Abstract
Objective
To evaluate the role of macrophage infiltration in the differentiation process of ureteral polyps and cancers.
Methods
This retrospective immunohistochemical study analysed archival samples of pathologically-confirmed specimens of low- and high-grade ureteral cancer, ureteral papilloma and ureteral polyps. The samples were immunohistochemically stained for cluster of differentiation (CD)4, CD8, CD16, CD25, CD56 and CD68 using immunofluorescence in order to identify different T-lymphocyte populations and macrophages.
Results
A total of 70 specimens were included in the analysis: 21 specimens of ureteral cancer, 17 specimens of ureteral papilloma, and 32 specimens of ureteral polyps. The largest proportion of CD4+CD25+ regulatory T cells was observed in the low-grade ureteral cancer group and almost none were observed in ureteral papillomas. The largest proportion of CD8+ cytotoxic T-lymphocytes was observed in the ureteral polyps. The largest proportion of CD56+ natural killer cells was detected in the ureteral polyps, with very low levels observed in the other three groups. The largest proportion of CD16+CD68+ macrophages was observed in the high-grade ureteral cancer group, which was significantly higher than that observed in the ureteral papillomas.
Conclusions
This study revealed that CD16+CD68+ macrophages appear to participate in ureteral neoplastic transformation.
Introduction
Cancer immunotherapy is becoming one of the most effective methods of cancer research and treatment.1,2 Cellular components within solid tumours are varied and they perform different protumorigenic or immunosuppressive functions.3,4 Increasing evidence suggests that macrophages function as specific effector cells in cancer immunotherapy, and their degree of tumour infiltration is correlated with prognostic outcomes in many different types of cancer.5,6
The primary aim of this present study was to investigate the extent of macrophage infiltration in benign and malignant ureteral polyps. The secondary aim was to observe the relationship between macrophages and T-lymphocyte subtypes in the pathological process of ureteral diseases.
Patients and methods
Patient population and clinical samples
This retrospective study analysed paraffin-embedded surgical specimens from ureteral cancer, ureteral papilloma, and ureteral polyps collected from patients who were admitted to the Department of General Surgery, the Central Hospital of Siping, Siping, Jilin Province, China between January 2002 and December 2015. The study was approved by the Ethics Committee at the Central Hospital of Siping, Siping, Jilin Province, China (no. SPZXYY20150106). Patient consent was not required as this was a retrospective study.
The surgical specimens were divided into four groups according to their pathological assessment and diagnosis: (i) ureteral papillomas (G1); (ii) ureteral polyps (G2); (iii) high-grade ureteral cancer (G3); and (iv) low-grade ureteral cancer (G4). The exclusion criteria were: (i) patients with active infection, including acute and chronic infections; and (ii) exposure to preoperative chemoradiotherapy. The tumour classifications were based on the World Health Organization/International Society of Urological Pathology classification system. 7 Additional clinical, demographic and pathological parameters were collected from the clinical data records.
Immunofluorescence analysis
To evaluate the presence of regulatory T (Treg) cells, anti-human cluster of differentiation (CD)4 and CD25 antibodies were used concomitantly. Anti-human CD8 antibodies were used to detect the presence of cytotoxic T-lymphocytes. Anti-human CD68 antibodies were used to identify macrophages in general; and anti-human CD16 antibodies were used to specifically confirm the presence of M1 macrophages (i.e. classically activated ‘killer’ macrophages). Anti-human CD56 antibodies were used to detect natural-killer T (NK) cells.
The cell surface staining for CD4, CD8, CD16, CD25, CD56, CD68 was undertaken as described previously. 8 For immunofluorescence staining of clinical tissue samples, formalin-fixed (10% formalin for 2 days at room temperature), paraffin-embedded samples were prepared as 3-µm-thick sections, and then washed with 0.01 M phosphate-buffered saline (PBS; pH 7.4) for 15 min at room temperature, and permeabilized with 0.2% Triton X-100 for 20 min at room temperature. Tissue sections were subsequently blocked with bovine serum albumin (Thermo Fisher Scientific, Inc., Waltham, MA, USA) at 37°C for 30 min, prior to incubation with primary antibodies against CD4 (rabbit, polyclonal, immunoglobulin G [IgG], at a dilution of 1:1000), CD8 (rabbit, polyclonal, IgG, at a dilution of 1:1000), CD25 (mouse, monoclonal, at a dilution of 1:1000), and CD56 (mouse, monoclonal, at a dilution of 1:1000) at 4°C overnight. The above primary antibodies were purchased from BD Company (Wuhan, China). CD16 (rabbit polyclonal, at a dilution of 1:500) and CD68 (mouse monoclonal, IgG, at a dilution of 1:500) were purchased from Abcam Company (Shanghai, China). The slides were washed with 0.01 M PBS (pH 7.4) three times for 5 min each wash. Tissue sections were then incubated with Alexa Fluor 594-conjugated goat anti-rabbit IgG (H+L) cross-adsorbed secondary antibody (at a dilution of 1:1000; Thermo Fisher Scientific, Inc.) for 1 h at room temperature. The slides were washed with 0.01 M PBS (pH 7.4) three times for 5 min each wash. The nuclei were counterstained with 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (DAPI; 1:10 000 dilution; Cell Signaling Technology, Inc., Danvers, MA, USA) at 37°C for 10 min.
Anti-mouse and anti-rabbit IgG secondary antibodies conjugated with Alexa dyes 488 or 568 (Invitrogen, Paisley, UK) were used for double fluorescent staining for 1 h at room temperature. DAPI was used to label cell nuclei (Roche, Mannheim, Germany) at 37°C for 10 min. Cell count was determined at a magnification of ×400 using a Nikon Eclipse E-800 fluorescence microscope (Nikon Instruments, Sendai, Japan). A labelling index (LI) was used to evaluate the proportion of positively-stained cells. Positive cells were randomly assessed in more than 1000 tumour cell nuclei for each tissue section. The LI was calculated randomly in each section as the proportion of positively-stained cells per 1000 tumour cells. The assessment of the positive cells was performed blindly by three independent observers (H.A.J., T.Q.Z. and J.I.F.).
Statistical analyses
All statistical analyses were performed using the SPSS® statistical package, version 19.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Data are presented as mean ± SD. Differences between groups were analysed using Fisher's exact test. A P-value < 0.05 was considered statistically significant.
Results
This retrospective study retrieved 70 archival paraffin-embedded specimens from patients with ureteral diseases, which were classified into four groups based on the pathological diagnosis: G1 consisted of 17 specimens of ureteral papillomas; G2 consisted of 32 specimens of ureteral polyps; G3 consisted of 12 specimens of high-grade ureteral cancer; and G4 consisted of nine specimens of low-grade ureteral cancer. The baseline characteristics of the patients in the four groups are summarized in Table 1.
Clinical and demographic characteristics of patients categorized according to the type ureteral disease.

