Abstract
Background:
Macrophages-mediated inflammation is involved in the progress of colitis. The present study aims to explore the roles of A20-binding inhibitor of NF-κB (ABIN1) in the macrophages and its underlying mechanisms.
Methods:
ABIN1 myeloid cell-conditional transgenic mice were established and genotyped by PCR and immunoblotting assays. Tumor necrosis factor (TNF)-α was applied to pre-treat bone marrow-derived macrophages (BMDMs) in the presence of lipopolysaccharide. The mRNA and protein levels of pro-inflammatory cytokines were determined by qRT-PCR and ELISA, respectively. Dextran sulfate sodium (DSS)-induced colitis was established to determine the effects of ABIN1 on the survival time, body weight, colon length, and colon histopathological changes. Western blotting was applied to determine the expressions of signaling proteins.
Results:
ABIN1 overexpression did not affect cell populations of macrophages and neutrophils in mice. Its overexpression reduced the productions of pro-inflammatory cytokines in BMDMs and ameliorated survival rate and colitis symptoms in the DSS-induced mouse model. The underlying mechanisms revealed that ABIN1 impaired macrophages-mediated inflammatory responses, in part by regulating the NF-κB signal pathway, and its ameliorated effects on the symptoms of DSS-induced colitis were associated with A20/tumor necrosis factor α-induced protein 3 (TNFAIP3).
Conclusion:
ABIN1 attenuated inflammatory responses and colitis by regulating A20/TNFAIP3 activities.
Introduction
Colitis is an inflammatory reaction that occurs in the inner lining of the colon. It can be caused by autoimmune disorders, infections, vascular diseases, certain medications, and other unknown reasons. 1 The symptoms of colitis include weight loss, abdominal pain, and diarrhea. 2 As a chronic disorder, colitis can gradually develop into more severe disease, including colon cancer. 3 Inflam-matory response plays an important role in the occurrence and development of colitis. 4 Many studies have demonstrated that inhibition of the inflammatory response is an effective strategy for the treatment of colitis. 5
The roles of macrophages in inflammation have been well elucidated. 6 Macrophages are important effector cells involved in innate immune responses via engulfing bacteria and secreting inflammatory cytokines and mediators. 7 In addition, it is well known that macrophages are responsible for maintaining immune homeostasis in the colon. 8 In healthy conditions, macrophages in the colon respond appropriately to stimuli and microbes in the luminal cavity. However, when homeostasis is disturbed, the activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) in the macrophages boost the transcription of inflammatory cytokines, including interleukin (IL)-6, IL-23 and inflammatory mediators such as nitric oxide synthase (NOS2).8–11 These processes thereby contribute to the pathogenesis of colitis. Many studies have supported the view that macrophages serve as a new class of target for the regulation of intestinal immunity. For instance, Jones and colleagues demonstrated that mouse macrophages are major resources of chemokines, including chemokine (C-C motif) ligand (CCL)-7 and CCL-8, and that targeting macrophages ameliorates the chronic inflammation symptoms of inflammatory bowel disease (IBD). 12 These results demonstrate that targeting macrophages might be an effective strategy for the treatment of colitis.
It is known that NF-κB regulates gene transcription of inflammatory cytokines and mediators, and functions as a master regulator in immune cells, including monocytes, macrophages, dendritic cells, and T cells.13,14 Activation of the IκB kinase (IKK) complex results in the phosphorylation and degradation of IκB proteins. IκB degradation reveals a nuclear localization sequence on NF-κB proteins, allowing the p50–p65 heterodimer to migrate into the nucleus, bind to specific κB-sites, and regulate target gene transcription. 15 However, the activities of NF-κB can be inhibited by A20-binding inhibitor of NF-κB (ABIN1) – also known as a polyubiquitin binding protein – serving as an inhibitor of NF-κB. 16 A20 (TNFAIP3) is a susceptibility gene for inflammation that is involved in regulation of ABIN1. Although the functional roles of A20 and polyubiquitin binding are unclear, many studies have suggested that A20 participates in the inhibitory mechanism of ABIN1. 17 Disruption of ABIN1–polyubiquitin binding promotes NF-κB and pro-inflammatory signaling, thereby enhancing production of pro-inflammatory mediators and cytokines. 17 Previous studies have also demonstrated that ABIN1 is involved in autoimmunity, allergic inflammation, and intrinsic glomerular inflammation.18–20 However, the roles of ABIN1 in colitis are still unknown. Therefore, the present study was designed to explore the role of ABIN1 in colitis and its underlying mechanisms. As discussed, macrophage-mediated inflammatory responses have been identified to be crucial for colitis.7,10 NF-κB serves as a master for the transcription of inflammatory cytokines and mediators in macrophages. 14 Therefore, the present study aimed to investigate the roles of ABIN1 in macrophage-mediated inflammatory responses in colitis.
