clinical differences between studies.
Subgroup analyses were performed in an effort to detect potential sources of heterogeneity among studies. Sensitivity analyses were performed to assess the robustness of combined results by removing individual studies and recalculating combined estimates. Whenever a subgroup contained more than 10 studies, it was tested for publication bias using Funnel plot and Egger’s test methods.
TSA (available at: https://ctu.dk/tsa/)19,20 is a cumulative meta-analysis method, meaning that after each study is included, an interim analysis is conducted to determine combined values. This not only corrects statistical significance but also calculates the required information size (RIS) for the determination of a conclusion to avoid random errors (false positive or false negative results) during repeated meta-analysis updates. 21 If the cumulative Z curve passed through the α-spanning and conventional boundary value (p = 0.05) before reaching the RIS, TSA statistical test results were generally considered stable. In this scenario, there would be no need to include additional samples in the analysis to verify or reject the effect of an intervention. If not, conclusions should be drawn carefully. In addition, if the cumulative Z curve crosses the futility area, expected results may be false. Meanwhile, to calculate diversity-adjusted required information size (DARIS), we set the following parameters as a priori assumptions:
the minimum relevant clinical difference (MIREDIF) or minimal clinically important improvement (MCII) from well-designed trails (if no relevant anticipated MIREDIF or MCII is found, we ½ standard deviation (SD) defined in trials was used as a parameter setting); 22
the SD of measured data;
two-sided testing with an I-type of error of 2%, 23 and a power of 90%; 24
diversity variation from the meta-analysis;
random-effects model.
Results
Search results and baseline
In total, 1795 records were searched. Of these, 501 records were excluded because they were duplicates. By considering titles and abstracts of remaining articles, 907 additional articles were excluded because they described animal experiments, were reviews, and so on. An examination of the full text of articles resulted in the exclusion of 382 that did not meet research style and method inclusion criteria. After all inclusion and exclusion criteria were applied, the meta-analysis included five eligible RCTs.25–29 The screening process is detailed in Figure 2.

A flow chart describing screening methodology is shown.
In total, 345 subjects from the five included studies were analysed. No studies mentioned numbers of cases that dropped out or were lost during follow-up. Participants were aged 27–74 years, and the average duration since disease diagnosis ranged from 3 to 7 years. There were 27 male patients and 318 female patients. Participants of the control group were prescribed either 200 mg or 400 mg oral HCQ sulphate daily, while those of the treatment group were prescribed the same daily dose of oral HCQ sulphate plus a Chinese herbal formula for nourishing Yin. The duration of treatment ranged from 1 to 3 months. Characteristics of included studies are described in Table 1 and the composition of herbal medicines prescribed in each study is presented in Table 2.
Characteristics of included studies.

AEs, adverse events; bid, bis in die; C, control; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; ESSDAI, European League Against Rheumatism Sjögren’s Syndrome Disease Activity Index; ESSPRI, European League Against Rheumatism Sjögren’s Syndrome Patient Reported Index; HCQ, hydroxychloroquine; IgG, immunoglobulin G; ms, months; SS, Sjögren’s syndrome; T, treatment; TCM, traditional Chinese medicine; tid, ter in die.
Compositions of formulas used in each of the five included studies.

Research quality assessment
According to bias risk assessment findings, all RCTs included in the meta-analysis were considered whole and of low quality. Results of the bias risk assessment are detailed in Figure 3.
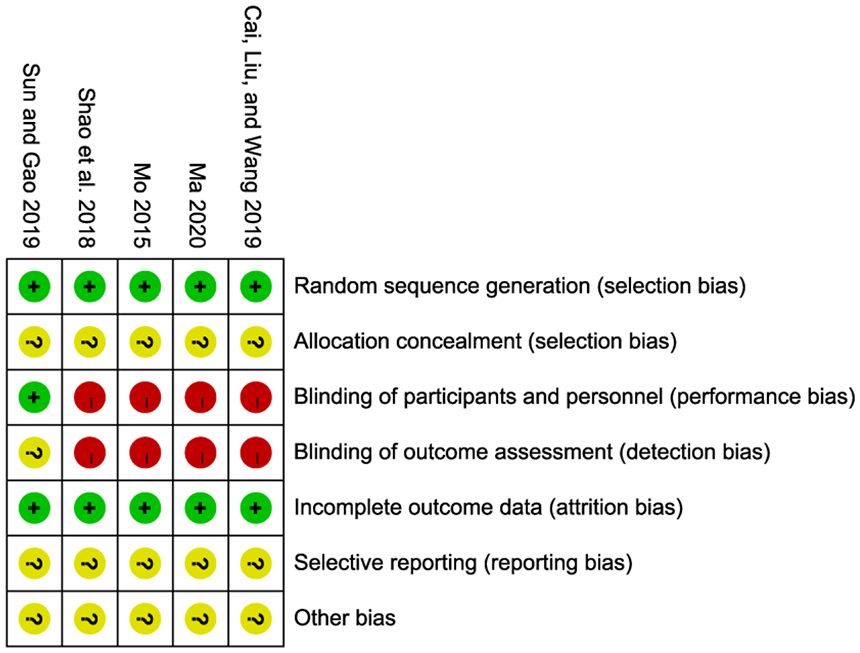
Risk of bias plot for the five included randomised controlled trials.
