Abstract
Objective
To determine whether empagliflozin reduces ventricular fibrillation and myocardial fibrosis after myocardial infarction via the transforming growth factor-β1/Smad3/miR-181a pathway.
Methods
Male nondiabetic Sprague–Dawley rats (n = 16) were randomized into sham, myocardial infarction, low-dose empagliflozin (10 mg/kg/day), and high-dose empagliflozin (30 mg/kg/day) groups. Myocardial infarction was induced via coronary artery ligation. After 4 weeks, ventricular fibrillation thresholds were assessed via electrical stimulation. Cardiac function (echocardiography), fibrosis (Masson’s trichrome staining), and molecular markers (transforming growth factor-β1, Smad3, miR-181a; assessed via western blotting/quantitative polymerase chain reaction) were analyzed.
Results
Empagliflozin-treated groups showed reduced left ventricular dilation (left ventricular internal diameters at end-diastole: 8.2 ± 0.3 vs. 9.1 ± 0.4 mm in myocardial infarction; left ventricular internal diameters at end-systole: 5.1 ± 0.2 vs. 6.0 ± 0.3 mm) and improved ejection fraction (45% vs. 38% in myocardial infarction). Ventricular fibrillation thresholds increased significantly with empagliflozin administration (p < 0.05). Myocardial fibrosis (collagen volume fraction: 12% vs. 25% in myocardial infarction) and transforming growth factor-β1/Smad3/miR-181a expression were downregulated in empagliflozin groups (p < 0.01). No dose-dependent differences were observed between the groups.
Conclusions
Empagliflozin attenuates postmyocardial infarction ventricular arrhythmias and fibrosis associated with the suppression of transforming growth factor-β1/Smad3 signaling and miR-181a expression, enhancing cardiac function. These findings highlight its therapeutic potential in postinfarct remodeling.
Keywords
Introduction
Myocardial infarction (MI) is a significant cardiovascular condition associated with elevated mortality and disability. 1 A prevalent complication of MI is ventricular arrhythmia (VA), with ventricular fibrillation (VF) being a critical contributor to mortality among affected patients. 2 The relationship between myocardial fibrosis and the onset of VAs following MI is well established. 3 During ventricular remodeling, cardiomyocyte fibrosis plays a protective role against myocardial rupture and aids in the healing of cardiac injury post-MI; however, it concurrently reduces myocardial compliance and disrupts both electrical conduction and standard ventricular diastolic function. 4 Central to this process is the transforming growth factor-β1 (TGF-β1)/Smad3 signaling pathway.5,6 TGF-β1 facilitates the synthesis of extracellular matrix (ECM) components, drives the transformation of cardiac fibroblasts into myofibroblasts, and augments collagen production. 7
Sodium–glucose cotransporter 2 inhibitors (SGLT2i), including agents such as empagliflozin (EMPA), dapagliflozin, canagliflozin, sotagliflozin, and ertugliflozin, represent a novel class of glucose-lowering medications that offer a safe and effective management option for patients with type 2 diabetes. 8 Many studies have indicated that SGLT2i plays a significant role in cardioprotection.8–11 These agents have demonstrated the capability to diminish postinfarction myocardial fibrosis, potentially through the inhibition of the TGF-β1/Smad3 signaling pathway.12,13 Furthermore, evidence suggests that SGLT2i decrease the incidence of arrhythmias. A meta-analysis conducted by Fernandes et al. and a cohort study reported that SGLT2i reduced the risk of atrial arrhythmias and sudden cardiac death in individuals with heart failure.14,15 Additionally, EMPA mitigated VF induction in a rabbit model of ischemia/reperfusion. 16 Nonetheless, the precise mechanisms through which SGLT2i alleviates arrhythmia remain unclear, and further investigation is warranted to ascertain whether these effects are mediated via glucose-lowering mechanisms.
MicroRNAs (miRNAs) are a class of noncoding RNAs that play a role in the translation process, thereby regulating gene expression by binding to messenger RNA (mRNA). These molecules are extensively implicated in various biological processes, including apoptosis, development, signal transduction, cellular proliferation, and immune responses. Recent research has highlighted the regulatory role of miR-181a in the development of myocardial fibrosis post-MI. 17 Furthermore, studies have indicated a potential connection between miR-181a and the TGF-β1/Smad signaling pathway.18,19 Chen et al. 20 demonstrated that miR-181a inhibits the expression of TGF-β receptor III, consequently promoting myocardial fibrosis in rat models post-MI.
