Abstract
Aims:
CD4+ T cells play crucial roles as both mediators and regulators of the pathogenesis of rheumatoid arthritis (RA). However, the characteristics of CD4+ T cell subpopulations in the earliest stage of RA development remain unclear. Hence, we determined the proportions and absolute counts of circulating CD4+ T cell subsets in patients with seropositive undifferentiated arthritis (SUA), the early and preclinical stage of RA.
Methods:
Peripheral blood samples and clinical information were collected from 177 patients with SUA, 104 patients with RA, and 120 healthy controls. All patients were newly diagnosed and untreated. Proportions and absolute counts of CD4+ T cell subpopulations were determined by flow cytometric analysis.
Results:
In patients with SUA, percentages and absolute counts of circulating regulatory T (Treg) cells were decreased significantly and Th17/Treg cell ratios were abnormally increased, whereas Th17 cell numbers were similar to those in healthy controls. In addition, sex-based differences in circulating Treg cells were observed, with female SUA patients having lower proportions and absolute counts of Treg cells than those in males. Moreover, female patients with SUA had higher erythrocyte sedimentation rates and 28-joint Disease Activity Scores than those in males.
Conclusion:
Immune tolerance deficiency resulting from an abnormal reduction in circulating Treg cells might be the most crucial immunological event in the earliest stage of RA. The sex-specific disparity in Treg cells should also be considered for immunoregulatory and preventive strategies targeting early RA.
Keywords
Introduction
Rheumatoid arthritis (RA) is a T cell-mediated autoimmune and inflammatory disease characterized by systemic inflammation, autoantibody production, and persistent synovitis, eventually resulting in joint degradation and disability. 1 Although the precise etiology of this disease remains unknown, it is likely that immune dysregulation, including the excessive activation of self-reactive effector T cells and the breakdown of self-tolerance, is the basis for the development of RA. 2 CD4+ T cells are central mediators of autoimmune pathology. 3 As predominantly identified CD4+ T cell subpopulations, the roles of helper T (Th) 1, Th2, Th17, and regulatory T (Treg) cells have been investigated extensively in immune-mediated diseases. 4 Th1, Th2, and Th17 cells are the leading effector cells in autoimmune and inflammatory diseases,5–7 whereas Treg cells suppress the autoreactive activities of effector CD4+ T cells and thus maintain immune tolerance. 8 CD4+ T cell subset quantification provides insight into the immune status of RA and has been demonstrated to be an important T cell biomarker. 9 However, the characteristics of CD4+ T cell subpopulations in the earliest stage of RA development remain unclear.
To address this knowledge gap, we determined the proportions and absolute counts of circulating CD4+ T cell subsets in patients with seropositive undifferentiated arthritis (SUA), the early and preclinical stage of RA. 10 We also compared the differences in CD4+ T cell subsets between patients with newly diagnosed, treatment-naive RA and healthy controls (HCs). SUA is characterized by the presence of RA-associated autoantibodies [anti-cyclic citrullinated peptide antibodies (anti-CCP) and/or rheumatoid factor (RF)] and by painful and tender joints, with or without oligo/polyarthritis, but not fulfilling the American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) 2010 RA criteria. 11 By exploring such characteristics in patients with SUA, we anticipate that it will eventually become possible to identify immunological predictors in the earliest stage of the disease, enabling more personalized preventive treatments for individuals who are at a high risk of developing RA.
Materials and methods
Study subjects
A total of 177 patients with SUA and 104 patients with RA were recruited for this study at the rheumatology outpatient clinic of the Second Hospital of Shanxi Medical University between January 2019 and June 2020. All patients were newly diagnosed and treatment-naive. Undifferentiated arthritis (UA) was defined as patients not fulfilling the 2010 ACR/EULAR RA criteria and who did not have a clinical diagnosis other than RA at baseline. 11 Seropositive status was defined as positive according to anti-CCP and/or RF (as described below). All patients diagnosed with RA fulfilled the ACR 1987 or 2010 criteria.11,12
Data on the clinical characteristics of patients with SUA and RA were collected retrospectively from their medical records, including demographics, disease characteristics, swollen joint counts (SJCs), tender joint counts (TJCs), erythrocyte sedimentation rate (ESR, mm/h), and C-reactive protein (CRP, mg/l). We also calculated the 28-joint Disease Activity Score (DAS28) based on 28-SJC, 28-TJC, and ESR values. Blood samples were drawn from all patients at the time of SUA or RA diagnosis before the start of treatment. In addition, 120 HCs, who had no infections, no recent vaccinations, and no use of immunosuppressive drugs, were included for a blood sample draw. All samples were analyzed at the Laboratory of Rheumatology of our hospital by the same staff to minimize variability.
