Abstract
Background:
Rituximab (RTX), a CD20 depleting agent, is a frequently used off-label treatment for multiple sclerosis (MS), while mitoxantrone (MTX) is approved, albeit rarely used for active relapsing MS (RMS). However, observational data comparing RTX and MTX effectiveness and safety are scarce.
Objective:
We aimed to compare effectiveness and safety of MTX and RTX in patients with active RMS.
Methods:
From combined retrospective clinical data of three MS centers, we selected patients who had received at least one infusion of RTX or MTX and had at least a 6-month clinical follow-up available. Treatment groups were compared by propensity score (PS)-adjusted regression and inverse PS-weighted generalized estimated equation models regarding disability progression, relapse activity, and adverse events (AEs).
Results:
We included 292 RMS patients (mean age 41.8 years, 71.6% female) who received RTX (119 patients, mean age 36.8 years, 74.8% female) or MTX (173 patients mean age 45.3 years, 69.4% female). Using both PS methods, we did not find a significant effect favoring RTX or MTX treatment regarding the probability of disability worsening or relapse occurrence. However, RTX treatment was associated with a significantly lower probability of severe AEs and AEs.
Conclusions:
RTX shows comparable effectiveness but a favorable safety profile compared with MTX in active RMS.
Introduction
Multiple sclerosis (MS) is a chronic autoimmune demyelinating disease that affects the brain and spinal cord. 1 An ever-increasing number of immunomodulatory or immunosuppressive disease-modifying drugs (DMD) have been shown to effectively reduce the number of relapses, lesion formation on magnetic resonance imaging (MRI), and, to a lesser extent, disability progression in relapsing MS (RMS). In contrast, treatment options are still limited for patients in later stages of the disease with longer disease duration and more advanced disability. 2
Based on the pathogenic role of B cells in MS, rituximab (RTX), an anti-CD20 monoclonal antibody, was tested with encouraging results in phase I and II clinical trials for RMS patients as well as in a subpopulation of primary progressive MS patients.3,4 Despite the promising data, no further clinical development and approval was sought, as patentable anti-CD20 monoclonal antibodies, ocrelizumab and ofatumumab, were approved for this indication.5–7 Nevertheless, rituximab is a frequently used off-label treatment, mostly for active RMS, including secondary progressive MS (SPMS) with superimposed relapses. Retrospective observational studies indicate a delay in disability progression with a reassuring safety profile.8–11
The cytotoxic anthracenedione mitoxantrone (MTX) is approved for treatment of active and progressive [defined as an Expanded Disability Status Scale (EDSS) score between 3.0 and 6.5] RMS. However, nowadays MTX is rarely used as its safety profile is limited by cardiotoxicity and long-term risk of developing malignancy, among other factors. 12
Given the discrepancy between label and frequency of practical use, there is a gap in knowledge regarding the effectiveness and safety profile of RTX compared with MTX in advanced RMS.
The aim of the present study was to compare the effectiveness and safety of RTX and MTX in patients with advanced RMS.
Methods
The STROBE guidelines were followed in this report. 13
Patients and definitions
The dataset was drawn from three hospital-based MS centers in Vienna, Austria (VIE-A, VIE-S) and Rostock, Germany (RO). Data were retrieved by request from the in-house pharmacy for RTX orders (time period: 1 January 2005 to 31 December 2018) (VIE-A, VIE-S) and by screening the in-house treatment documentation for MTX (time period: 13 March 1998 to 28 February 2015) (RO).
Clinical charts and patients’ files were reviewed and data retrieved, including demographic data, details of MS course (time to diagnosis, disease onset, EDSS at treatment initiation and last follow-up during therapy), DMD history, DMD switch in case of RTX/MTX termination, disease activity prior to treatment initiation (EDSS worsening, number of relapses and MRI activity within the last 2 years), and adverse events (AEs) reported in the clinical charts. AEs during RTX/MTX therapy mentioned in patient’s charts were retrieved and were divided in infusion and non-infusion-related AEs, the latter including malignancies, infections, gastrointestinal disorders, and cardiac disorders. Severe AEs (SAEs) were defined according to the Common Terminology Criteria for Adverse Events (CTCAE) V4.0 grading system, 14 grade 3–5 if detailed documentation was available; otherwise, they were considered as non-severe AEs.
Patients with RMS aged >18 years who had been diagnosed according to the respective McDonald criteria15–18 and who received at least one dose of either MTX or RTX were included in this analysis. The inclusion process is shown in Figure 1.