Meta-analysis findings
Disease activity indices
In Figure 4, data show that YNT plus hydroxychloroquine (YNTPH) administration resulted in decreased ESSPRI and ESSDAI scores versus HCQ administration alone (MD = −2.15, 95% CI = −2.60 to −1.69; p < 0.00001; I2 = 0%; fixed-effects model; two studies with 115 participants; MD = −1.76, 95% CI = −2.30 to −1.21; p < 0.00001; I2 = 0%; fixed-effects model; two studies; 115 participants, respectively).26,29
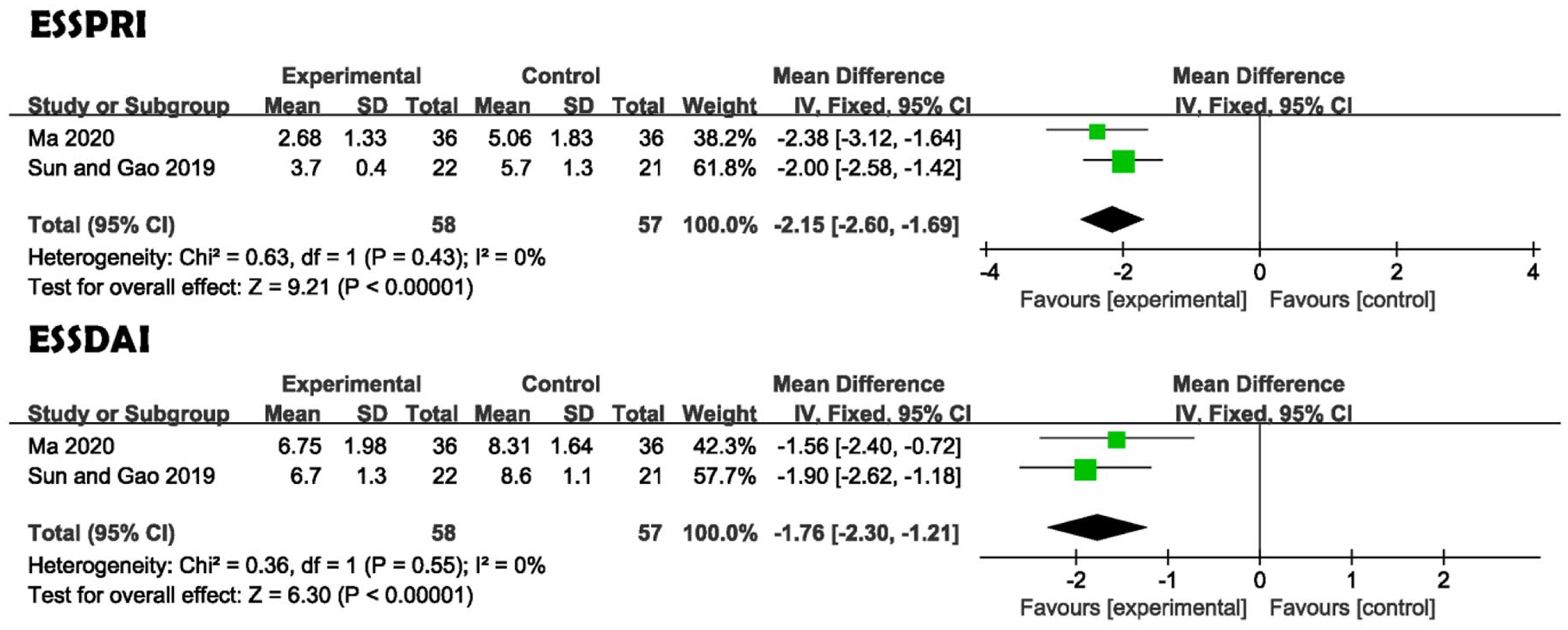
Forest plots of disease activity index.