Materials and methods
Antibody and reagents
Anti-IκBα (C-21, 1:1000), anti-JNK2 (C-17,1:1000), and anti-IKKα (H-744, 1:1000) were from Santa Cruz Biotechnology (Dallas, TX, USA). Anti-phospho-IκBα (Ser32, 14D4, 1:1000), anti-phospho-p38 (Thr180/Tyr182, 3D7, 1:1000), phospho-JNK (Thr180/Tyr185, 1: 1000), anti-phospho-IKKα/β (Ser176/180, 16A6, 1:1000), and anti-ABIN-1 (1:1000) were purchased from Cell Signaling Technology (Danvers, MA, USA). Anti-actin (C-4, 1:10000) and lipopolysaccharides (LPS, Escherichia coli 055: B5) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Tumor necrosis factor (TNF) was purchased from Peptech (Burlington, MA, USA).
Generation of transgenic mice
Tnfaip3fl/fl (A20) mice with the C57BL/6J genetic background were constructed by the Shanghai Yunhao Biotechnology Center (Shanghai, China). Lyz2-Cre mice with the C57BL/6J genetic background were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). The animals were kept under temperature- and humidity-controlled conditions (temperature 22–24°C, humidity 60 ± 5%).
As shown in Figure 1A, to generate myeloid cell conditional Tnfaip3 KO (Tnfaip3f/fLyz2Cre/+) mice, Tnfaip3fl/fl mice were crossed with Lyz2-Cre mice. The ABIN1 transgenic mice were generated by the pronuclear microinjection of fertilized FVB/N eggs. Mice homozygous for the ABIN1 allele are viable and fertile, with a loxP-flanked Neo-STOP cassette preventing transcription of downstream bicistronic ABIN1 sequences. Tnfaip3fl/fl mice were recognized by PCR using the following primers: forward: 5′ ATAACTTCGTATAATGTATGCTATACGAAGTTAT 3′; reverse: 5′ CCTCCAGAAGCTCCCCTAACATCTT 3′. Reactions were carried out in a T3000 thermocycler (Biometra, Göttingen, Germany) using the following cycling conditions: 2 min at 95°C followed by 40 cycles of 95°C for 30 s, 55°C for 1 min, and 72°C for 1 min. A final extension step at 72°C for 5 min was performed after the cycles. When bred to mice that express Cre recombinase, offspring will have the STOP cassette deleted, with subsequent expression of ABIN1 in Cre-expressing cells. The transgenic mice were bred in a specific pathogen-free facility. PCR analysis was applied for genotyping. Animal studies were performed in strict accordance with National Institutes of Health (NIH) guidelines for the care and use of laboratory animals. All efforts were made to minimize the number of animals used and their suffering. Animal studies were approved by the ethics committee of the First Affiliated Hospital of Zhengzhou University (ZDYFY-2432).

ABIN1 overexpression does not affect the development of BMDMs and neutrophils. (A) Schematic diagram showed the process of generation of ABIN1 myeloid cell-conditional transgenic mice. (B) PCR was applied to identify the genotype of ABIN1 myeloid cell-conditional transgenic mice. (C) Immunoblotting assays were performed to determine the specific expression of ABIN1 in the macrophages and T cells from MTg mice. (D) Flow cytometry analysis was performed to identify BMDMs and neutrophils. Data were presented as mean ± SEM.