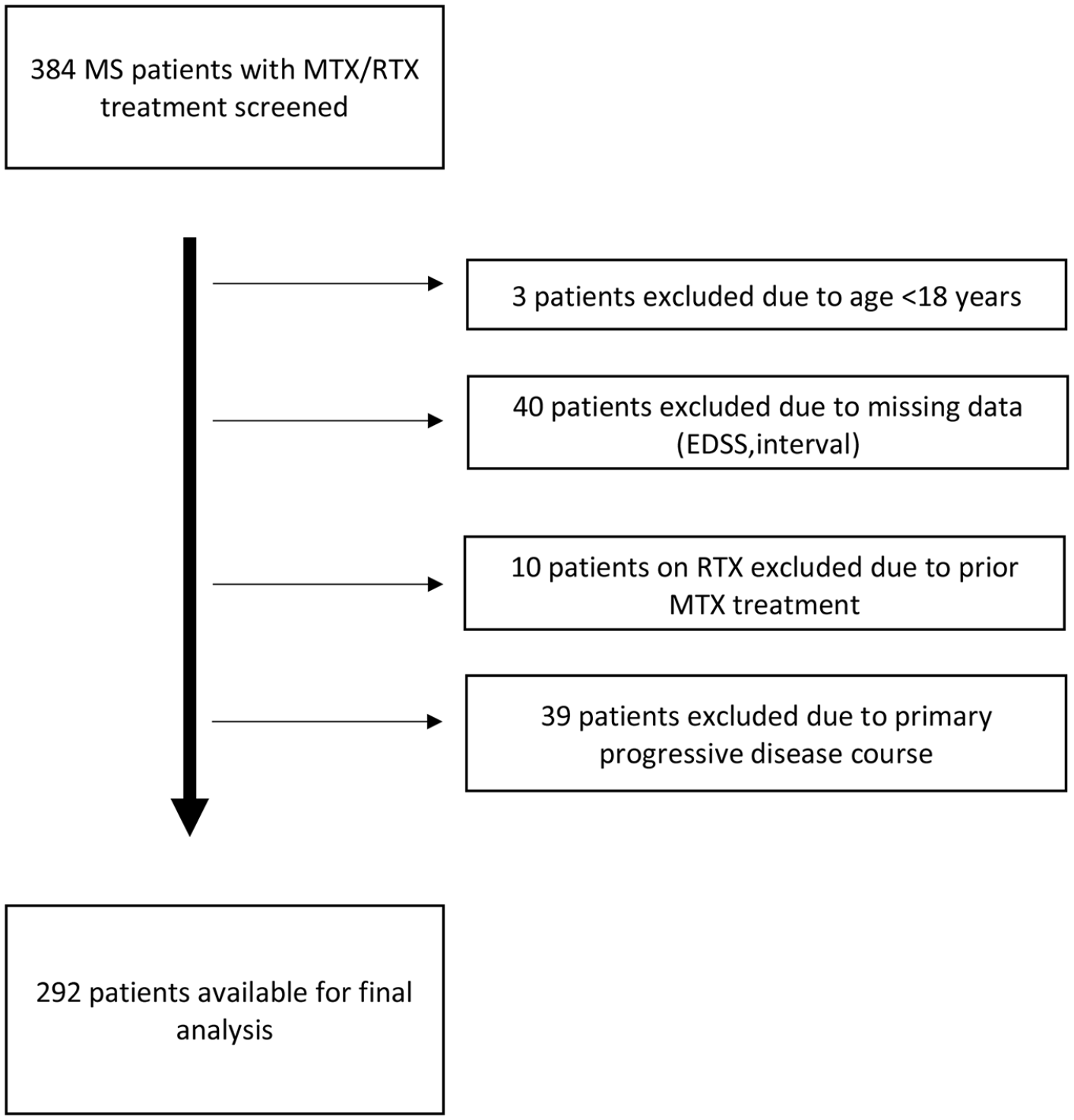
Inclusion flow chart.
Treatment protocols
RTX was administered at either 375 mg/m2 body surface area (70.6% of patients) or a total dose of 1000 mg (29.4% of patients) according to the choice of the treating physician. At treatment start, patients received the respective dose at day 0 and day 14, and afterward, treatment intervals were based on reappearance of CD19+ cells using flow cytometry monitoring (minimum 6 months, maximum 12 months). Pretreatment consisted of paracetamol, steroids, and diphenhydramine.
MTX was administered at 10–12 mg/m2 body surface area every 3 months. Upon clinical judgment, MTX was reduced to 5 mg/m2 body surface area per infusion as a maintenance dose according to the German guidelines with a maximum cumulative dose of 140 mg/m2 body surface area. 19 Pretreatment consisted of ondansetron, heparin, and a proton pump inhibitor.
