Abstract
Background:
Patients with type 2 diabetes mellitus (T2DM) have an increased risk of fracture compared with those without T2DM. Some oral glucose-lowering agents may increase the incidence of fracture. Whether sodium-glucose co-transporter 2 inhibitors (SGLT2is) are associated with increased risk of fracture remains unclear.
Methods:
We retrieved articles from PubMed, Embase, Cochrane Library database, and other sources up to 24 October 2019. We included randomized controlled trials (RCTs) that reported fractures and analyzed the fracture incidence of SGLT2i, canagliflozin, dapagliflozin, and empagliflozin. Subgroup analysis was also performed based on baseline characteristics.
Results:
A total of 78 RCTs with 85,122 patients were included in our analysis. The overall SGLT2i fracture incidence was 2.56% versus 2.77% in the control group [odds ratio (OR), 1.03; 95% confidence interval (CI) (0.95, 1.12); p = 0.49]. Compared with the control treatment, treatment with canagliflozin led to a higher rate of fractures [OR, 1.17; 95% CI (1.00, 1.37); p = 0.05], but no significant difference was observed when compared with dapagliflozin [OR, 1.02; 95% CI (0.90, 1.15); p = 0.79] or empagliflozin [OR, 0.89; 95% CI (0.73, 1.10); p = 0.30]. Subgroup analysis showed that, in a follow-up of less than 52 weeks, SGLT2i decreased the incidence of fracture by 29% [OR, 0.71; 95% CI (0.55, 0.93); p = 0.01], but this benefit was lost when the follow-up extended to more than 52 weeks [OR, 1.08; 95% CI (0.98, 1.18); p = 0.12].
Conclusion:
Canagliflozin seems to increase the risk of fracture, while other SGLT2is do not result in a higher incidence of fracture.
Introduction
Patients with type 2 diabetes mellitus (T2DM) have an increased risk of fracture compared with those without T2DM. In addition to common hip and spine fractures, femur, femoral neck, and pelvic fractures are often reported in observational studies.1–3 This could be attributed to many factors, including bone mineral density (BMD), bone turnover, microarchitecture, and material properties.4,5 Elderly individuals are more likely to experience both fractures and T2DM. Complications of T2DM can lead to delayed union or to non-union of fractures.6,7 Some oral glucose-lowering agents such as thiazolidinediones (especially rosiglitazone)8–10 may increase the incidence of fracture, and further study into their effects in T2DM patients is warranted to mitigate this risk.
Sodium-glucose co-transporter 2 inhibitors (SGLT2is) are novel glucose-lowering agents that lower blood glucose by inhibiting glucose and sodium reabsorption in the proximal tubule of the kidney. In addition to lowering glucose, SGLT2is can also lead to weight loss, decreased blood pressure, and reduction in serum uric acid.11–13 Recent clinical studies have demonstrated that SGLT2is can lower the risk of mortality, heart failure, renal failure, and cardiovascular events.14,15 Indeed, the 2019 European Society of Cardiology Guidelines recommend SGLT2i as a priority for patients with T2DM and cardiovascular diseases (CVDs), or at very high/high cardiovascular (CV) risk, to reduce CV events. 16 However, the CANVAS study found that the canagliflozin group had a higher incidence of bone fracture than the placebo group (15.4% versus 11.9% per year, p = 0.02). 17 A subsequent study attempted to determine the reasons for this increase in fracture risk, but did not succeed. 18 A study with a follow-up period of 104 weeks found that SGLT2i may be related to an elevated incidence of fracture (7.7% versus 0%). 19 Another study indicated that the decrease in BMD in the canagliflozin group appears to be associated with increased fracture risk, 20 while other studies found that SGLT2is could lower the incidence of fracture.21,22 Given the controversial results of SGLT2is on fracture events, we sought to synthesize all available data to investigate the safety of SGLT2is.