Dextran sulfate sodium-induced colitis mouse model
A dextran sulfate sodium (DSS)-induced colitis mouse model was established according to a previously reported method. 21 In brief, the mice were administered autoclaved drinking water containing 3.5% DSS for 6 days. After that, the DSS water was replaced by normal drinking water. The mice were monitored every 2 days and body weight was recorded. The experimental period was 10 days. At the end of the experimental period, the mice were sacrificed and the colon was collected. Colon length was measured and histopathology examinations were performed. The histopathological evaluation of the specimens was performed by a trained and blinded gastrointestinal (GI) pathologist using the following scoring: Erosion/ulceration: 5–10% (1), 20–30% (2), 40–50% (3), 60–70 (4), 80–90 (5), 100% (6); inflammation severity: minimal (1), mild (2), moderate (3), severe (4), very severe (5); mural involvement: mucosa (1), submucosa (2), tunica muscularis (3), serosa (transmural) (4), transmural reaching mesentery (steatitis) (5); damage distribution:,25% (1); 26–50% (2); 51–75% (3); 75% (4).
For the survival experiment, the mice were administered autoclaved drinking water containing 3.5% DSS for 6 days. After that, the DSS water was replaced by normal drinking water. The observations were performed every 2 days and the survival rate was recorded.
Bone marrow-derived macrophage isolation and culture
Bone marrow-derived macrophages (BMDMs) were isolated from the wild-type C57BL/6J mice or ABIN1-transgenic mice according to a previously reported method. 22 The cells were cultured in Dulbecco’s modified eagle medium (DMEM) containing 10% fetal bovine serum with macrophage colony-stimulating factor M-CSF (10 ng/ml) for 5 days. After that, the differentiated cells were stained with Pacific blue-conjugated anti-F4/80 and analyzed using Flow cytometry (BD FACSCalibur, San Jose, CA, USA).
BMDMs were treated with LPS for the indicated time according to the experimental design. Besides, BMDMs were pretreated with TNFα for the indicated time followed by the stimulation of LPS.
qRT-PCR
An RNA extraction kit (Qiagen, Chatsworth, CA, USA) was used to isolate RNA from the BMDMs, according to the manufacturer’s protocol. Reverse transcriptase was used in the room temperature reaction. The primers for the target genes were as follows: IL-6 forward: 5′-CAC AGA GGA TAC CAC TCC CAA CA-3′, and reverse: 5′-TCC ACG ATT TCC CAG AGA ACA-3′; Nos2 forward: 5′-GTG ACA AGC ACA TTT GG-3′, and reverse: 5′-AAG GCC AAA CAC AGC ATA CC-3′; IL-23α forward: 5′-GCC AAG ACC ATT CCC GA-3′, and reverse: 5′-TCA GTG CTA CAA TCT TCA GAG GAC A-3′. Melt curves were used to analyze accuracy. The expression of each gene was calculated using 2−△△Ct values. The mRNA expression values of target genes were normalized to that of glyceraldehyde 3-phosphate dehydrogenase (GAPDH).
Enzyme-linked immunosorbent assay
The levels of IL-6 and IL-23 in the supernatant were determined using enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions (R&D Biosystems, Minneapolis, MN, USA).
Western blotting
Protein was extracted according to previously described methods.23,24 In brief, a cold radioimmunoprecipitation assay buffer (RIPA) buffer containing protease inhibitor was used to lyse the cells. After that, the extraction buffer was centrifuged at 13,000 g for 15 mins to remove the sample debris and other insoluble materials. Bicinchonic acid (BCA) protein assay kits were applied to quantify the concentrations of extracted proteins.
An equal amount of proteins was loaded and separated using 10% SDS gel. After that, the gel was transfer to a polyvinylidene fluoride (PVDF) membrane, which was blocked with 5% non-fat milk at room temperature for 2 h. Next, a primary antibody was used to incubate with the membrane at 4°C overnight. Appropriated secondary antibodies conjugated with HRP were used. β-actin was applied as an internal control. The imaging system was applied to qualify the expression of target genes.
Statistical analysis
Data were shown as mean ± standard error of the mean (SEM). One-way analysis of variance (ANOVA) with multiple comparisons and the Student-Newman-Keuls (SNK) test were performed. A p value <0.05 was taken to indicate statistical significance between the two groups.