Outcome
The primary effectiveness endpoint was disability worsening, defined as an EDSS increase of ⩾1.5 points when the baseline score was 0, ⩾1.0 point when the baseline score was 1–5.5, or ⩾0.5 points when the baseline score was >5.5. 20 Secondary effectiveness endpoints were annualized change in EDSS under treatment and occurrence of relapse.
The primary safety endpoint was occurrence of SEAs. Secondary safety endpoints were occurrence of malignancies, infections, cardiac AEs, and overall AEs after treatment initiation.
Ethics
The ethics committees of the Medical University Vienna and of the Rostock University Medical Center approved the study (ethical approval number: 1731/2018 and A 2014-0002). Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements and was waived by the ethics committees.
Data availability
De-identified data can be made available upon reasonable request from qualified researchers and after approval from the ethics review board at the Medical University of Vienna and Rostock University Medical Center.
Statistics
Statistical analysis was performed using R statistical software (Version 4.0.0). Univariate group comparisons were done by Chi-square test, Mann–Whitney U test, or independent t-test (with Welch’s correction in case of unequal standard deviations in the groups) as appropriate.
To control the bias for non-randomized treatment assignment, we used propensity scoring (PS)-based methods. First, individual PS was calculated using a multivariate binary logistic regression model with treatment group (RTX or MTX) as the dependent variable, including as independent variables demographic and clinical variables that had been selected on the basis of their a priori possibility of confounding treatment assignation and their availability at the time of treatment assignation: sex, age at onset, age at treatment initiation, disease duration, MS disease course [relapsing–remitting MS (RRMS) or SPMS], EDSS, number of relapses and disability worsening in the last 2 years, and most effective prior MS therapy used. Then, PS-weights were calculated by assigning patients in the MTX group a weight of 1/PS and those in the RTX group a weight of 1/(1-PS). 21
Treatment groups were compared by two separate PS-based methods in order to minimize the potential impact of model-inherent bias: PS-adjusted regression and inverse PS weighting (IPSW) generalized estimated equation (GEE) models. 22
We calculated binary logistic regression models with the primary endpoints and binary secondary endpoints as the dependent variable and treatment group as the independent variable adjusting for PS and observation time as well as correcting for clinically relevant covariates. Regarding the secondary endpoint annualized change in EDSS under treatment, we used a linear regression model set up analogously.
Also, we calculated IPSW-GEE models with the primary and secondary endpoints as dependent variables and treatment group as the independent variable, adjusted for observation time in years and clinically relevant covariates as cofactors.
For both methods, we also conducted pre-planned subgroup analyses of patients with RRMS and SPMS.
We conducted sensitivity analyses evaluating the robustness of results to Will Rogers phenomenon by separating patients according to date of diagnosis (before and after the introduction of the McDonald criteria in 2001). Robustness of the statistically significant differences to unidentified confounders not accounted for by the PS was quantified with Rosenbaum sensitivity test for Hodges–Lehmann Γ. 23
We tested all variables for normal distribution by Lilliefors test and for collinearity by variance inflation factor (VIF) and excluded all variables from the regression analysis if the VIF was >2.0, corresponding to an R2 of 0.60. Missing values were handled by multiple (20 times) imputation using the missing not at random approach with pooling of estimates according to Rubin’s rules. 24 A two-sided p-value < 0.05 was considered statistically significant.
Results
Demographics and characteristics of the whole cohort and the specific treatment groups (RTX and MTX) are shown in Table 1. As expected, there were significant imbalances between treatment groups, for example, mean age at treatment initiation [RTX 36.8 years (10.2); MTX: 45.3 (10.0)]. The multivariate logistic regression model underlying PS calculation is shown in Supplemental Table 1.
Demographic and clinical characteristics of the whole cohort and treatment groups.

Number (percentage).
Mean and standard deviation.
Median and IQR.
p-Values calculated for comparing stable and clinically progressing patients using dChi-square-test, eTwo-sample t-test, or fMann–Whitney U test as appropriate.
Available for 162 patients.
Available for 257 patients.
DMD, disease-modifying drug; EDSS, Expanded Disability Status Scale; H-DMD, defined as patients receiving one or more DMD of either cyclophosphamide, fingolimod, or natalizumab prior to initiation of RTX/MTX; M-DMD, defined as patients receiving one or more DMD of either interferon beta preparations, glatiramer acetate, dimethyl fumarate, teriflunomide, azathioprine or methotrexate prior to initiation of RTX/MTX; MS, multiple sclerosis; MTX, mitoxantrone; RRMS, relapsing–remitting MS; RTX, rituximab; SPMS, secondary progressive MS.