This study aimed to examine the impact of EMPA on the mitigation of VAs and cardiac fibrosis by downregulating miR-181a and inhibiting the TGF-β1/Smad3 signaling pathway following MI.
Material and methods
Establishment of the animal model
Twenty male nondiabetic Sprague–Dawley rats, each weighing approximately 200 g, were obtained from the Animal Center of Southwest Medical University, Luzhou, China. Sixteen of these rats underwent ligation of the left anterior descending coronary artery to induce MI; however, four died during the surgical procedure. The remaining 12 MI rats were further categorized into three groups: MI, low-dose EMPA (low-EMPA), and high-dose EMPA (high-EMPA), with four rats designated to the sham group. All animals were maintained at the Heart Research Institute of Southwest Medical University in a controlled environment, characterized by a temperature of approximately 25°C, humidity levels ranging from 40% to 50%, and a 12-h light/dark cycle. Food and water were provided ad libitum, and bedding was routinely replaced. All experimental protocols were conducted in accordance with the US National Institutes of Health’s Guide for the Care and Use of Laboratory Animals (8th edition, 2011) as well as Helsinki Declaration of 1975, as revised in 2013. Furthermore, these experiments were approved by the Institutional Animal Care and Treatment Committee of Southwest Medical University, China (Approval No. 20221026-008, Date: 2022-10-26).
Animal treatment
Following a fasting period of 12 h, during which only food was withheld, the rats were anesthetized using an intraperitoneal injection of sodium pentobarbital at a dose of 50 mg/kg. The animals were subsequently secured on a surgical table for skin preparation and disinfection. Oral endotracheal intubation was performed, and a ventilator was connected to facilitate mechanical ventilation, characterized by a tidal volume of 0.8 mL and a respiratory rate of 80 breaths per minute, with a respiratory time ratio of 5:4. Electrocardiographic electrodes were inserted subcutaneously in the limbs of the rats, allowing electrocardiogram recordings via standard leads I and II and a lead for VF. A longitudinal incision was made on the left thorax, followed by meticulous layer-by-layer blunt dissection of the muscle tissue, culminating in a thoracotomy at the fourth intercostal space. The pericardium was carefully separated to allow access to the heart. Ligation was performed 2 mm distal to the junction of the pulmonary conus and left atrial appendage using a 6-0 suture. Successful establishment of the MI model was confirmed by observing a color change in the myocardium from red to white or by elevation of the S-T segment. In the sham group, rats were subjected to thoracotomy without ligation, and the chest was subsequently closed in layers. On the day following model establishment, all rats received a gavage of EMPA or distilled water. The low-EMPA group was administered EMPA (10 mg/kg/day; Jardiance, Boehringer Ingelheim International GmbH, Germany) intragastrically, whereas the high-EMPA group received EMPA at a higher dosage of 30 mg/kg/day. EMPA was dissolved in 0.9% normal saline. The final concentration of EMPA in the administered solution was 1 mg/mL (low dose) and 3 mg/mL (high dose). Prior to administration, the solution was vortexed for 5 min and centrifuged at 3000 rpm for 10 min to ensure complete dissolution. The sham and MI groups received 3 mL of distilled water via oral gavage daily, using the same administration method as the EMPA-treated groups. The treatment regimen was continued for 28 days.
Echocardiography
In the fourth week following skin preparation, parameters including left ventricular (LV) internal diameter at end-diastole (LVIDd), LV internal diameter at end-systole (LVIDs), LV fractional shortening (LVFS), and LV ejection fraction (LVEF) were assessed using a small animal echocardiography system (Vevo2100, FUJIFILM, Japan) under gaseous anesthesia with isoflurane at a maintenance concentration of 1.5%.
Electrical stimulation
Following anesthesia induction, skin preparation, mechanical ventilation, and thoracotomy as previously outlined, a bipolar pacemaker electrode was implanted in the peripheral region of the MI in rats. The electrode was positioned at a depth of 1 mm with an interelectrode distance of 2 mm, ensuring that the electrodes were placed at a distance of no more than 3 mm from the infarction border. Burst stimulation was conducted via a multiguide electrocardiographic recording stimulation system (with a cycle length of 60 ms, pulse width of 10 ms, and duration of 30 s; NIHON KOHDEN), followed by a 1-min stimulation period. The pacing voltage commenced at 4 V and was incrementally increased by 1 V until VF was induced, which was defined as the minimum intensity that elicited VF.