Analysis of lymphocyte subsets by flow cytometry
Absolute counts and percentages of lymphocyte subsets were determined by flow cytometry (FACS Calibur, BD Biosciences, San Jose, CA, USA) in EDTA anti-coagulated whole blood. Quantification beads (Trucount, BD) in combination with peridinin chlorophyll protein (PerpCP)-conjugated CD45, fluorescein isothiocyanate (FITC)-conjugated CD3, phycoerythrin (PE)-conjugated CD8, and allophycocyanin (APC)-conjugated CD4 were used to measure the absolute counts and percentages of CD3+/CD4+/CD8+ T lymphocytes according to the manufacturer’s instructions. The percentages and absolute counts of CD3+ T cells, CD3+CD4+ T cells, and CD3+CD8+ T cells were automatically calculated using BD Multitest software (BD Biosciences).
CD4+ T cells are typically categorized by the expression of surface molecules and intracellular proteins that reflect functionally distinct cell subtypes, as described previously in detail, 13 as follows: FITC-CD4 and APC-interferon (IFN)-γ (intracellular staining) for Th1 cell analysis; FITC-CD4 and PE-interleukin (IL)-4 (intracellular staining) for Th2 cell analysis; FITC-CD4 and PE-IL-17A (intracellular staining) for Th17 cell analysis; and FITC-CD4, APC-CD25, and PE-Foxp3 (intracellular staining) for Treg cell analysis. The percentages of CD4+ T cell subpopulations were calculated automatically using BD Multitest software (BD Biosciences), and the absolute counts were calculated using the percentages of CD4+ T cell subsets and the absolute counts of CD3+CD4+ T cells. All immunofluorescence antibodies were purchased from BD Biosciences, and the results are expressed as percentages of the parental lineage gate and as absolute counts (cells/µl). Gating strategies were described previously. 14
Analysis of cytokines by flow cytometry
The levels of serum IL-2, IL-4, IL-6, IL-10, IL-17, IFN-γ, and tumor necrosis factor-α (TNF-α) were determined by magnetic bead-based multiplex immunofluorescence assays using the Human Th1/Th2/Th17 Subpopulation Test Kit (Jiangxi Cellgene Biotech Co., Ltd., China). Data on cytokines were acquired using the Bio-Plex 200 reader and Bio-Plex Manager software, which provide output in terms of concentrations (pg/ml).
Autoantibody measurements
The serum levels of total immunoglobulin (Ig) RF were determined by turbidimetry with a modular analyzer (Roche, Mannheim, Germany) and a cut-off level for RF-seropositivity ⩾20 IU/ml. IgG anti-CCP levels were measured by a fluorescent enzyme immunoassay using the Addcare ELISA400 system (Addcare Laboratory Systems, Yantai Biotech Co., Ltd., Shandong, China) with a cut-off level for anti-CCP seropositivity of ⩾25 RU/ml.
Statistical analysis
Statistical analysis was performed with SPSS 20.0 statistical software (SPSS Inc., Cary, NC, USA). Data are expressed as the number of cases for category-based variables, as well as the median and 25th and 75th percentiles [interquartile range (IQR)] for continuous variables with non-normal distributions. The differences between groups were statistically analyzed by the χ2 test or the non-parametric Wilcoxon rank-sum test as appropriate. Spearman correlation coefficients were used for correlation analysis. A p value of <0.05 (two-sided) was considered statistically significant.
Results
Clinical and serological characteristics of patients with SUA and RA
Clinical and serological characteristics of patients with SUA (n = 177) and RA (n = 104), including age, sex, disease duration, ESR, CRP, DAS28 score, and autoantibodies, are summarized in Table 1. There were no significant differences in the distributions of age and sex between the HCs and patients with SUA or RA. In addition, the median disease duration was not significantly different between patients with SUA and RA. However, the median values of ESR, CRP, and DAS28 scores in SUA patients were significantly lower than those in RA patients. Moreover, the median serum levels of RF and anti-CCP antibodies were significantly increased in RA patients when compared with those in SUA patients.
Clinical and serological characteristics of patients with SUA and RA and HCs.

The chi-squared test was performed for categorical variables. The Wilcoxon rank-sum test was performed for quantitative variables with non-normal distributions.
p < 0.01, #p < 0.05 versus patients with SUA.
Anti-CCP antibody, anti-cyclic citrullinated peptide antibody; CRP, C-reactive protein; DAS28, 28-joint disease activity score; ESR, erythrocyte sedimentation rate; HCs, healthy controls; IQR, interquartile range; IU, international units; RA, rheumatoid arthritis; RF, rheumatoid factor; SUA, seropositive undifferentiated arthritis.
