Abstract
Background:
We investigated the associations between glycated hemoglobin (HbA1c) trajectories and cardiovascular outcomes using data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study.
Methods:
We used HbA1c values within the first 2 years of treatment for modeling with a latent class growth model. Groups of HbA1c trajectories were modeled separately in the standard (group 1–group 4) and intensive (group 5–group 8) treatment arms. The primary outcome in the ACCORD study was a composite of non-fatal myocardial infarction, non-fatal stroke, or death from cardiovascular causes. Effects of HbA1c trajectories on cardiovascular outcomes were analyzed using a Cox-proportional hazard model.
Results:
Baseline HbA1c levels for the eight trajectories (group 1–group 8) were 7.8 ± 0.8, 8.2 ± 0.9, 9.3 ± 1.1, 9.6 ± 1.2, 7.8 ± 0.7, 10.1 ± 0.8, 8.3 ± 0.7, and 9.5 ± 1.1%, respectively. The respective values after 2 years of treatment were 7.0 ± 0.6, 7.7 ± 0.7, 8.5 ± 0.9, 10.3 ± 1.3, 6.2 ± 0.4, 6.5 ± 0.6, 7.2 ± 0.6, and 8.5 ± 1.1%. After a median follow-up of 4.8 years, group 5 and group 6 had similar outcomes compared with group 1 (reference group). In contrast, group 3, group 4, and group 8 had higher risks of the primary composite outcome compared with group 1.
Conclusion:
HbA1c trajectory was associated with cardiovascular outcomes in type 2 diabetes patients with high cardiovascular risk.
Introduction
Type 2 diabetes (T2D) is a serious health problem with a growing prevalence worldwide. 1 People with T2D are at an increased risk of death and chronic complications,2,3 which can lead to an impaired quality of life. T2D is characterized by abnormalities in glucose homeostasis,4,5 and the degree of hyperglycemia, usually assessed by glycated hemoglobin (HbA1c), 6 has been associated with risk of chronic diabetes complications. In a prospective observational study 7 of patients with recently diagnosed T2D, each 1% reduction in HbA1c was associated with reductions in risk of 37% and 14%, respectively, for microvascular complications and myocardial infarction.
However, findings from prospective clinical trials regarding the effects of intensive glycemic control on cardiovascular outcomes in patients with T2D are inconsistent. In patients with newly diagnosed T2D, intensive glycemic control for a median of 10 years 8 led to long-term risk reduction in myocardial infarction and all-cause mortality. 9 In contrast, intensive glycemic control did not reduce the risk of composite cardiovascular endpoints and all-cause mortality in T2D patients with long disease duration.10–12 The risk of all-cause mortality was even increased significantly [hazard ratio (HR) 1.22, 95% confidence interval (CI) 1.01–1.46, p = 0.04] in the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study. 10
People with T2D are a heterogeneous population 13 and there may be considerable between-individual variations in responses to glucose-lowering treatment.14–16 Moreover, the associations of between-individual variations in response to glycemic control with cardiovascular outcomes in patients with T2D are not yet clear. Using latent class analysis 17 may help identify patients with distinct HbA1c trajectories that are different from groupings using artificial HbA1c cut-off levels. In this study, we aimed to investigate the associations between HbA1c trajectories determined using latent class analysis 17 and cardiovascular outcomes using data from the ACCORD study. 10
Methods
The study design and the primary results of the ACCORD study10,18 have been published previously (ClinicalTrials.gov identifier: NCT00000620). The purpose of the ACCORD study 10 was to determine whether major cardiovascular events in people with T2D could be prevented by intensive glycemic control (HbA1c target 6.0% versus 7.0–7.9%). The ACCORD study patients were recruited between January 2001 and October 2005, 18 and the primary results were reported in 2008 after a median follow-up of 3.5 years (until December 2007). 10 The main inclusion criteria were patients with type 2 diabetes with an HbA1c of 7.5% or more and high cardiovascular risk. The definition of high cardiovascular risk included (1) age 40–79 years with a history of cardiovascular disease, (2) age 55–79 years with evidence of atherosclerosis, albuminuria, or left ventricular hypertrophy, or (3) age 55–79 years with two or more cardiovascular risk factors (dyslipidemia, hypertension, current smoker, or obesity).10,18 The key exclusion criteria included frequent or recent events of severe hypoglycemia, unable to do home glucose monitoring or insulin injection, body mass index >45 kg/m2, serum creatinine >132.6 µmol/l (1.5 mg/dl), or other serious illness.10,18
All patients in the ACCORD study provided written informed consent. 10 Our study was a secondary analysis using data from the ACCORD study, which are available for research use (https://biolincc.nhlbi.nih.gov/studies/accord/, accessed on 19 March 2021). In this study, we aimed to investigate the associations between HbA1c trajectories and cardiovascular outcomes. Our proposal was approved by the Institutional Review Board of Taichung Veterans General Hospital, Taichung, Taiwan (approval number CE17112A). Our proposal requesting the data was approved by the National Heart, Lung, and Blood Institute in May 2017, and an extension was permitted in March 2020. Informed consent is not required for our secondary analysis since we have de-identified data approved and released by the National Heart, Lung, and Blood Institute (https://biolincc.nhlbi.nih.gov/studies/accord/).