Effectiveness
The median treatment duration in the entire cohort was 21.5 months and ranged from 1 to 134 months. Progression of disability occurred in 82 (28.1%) patients of the entire cohort, relapses in 81 (27.7%) patients. Treatment was discontinued in 112 (38.4%) of patients.
Disability progression was observed in 21.0% and 32.9% of the analyzed patients in the RTX group and in the MTX group, respectively (p = 0.034). A relapse in the follow-up occurred in 27.4% of the patients in the RTX group and in 29.9% of the patients in the MTX group (p = 0.690). Median treatment duration was 16 months (1–90) in the RTX group compared with 31 months (1–134) in the MTX group (p < 0.001).
We found no significant difference favoring RTX or MTX treatment in terms of likelihood of disability worsening, either with PS-adjustment or IPSW. This was also true for relapse occurrence and EDSS change rates (Figure 2). Subgroup analyses of RRMS and SPMS patients also did not show significant differences between the RTX or MTX treatment groups with all confidence intervals clearly reaching over the limes of an odds ratio (OR) = 1.

Comparison of effectiveness of RTX and MTX.
Safety
In the entire cohort, 23 SAEs and 131 non-infusion-related AEs were observed, with infections (28.8%) being the most frequent ones (Table 2).
Safety of RTX and MTX.

n (%).
Chi-square test.
Available from 119 patients (=only for RTX patients) Sensitivity analyses confirmed the results of the primary analyses. According to Rosenbaum sensitivity tests for Hodges–Lehmann Γ, vulnerability of treatment group comparisons to potential unmeasured confounders was low.
AE, adverse event; MTX, mitoxantrone; n.a., not available; n.k., not known; RTX, rituximab.
SAEs were reported more frequently in the MTX group (p = 0.05) and AEs were significantly more common in the MTX group than in the RTX group (p < 0.001). Infections and cardiac side effects were more likely to occur in the MTX group (p < 0.001 each). Three patients treated with MTX were diagnosed with malignancy (myelodysplastic syndrome, acute myeloid leukemia, and renal cell carcinoma), while no malignancies occurred in the RTX group. Infusion-related reactions occurred in 21 (17.8%) of the RTX-treated patients. Those were mostly mild (nausea, flush syndrome, chills), but led to the discontinuation of treatment in four patients (3.4%).
Using both PS-adjustment and IPSW, RTX treatment was associated with a significantly lower probability of SAEs [OR: 0.32, 95% confidence interval (CI): 0.07–0.92, p = 0.023 and OR: 0.37, 95% CI: 0.09–0.64, p < 0.001, respectively] and AEs (OR: 0.06, 95% CI: 0.02–0.20, p < 0.001 and OR: 0.01, 95% CI: 0.00–0.04, p < 0.001, respectively) (Figure 3). This was further substantiated in the analysis of disease course subgroups, where both PS-based methods significantly favored RTX treatment in terms of lower probability of AEs in RRMS (OR: 0.12, 95% CI: 0.02–0.72, p = 0.020 and OR: 0.01, 95% CI: 0.00–0.07, p < 0.001, respectively) as well as in SPMS (OR: 0.02, 95% CI: 0.00–0.26, p = 0.003 and OR: 0.01, 95% CI: 0.00–0.05, p < 0.001, respectively). With regard to the probability of SAEs, RTX was associated with a significantly lower probability in SPMS (OR: 0.16, 95% CI: 0.01–0.97, p = 0.048 and OR: 0.06, 95% CI: 0.01–0.48, p = 0.008), while no statistically significant differences were seen in RRMS.

Comparison of safety of RTX and MTX.
Discussion
Considering that (i) B-cell depletion by RTX is frequently used off-label in active RMS, and (ii) MTX is an effective and approved DMD for active and/or progressive RMS/SPMS but rarely used due to its safety profile, we aimed here to compare the effectiveness and safety of RTX and MTX in a retrospective three-center real-world cohort.
The main finding of this study is that we did not find a significant difference in the effectiveness of RTX versus MTX in terms of disability progression, relapses or changes on EDSS; however, RTX was associated with significantly fewer SAEs and AEs overall.
The effectiveness of MTX has been demonstrated in both randomized controlled trials (RCTs) and retrospective observational studies. The largest phase III study (MIMS) in 194 RRMS and SPMS patients comparing MTX with placebo showed a significant reduction in both relapse rate (66%; p = 0.001) and disability worsening (64%; p = 0.036). 25 A Cochrane review including 221 RRMS and SPMS patients from three RCTs indicated a significantly increased likelihood of remaining free of relapse (OR 2.8, p < 0.001) and disability progression (OR 3.3; p = 0.04) at 2 years follow-up compared with placebo. 26 Subgroup analyses suggest that MTX is most effective in RRMS and SPMS patients with active disability progression, 27 a predominant feature of our cohort.