Decreased numbers of circulating Treg cells and increased Th17/Treg cell ratios in patients with SUA
As displayed in Figure 1, the proportions and absolute counts of circulating Treg cells were decreased and the Th17/Treg cell ratios were increased in SUA patients as compared with those in HCs. However, there was no significant difference in the levels of circulating Th17 cells between HCs and SUA patients, indicating that a reduction in Treg cells might be the leading cause of the Th17/Treg imbalance in SUA patients. Moreover, abnormally increased Th1 cell numbers and decreased Th2 cell numbers were observed in SUA patients, and the differences were significant. Additionally, the Th1/Th2 cell ratios were significantly elevated in SUA patients. Similar to those observed in SUA patients, the frequencies and absolute counts of circulating Treg and Th2 cells were decreased, and the ratios of Th1/Th2 and Th17/Treg cells were increased in RA patients when compared with those of HCs. Although the distributions of Th1 cells were higher in RA patients than those of HCs, the differences were not statistically significant.
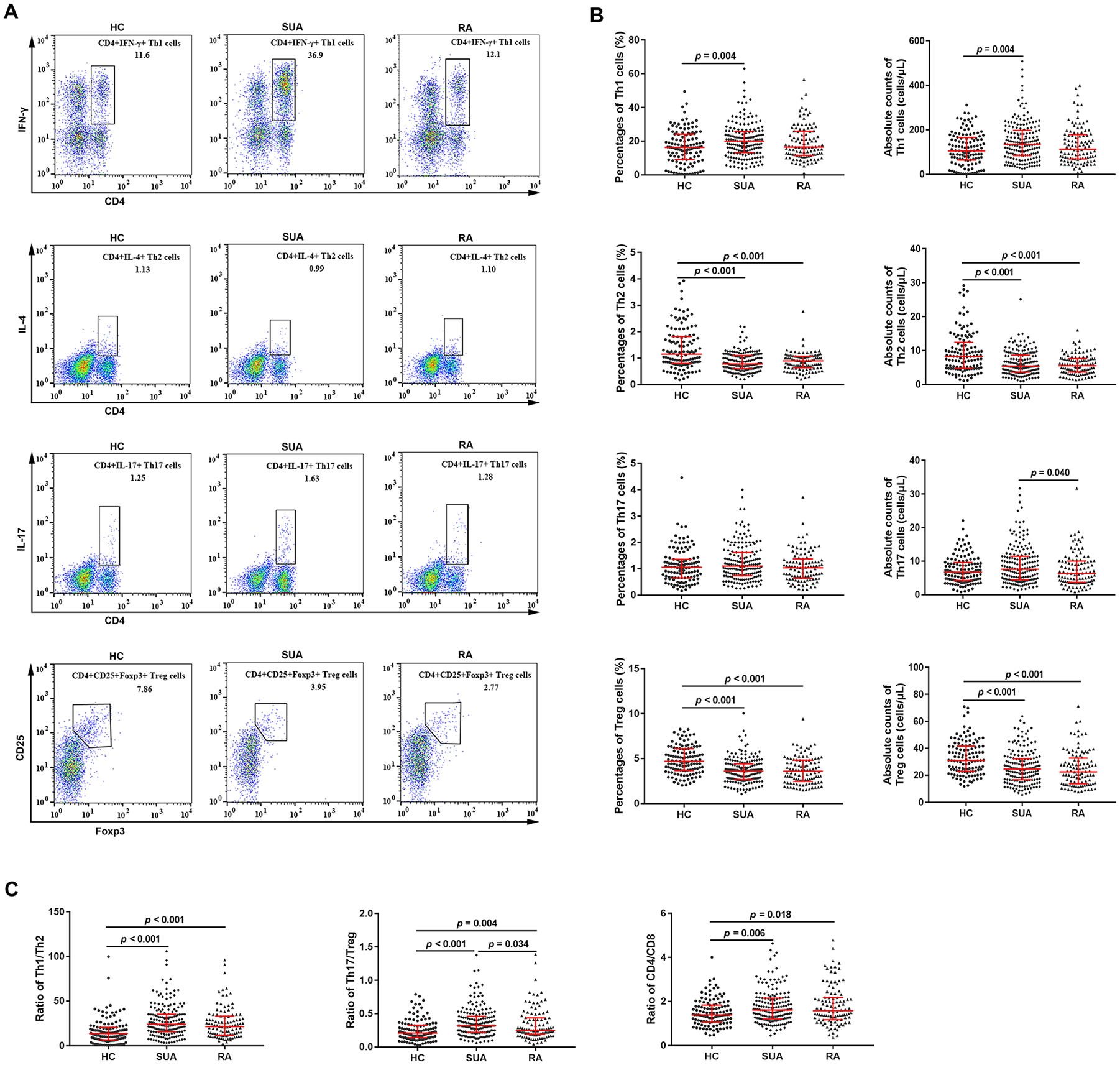
Analysis of circulating Th1, Th2, Th17, and Treg cells, and the ratios of Th1/Th2, Th17/Treg, and CD4/CD8 cells in HCs, and in patients with SUA and RA. (A) Three representative FACS analysis of Th1, Th2, Th17, and Treg cells gated on CD4+ T cells in HCs and patients with SUA and RA. (B) Percentages and absolute counts of Th1, Th2, Th17, and Treg cells in HCs and patients with SUA and RA. (C) Th1/Th2, Th17/Treg, and CD4/CD8 ratios in HCs and patients with SUA and RA. Red bars and errors: median and 25th and 75th percentiles. Statistics: Wilcoxon rank-sum test.