We established HbA1c trajectories by using HbA1c values within the first 2 years of the ACCORD study (Figure 1). To avoid possible confounding on glycemic control by cardiovascular events, patients who had the composite primary outcome within the first 2 years of treatment were excluded from the analyses. Finally, a total of 9752 patients were analyzed (Figure 1). We modeled groups of HbA1c trajectories separately in the standard and intensive treatment arms using a latent class growth model.19,20 As patients may have similar HbA1c levels at baseline but quite different individual responses to treatment, this method 17 may help identify “latent” groups that are not directly observable. The number of HbA1c trajectories was decided according to Information of Bayesian Information Criterion. 21 We assigned patients to different trajectory groups by posterior probabilities from the trajectory model. The use of a censored normal model was appropriate due to the continuous outcomes of HbA1c. The number of patients in each trajectory group was set to exceed 3% of the study population.21,22 The models were fitted using the SAS ProcTraj procedure. 21 Finally, four distinct HbA1c trajectories were identified in both of the standard and intensive treatment arms (Figure 2).

Flow diagram displaying the number of study participants for analyses.

HbA1c trajectories during the first 2 years of the ACCORD study. Dotted line, HbA1c trajectories in the standard treatment arm (group 1–group 4). Solid line, HbA1c trajectories in the intensive treatment arm (group 5–group 8).
The primary outcome in the ACCORD study 10 was a composite of non-fatal myocardial infarction, non-fatal stroke, or death from cardiovascular causes. The other outcomes of interest included all-cause mortality and fatal or non-fatal heart failure. The definitions of these outcomes were reported previously. 10 Risks of these cardiovascular endpoints were compared among the HbA1c trajectory groups with adjustment of age, sex, race, body mass index, smoking, history of cardiovascular disease, chronic kidney disease, and baseline systolic blood pressure, low-density lipoprotein cholesterol and HbA1c in the multivariable analyses.
Statistical analyses
Chi-square test and one-way ANOVA were used to determine the statistical significance of the differences in categorical and continuous variables, respectively, among the HbA1c trajectory groups. Kaplan–Meier survival curves were plotted for the HbA1c trajectory groups. The effects of HbA1c trajectory on the primary composite outcome and other outcomes of interest, including all-cause mortality and fatal or non-fatal heart failure, were analyzed using a Cox-proportional hazard model. A two-sided p value of <0.05 was considered statistically significant. SAS software (Version 9.4; SAS Institute, Inc., Cary, NC, USA) was used to conduct all of the statistical analyses.