RTX has been successfully tested in phase I and phase II clinical trials, demonstrating a significant reduction in lower annualized relapse rate (56%; p = 0.04) and gadolinium-enhancing lesions (91%; p < 0.001) at 24 weeks compared with placebo.3,28 Still, regulatory approval was never sought, not least because newer and thus patentable B-cell depleting agents were already in the pipeline, and expensive clinical developmental programs were not continued.3,4,28
Retrospective observational studies already indicated RTX to be effective in reducing relapse rate, disability progression, and MRI activity without major safety problems. Notwithstanding, these studies were limited by lacking control groups or small sample size.8,9,29 Noteworthy, a recently published retrospective PS-matched study showed a reduction of disability progression treated with RTX compared with no treatment specifically in SPMS with and without relapses (HR: 0.49; p = 0.03). 10 As a novel finding, our study adds evidence that RTX is comparable in all relevant clinical parameters of effectiveness to a potent active comparator (MTX), in a cohort comprising active RMS and SPMS.
Looking at safety, AEs occurred in more than half of the patients receiving MTX, with almost one in 10 patients experiencing SAEs. This is within the known spectrum and frequency of AEs such as toxic cardiomyopathy.12,30 Non-infusion-related AEs in RTX-treated patients were observed in a similar frequency as reported in previous studies, with infections being the most prominent.8,10,31
Thus, in line with the general use of RTX as an off-label treatment option for patients with MS, our data add to the available literature that RTX is a relatively safe treatment in MS. In contrast, although effective, MTX has a rather unfavorable benefit–risk ratio and should only be considered after careful consideration of patients’ individual benefit–risk profile and in the absence of alternatives. Under these circumstances, a MTX dosing regimen with a more balanced risk/benefit ratio, such as the French protocol (8 mg/m2), might be preferred. 32
Limitations
The retrospective analysis of data collected in clinical routine creates a variety of possible biases: Most important is the inherent bias of indication, as the choice of treatment is not random and is determined by the characteristics and preferences of patients and the prescribing practices of clinicians. 22 This bias has been effectively controlled using PS explaining 83% of variation in treatment group allocation (Supplemental Table 1). Also, different frequencies of clinical visits and differing periods of follow-up might induce detection bias and attrition bias, respectively. The latter was corrected by the inclusion of follow-up duration in the analyses. As data were collected retrospectively, potentially causing reporting bias, we have considered SAEs as such only if there is clear documentation, and otherwise classified them as AEs. While this might have resulted in underrepresentation of SAEs, AE rates are consistent with the published data.8,10,30,31 However, safety profiles are in general not comprehensively captured in retrospective studies. Varying disease durations at baseline may cause an immortal time bias, which was mitigated by adjusting for including disease duration in the PS estimation and in the regression models. Evolving diagnostic MS criteria might induce Will Rogers phenomenon. However, sensitivity analyses for the era before and after the introduction of the McDonald criteria did not show significant differences, probably because the growing sensitivity of diagnostic criteria does have little effect in cohorts of highly active and physically affected patients. Further, we did not have sufficient data for analyzing MRI data, which reduces sensitivity to detect differences in effectiveness. Finally, MRI lesion burden would typically influence treatment choice and could create unidentified bias. Still, Rosenbaum bounds did indicate only a small potential impact of hidden bias not accounted for by the PS.
In summary, RTX has comparable effectiveness in active RMS but a more favorable safety profile compared with MTX. MTX should only be used in rare and exceptional cases, while RTX is a treatment option worth considering on an individualized basis.
Supplemental Material
sj-pdf-1-taj-10.1177_20406223211024366 – Supplemental material for Rituximab versus mitoxantrone: comparing effectiveness and safety in advanced relapsing multiple sclerosis
Supplemental material, sj-pdf-1-taj-10.1177_20406223211024366 for Rituximab versus mitoxantrone: comparing effectiveness and safety in advanced relapsing multiple sclerosis by Tobias Zrzavy, Esther Daniels, Niklas Stuka, Dennis Weber, Alexander Winkelmann, Helmut Rauschka, Michael Hecker, Fahmy Aboulenein-Djamshidian, Stefanie Meister, Fritz Leutmezer, Thomas Berger, Gabriel Bsteh, Uwe Klaus Zettl and Paulus Rommer in Therapeutic Advances in Chronic Disease
Footnotes
Conflict of interest statement
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