Inflammatory marker and immunoglobulin levels
Three studies reported changes in serum ESR levels after Chinese herbal formula for Yin-nourishing treatment.26–28 The overall MD was −4.59 and the 95% CI ranged from −7.32 to −1.86 (p = 0.001), indicating that ESR was significantly reduced in the YNTPH versus HCQ group. Three trials26–28 measured changes in CRP to indicate outcomes. The pooled MD was −0.78 with a 95% CI that ranged from −1.40 to −0.16. These data suggested treatment resulted in a statistically significant decline in CRP levels in YNTPH groups. No statistically significant degree of heterogeneity was observed between the studies (I2 = 0%). Four trials estimated the effect of YNTPH versus HCQ alone on serum IgG levels.25–28 Results indicated that the YNTPH group had significantly improved serum IgG levels compared with those who were given HCQ alone (MD = −2.98, 95% CI = −5.20 to −0.77; p = 0.008; Figure 5).

Forest plots of levels of acute inflammatory reactants and immunoglobulins.
Exocrine gland secretion function
Compared with HCQ alone, the combined treatment of YNT and HCQ likely improved tear levels (MD = 3.08, 95% CI = 0.30–5.85; p = 0.03; I2 = 96%; random-effects model; four studies; 225 participants; Figure 6).26–29 No statistical between-group difference regarding salivary secretion, however, was observed (p = 0.05).

Forest plots of exocrine gland function.
Subgroup analysis and sensitivity analysis
The overall heterogeneity of IgG levels was high (I2 = 92%). Therefore, a random-effects model was applied. To reduce interference due to clinical heterogeneity, a subgroup analysis according to prescription guidelines and treatment duration of YNT was carried out. Subgroup analyses by different herbs did not reveal a significant difference with regard to effect size (p = 0.90). Subgroup analyses by treatment duration suggested clinical effects significantly differed (p < 0.00001) (Figure 7, Supplemental Table S3). Schirmer’s test also identified heterogeneity. When a meta-analysis was conducted using duration of treatment to define subgroups, a significant between-group difference was shown (p = 0.003). Changes based on TCM syndrome differentiation, however, did not significantly differ when a subgroup analysis was conducted to compare effects of TLKT and RQNYT (Figure 7, Supplemental Table S3; p = 0.26). Moreover, salivary secretion results were determined to display significant heterogeneity. Subgroup analysis results showed that it eliminated salivary flow rate heterogeneity. TLKT plus HCQ (TLKTPH) appeared to be more effective than RQNYT plus HCQ (RQNYTPH) for improving salivary flow rate (Figure 7, Supplemental Table S3). It is worth noting subgroup analyses of the three groups of indicators suggested that YNTPH mainly resulted in the short-term effects. Sensitivity analysis was performed by sequentially omitting each study. Results of the analysis failed to suggest significant variation from overall IgG level and stimulated salivary flow rate estimates. Sensitivity analysis via Schirmer’s test indicated that pooled values were less than sure-footed (Figure 8).

Subgroup analysis based on different potential sources of heterogeneity.

Sensitivity analysis in terms of Schirmer’s test, salivary flow rate and IgG.
Results of TSA
The minimal clinically important improvement (MCII) of ESSPRI scores was defined as a decrease of at least one point, or 15%. 30 Before reaching the anticipated sample (RIS = 171), the cumulative Z curve crossed the monitoring boundary for benefit, indicating the stability of results. The MCII of ESSDAI scales was −3.0 points. 30 The RIS was not renderable because the first information fraction exceeded 100% of the RIS, showing that YNT group findings were clinically significant (Figure 9). In the TSA of acute inflammatory reactant and immunoglobulin-level changes, we regarded ½ of the SD as the MCII or MIREDIF. Findings indicated that ESR results were considered stable: An assessment of CRP and IgG findings, however, demonstrated that more trials will be needed to determine YNTPQ is more effective than HCQ therapy alone (Figure 10). Regarding the improvement of the exocrine gland function, in which ½ SD was set as the MCII or MIREDIF, the TSA indicated that sample size was not large enough to show that YNT is beneficial as a supplement to HCQ because the Z curve intersected the futility area or failed to cross the conventional boundary with p value of 0.05 (Figure 11).

Trial sequential analysis of disease activity index.

Trial sequential analysis of levels of acute inflammatory reactants and immunoglobulins.

Trial sequential analysis of exocrine gland function.