Results
ABIN1 overexpression did not affect development of BMDM and neutrophils
The LysMcre knock-in allele has a nuclear-localized Cre recombinase inserted into the first coding ATG of the lysozyme 2 gene (Lyz2) that abolishes endogenous Lyz2 gene function and places NLS-Cre expression under the control of endogenous Lyz2 promoter/enhancer elements. We first determined genotypes using PCR and immunoblotting assays, respectively. The PCR results showed genotypes of Abin1Tgfl/flLyz2cre/+(MTg), Abin1Tg+/+Lyz2cre/+ (WT), and Het (Abin1Tgfl/+Lyz2cre/+). The upper band indicates the targeted genome with transgenic Abin1; without this sequence, the band appears at a low molecule weight (Figure 1B). Interestingly, we observed that ABIN1 was expressed only in the BMDMs but not in T cells from MTg mice, indicating that an ABIN1 myeloid cell-conditional transgenic mouse had been established successfully (Figure 1C). In order to determine the effects of ABIN1 overexpression on cell populations, we then analyzed populations of CD11b+B220+ macrophages and CD11b+Gr1+ neutrophils in MTg and WT mice (Figure 1D). Populations of CD11b+B220+ macrophages and CD11b+Gr1+ neutrophils showed no significant difference between the MTg and WT mice (Figure 1D), suggesting that ABIN1 overexpression did not affect the development of BMDM and neutrophils.
ABIN1 impaired the induction of pro-inflammatory cytokines
Next, BMDMs from AbinMTg and AbinWT mice were isolated. The cells were induced by LPS for 2 and 6 h, respectively. mRNA levels of IL-6, IL-23α, IL-12α, and Nos2 in the BMDMs between AbinMTg and AbinWT mice showed no significant difference (Figure 2A). It is known that BMDMs exhibit low expression of A20/TNF α-Induced Protein 3 (TNFAIP3). ABIN1 overexpression did not enhance expression of inflammatory cytokines. Therefore, to induce expression of A20/TNFAIP3, we pre-treated BMDMs with TNFα prior to incubation with LPS. Interestingly, the results showed that the mRNA levels of IL-6, IL-23α, IL-12α, and Nos2 in BMDMs were decreased significantly in AbinMTg mice compared with those in AbinWT mice (Figure 2B). Moreover, we also determined secretion of IL-6 and IL-23. The results demonstrated that the protein levels of IL-6 and IL-23 in BMDMs decreased significantly in AbinMTg mice compared with those in AbinWT mice (Figure 2C). These results suggested that ABIN1 overexpression impaired the production of pro-inflammatory cytokines.

ABIN1 impairs the induction of pro-inflammatory cytokines. (A) qRT-PCR was performed to determine the mRNA levels of pro-inflammatory cytokines in primary BMDMs induced by LPS (100 ng/ml). (B) In addition, WT and ABIN1-transgenic BMDMs were pretreated with TNF for 24 h. After that, the mRNA levels of il6, il23, il12 and Nos2 in BMDMs induced by LPS (100 ng/ml) were determined by qRT-PCR. (C) ELISA was performed to determine the protein levels of IL-6 and IL-23. All data were presented as mean ± SEM.
ABIN1 overexpression in macrophages ameliorated symptoms of DSS-induced colitis
To confirm the effects of ABIN1 overexpression on the expression pattern of pro-inflammatory cytokines in vitro, we further investigated the effects of ABIN1 overexpression on the symptoms of DSS-induced colitis in mice. First, we observed that the survival time of AbinMTg mice was significantly prolonged as compared with that of AbinWT mice (Figure 3A, p = 0.014). We then measured the body weight and colon length in the AbinMTg and AbinWT mice induced by DSS. The results showed that both body weight and colon length were significantly decreased in AbinWT mice compared with AbinMTg mice (Figure 3B and C). Disease activity index, taking into account body weight loss, severity of diarrhea, and blood in the stool confirmed the conclusion that AbinMTg mice displayed a resistance to DSS-induced colitis (Figure 3D). In addition, histopathology examinations also showed that severe infiltration of leukocytes in the colon tissue of AbinWT mice, whereas fewer numbers of leukocytes were observed in the colon tissue of AbinWT mice (Figure 3E). Furthermore, we sorted colon CD11b+F4/80+ macrophages using flow cytometry, and determined the mRNA levels of IL-6, IL-23α, and Nos2. The results showed that mRNA levels of IL-6, IL-23α, TNF-α, IL-1β, and Nos2 were significantly decreased in the AbinMTg mice as compared with those in the AbinWT mice (Figure 3F). However, IL-10 displayed an increase in AbinMTg mice. These results support the view that ABIN1 overexpression in macrophages ameliorated the symptoms of DSS-induced colitis.