Hematoxylin and eosin (H&E) and Masson’s trichrome staining
Tissue samples were collected from the border zone of the LV infarct region. LV tissue specimens were fixed in a 4% tissue fixation solution for 24 h, followed by dehydration, embedding, and sectioning at a thickness of 5 μm. These sections were subsequently stained with H&E and Masson’s trichrome stain in accordance with established protocols. Images were captured using a digital imaging system, and the results of Masson’s trichrome staining were quantitatively analyzed using ImageJ software at a magnification of 20×.
Quantitative polymerase chain reaction (qPCR)
Total mRNA was extracted from the border zone of the LV infarct region using commercial RNA extraction kits (AG21101; Accuratr Biology, China). The RNA was transformed into cDNA using the Evo M-MLV RT Mix Kit (AG11728, Accuratr Biology, China), following the manufacturer’s instructions. A qPCR assay was conducted to determine miRNA-181a1 expression using SYBR® Green Premix Pro Taq HS qPCR Kit (AG11701, Accuratr Biology, China). The primers used in this study were as follows: miRNA-181a1 forward: 5′-CGAACATTCAACGCTGTCG-3′ and reverse: 5′-AGTGCAGGGTCCGAGGTATT-3′; GAPDH forward: 5′-GAAGGTCGGTGTGAACGGAT-3′ and reverse: 5′-CCCATTTGATGTTAGCGGGAT-3′; and U6 forward: 5′-CTCGCTTCGGCAGCACA-3′ and reverse: 5′-AACGCTTCACGAATTTGCGT-3′. The relative expression levels of specific mRNAs and miRNAs were normalized to those of U6 or GAPDH using the 2−ΔΔCq method.
Western blotting
Proteins were isolated from the margins of the LV infarct region and their concentrations were determined using a bicinchoninic acid assay (P0010, Beyotime, Shanghai, China). The samples were diluted to a concentration of 5 mg/mL using radioimmunoprecipitation assay lysis buffer (P0013B, Beyotime) along with a 6× sodium dodecyl sulfate polyacrylamide gel electrophoresis protein loading buffer (P0015F, Beyotime) and subsequently subjected to boiling for 10 min at 100°C. Subsequently, 10% polyacrylamide gels (PG112, EpiZyme, USA) were cast, and 10 µL of each protein sample was loaded into the designated wells. Electrophoresis was performed using a Bio-Rad system (California, USA) at an initial voltage of 80 V until the proteins migrated into the lower gel region, followed by an increase to 150 V for 40 min. The polyvinylidene fluoride (PVDF) membrane was preactivated with methanol, and a sandwich structure was established in accordance with the positive and negative electrodes. Proteins were transferred onto a PVDF membrane for 90 min at a current of 250 mA using a transfer buffer. Following a 15-min blocking period with a protein-free rapid blocker, the membrane was incubated overnight at 4°C with the following primary antibodies: anti-TGF-β1 (1:1000, ab215715, Abcam), anti-Smad-3 (1:1000, 9523 T, CST), and anti-Smad-2 (1:1000, 5339 T, CST). After three washes for 10 min each with Tris-buffered saline with Tween-20 (TBST), the membrane was incubated with goat anti-rabbit secondary antibody (1:1000, 7074P2, Cell Signaling Technology) for 90 min at room temperature. Following three additional washes for 10 min each with TBST, immunoreactive bands were visualized using a chemiluminescent substrate (P0018AS, Beyotime). The procedures for measuring GAPDH (1:2000, ab181602, Abcam) were the same as described previously, and the protein bands were analyzed and quantified using ImageJ software.
Statistical analysis
Data analysis was performed using SPSS 17.0, and the measurement data were presented as mean ± standard deviation. Comparisons between groups were conducted using two-way repeated measures analysis of variance, and a p value of less than 0.05 was considered to indicate statistical significance.
Results
Establishment of the MI model and survival condition
Establishment of MI model
The ligation site was determined to be 2 mm beneath the junction of the pulmonary tricuspid valve and left atrial entrance. Successful model establishment was indicated by a color change in the LV myocardium from red to white, reduction in myocardial contractility, or elevation in the S-T segment on the electrocardiogram, as illustrated in Figure 1.