Results
Table 1 shows the baseline characteristics of the study patients in the two treatment arms by HbA1c trajectory. The baseline HbA1c levels for group 1–group 8 were 7.8 ± 0.8, 8.2 ± 0.9, 9.3 ± 1.1, 9.6 ± 1.2, 7.8 ± 0.7, 10.1 ± 0.8, 8.3 ± 0.7, and 9.5 ± 1.1%, respectively. The respective HbA1c levels after 2 years of treatment were 7.0 ± 0.6, 7.7 ± 0.7, 8.5 ± 0.9, 10.3 ± 1.3, 6.2 ± 0.4, 6.5 ± 0.6, 7.2 ± 0.6, and 8.5 ± 1.1% (Figure 1). As patients in group 1 had a mean HbA1c level around 7% during the first 2 years of study, they were used as the reference group when comparing cardiovascular endpoints risks among the HbA1c trajectory groups. Prescribed glucose-lowering drugs and hypoglycemia after randomization by HbA1c trajectory are given in Table 2. Compared with patients in the standard treatment arm (HbA1c trajectory group 1–group 4), patients in the intensive treatment arm (HbA1c trajectory group 5–group 8) had higher rates of being prescribed with oral glucose-lowering drugs and insulin, as well as higher rates of hypoglycemia.
Baseline characteristics of study subjects in the two treatment groups by HbA1c trajectory. a

Values are mean ± SD or n (%).
The eight HbA1c trajectory groups were modeled using HbA1c values in the first 2 years of treatment. Treatment targets for the standard and intensive arm were HbA1c 7.0–7.9% and <6.0%, respectively.
Baseline HbA1c level.
Overall (eight subgroups) analysis of variance.
BP, blood pressure; CKD, chronic kidney disease (eGFR <60); CVD, cardiovascular disease; eGFR, estimated glomerular filtration rate; HbA1c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; UACR, urinary albumin to creatinine ratio.
Prescribed glucose-lowering drugs and hypoglycemia after randomization by HbA1c trajectory.
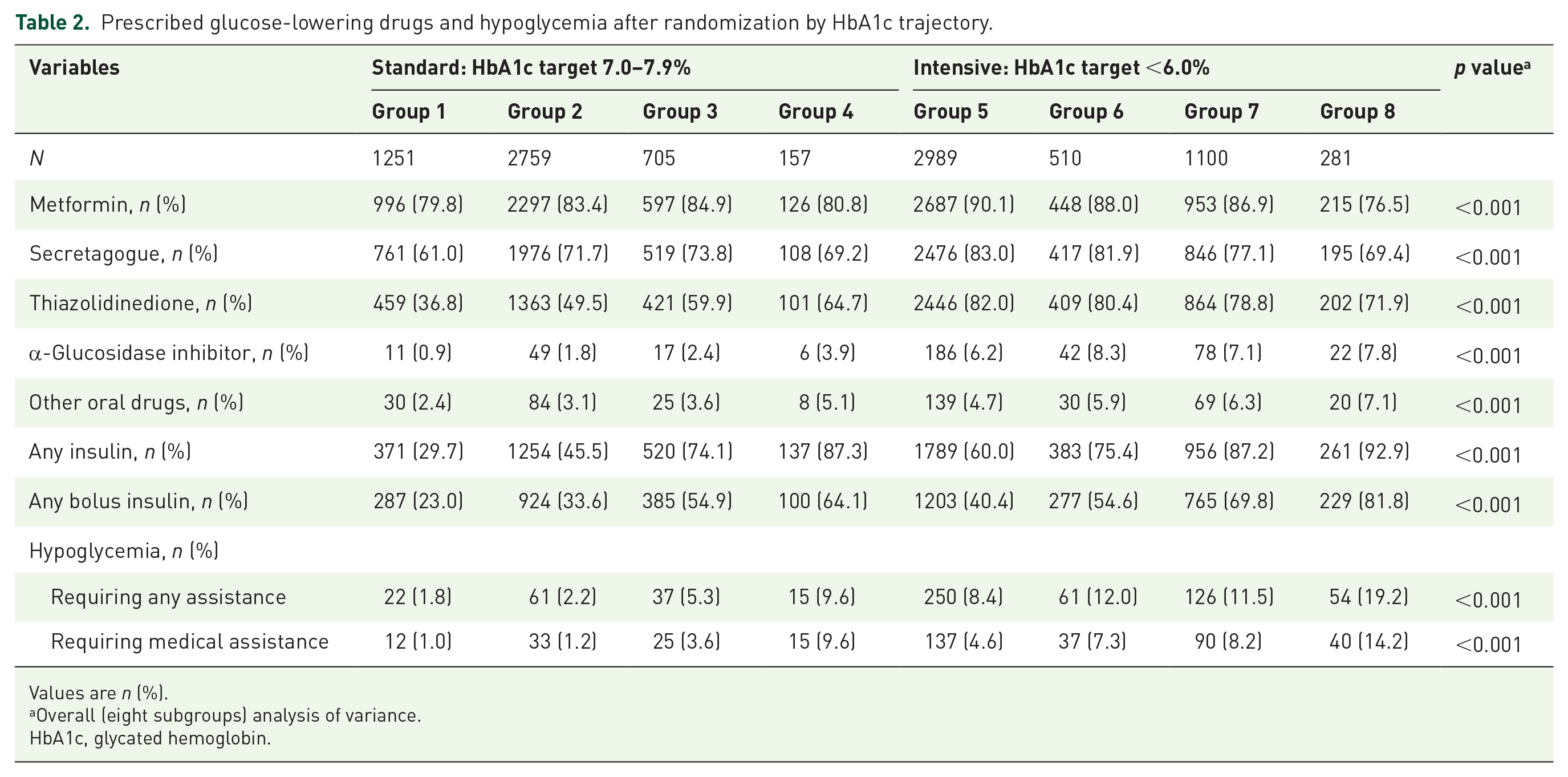
Values are n (%).
Overall (eight subgroups) analysis of variance.
HbA1c, glycated hemoglobin.