Reports of adverse events
Three studies26,28,29 reported the following adverse events, which occurred in both experimental and control groups: gastrointestinal reaction, cold, blurred vision, mildly abnormal liver function, headache, increased blood pressure, rush, and so on. Adverse events were reported in seven of 94 patients (7.4%) of the experimental group and eight of 91 patients (8.8%) of the control group. One study described noted that only four cases of gastrointestinal reactions were reported during the study, but did not describe the group from which the cases were reported. 27
Publication bias and quality of evidence
As less than 10 studies were included in the meta-analysis, the publication bias of this study was not assessed. Furthermore, because the five studies included in the analysis failed to describe trial protocol registration information, and, in general, positive results are easier to publish than negative results, we were unable to evaluate the integrity of outcome indicators. In conclusion, we were unable to rule out the possibility of publication bias. In this study, the quality of evidence presented in each included trial was determined to be low or very low. Reasons for downgrading study quality included poor methodological quality, small sample size, substantial heterogeneity or a lack of statistically significant results (Supplemental Table S4).
Discussion
In China, herbal formulae have in recent decades been used as an adjuvant medical therapy for patients with pSS to relieve symptoms such as dry mouth, dry eyes and joint pain, often with promising results. 31 A previously published meta-analysis analysed the combined effect of HCQ and traditional Chinese medicine. The study revealed that the combination of drugs was more effective than HCQ alone, with improvements in saliva flow rate, ESR and CRP; furthermore, no significant differences in treatment safety were observed. 12 If the treatment group is not treated according to syndrome differentiation, the quality of evidence, however, is difficult to control. According to proponents of traditional Chinese medicine, SS is caused mainly by Qi–Yin deficiency and liver and kidney Yin deficiency.32,33 Our prior data mining findings revealed that there is a certain degree of credibility attached to the treatment of SS with Yin-nourishing herbs. Therefore, of the many different treatments and traditional Chinese medicines, we decided to focus on nourishing Yin medicine, specifically its safety and effectiveness in the treatment of pSS.
Seven outcomes were considered in this study, including disease index, serological indicators (IgG, ESR and CRP), exocrine gland secretion function and adverse events. Findings of the five RCT studies selected revealed evidence of positive effects of the administration of HCQ with Yin-nourishing Chinese herbs in the treatment of pSS, including reduced inflammation, and improved exocrine gland function. Recent studies34,35 have confirmed that the correlation between ESSDAI and objective disease change is stronger than that of other indicators of disease activity. Furthermore, ESSPRI is more accurate for evaluating subjective symptoms of patients than other disease indicators. Both ESSPRI and ESSDAI are strong and sensitive indicators of pSS disease activity and are good outcome indicators in clinical efficacy evaluations. According to our meta-analysis of ESSDAI and ESSPRI, YNTPH may confer some advantages in terms of alleviating pSS disease activity.
A prior meta-analysis 10 demonstrated that those receiving HCQ had reduced ESR levels when compared with patients of the placebo group. Findings of this study indicated that YNT synergistically affected the anti-inflammatory activity of HCQ, promoting the alleviation of acute inflammatory reactions. Our findings demonstrated that YNTPH may reduce IgG levels, suggesting that YNT synergistically enhances the immunomodulatory activity of HCQ. Due to the small sample size and insufficient information regarding other immune complexes, the result, however, may be a false positive. Previous clinical trial evidence 11 has shown no significant difference in Schirmer test results of HCQ and control groups. Our findings showed that YNTPQ improved lacrimal gland secretion to a certain extent, suggesting that YNT may slightly improve symptoms versus HCQ alone. Recently published studies10,11 have indicated that HCQ has little or no effect on salivary flow. Our results also show that effects of YNTPQ on salivary gland function seem to be limited.
In the safety assessment, there were no serious adverse events reported among four RCTs and gastrointestinal reactions were the main type of adverse events reported among the two groups. Subgroup analysis results demonstrated that different courses of treatment might determine potential heterogeneity of IgG level and exocrine gland secretion function test results. Furthermore, we found that the patients who received the course of treatment within 3 months had statistically significantly improved immunoglobulin and lacrimal and salivary gland function levels. There is a need for researchers to further investigate short-term mechanistic details of YNT. In addition, when statistics of stimulated salivary flow rate were assessed did we find evidence showing that TLKTPH may increase salivary secretion to a greater degree than RQNYTPH. In order to deeply understand the influence of sample size on the conclusion, we, however, conducted a TSA. The analysis showed that the possibility of type I errors was avoided and the sample size was sufficient for only three outcome measures: ESSPRI, ESSDAI, and ESR. Furthermore, according to Cochrane Collaboration deviation risk tool and GRADE evidence classification findings, many of the selected studies possess low-quality evidence. In other words, currently available evidence is insufficient for assessing synergistic effects of YNT and HCQ and additional high-quality RCTs are needed to verify current findings.