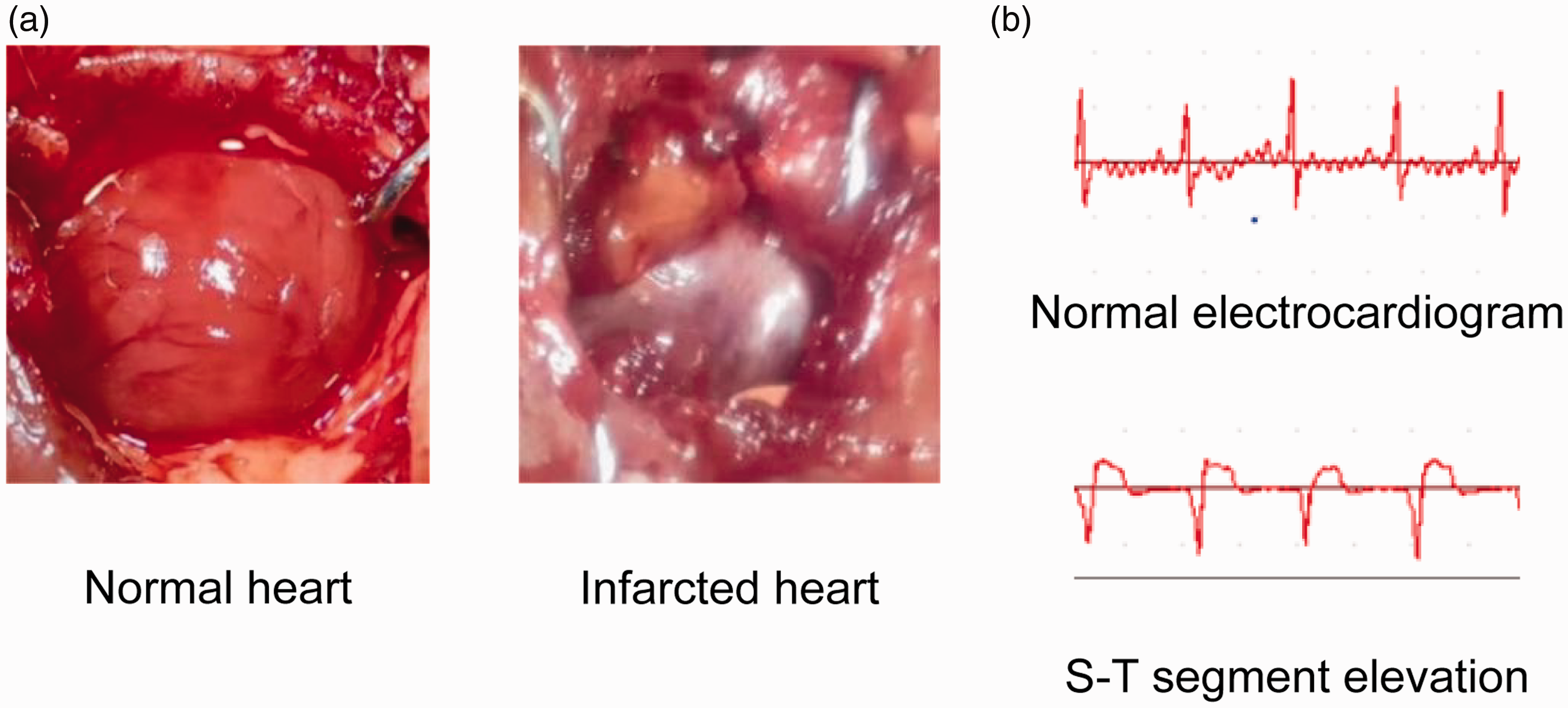
Signs of successful modeling. (a) The myocardial color changed from red to white and (b) S-T segment was elevated.
Survival condition
During the ligation of the left anterior descending coronary artery, four rats died; however, all rats survived throughout the gavage phase.
Echocardiography
Structural alterations in rat hearts were assessed using M-mode echocardiography after 4 weeks. Heart rates during echocardiography were monitored and showed no significant differences between groups. The evaluated critical parameters are shown in Figure 2. The administration of EMPA notably improved the structural changes observed in the left ventricle. Both LVIDd and LVIDs improved in the low-EMPA and high-EMPA groups compared with the MI group, with no statistically significant difference between the low-EMPA and high-EMPA groups (p > 0.05). Additionally, both the low-EMPA and high-EMPA groups showed a significant enhancement in EF compared with the MI group (p < 0.05). The MI, low-EMPA, and high-EMPA groups showed significantly lower LVFS than the sham group (p < 0.05), with no significant difference in efficacy between the high- and low-EMPA groups (p > 0.05).