Materials and methods
Search strategy
The following keywords were used in the literature search: “Sodium-Glucose Transporter 2 Inhibitors”, “Dapagliflozin”, “Canagliflozin”, “Empagliflozin”, “Ipragliflozin”, “Sergliflozin”, “Remogliflozin”, “Tofogliflozin”, “Luseogliflozin”, “Sotagliflozin”, “Ertugliflozin”, “Velagliflozin”, “Licogliflozin”, and “Mizagliflozin”. Databases such as PubMed, Embase, Cochrane Central Register of Controlled Trials (CENTRAL), and the clinical trial registration website https://www.clinicaltrials.gov/ were searched to identify randomized controlled trials (RCTs) whose comparators were SGLT2i and other treatments (including placebo) until 24 October 2019. We searched only articles published in English (further details are available in the Supplemental Material online).
Eligibility criteria
Studies with the following criteria were eligible for inclusion:
RCTs;
Fractures reported in the Results or in the section of adverse events;
Intervention group with a single medication (SGLT2i) or a mixture (containing SGLT2i and other hypoglycemic drugs), with a placebo control group or other active treatment. Trials were included irrespective of the dosage of SGLT2i and active treatment.
Animal experiments, case reports, cohort studies, pooled analyses, and studies with a sample size less than 50 were excluded.
Data extraction and quality assessment
Four authors (YL, YY, JD, and SB) screened the retrieved citations and selected the potential references. In the case of disagreements, other authors (WL and XN) were consulted until a consensus was reached. All potential studies were further analyzed with full text. Baseline characteristics, follow-up period, outcome, and adverse events data were extracted by four authors (KNCS, ZX, YG, and YZ). If the data were incomplete or unclear, the study details were searched in clinitrialtrials.gov website or other published articles.
Quality assessment
We evaluated the risk of bias for every study according to the Cochrane handbook for systematic reviews of interventions (version 5.1.0). Two authors independently examined the references and classified studies into low risk, unclear risk, and high risk through random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias), and other bias.
Outcomes
The primary endpoint was the overall fracture incidence of SGLT2i, and the secondary outcomes were fracture incidence with canagliflozin, dapagliflozin, and empagliflozin.
Statistical analysis
All analyses were performed using Stata 15.1 software (StataCorp, College Station, TX, USA) and ReviewManager (RevMan) version 5.3 (The Cochrane Collaboration, Copenhagen, Denmark). The study was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 23 The odds ratios (ORs) and 95% confidence intervals (CIs) were calculated by Mantel–Haenszel analysis to compare the safety of SGLT2is. To compare the real effects of SGLT2i on fractures, all doses of SGLT2i and all control groups were included. We also evaluated the fracture incidence of canagliflozin, dapagliflozin, and empagliflozin. Considering the influence of other factors on fractures, we performed subgroup analysis based on control type (placebo or other active comparators), follow-up period, ethnicity, age, data source, presence or absence of chronic kidney diseases (CKDs), history of CVD, and T2DM duration. We used the I2 statistic and chi-square test to evaluate heterogeneity across trials; I2 > 50% was considered to indicate substantial heterogeneity. 24 The Mantel–Haenszel fixed-effects model was used where I2 < 50%; otherwise, the Mantel–Haenszel random-effects model was used. By excluding each trial subsequently, we performed sensitivity analyses to evaluate the stability and reliability of the results. A visual funnel plot was used to evaluate publication bias.
Results
Study selection
Using the above-mentioned keywords, we identified 9750 potential references. A total of 4791 duplicates were excluded manually and using software, and the remaining 4959 manuscripts were screened by browsing titles and abstracts. Subsequently, the full text of 452 potentially eligible references was searched; of which, 374 articles were excluded as duplicate articles, not reporting fracture, and due to improper comparators. Finally, the remaining 78 RCTs of 85,122 patients were included in our meta-analysis (flowchart in Figure 1).