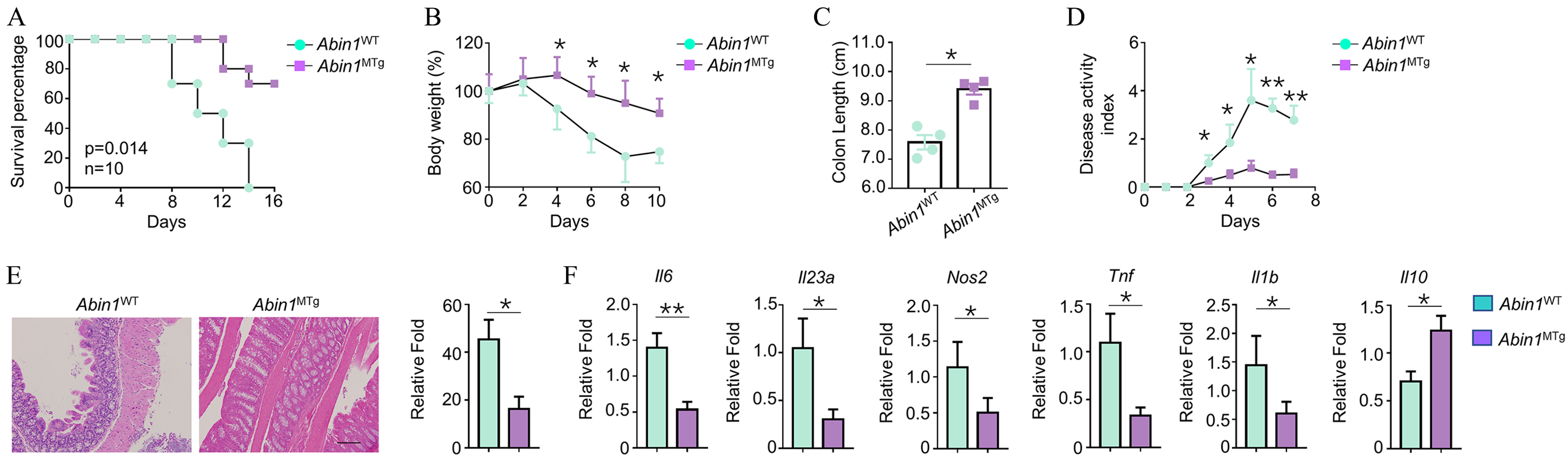
ABIN1 overexpression ameliorates symptoms of DSS-induced colitis. WT and ABIN1MTg mice were treated with 3% (for body weight loss) or 3.5% DSS (for survival rate) (in drinking water) for 5 days and then supplied with normal drinking water. After that, survival rate (A) and body weight loss (B) were recorded. In addition, after the mice were sacrificed, colon length was measured as indicated (C). (D) WT and ABIN1MTg mice were treated with 3% DSS as described in (B). Disease activity index of DSS-treated mice was evaluated daily. (E) Histopathology examinations were performed on day 10 (scale bar, 100 μm). (F) qRT-PCR was performed to determine the mRNA levels of il6, il23, Tnf, Il1b, Il10, and Nos2 in CD11b+F4/80+ macrophages. All data are presented as mean ± SEM.