Structural changes in the heart measured using M-mode echocardiography and electrical stimulation results. (a) Images of the ultrasonic cardiogram. (b) Cardiac echocardiography parameters of the experimental rats, including LVIDd, LVIDs, EF (%), LVFS (%), (n = 4 per group). (c) Electrocardiogram of ventricular fibrillation after electrical stimulation and the ventricular fibrillation threshold of MI rats. The data are presented as the mean ± SD. n1 (sham group) = 4; n2 (MI group) = 10; n3 (low-EMPA group) = 4; n4 (high-EMPA group) = 4. Two-way repeated measures ANOVA was performed. # Means p ≤ 0.05 vs. sham group, *Means p ≤ 0.05 vs. MI group. ANOVA: analysis of variance; EF: ejection fraction; LVIDd: left ventricular internal diameter at end-diastole; LVIDs: left ventricular internal diameter at end-systole; LVFS: left ventricular fractional shortening; VF: ventricular fibrillation.
VF threshold
As shown in Figure 2, the experimental findings indicated that the VF thresholds in the MI group were significantly lower than those observed in the sham group (p < 0.05). A comparison between the MI and treatment groups (low-EMPA and high-EMPA) revealed that both EMPA treatment groups exhibited higher VF thresholds than the MI group. However, the difference in VF thresholds between the low- and high-EMPA groups was not statistically significant (p > 0.05).
Histomorphology
As illustrated in Figure 3, H&E staining demonstrated that the cardiomyocytes in the sham group exhibited a well-organized arrangement, with centrally positioned and distinctly visible nuclei, accompanied with minimal inflammatory cell infiltration. Conversely, the MI group displayed a disorganized architecture of cardiomyocytes, solidified nuclei, a distorted structure of myocardial fibers, and a notable infiltration of inflammatory cells. Both the low-EMPA and high-EMPA groups demonstrated enhancements in myocardial organization and a reduction in the number of inflammatory cells, with the high-EMPA group exhibiting more pronounced benefits.

Empagliflozin (EMPA) ameliorated myocardial inflammatory cell infiltration and fibrosis in MI rats and regulated the expression of miR-181a, TGF-β and Smad3. (a) Representative images of the left ventricle stained with hematoxylin and eosin. (b) Masson’s trichrome staining of myocardial fibrosis and CVF. (c) Relative miR-181a mRNA expression and (d) Western blotting of TGF-β and Smad3. The data are presented as the mean ± SD. n1 (sham group) = 4; n2 (MI group) = 10; n3 (low-EMPA group) = 4; n4 (high-EMPA group) = 4. Two-way repeated measures ANOVA was performed. # Means p < 0.05 vs. sham group, * Means p < 0.05 vs. MI group. ANOVA: analysis of variance; CVF: collagen volume fraction; MI: myocardial infarction; TGF-β: transforming growth factor β.
Masson’s trichrome staining of the rat myocardium, observed under a light microscope, is shown in Figure 3. In the sham group, cardiomyocytes were arranged in a regular pattern, exhibiting minimal collagen fibers and slight myocardial fibrosis. In contrast, the MI group showed a disordered arrangement of cardiomyocytes, increased collagen fibers, and significant myocardial fibrosis. Treatment with low EMPA resulted in an improved arrangement of cardiomyocytes and a reduction in the area occupied by collagen fibers, consequently decreasing myocardial fibrosis. High-EMPA treatment further diminished the collagen fiber area. Statistical analysis indicated that the level of myocardial fibrosis was significantly higher in the MI group than in the sham group (p < 0.05). Both the EMPA-treated groups demonstrated a reduction in myocardial fibrosis relative to the MI group (p < 0.05), with no statistically significant difference observed between the two EMPA groups (p > 0.05).
TGF-β1 and Smad3 expression
As illustrated in Figure 3, western blot analysis indicated that the expression levels of TGF-β1 and Smad3 markedly increased in the myocardial tissues of rats in the MI group compared with those in the sham group (p < 0.05). This finding suggests the upregulation of TGF-β1 and Smad3 expression in the presence of MI lesions. When comparing the MI group with the low- and high-EMPA groups, a reduction in the expression of TGF-β1 and Smad3 was observed in the myocardial tissues at the LV infarct margins of the treated rats (p < 0.05). This indicated that varying doses of EMPA were effective in inhibiting the expression of TGF-β1 and Smad3, although no significant difference was found between the low- and high-EMPA groups (p > 0.05).