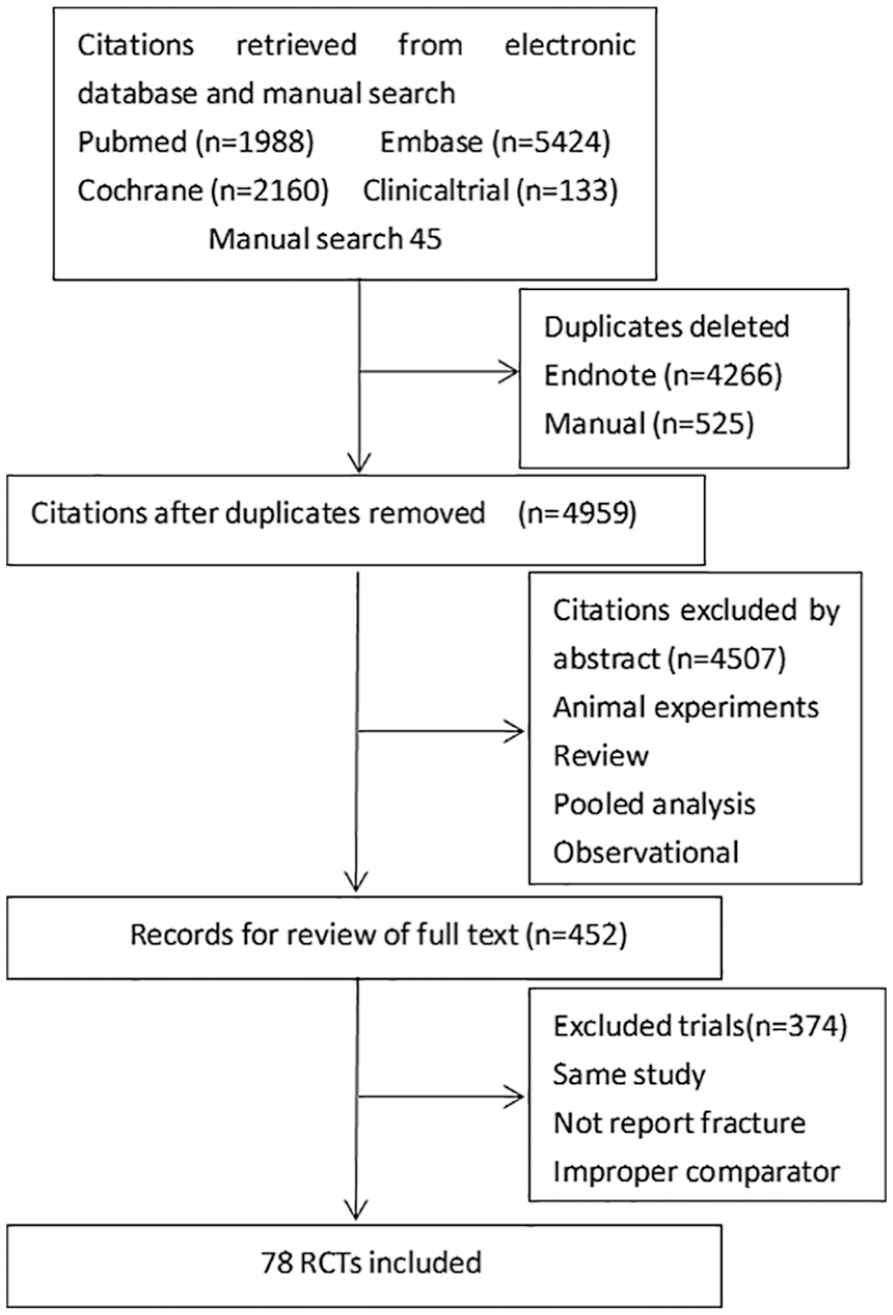
Flowchart of study selection.