ABIN1 impairs activation of the NF-κB signal pathway
We noticed that TNF-α induced expression of A20/TNFAIP3. Also, the effects of ABIN1 on the expression of inflammatory cytokines are associated with A20/TNFAIP3. Therefore, we first determined the effects of ABIN1 on expression of A20/TNFAIP3 in the presence of TNF-α. The mRNA levels of A20/TNFAIP3 in AbinMTg and AbinWT showed no significant difference (Figure 4A). Accordingly, we also found that phosphorylation of mitogen-activated protein kinase (MAPK) family proteins, including p38, ERK1/2, and JNK2 was similar in the TNF-α-pretreated BMDMs from AbinMTg and AbinWT mice (Figure 4C). Interestingly, we noticed that the phosphorylation of NF-κB signaling proteins including IκBα and IKKα/β was significantly reduced in BMDMs from AbinMTg mice (Figure 4B), suggesting that ABIN1 regulated the expression of inflammatory cytokines by impairing activation of the NF-κB signal pathway.
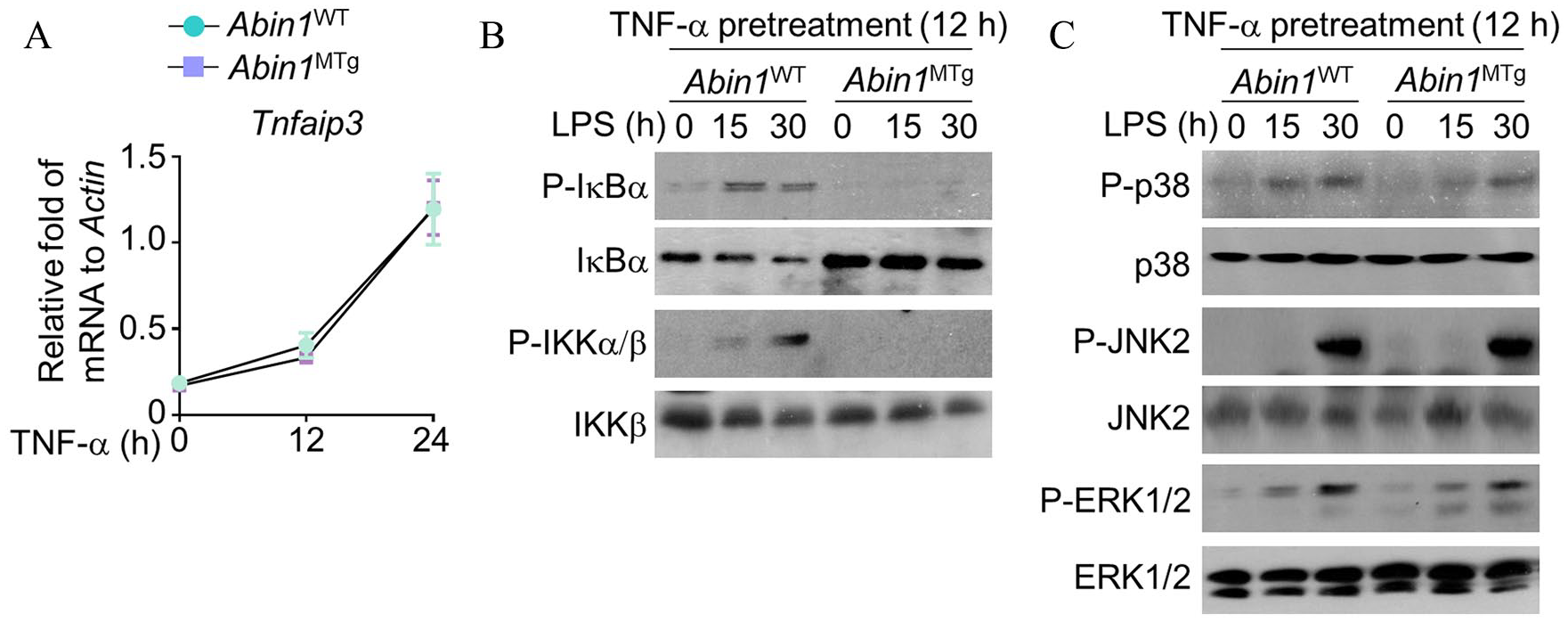
ABIN1 impairs activation of the NF-κB signal pathway. (A) qRT-PCR was performed to determine mRNA levels of A20 in primary BMDMs induced by TNF. (B,C) WT and ABIN1-transgenic BMDMs were pretreated with TNF for 12 h. After that, the cells were stimulated by LPS (1 μg/ml) at different time points, and the activation of NF-κB associated proteins, p38, ERK1/2, and JNK2, was determined using Western blotting.