Figure 3 shows the Kaplan–Meier curves of the cumulative incidence of primary composite outcome according to HbA1c trajectory. After a median follow-up of 4.8 years, patients in group 5 (HbA1c change from 7.8 ± 0.7% at baseline to 6.2 ± 0.4% at 2 years) had the lowest incidence of primary composite outcome. In contrast, patients in group 8 (HbA1c change from 9.5 ± 1.1% at baseline to 8.5 ± 1.1% at 2 years) had the highest incidence, followed by group 3 (HbA1c change from 9.3 ± 1.1% at baseline to 8.5 ± 0.9% at 2 years) and group 4 (HbA1c change from 9.6 ± 1.2% at baseline to 10.3 ± 1.3% at 2 years).

Kaplan–Meier curves of the cumulative incidence of primary composite outcome by HbA1c trajectory.
Figure 4 shows the adjusted HR for the primary composite outcome, all-cause mortality, and primary composite outcome or death using group 1 (HbA1c change from 7.8 ± 0.8% at baseline to 7.0 ± 0.6% at 2 years) as the reference group. Only group 5 and group 6 had an HbA1c level lower than group 1 during the first 2 years of treatment (Figure 2), while there was no significant between-group difference in the risks of the primary composite outcome or all-cause mortality (Figure 4). In contrast, patients who had a mean HbA1c level higher than 8% during the first 2 years of treatment (group 3, group 4, and group 8; Figure 2) had higher risks of the primary composite outcome and all-cause mortality compared with group 1 (p < 0.05 for group 3 and group 8; Figure 4).

Adjusted HR of the primary composite outcome and all-cause mortality by HbA1c trajectory.
Figure 5 shows the adjusted HR for cardiovascular death, non-fatal myocardial infarction, non-fatal stroke, and heart failure using group 1 (HbA1c change from 7.8 ± 0.8% at baseline to 7.0 ± 0.6% at 2 years) as the reference group. Patients in group 5 had a lower risk of non-fatal stroke (HR 0.423, 95% CI 0.190–0.942, p = 0.035) compared with group 1, while there was no significant between-group difference in the risks of cardiovascular death and other outcomes (Figure 5). In contrast, patients in group 3, group 4, and group 8 had worse outcomes than those in group 1. Similar findings were observed even if we did not exclude patients who had the composite primary outcome within the first 2 years of treatment from our analyses (Supplemental material Figures 1–4 online).

Adjusted HR of individual components of the secondary outcomes by HbA1c trajectory.