Characteristics of eligible studies
In total, 78 RCTs of 85,122 patients were included in our final analysis; 65 RCTs were selected from published articles and the other 13 trials were retrieved from the https://www.clinicaltrials.gov/ website. A total of 50,471 (59.3%) patients were treated with SGLT2is, and 34,651 (40.7%) were treated with other drugs or placebo. The shortest and the longest follow-up periods were 12 weeks and 296 weeks, respectively, and the sample size in each study ranged from 121 to 17,160 patients. Canagliflozin was used in the treatment group in 16 studies, dapagliflozin in 33 studies, empagliflozin in 17 studies, bexagliflozin in two studies, ertugliflozin in six studies, ipragliflozin in two studies, remogliflozin in one study, and tofogliflozin in one study. Control group agents included exenatide, glimepiride, metformin, linagliptin, pioglitazone, sitagliptin, and placebo. The baseline characteristics and other data of the included studies are listed in the Supplemental Material.
Primary endpoint
The primary endpoint was the overall fracture incidence of SGLT2i. A total of 78 trials in 85,122 patients reported treatment with SGLT2i or control (including placebo). A total of 1294 fractures occurred in 50,471 patients treated with SGLT2i, and 961 fractures occurred in 34,651 patients treated with control agents (including placebo); SGLT2i did not increase the risk of fracture [2.56% versus 2.77%; OR, 1.03; 95% CI (0.95, 1.12); p = 0.49]. Subgroup analysis showed that in a follow-up of no more than 52 weeks, patients treated with SGLT2i showed a 29% decrease in the incidence of fracture [OR, 0.71; 95% CI, (0.55, 0.93); p = 0.01], but this benefit was lost when the follow-up extended to more than 1 year [OR, 1.08; 95% CI (0.98, 1.18; p = 0.12)]. There was no significant difference in the other subgroup analyses based on control group agents (active comparator versus placebo), ethnicity (White versus Asian), history of CVD, age (<60 years old or >60 years old), data source (published article versus website registration information), patients with or without CKD, and duration of T2DM (fracture risk is shown in Figure 2, subgroup analysis is listed in Table 1).