Data presented as median (range), n of patients or n of patients (%).
The distribution of different immune cells in the four types of ureteral disease.
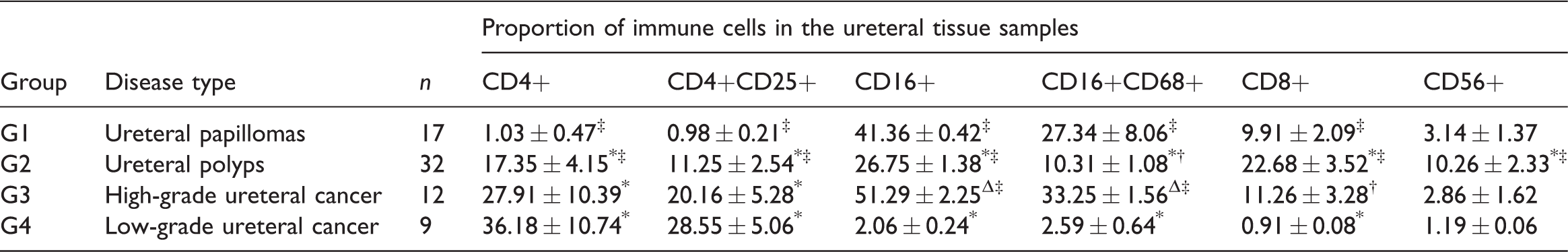
Data presented as mean ± SD.
Compared with G1: *P < 0.01, ΔP < 0.05; Fisher's exact test.
Compared with G4: †P < 0.05, ‡P < 0.01; Fisher's exact test.
Immune cell infiltration was detected in the four ureteral disease tissue types (Figure 1). The extent of CD4+CD25+ Treg cell infiltration was different in the four groups. The largest proportion of CD4+CD25+ Treg cells was observed in the low-grade ureteral cancer group (G4) and almost none were observed in the ureteral papillomas (G1) (Table 2; Figure 2).

Representative photomicrographs showing immune cell infiltration into the four ureteral disease types (haematoxylin and eosin): (a) ureteral polyps; (b) ureteral papilloma; (c) high-grade ureteral cancer; and (d) low-grade ureteral cancer. Arrows show immune cell infiltration. Scale bar 50 µm. The colour version of this figure is available at: http://imr.sagepub.com.

Representative fluorescent photomicrographs showing the presence of CD4+CD25+ regulatory T cells in the four ureteral disease types: (a) ureteral polyps; (b) ureteral papilloma; (c) high-grade ureteral cancer; and (d) low-grade ureteral cancer. CD25 is stained red and CD4 is stained green. The nuclei are stained blue with DAPI. Arrows show the double-positive stained cells. Scale bar 50 µm. The colour version of this figure is available at: http://imr.sagepub.com. DAPI, 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride.
Only limited numbers of CD8+ cytotoxic T-lymphocytes were observed in the low-grade ureteral cancer group (G4), in contrast with the group of ureteral polyps (G2) (Table 2; Figure 3). A significantly larger proportion of CD8+ cytotoxic T-lymphocytes was observed in the ureteral polyps compared with the ureteral papillomas (G1) (P < 0.01). A significantly lower proportion of CD8+ cytotoxic T-lymphocytes was observed in the low-grade ureteral cancer group (G4) compared with the ureteral papillomas (G1) (P < 0.01).