Abnormal numbers of circulating CD4+ T and CD8+ T cells and increased ratios of CD4/CD8 cells in patients with SUA
We also analyzed the characteristics of T lymphocyte subsets in patients and HCs. As shown in Figure 2, the circulating CD4+ T cell frequencies were higher and the CD8+ T cell absolute counts were lower in SUA patients compared with those in HCs, and the same differences were found in RA patients. Additionally, the absolute counts of T cells were significantly decreased in patients with RA. Moreover, the CD4/CD8 ratios were higher in patients with both SUA and RA than in HCs (Figure 1).

Analysis of circulating T, CD4+ T, and CD8+ T cells in HCs and patients with SUA and RA. (A) Three representative FACS analysis of T, CD4+ T, and CD8+ T cells gated on CD45+ lymphocytes in HCs and patients with SUA and RA. (B) Percentages and absolute counts of T, CD4+ T, and CD8+ T cells in HCs and patients with SUA and RA. Red bars and errors: median and 25th and 75th percentiles. Statistics: Wilcoxon rank-sum test.
Increased serum concentrations of inflammatory cytokines in patients with SUA
RA is a chronic and systemic inflammatory disease, but the status of inflammatory cytokines in the early stage of the disease is not clear. Among all patients, there were 89 patients with SUA and 55 patients with RA for whom serum cytokines were determined. Thus, we analyzed the differences in multiple serum cytokines between patients and HCs (n = 72). As shown in Figure 3, the serum levels of IL-2, IL-4, IL-6, IL-10, IL-17, TNF-α, and IFN-γ were significantly increased in SUA patients when compared with those in HCs (all p < 0.001), and the same differences were observed in RA patients (all p < 0.001). Moreover, the levels of IL-6 and TNF-α in RA patients were significantly higher than those in SUA patients (p < 0.001 and p < 0.05, respectively).
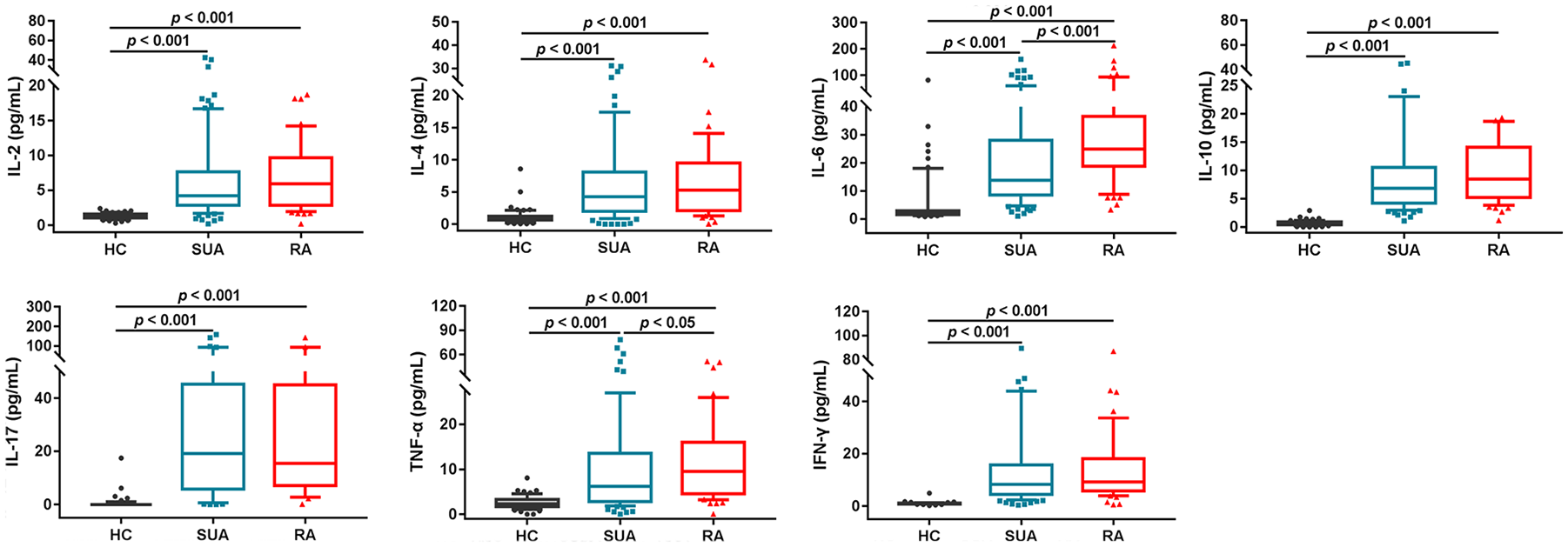
Analysis of serum IL-2, IL-4, IL-6, IL-10, IL-17, TNF-α, and IFN-γ levels in HCs and patients with SUA and RA. Statistics: Wilcoxon rank-sum test.
Correlation of clinical parameters with various circulating lymphocyte subpopulations or serum cytokines in patients with SUA