Overall, study findings obtained using data mining and systematic review methodology (Figure 1) suggest that Chinese herbal medicines, particularly commonly prescribed nourishing Yin, may effectively improve pSS treatment. It is hoped that findings serve as a valuable benchmark for clinical syndrome differentiation. Among them, Rehmannia glutinosa (Gaertn.) DC. and Lycium barbarum L. are commonly used to tonify the liver and kidney and Ophiopogon japonicus (Thunb.) Ker Gawl. and Astragalus Mongholicus Bunge are commonly used as Qi and Yin tonifying remedies. Recent studies have shown that catalpol in Rehmanniae Radix 36 can regulate the interaction between T cells and B cells to reduce lymphocyte infiltration, thereby improving exocrine gland function. It has been reported that Ophiopogon japonicus polysaccharide may inhibit TLR4/MyD88/NF-κB-related signalling pathways, reduce oxidative stress damage and macrophage inflammatory factor secretion and reduce inflammation caused by SS.37,38 Ophiopogonis Radix and Rehmanniae Radix are often used in combination with other therapies to treat Yin deficiency and improve fluid production, which is equivalent to increasing the levels of salivary gland secretion. Astragali Radix may promote the secretion of cytokines via astragalus polysaccharides, which stimulate the body’s immune response, and have significant anti-inflammatory and immunomodulatory effects. 39
Lycium barbarum polysaccharide, which is involved in the regulation of T-cell differentiation, inhibited the development of pSS in a murine model. 40 Other studies revealed that Dendrobium officinale polysaccharides inhibit tumour necrosis factor (TNF)-α-induced apoptosis in human salivary gland cells.41,42 Moreover, studies have indicated that Dendrobium officinale Kimura et Migo extract may regulate the expression of aquaporin protein-5 polypeptides in the labial gland of pSS patients, thus promoting salivary secretion and improving dry mouth symptoms. According to prior clinical findings, total glucosides of paeony extracted from the dried root of Paeoniae Radix Alba, when combined with an immunosuppressant, alleviate lacrimal and salivary gland secretion symptoms, and inflammatory index and immune disorder among pSS patients to a certain extent. 13 Another study showed that total glucosides of paeony regulated inflammatory factors related to intestinal movement and reduced gastrointestinal reactions in SS-mediated mice, although the study also reported mild toxicity and side effects. 43 The traditional Chinese medicines mentioned previously are commonly used and several studies that assessed pharmacological effects, animal experiments and clinical trials have indicated that effects of therapies on pSS are positive. Therefore, there is a broad consensus that Yin-nourishing Chinese medicines significantly reduce the manifestation of pSS.
Limitations and future prospects
Overall poor design of trials
First, only one of the studies included in the meta-analysis evaluated a placebo-controlled group. Furthermore, none mentioned that patients and researchers strictly double-blinded and no third-party blinding was set up. Second, studies contained few detailed descriptions of random allocation. It should be noted that many of the cited studies failed to be of good methodological quality and had small sample sizes, in particular. For example, randomness was only sometimes considered and allocation concealment was not considered in any included studies. Given that the disease is highly prevalent in women, it is perhaps understandable that the proportion of women assessed was high in many studies, some of which were included all-female cohorts. Although gender is not a statistically significant factor in this context, risk of bias remains. Furthermore, most studies did not consider internationally recognised disease activity standards, such as ESSPRI and ESSDAI. Few studies detailed YNTPH safety reports and protocols. Therefore, to improve evidence quality, future trials should address the issue of study design.
Heterogeneity of YNT
Forms of YNT considered in studies include decoctions, granules and traditional Chinese medicine preparations. Oral absorption rates and component concentrations of types of preparations differ. This might explain the high degree heterogeneity observed among test results. Although all TCM treatment schemes cited in this study are based on the dialectical treatment of YNT, Chinese herbal formulas are generally composed of multiple traditional Chinese medicines. Because each prescription of herbal medicine is not that of a single component, it is difficult to arrive at a specific assessment index and quantitative standard in the research of TCM. Importantly, different doses, dosage forms and even pharmaceutical modes of single herbs affect the overall efficacy, and therefore, heterogeneity between studies is inevitable. In addition, Chinese medicines and other traditional therapies are mainly based on syndrome differentiation. A variety of syndrome types can be summarised for the same disease and medications given for different syndrome types of the same disease are highly variable. Due to the lack of prescription standardisation, herbal composition and dosage variability may affect the interpretation of results.