Discussion
In this study, we demonstrated that HbA1c trajectory in a prospective randomized trial of glycemic control in T2D patients with high cardiovascular risk was associated with cardiovascular outcomes. Patients in group 5 and group 6 (both were in the intensive treatment arm) had the lowest mean HbA1c level during the first 2 years of treatment (mean HbA1c at 2 years 6.2 ± 0.4 and 6.5 ± 0.6%, respectively; Figure 2). Nevertheless, they had similar primary composite outcome and all-cause mortality compared with patients in group 1 (in the standard treatment arm), who had a mean HbA1c level just below 7% during the first 2 years of treatment (mean HbA1c 7.0 ± 0.6% at 2 years; Figure 2). In contrast, patients in group 3, group 4, and group 8 who had poor glycemic control despite treatment (mean HbA1c at 2 years 8.5 ± 0.9, 10.3 ± 1.3, and 8.5 ± 1.1%, respectively; Figure 2) had worse outcomes than those in group 1 (Figures 3 and 4). These findings suggest that compared with patients who had an HbA1c around 7% (group 1), patients with persistent poor glycemic control had worse outcomes, while those with a HbA1c less than 6.5% by intensive treatment had similar outcomes.
Our results suggest that glycemic response to treatment in patients with T2D was associated with patients’ outcomes. 23 Patients in group 1 with a mean HbA1c around 7% by standard treatment had similar outcomes compared with those who had a mean HbA1c less than 6.5% (group 5 and group 6) by intensive treatment. It is worth noting that both patients in group 6 (HbA1c 10.1 ± 0.8%) and group 8 (HbA1c 9.5 ± 1.1%) had significantly worse glycemic control at baseline compared with group 1 (HbA1c 7.8 ± 0.9%; Table 1). Patients in group 6 had good response to intensive treatment (HbA1c 6.5 ± 0.6% after treatment; Figure 2), and they had similar outcomes compared with patients in group 1 (Figures 4 and 5). In contrast, patients in group 8 had poor response to intensive treatment (HbA1c 8.5 ± 1.1% after treatment; Figure 2), and they had higher risks of cardiovascular events and mortality compared with patients in group 1 (Figures 4 and 5). Patients in group 5 with a mean HbA1c of 6.2 ± 0.4% at 2 years had a lower risk of non-fatal stroke (HR 0.423, 95% CI 0.190–0.942, p = 0.035; Figure 5) compared with patients in group 1. This finding was similar to a recent meta-analysis 24 which revealed a modest risk reduction in stroke by intensive glycemic control. These findings suggest that intensive glycemic control may improve outcomes in T2D patients with good response to treatment, although there may be an increase in risk of hypoglycemia.10–12,25
In contrast, poor response to glycemic treatment was associated with adverse outcomes.26–28 Compared with patients in group 1, those in group 3, group 4, and group 8 had higher risks of the primary composite outcome and all-cause mortality (p < 0.05 for group 3 and group 8; Figure 4), and some of the secondary outcomes (Figure 5). It is interesting to note that patients in group 3 (standard treatment) had a similar HbA1c trajectory (Figure 2) to those in group 8 (intensive treatment). However, patients in group 8 had a higher risk of the primary composite outcome (HR 1.439, 95% CI 0.946–2.188, p = 0.089), all-cause mortality (HR 2.576, 95% CI 1.648–4.025, p < 0.001), and cardiovascular mortality (HR 2.178, 95% CI 1.111–4.268, p = 0.023), compared with patients in group 3 (data not shown in Results). Moreover, there was a higher rate of insulin therapy (92.9% versus 74.1%) and more than triple the risk of hypoglycemia (19.2% versus 5.3%; Table 2). These findings suggest that patients with T2D who had poor response to treatment were at high risks of adverse outcomes, especially in those who had poor glycemic control with an intensive glycemic target (group 8 versus group 3). Taken together, we suggest an initial treatment target of HbA1c less than 7% for T2D patients with high cardiovascular risk. Poor response to treatment was associated with adverse outcomes (group 3 and group 4). An intensive treatment target of HbA1c less than 6.5% (or even 6.0%) may not be appropriate in those who had poor response to treatment (e.g. group 8), as the treatment may increase risk of hypoglycemia without significant benefit in cardiovascular outcomes.29,30
It is worth noting that patients in group 4 had a mean HbA1c higher than 9% during the first 2 years of treatment (Figure 2). They also had the highest rate of insulin therapy and hypoglycemia (Table 2) in the standard treatment arm. There might be some factors that contribute to the poor glycemic response to treatment and high risk of hypoglycemia in this group, such as high glucose fluctuations.31,32 All of the aforementioned factors may increase risk of adverse cardiovascular events in this group (Figures 3–5). This issue merits further investigation.