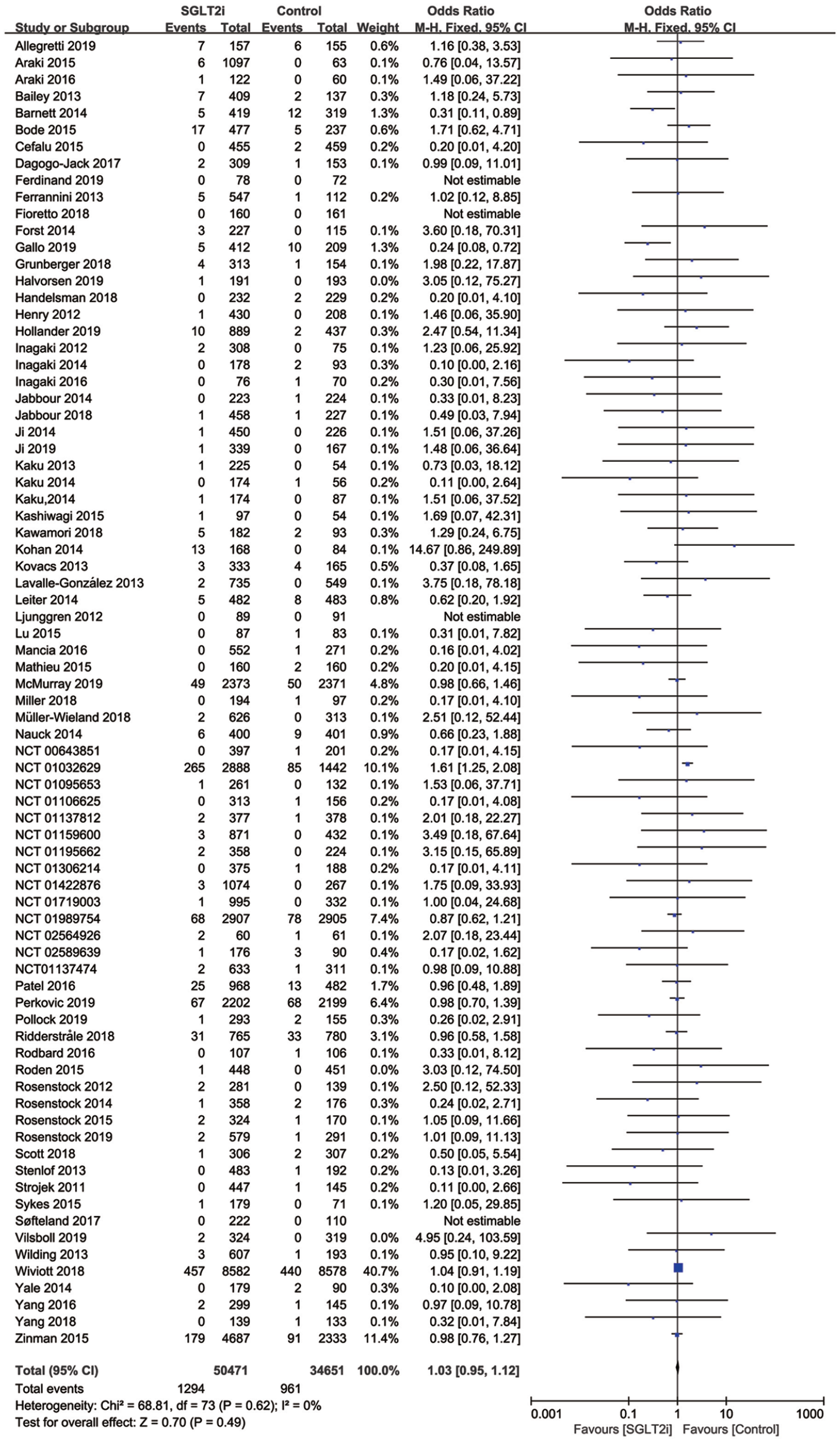
Forest plot of fracture incidence between SGLT2is and other treatment.
Subgroup analysis of fracture incidence between SGLT2i and other treatment.

CKD, chronic kidney disease; CVD, cardiovascular disease; DM, diabetes mellitus; OR, odds ratio; SGLT2i, sodium-glucose co-transporter 2 inhibitor.
Secondary outcomes
The secondary outcomes were fracture incidences of canagliflozin, dapagliflozin, and empagliflozin. In the present meta-analysis, canagliflozin tended to increase the risk of fracture [3.51% versus 2.77%; OR, 1.17; 95% CI (1.00, 1.37); p = 0.05]. With regard to subgroup analysis, canagliflozin increased the risk of fracture in patients with a follow-up ⩾104 weeks, mean age >60 years old, duration of T2DM >10 years, or whose ethnicity is White (4.92% versus 3.48%, p = 0.02; 4.82% versus 3.46%, p = 0.03; 4.69% versus 3.39%, p = 0.02; 3.78% versus 2.88%, p = 0.03, respectively). Dapagliflozin and empagliflozin were not associated with a higher incidence of fracture (Figures 3–5 and Table 1).

Forest plot of fracture incidence between canagliflozin and other treatment.

Forest plot of fracture incidence between dapagliflozin and other treatment.

Forest plot of fracture incidence between empagliflozin and other treatment.
Publication bias and quality assessment
We observed no obvious publication bias from the funnel plot (funnel plot is shown in the Supplemental Material).
Sensitivity analysis
There was no obvious heterogeneity in the overall SGLT2i analysis or the dapagliflozin and empagliflozin group analysis (I2 = 0). In analysis of the canagliflozin group, I2 was 32%, but this dropped to 0 if the CANVAS study was omitted.