We next analyzed the correlations between clinical parameters and immunological indicators in patients with SUA. We found negative correlations between duration of disease and Treg cell percentages (r = −0.345, p < 0.001) or Treg cell absolute counts (r = −0.214, p = 0.004), and weak positive correlations between disease duration and CD4+ T cell percentages or T cell proportions (r = 0.220, p = 0.003; r = 0.182, p = 0.016, respectively). In addition, correlations between disease activity (ESR, CRP, or DAS28) and immune cell subtypes (Th1 or Th17 cell numbers) were significantly weak and negative. The serum concentrations of RF were correlated negatively with Th17 cell proportions or the Th17/Treg ratio (r = −0.153, p = 0.041; r = −0.151, p = 0.036, respectively). However, there were no significant correlations between serum concentrations of anti-CCP antibodies and T cell subsets. Moreover, the concentrations of RF were correlated positively with IL-2, IL-4, IL-6, IL-10, IFN-γ, and TNF-α levels (r = 0.302, p = 0.004; r = 0.281, p = 0.008; r = 0.237, p = 0.025; r = 0.315, p = 0.003; r = 0.344, p = 0.001; r = 0.340, p = 0.001, respectively). Correlations between CRP and IL-6 were moderately significant and positive (r = 0.446, p = 0.004) (Figure 4A). There was no significant correlation between the levels of anti-CCP antibodies and multiple cytokines.
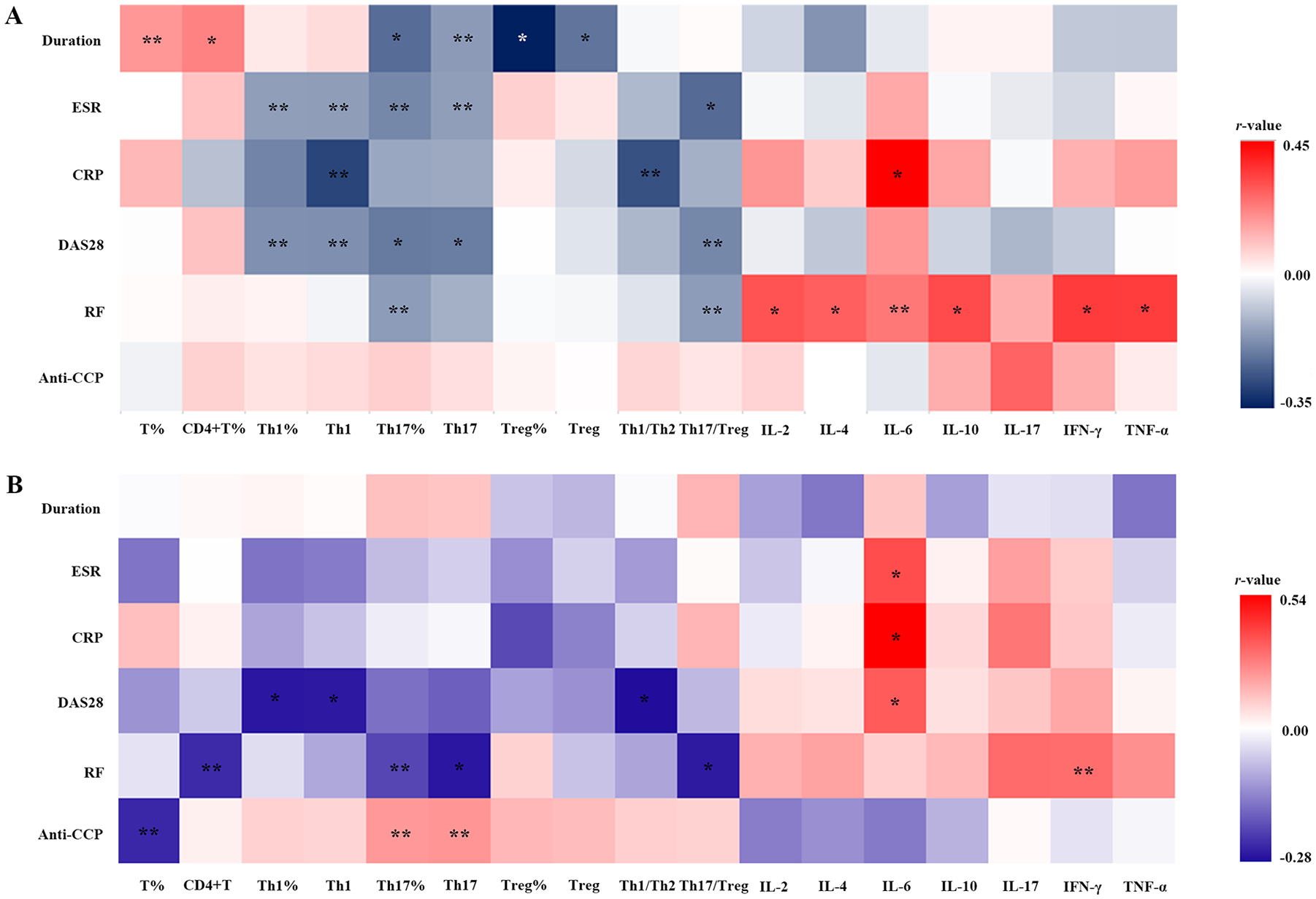
Heatmaps of correlations between clinical parameters and immunological indicators. (A) Correlation of clinical parameters against circulating immune cell subpopulations and serum inflammatory cytokines in patients with SUA. (B) Correlation of clinical parameters against circulating immune cell subsets and serum inflammatory cytokines in patients with RA.
The correlations between clinical parameters and immunological indicators in patients with RA were also determined. As shown in Figure 4B, correlations between the DAS28 score and Th1 proportions, absolute counts, and Th1/Th2 ratio were significantly weak and negative (r = –0.259, p = 0.008; r = –0.257, p = 0.009; r = –0.271, p = 0.005, respectively). The serum concentrations of RF were correlated negatively with CD4+ T counts, Th17 proportions and counts, and the Th17/Treg ratio (r = –0.237, p = 0.016; r = –0.206, p = 0.036; r = –0.259, p = 0.008; r = –0.255, p = 0.009, respectively). The serum concentrations of anti-CCP antibodies were correlated negatively with T cell percentages (r = –0.240, p = 0.014), but positively with Th17 proportions and counts (r = 0.216, p = 0.028; r = 0.224, p = 0.022, respectively). Furthermore, positive correlations were found between disease activity (ESR, CRP, or DAS28) and serum IL-6 concentrations (r = 0.377, p = 0.005; r = 0.538, p = 0.001; r = 0.347, p = 0.010, respectively). The concentrations of RF were also correlated positively with IFN-γ (r = 0.310, p = 0.021) (Figure 4B). There was no significant correlation between anti-CCP antibodies and multiple cytokines in RA patients.
Sex-based disparities in circulating Treg cells in patients with SUA
RA is a progressive autoimmune disease that is more common in women than in men. In line with this general statistic, more female SUA patients were enrolled in the present study (130 female patients versus 47 male patients). Thus, the sex-specific characteristics of various T cell subtypes were determined. As shown in Figure 5A, the absolute counts and proportions of Treg cells were decreased significantly in female SUA patients as compared with those in males (median percentage: 3.27 versus 3.75%, Z = −1.991, p = 0.046; median absolute counts: 22.64 versus 29.73 cells/μl, Z = −2.647, p = 0.008). There were no sex-based differences in circulating Th1, Th2, and Th17 cells in SUA patients (data not shown). Moreover, there were no significant sex-based differences in CD4+ T cell subsets in newly diagnosed patients with RA (data not shown). In addition, the median ESR and DAS28 scores in female patients with SUA were significantly higher than those in male patients (Z = −3.387, p = 0.001; Z = −2.593, p = 0.010, respectively). Among the inflammatory cytokines tested, only the serum level of IL-6 showed sex-related disparities in SUA patients, with a lower median level in female SUA patients (n = 63, 12.12 pg/ml) than that in males (n = 26, 19.09 pg/ml) (Z = −1.976, p = 0.048; Figure 5B). No significant sex-based differences in serum IL-2, IL-4, IL-10, IL-17, IFN-γ, and TNF-α levels were observed.