We also, however, believe that the therapeutic theory of Chinese medicine is similar to the theory of synergism: The positive effects of TCM are more pronounced when the right Chinese medicine combinations, selected in accordance with certain therapeutic principles, are combined. For patients with complex forms of pSS, TCM syndrome differentiation (a kind of individualised treatment scheme) may be still needed to confirm the add-on effects of YNT. In the future, methodology for evaluating individualised Chinese medicine therapies and the establishment of real-world research strategies require further exploration. Furthermore, we may benefit from the identification of new medications via data mining of currently published herbal prescriptions, so as to simplify prescriptions included in assessments to and facilitate the formulation of standardised Chinese medicine preparations with the best curative effects.
Lack of long-term follow-up
The pSS is characterised by clinical symptoms of varying severity. Therefore, patients with pSS are advised to undergo systematic and long-term therapy, and regularly review relevant indicators to evaluate disease progress. If endocrine glands become increasingly damaged throughout pSS disease progression, symptoms of dry mouth and eyes will increase in severity, which may cause irreversible systemic damage. That is, in severe and particular cases, YNTPH may have very limited efficacy. The lack of long-term follow-up of patients and the lack of detailed case analyses of shedding limit the clinical applicability of findings. In addition, the duration for which efficacy and adverse reactions are assessed tends to be short and many studies lack detailed descriptions of findings. We suggest that studies of Chinese medicine in the treatment of pSS should include long-term follow-up observations to evaluate the long-term efficacies of therapies.
Conclusion
This article is a meta-analysis of five RCTs that evaluated YNTPH in patients with pSS. Results of these studies broadly suggest that YNTPH is likely effective for reducing levels of immune inflammation and improving exocrine gland function. The methodological quality of the studies considered, however, was relatively low. Meanwhile, TSA indicated that most of study outcomes did not meet RIS criteria, suggesting that some results may be false positives. In addition, the safety of YNTPH could not be clearly evaluated based on the included studies alone due to insufficient information provided regarding the presence of adverse events. The long-term clinical efficacy and safety of YNTPH require further confirmation via carefully designed, high-quality RCTs.
Supplemental Material
sj-doc-1-taj-10.1177_20406223221077966 – Supplemental material for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials
Supplemental material, sj-doc-1-taj-10.1177_20406223221077966 for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials by Shaowei Li, Rui Ou, Dandan Liu, Zhihuang Chen, Song Wei, Xiaohao Li, Xianxian Zhang, Yingwan Liu and Chunfu Hou in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-doc-2-taj-10.1177_20406223221077966 – Supplemental material for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials
Supplemental material, sj-doc-2-taj-10.1177_20406223221077966 for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials by Shaowei Li, Rui Ou, Dandan Liu, Zhihuang Chen, Song Wei, Xiaohao Li, Xianxian Zhang, Yingwan Liu and Chunfu Hou in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-doc-3-taj-10.1177_20406223221077966 – Supplemental material for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials
Supplemental material, sj-doc-3-taj-10.1177_20406223221077966 for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials by Shaowei Li, Rui Ou, Dandan Liu, Zhihuang Chen, Song Wei, Xiaohao Li, Xianxian Zhang, Yingwan Liu and Chunfu Hou in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-doc-4-taj-10.1177_20406223221077966 – Supplemental material for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials
Supplemental material, sj-doc-4-taj-10.1177_20406223221077966 for Is Chinese herbal formula (nourishing Yin therapy) effective and well tolerated as an adjunct medication to hydroxychloroquine in the treatment of primary Sjögren’s syndrome? A meta-analysis of randomised controlled trials by Shaowei Li, Rui Ou, Dandan Liu, Zhihuang Chen, Song Wei, Xiaohao Li, Xianxian Zhang, Yingwan Liu and Chunfu Hou in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Natural Science Foundation of China Project (81573883), Military Logistics Research Project (CLB18J039 and CGZ16C007), Science and Technology Projects of Guangdong Province (201704020160), Science and Technology Projects of Guangzhou (202102080596), and Guangdong Medical Science and Technology Research Fund Project (B2021359 and C2021117). We are extremely grateful to Vikas Narang and Malcolm Sutherland for supporting this article as the language editors.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