Figure 3 shows the levels of miR-181a mRNA quantified using qPCR. The expression of miR-181a was significantly elevated in the MI group compared with that in the sham group (p < 0.05). In addition, both EMPA-treated groups displayed a statistically significant decrease in the expression levels of miR-181a-1 compared with the MI group (p < 0.05).
Discussion
MI is a cardiovascular condition characterized by significant mortality and disability rates. 1 Among the various complications associated with MI, VAs, particularly VF, are a critical factor contributing to mortality in affected patients. 2 The development of myocardial fibrosis is intricately linked to the occurrence of VAs following MI. 3 As a novel class of hypoglycemic agents, SGLT2i not only demonstrate considerable efficacy in reducing blood glucose levels but also exhibit protective cardiovascular effects. 21 The cardiovascular benefits associated with SGLT2i manifest in several dimensions. 22 Nonetheless, there is a lack of research dedicated to examining the influence of EMPA on arrhythmias subsequent to MI. Our findings indicated that EMPA can significantly mitigate myocardial fibrosis following MI, leading to marked enhancements in cardiac functionality and a decrease in the incidence of VF.
The TGF-β1/Smad signaling pathway is crucial for the promotion of myocardial fibrosis. TGF-β1 facilitates the production of ECM, drives the differentiation of myocardial fibroblasts, and enhances collagen synthesis. 3 Myocardial fibrosis serves as the foundation for structural remodeling of the heart, which not only diminishes cardiac efficiency but also disrupts the organization of cardiomyocyte arrangement, resulting in abnormal local myocardial conduction. 2 This conduction heterogeneity increases the likelihood of arrhythmia. Recent investigations have illustrated that SGLT2i can attenuate the extent of fibrosis following MI by inhibiting the TGF-β1/Smad3 pathway, suggesting a prospective role for SGLT2i in counteracting myocardial fibrosis. However, despite the growing body of research on the antifibrotic effects of SGLT2i, no study has conclusively demonstrated that SGLT2i can diminish the occurrence of postinfarction VF by ameliorating myocardial fibrosis.
miRNAs, which measure approximately 20–22 nucleotides in length, are small noncoding RNA molecules that are observed in various organisms, including animals, plants, and viruses. 23 miRNAs play a pivotal role in regulating essential genes and signaling pathways associated with myocardial fibrosis by influencing the activation, differentiation, and ECM production of fibroblasts.24,25 Among these, miR-181a has emerged as a significant miRNA that has a marked effect on gene regulation. Recent investigations have underscored the vital contributions of miR-181a to the progression of cancer and cardiovascular conditions. 26 Notably, a study by Zhu et al. 27 revealed that circulating levels of miR-181a in patients with MI exhibit significant temporal fluctuations, suggesting that plasma concentrations of miR-181a serve as a promising biomarker for MI. Furthermore, research has highlighted the involvement of miR-181a in the TGF-β1/Smad signaling pathway.18,19 In a study conducted by Chen et al., it was demonstrated that miR-181a exacerbates myocardial fibrosis following infarction in rat models by inhibiting TGF-β receptor III (TGF-βRIII), thus emphasizing the critical role of miR-181a in the context of myocardial fibrosis. Interestingly, miR-181a has also been noted for its varied functions in the proliferation and differentiation of tumor cells. Bi et al. 18 reported that overexpression of miR-181a-5p inhibited the expression of early growth response factor 1, which resulted in downregulation of the TGF-β1/Smad pathway and subsequent suppression of tumor cell proliferation in hepatocellular carcinoma. In contrast, Xu et al. 19 found that miR-181a promotes epithelial–mesenchymal transition in esophageal squamous cell carcinoma through the TGF-β/Smad pathway, where elevated levels of miR-181a are associated with increased TGF-β1 expression, thereby facilitating enhanced cell migration and proliferation. Although these studies have provided evidence for the involvement of miR-181a in myocardial fibrosis, none of them have explicitly confirmed its effects via the TGF-β1/Smad3 pathway. Additionally, there is a lack of research addressing the potential role of SGLT2i in the downregulation of miR-181a in cardiomyocytes.