Discussion
To the best of our knowledge, the present study is the largest meta-analysis to directly compare SGLT2is with other hypoglycemic agents. In our analysis of 78 RCTs, we found that canagliflozin seems to increase the risk of fracture, while other SGLT2is are not associated with a higher incidence of fracture.
It has been reported that there is increased risk of fracture in elderly T2DM patients that can be lowered by glycemic control. 25 SGLT2i can achieve a 0.7% reduction in glycated hemoglobin, 12 which may explain why SGLT2i leads to a lower fracture incidence over a follow-up of less than 52 weeks. However, when this period is extended to more than 52 weeks, the impact of SGLT2i on phosphate metabolism may not be omitted. SGLT2is mediate their effects by inhibiting the reabsorption of glucose in the proximal tubule of the kidney; 11 this increases the concentration of serum phosphate, likely through an increase in tubular reabsorption, which may result in a higher parathyroid hormone (PTH) concentration and increased fracture incidence. In addition, the change in phosphate concentration may provoke the secretion of fibroblast growth factor 23 (FGF23). Together, these factors may have a combined action on BMD and increase the fracture incidence.26,27 What is more, a meta-analysis including 43 RCTs demonstrated that SGLT-2i can lower systolic blood pressure (BP) by 2.46 mmHg and diastolic BP by 1.46 mmHg. 28 Scheen pointed out that SGLT-2is have a higher incidence of orthostatic hypotension compared with other hypoglycemic drugs in elderly populations, who have a higher incidence of fracture. 29 The antihypertensive effect of SGLT2 may play an important role in the occurrence of fracture. In summary, this combination effect of BP lowering and BMD change may neutralize the advantage of glycemic control and cause SGLT2i to have an effect on fracture incidence, comparable to that observed in other treatments.
Our results are consistent with those of previous meta-analyses30,31 in that we find that SGLT2i may have a beneficial effect on fracture in a follow-up of less than 52 weeks, but when this period is extended to more than 52 weeks, this benefit disappears. However, we also found that canagliflozin may increase the incidence of fracture, which is different from the findings of the previous study; this is likely to be because we included the CANVAS study using the newest follow-up data,17,18 which showed an obvious increase in fracture. Because the CANVAS study introduces heterogeneity to the subgroup analysis of canagliflozin, the heterogeneity drops from 32% to 0 if the CANVAS study is excluded. Correspondingly, the OR of the canagliflozin group versus other hypoglycemic agents drops from 1.17 (1.00, 1.37) to 0.92 (0.75, 1.14) if the CANVAS study is excluded. Furthermore, it is important to note that the CANVAS study had the longest mean follow-up period, of 296 weeks, with a large sample size of 4330, and the baseline risk for fracture is higher than that reported in other studies.32–34 In addition, the primary outcome in CANVAS was low-trauma fractures as judged by the trial adjudication committee. 17 These factors may explain why patients treated with canagliflozin in the CANVAS study had a higher incidence of fracture. 35 A real-world study that enrolled 159,928 patients and compared canagliflozin with glucagon-like peptide-1 (GLP-1) receptor agonist suggested that canagliflozin is not associated with a higher risk of fracture. 32 Because we observed that the real-world study had a mean follow-up of 34 weeks, it is difficult to conclude the long-term safety. Similar to our results, a previous study that pooled the results of 10 trials showed that fracture risk in the canagliflozin group was increased. 35 Therefore, the long-term fracture incidence of canagliflozin deserves further attention.