Representative fluorescent photomicrographs showing the presence of CD8+ cytotoxic T-lymphocytes and CD56+ natural-killer T cells in the four ureteral disease types: (a) ureteral polyps; (b) ureteral papilloma; (c) high-grade ureteral cancer; and (d) low-grade ureteral cancer. CD8 is stained red and CD56 is stained green. The nuclei are stained blue with DAPI. Arrows show the double-positive stained cells. Scale bar 50 µm. The colour version of this figure is available at: http://imr.sagepub.com. DAPI, 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride.
The highest proportion of CD56+ NK cells was detected in the ureteral polyps (G2) (Table 2; Figure 3), with very low levels observed in the other three groups.
The largest proportion of CD16+CD68+ macrophages was observed in the high-grade ureteral cancer group (G3), which was significantly higher than that observed in the ureteral papillomas (G1) (P < 0.05) (Table 2; Figure 4). Compared with the ureteral papillomas (G1), there were significantly lower proportions of CD16+CD68+ macrophages observed in the ureteral polyps (G2) (P < 0.01) and the low-grade ureteral cancers (G4) (P < 0.01).

Representative fluorescent photomicrographs showing the presence of CD16+ M1 macrophages and CD68+ general macrophages in the four ureteral disease types: (a) ureteral polyps; (b) ureteral papilloma; (c) high-grade ureteral cancer; and (d) low-grade ureteral cancer. CD16 is stained green and CD68 is stained red. The nuclei are stained blue with DAPI. Arrows show the double-positive stained cells. Scale bar 50 µm. The colour version of this figure is available at: http://imr.sagepub.com. DAPI, 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride.
Discussion
This current study investigated the infiltration of macrophages into four different ureteral tissues. These current data suggest that CD16+ M1 macrophages are involved in the differentiation of ureteral polyps into ureteral papilloma (i.e. benign lesion). To the best of our knowledge, this is the first report about the role of macrophages in the ureteral polyp differentiation process.
Macrophages that are CD16+CD68+ are usually referred to as M1 macrophages. Ureteral polyps can undergo either benign or malignant transformation. 9 The process of neoplastic transformation is affected by the local microenvironment, in particular by regulation by the immune system. 10 In the current study, the role of macrophages in different ureteral tissues was examined and CD16+CD68+ M1 macrophages were present at significantly higher proportions in high-grade ureteral cancers compared with ureteral papillomas. These current data suggest that CD16+CD68+ M1 macrophage phagocytosis participates in the neoplastic transformation in the ureter and that this role is limited to advanced disease.
In this present study, Treg cells were identified by the coexpression of CD4 and CD25 in ureteral tissue samples. CD4+CD25+ Treg cells were observed in ureteral polyps and low- and high-grade ureteral cancers, but were almost absent in ureteral papillomas. Taken together, these data suggest that CD4+CD25+ Treg cell infiltration is meaningful in terms of ureteral polyps.
In order to investigate the role of cytotoxic T-lymphocyte and/or NK cell infiltration in tumour progression, tissue samples were labelled for CD8 and CD56. CD8+ cytotoxic T-lymphocytes were rarely observed in the low-grade ureteral cancers, while detected in the other studied tissue types. These data suggest that the potential anti-tumour effects of CD8+ cytotoxic T-lymphocytes do not appear to play a role in ureteral cancer. The highest levels of CD56+ NK cells were observed in ureteral polyps, with very low levels observed in the other three tissue types, suggesting that their potential killing function plays a limited role in ureteral neoplastic transformation. The role of NK cells in adaptive immunity needs to be further explored.
Tumour differentiation is a complex process and abnormalities in the pathophysiology of the B cell and T cell compartments have been identified. 11 Immune dysfunction in cancer patients leads to a reduced immunosuppressive capacity and a lower inhibitory effect on activated neoplastic proliferation.12,13 These findings suggest that there is an immune cell malfunction in the pathophysiology of cancer and this may have therapeutic implications. 14 The current study did not measure the B cell populations in the ureteral tissue samples, so it is not possible to draw any conclusions about their role in the immune regulation in ureteral lesions. Future research is required to investigate more fully the immunomodulatory characteristics of ureteral lesions.
In conclusion, these current data demonstrated that the co-expression of CD16+CD68+ macrophages and CD8+ cytotoxic T-lymphocytes is related to ureteral neoplastic transformation. These findings suggest that CD16+CD68+ macrophages and CD8+ cytotoxic T-lymphocytes were involved in the poor differentiation of ureteral neoplastic tissue. In our opinion, macrophages are often under-appreciated as effector cells that can target cancer. Future large-scale clinical studies are needed to investigate the role of macrophages as a potential treatment for ureteral cancer.
Footnotes
Author contributions
Hai-Yan Jia completed the statistical analyses. Tian-Qi Zhang completed the immunofluorescence analyses. Ji-Ning Fu collected the case information. Ying Liu designed the experiments and wrote the manuscript. Yue-Hui Wang designed the experiments and contributed to the discussion.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research received a grant from the Science and Technology Department of Jilin Province, China (no. 201105100).