In the present study, echocardiographic assessments conducted on rats treated with EMPA revealed notable enhancements in parameters such as LVIDs, LVIDd, LVEF, and LVFS. This beneficial effect is likely attributable to the attenuation of myocardial fibrosis. The results of Masson’s trichrome staining further indicated that EMPA significantly diminished both the area occupied by collagen fibers and the collagen volume fraction within the myocardial tissues of postinfarction rats. Additionally, our findings suggest that EMPA can lower the VF threshold in postinfarction rats through its action on myocardial fibrosis. Electrical stimulation experiments demonstrated that EMPA markedly elevated the VF threshold in these rats, thereby mitigating the risk of arrhythmia. This observation aligns with the varying degrees of myocardial fibrosis recorded in each subgroup, as indicated by the Masson’s trichrome staining results obtained in our experiments.
Further analyses confirmed that EMPA inhibited the TGF-β1/Smad3 signaling pathway by downregulating miR-181a. Western blotting and qPCR assays revealed that the expression levels of miR-181a, TGF-β1, and Smad3 were significantly elevated in the myocardial tissue cells of rats in the infarction group compared with those in the sham group. However, following EMPA treatment, a significant reduction in TGF-β1 and Smad3 expression was observed in both the low- and high-EMPA treatment groups. Additionally, the notable decrease in miR-181a expression within the EMPA-treated cohorts further supports the notion that EMPA exerts its inhibitory effects on the TGF-β1/Smad3 pathway by downregulating miR-181a.
In summary, our study shows that EMPA has the potential to ameliorate myocardial fibrosis subsequent to MI by modulating the expression of miR-181a and the TGF-β1/Smad3 signaling pathways. Compared with previous studies, our study identifies miR-181a as a novel mediator of TGF-β1/Smad3-driven fibrosis in post-MI arrhythmogenesis. This leads to marked improvements in cardiac functionality and a reduction in the incidence of VF. Thus, EMPA may represent a promising therapeutic option for enhancing the outcomes of VF post-MI, providing a robust theoretical foundation for clinical application.
Study limitations
There may be an interaction between SGLT2i and miR-181a in myocardial fibrosis therapy. The current study revealed the potential roles of SGLT2i, miR-181a, and the TGF-β1/Smad3 pathway in myocardial fibrosis therapy. However, these studies are in their preliminary stages, and more experiments are needed to confirm these findings. In addition, the small sample size (n = 4/group) limits the generalizability of our findings. Future studies should validate these findings in larger cohorts and explore sex-specific effects. The next steps include investigating EMPA’s impact on long-term arrhythmia recurrence in diabetic MI models.
Conclusion
Empagliflozin ameliorated myocardial fibrosis after MI associated with modulation of miRNA-181a and TGF-β1/Smad3 pathways, resulting in significant improvement in cardiac function and reduction in the occurrence of VF.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251353040 - Supplemental material for Empagliflozin attenuates ventricular fibrillation postmyocardial infarction associated with reduced transforming growth factor-β 1/Smad3 signaling and miR-181a expression
Supplemental material, sj-pdf-1-imr-10.1177_03000605251353040 for Empagliflozin attenuates ventricular fibrillation postmyocardial infarction associated with reduced transforming growth factor-
Footnotes
Acknowledgement
The authors thank Dr. Xiaoqiu Tan (Key Laboratory of Medical Electrophysiology of Ministry of Education and Medical Electrophysiological Key Laboratory of Sichuan Province, Collaborative Innovation Center for Prevention and Treatment of Cardiovascular Disease, Institute of Cardiovascular Research, Southwest Medical University) for providing the experimental platform.
Author contributions
JS was responsible for designing experiments, conducting experiments, and writing articles. MH, YJ, MD, ZX, and ZY performed the experiments. TL was responsible for guiding the experiment and reviewing the article. QY was responsible for designing experiments, guiding the experiment, and reviewing the article.
Availability of data and materials
The data used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors have no relevant financial or nonfinancial interests to disclose.
Ethics approval and consent to participate
All animal procedures conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institute of Health (8th edition, 2011) and were approved by the Institutional Animal Care and Treatment Committee of Southwest Medical University, China.
Funding
This study was supported by the Medical Research Project of Sichuan Province (S22091), Medical Research Project of Sichuan Province (S21111), and Open Fund of Medical Electrophysiological Key Laboratory of Sichuan Province (KeyME-2018-10).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