The mechanism through which canagliflozin increases the risk of fracture is still unclear. SGLT-2i agents have different selectivity for SGLT-2 versus SGLT-1. Empagliflozin has the highest SGLT-2/SGLT-1 affinity ratio and canagliflozin the lowest. 11 Masiukiewicz and Ljunggren et al. suggest that ertugliflozin and dapagliflozin had no significant effect on BMD and other bone biomarkers,36,37 while Bilezikian et al. found that canagliflozin can lower the hip BMD and increase bone biomarkers. 20 Animal experiments also showed that canagliflozin can increase the concentration of phosphate, FGF23, and PTH, whereas tofogliflozin has been shown to have no clear effect on bone mass by microcomputed tomography. 38 Whether the difference in selectivity for SGLT-2/SGLT-1 and different bone biomarkers lead canagliflozin to have a higher fracture incidence remains uninvestigated.
With regard to subgroup analysis, we found no obvious difference in SGLT2i and other treatment, with the exception of the canagliflozin group. Tang et al. found that SGLT2is had a tendency to increase the risk of fracture in the Asian population, 39 but we did not observe that phenomenon in our subgroup analysis. Tang et al. included 2819 patients and found no significant difference [OR, 2.05; 95% CI (0.86, 4.87)], while we included 18 studies of 5279 patients (mainly Asians) and observed no significant difference [OR 1.03, 95% CI (0.59, 1.79)]. Furthermore, a study mainly including CKD patients showed that empagliflozin had a reduced incidence of fracture in stage 3 CKD 21 (1.6% versus 4.8%), but another study with a follow-up period of 104 weeks that mainly included moderate renal impairment CKD patients showed an obvious increase in fracture incidence for dapagliflozin (7.74% versus 0%). 19 Our meta-analysis included eight trials of CKD patients and found that SGLT2is had no obvious effects on fracture incidence [OR, 0.84; 95% CI (0.49, 1.43), p = 0.53]. However, because the heterogeneity across trials was high, we could not eliminate the influence on analysis; thus, further studies are needed to determine the fracture risk of SGLT2i.
Limitations
The present meta-analysis has some limitations in addition to the disadvantages in the original research. First, some data are acquired from the clinical registration website and not from the published article; this may introduce bias. However, there was no difference in our conclusion when we performed subgroup analysis based on data source and subgroup analysis. Second, some events data are not presented in the article, and, if after contact with the authors we were still unable to gain access to the raw data, the events data were transformed from the published data. Third, we were unable to access some baseline characteristics, which limited our ability to perform subgroup analysis.
In summary, canagliflozin seems to increase the risk of fracture, while other SGLT2is do not result in a higher incidence of fracture.
Supplemental Material
Supplymentary_Materials-Baseline_characteristics – Supplemental material for Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials
Supplemental material, Supplymentary_Materials-Baseline_characteristics for Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials by Yake Lou, Ying Yu, Junchao Duan, Sining Bi, Khaing Nyein Chan Swe, Ziwei Xi, Yanan Gao, Yujie Zhou, Xiaomin Nie and Wei Liu in Therapeutic Advances in Chronic Disease
Supplemental Material
Supplymentary_Materials-Primary_outcomes_and_eligiblity_criteria – Supplemental material for Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials
Supplemental material, Supplymentary_Materials-Primary_outcomes_and_eligiblity_criteria for Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials by Yake Lou, Ying Yu, Junchao Duan, Sining Bi, Khaing Nyein Chan Swe, Ziwei Xi, Yanan Gao, Yujie Zhou, Xiaomin Nie and Wei Liu in Therapeutic Advances in Chronic Disease
Supplemental Material
Supplymentary_Materials-Search_strategtplusInfluence_analysisplusFunnel_plot – Supplemental material for Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials
Supplemental material, Supplymentary_Materials-Search_strategtplusInfluence_analysisplusFunnel_plot for Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials by Yake Lou, Ying Yu, Junchao Duan, Sining Bi, Khaing Nyein Chan Swe, Ziwei Xi, Yanan Gao, Yujie Zhou, Xiaomin Nie and Wei Liu in Therapeutic Advances in Chronic Disease
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NO. 81470429) and Beijing Science and Technology Development Fund of Traditional Chinese Medicine (NO. JJ2016-31)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