Sex-related disparities in Treg cells, serum inflammatory cytokines, and disease activity index in patients with SUA. (A) Sex-based differences in Treg cells and disease activity index in patients with SUA. (B) Sex-related disparities in serum inflammatory cytokines in patients with SUA. Statistics: Wilcoxon rank-sum test.
We also compared the differences in various T lymphocyte subsets between male or female SUA patients and their respective HCs, revealing a sex-specific profile of peripheral CD4+ T cell subsets. As shown in Figure 6A, the proportions and absolute counts of Treg cells were lower in female SUA patients than those of female HCs. Moreover, the circulating Th1 cell numbers were increased and the Th2 cell numbers were decreased in female SUA when compared with those in female HCs. However, only Treg cell numbers and Th2 percentages significantly differed between male SUA patients and male HCs (Figure 6B). In addition, the CD4+ T cell percentages, Th1/Th2 ratios, and Th17/Treg ratios were increased in both male and female SUA patients when compared with those in their respective HCs. A reduction in CD8+ T cell absolute counts and an increase in CD4/CD8 ratios were only observed in male SUA patients (Figure 6).

Sex-specific differences in various T lymphocyte subsets between patients with SUA and their respective HCs. (A) Differences in multiple T cell subpopulations between female patients with SUA and female HCs. (B) Differences in multiple T cell subpopulations between male patients with SUA and male HCs. Red bars: median. Statistics: Wilcoxon rank-sum test.
Discussion
UA is a common type of inflammatory arthritis that cannot be diagnosed as a specific rheumatic disease. 15 As one form of UA characterized by autoantibody production, SUA will progress to RA as the most severe and persistent form. 16 RF and anti-CCP antibodies are the two most remarkable autoantibodies in RA and can be detected in serum several years before disease onset. 17 However, the presence of autoantibodies alone is not sufficient to predict progression to RA, with only 43% of anti-CCP-positive individuals fulfilling the 2010 ACR/EULAR criteria over 4 years. 18 Another study reported that the positive predictive value for RA development was 64% when both anti-CCP-2 and RF were positive and was 58% when only anti-CCP-2 was positive during a 5-year follow up. 19 Hence, there has been an increasing interest in the identification of other biomarkers that can assist in the prediction of disease onset.
CD4+ T cells play crucial roles as both mediators and regulators of RA pathogenesis. 20 Immuno-logical dysregulations, including the excessive activation of effector CD4+ T cells and dysfunction of immunosuppressive CD4+ T cell subsets, might be other key factors that accelerate the development of RA at the earliest phase. In this study, we determined the characteristics of various immune cell subtypes in patients with SUA, showing extensive abnormalities in circulating CD4+ T cells and subpopulations, with particularly strong reductions in the Treg cell proportions and absolute counts. Additionally, abnormal immune imbalances between Th17 and Treg cells, Th1 and Th2 cells, or CD4+ T and CD8+ T cells were apparent in patients with SUA. These results suggest that distinct dysregulation of the immunological network occurs in the earliest stage of RA.
The imbalance between Th17 and Treg cells has been identified as a crucial aspect of RA pathogenesis.21,22 Notably, the circulating Th17 cell numbers in SUA patients were similar to those in HCs in this study, indicating that a reduction in Treg cells might be the leading cause of the Th17/Treg imbalance in patients with SUA. Treg cells are an immunosuppressive subset of CD4+ T cells characterized by the expression of the X-chromosome-encoded transcription factor Foxp3, and these cells have an essential function in preventing autoimmunity and maintaining self-tolerance. 23 Therefore, deficiencies in either the number or function of Treg cells can lead to the breakdown of immune homeostasis and the development of autoimmune diseases. 24 The present results suggest that immunological dysregulation, especially the breakdown of immune tolerance resulting from the abnormal reduction in Treg cells, might be the most crucial immunological event in the earliest stage of RA. Furthermore, in line with the findings of previous studies on patients diagnosed with RA,25–27 the current study also suggests a decrease in the numbers of circulating Treg cells in patients newly diagnosed with RA, providing further evidence that self-tolerance deficiency contributes to the progression of SUA to RA. Th1 cells are primarily involved in host defense and autoimmunity by acting as proinflammatory effectors. 5 In this study, increased circulating Th1 cells were observed in SUA patients, whereas no significant differences were found in Th1 cell counts between RA patients and HCs. This discrepancy should be further investigated using other molecular markers in future studies with a larger sample size.