There are some limitations in this study. First, we re-analyzed the data from a prospective randomized trial 10 and stratified the patients by their HbA1c trajectories using a latent class growth model.19,20 Our method disturbed the well-balanced baseline characteristics between standard and intensive treatment arms by randomization. It is possible that some imbalanced baseline factors confounded our results, although we adjusted for age, sex, and some well-known risk factors for cardiovascular events. Nevertheless, our method identified some latent HbA1c trajectory groups 17 which may not be observed with other analytic methods. We demonstrated significant differences in cardiovascular outcomes among the HbA1c trajectory groups, despite the same HbA1c target within the standard (HbA1c target 7.0–7.9%, group 1–group 4) and the intensive (HbA1c target <6.0%, group 5–group 8) treatment arm. The between-group differences in baseline characteristics were only modest, except in glucose and HbA1c levels (Table 1). Nevertheless, patients with a significantly higher baseline glucose and HbA1c level may have similar outcomes to those in group 1 (e.g. group 6). Our findings highlight the association between glycemic response to treatment and patients’ outcomes. Second, the causal relationship between HbA1c trajectory and cardiovascular outcomes could not be confirmed by our analysis. However, the data were obtained from a prospective study. These patients were deemed to be eligible for intensive glycemic control at the study entry. Hence, it is more likely that between-individual variations in response to treatment led to different HbA1c trajectories and cardiovascular outcomes, rather than reverse causality, that is, adverse outcomes led to unfavorable HbA1c trajectories. Last, the ACCORD study 10 included T2D patients with high cardiovascular risk. Extrapolation of our results and application of these findings to other populations (such as patients with newly diagnosed T2D) should be performed with caution.
Conclusion
In conclusion, we demonstrated that HbA1c trajectories in T2D patients with high cardiovascular risk were associated with cardiovascular outcomes. We suggest an initial treatment target of HbA1c less than 7% for these patients. An intensive treatment target of HbA1c less than 6.5% (or even 6.0%) may not be appropriate in those who had poor response to treatment.
Supplemental Material
sj-tif-1-taj-10.1177_20406223211026391 – Supplemental material for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study
Supplemental material, sj-tif-1-taj-10.1177_20406223211026391 for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study by Jun-Sing Wang, Wei-Ju Liu and Chia-Lin Lee in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-2-taj-10.1177_20406223211026391 – Supplemental material for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study
Supplemental material, sj-tif-2-taj-10.1177_20406223211026391 for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study by Jun-Sing Wang, Wei-Ju Liu and Chia-Lin Lee in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-3-taj-10.1177_20406223211026391 – Supplemental material for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study
Supplemental material, sj-tif-3-taj-10.1177_20406223211026391 for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study by Jun-Sing Wang, Wei-Ju Liu and Chia-Lin Lee in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-4-taj-10.1177_20406223211026391 – Supplemental material for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study
Supplemental material, sj-tif-4-taj-10.1177_20406223211026391 for HbA1c trajectory and cardiovascular outcomes: an analysis of data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study by Jun-Sing Wang, Wei-Ju Liu and Chia-Lin Lee in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
This manuscript was prepared using ACCORD Research Materials obtained from the NHLBI Biologic Specimen and Data Repository Information Coordinating Center and does not necessarily reflect the opinions or views of the ACCORD or the NHLBI.
Author contributions
Conception and design: JSW and CLL. Acquisition of data: WJL and CLL. Analysis and interpretation of data: all authors. First draft of the manuscript: JSW and CLL. Critical revision for intellectual content: WJL. Final approval of the version to be published: all authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by research grant from Taichung Veterans General Hospital, Taichung, Taiwan (TCVGH-1083505C, 2019; TCVGH-1093504C, 2020). The funder was not involved in the study design, data collection, analysis, interpretation of the results, or preparation of the article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