RA is a chronic and progressive autoimmune disease that is more common in women than in men (3:1 ratio). 28 Hence, we also compared the sex-based disparities of various immune cells and found significantly different distributions of circulating regulatory CD4+ T cells, with lower proportions and absolute counts of Treg cells in female SUA patients than in males. Additionally, we compared the differences in CD4+ T cell subpopulations between male or female SUA patients and their respective HCs, and found more extensive abnormalities in circulating CD4+ T subpopulations in female SUA patients. Moreover, female SUA patients had higher disease activity than those in males. CD4+ Treg cells are dominant negative regulators of many facets of the immune system, controlling immune responses and enforcing peripheral tolerance to self-antigens. 29 The significantly reduced number of CD4+CD25+Foxp3+ Treg cells in female SUA patients indicates that the initial immunological mechanisms responsible for RA might differ between males and females.
Cytokine networks modulate the immune cell response at a systemic level. Abnormalities in cytokines, particularly in Th1/Th2/Th17 cytokines, might trigger the transition from systemic immunity to arthritis. 30 In this study, we found excessive upregulation of the expression of various inflammatory cytokines in patients with SUA, and this abnormality in the inflammatory cytokine network was also observed in RA patients. The results of our study are in accordance with those of previous reports. 16 These conclusions suggest that dysregulation of the cytokine profile contributes to autoimmune processes, and undoubtedly plays crucial roles in the pathogenesis of SUA and its progression to RA. Moreover, the serum concentrations of IL-6 and TNF-α were significantly higher in RA patients than in SUA patients, further indicating the critical roles of these two proinflammatory cytokines in the development of RA. Certainly, the effects of IL-6 and TNF-α on RA have been confirmed in previous studies, and earlier introduction of anti-IL-6R or anti-TNF-α agents result in enhanced remission in patients with RA and UA.31,32 However, unlike the sex-based disparities found in T cell subpopulations, sex-specific differences were only observed in serum IL-6 levels, with lower levels observed in female SUA patients than in males. IL-6 is a proinflammatory cytokine and plays important roles in RA pathogenesis. We also observed that female SUA patients had high disease activity. Because serum cytokine data were only available for 26 male patients with SUA, the discrepancy might be associated with the smaller sample size and thus should be further investigated in future studies with a larger sample size.
Conclusion
This study provides reference data to evaluate alterations in various CD4+ T cell subpopulations and proinflammatory cytokines in patients with SUA. To the best of our knowledge, this is the first study demonstrating a reduction and sex-based difference in peripheral Treg cells in SUA patients with a large sample size. An abnormal reduction in circulating Treg cells could lead to an immune tolerance deficiency, which might be the initial immunological mechanism underlying the earliest stage of RA development. Moreover, the lower number of Treg cells in female SUA patients than in males might imply that sex-specific disparities in CD4+ Treg cells should be taken into account for immunoregulatory and preventive strategies targeting the early and preclinical stages of RA. The main limitation of this study was that we were not able to evaluate the function of Treg cells in SUA patients, which will need to be clarified in future studies. Human Treg cells can be divided into several fractions; therefore, other specific molecular markers, including CD45RA, CTLA-4, PD-1, LAG3, TIM3, and GITR, should be used to analyze the distinct Treg cell fractions in future studies. In addition, the associated molecular mechanisms responsible for deficiencies in Treg cell numbers/function and RA development require further investigation.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to all our co-workers and collaborators, and to Jing Luo, Xiangcong Zhao, and Chen Zhang for their technical support.
Author contributions
Study design, data collection and analysis, and article writing: HQN, JR, and XFL. Data collection, analysis and interpretation, and diagram drawing: CrY, ClY, NL, YSL, and XL. All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final version for publication. JR had full access to all data in the study, and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Second Hospital of Shanxi Medical University (2019YX-144). Informed consent was obtained from each patient and healthy participant at the time of study enrolment. All methods were performed in accordance with the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81871295), Key Research and Development Projects in Shanxi Province (201903D321026), and Scientific Research Foundation of Health and Family Planning Commission of Shanxi Province, China (No. 201601136).