ABIN1 affects colitis via regulation of A20/TNFAIP3 activity
To identify the roles of A20/TNFAIP3 in ABIN1-mediated regulatory effects in inflammatory responses, we blocked expression of A20/TNFAIP3 in the mice by crossing ABIN1MTg mice with A20MKO mice. The results showed the A20 was expressed only in A20WT ABIN1MTg mice, but not in A20KO ABIN1MTg mice (Figure 5A). We then analyzed the mRNA levels of IL-6, IL-23α, and Nos2. The results showed that mRNA levels of IL-6, IL-23α, and Nos2 were decreased significantly in the ABIN1MTg A20WT mice as compared with those in the ABIN1WT A20WT mice. However, we did not observe a significant difference of IL-6, IL-23α, and Nos2 mRNA levels in ABIN1MTg A20MKO and ABIN1WT A20MKO mice (Figure 5B). These results support the view that ABIN1 affects inflammatory responses by regulation of A20/TNFAIP3 activity. Moreover, we observed that the survival time of DSS-induced ABIN1MTg A20MKO mice was prolonged as compared with that of ABIN1WT A20MKO mice, but no significance was observed (Figure 5C, p = 0.267). However, the body weight and colon length between ABIN1MTg A20MKO and ABIN1WT A20MKO mice showed no significant difference (Figure 5D and 5E). As observed in vitro, no significant difference in IL-6, IL-23α, Tnf, Il1b, and Nos2 mRNA levels in isolated macrophages from colon were found between ABIN1MTg A20MKO and ABIN1WT A20MKO mice (Figure 5F).

ABIN1 affects colitis by regulation of A20 activity. (A) Primary BMDMs from A20MKO ABIN1MTg or A20WT ABIN1MTg were stimulated with TNF. Next, A20 protein levels were determined by Western blotting. (B) mRNA levels of il6, il23, and Nos2 were determined using qRT-PCR in TNF-pretreated BMDMs stimulated with LPS (100 ng/ml). (C–E) WT and ABIN1MTg A20MKO mice were treated with 3% (for body weight loss) or 3.5% DSS (for survival rate) (in drinking water) for 5 days and then supplied with normal drinking water. Survival rate (C), body weight loss (D), and colon length (E) were evaluated. (F) qRT-PCR was performed to determine the mRNA levels of il6, il23 Tnf, Il1b, and Nos2 in CD11b+F4/80+ macrophages. Data are presented as mean ± SEM.
Discussion
In the present study, for the first time, we explored the roles of ABIN1 in the DSS-induced colitis mouse model. The results demonstrated that ABIN1 overexpression reduced the production of inflammatory cytokines, including IL-6 and IL-23α and inflammatory mediator Nos2, via inhibition of the NF-κB signaling pathways. Interestingly, we also revealed that the effects of ABIN1 on inflammation are A20/TNFAIP3 dependent. In an in vivo study, we observed that ABIN1 overexpression was able to ameliorate the symptoms of colitis, and to prolong the survival time of DSS-induced colitis. These results supported the view that targeting ABIN1 might be a good strategy for the treatment of colitis.
Before we explored the roles of ABIN1 overexpression on macrophage-mediated inflammatory responses, our study first investigated the effects of ABIN1 overexpression on the development of macrophages and neutrophils. This was to exclude that the effects of ABIN1 on macrophage-mediated inflammatory responses are due to changes in the macrophage and neutrophil cell populations. Both macrophages and neutrophils have been demonstrated to play crucial roles in inflammatory responses. 25 Macrophages are the major cell population in the intestinal tissue and their balance is important for maintaining homeostasis immunity. 11 Neutrophils are another myeloid cell compartment in the intestinal tissue, 26 so we also determined the population of neutrophils. The results showed that the populations of CD11b+B220+ macrophages and CD11b+Gr1+ neutrophils between the ABIN1MTg and ABIN1WT mice showed no significant difference, suggesting that ABIN1 overexpression did not affect the development of macrophages and neutrophils.
Macrophages are the gatekeepers of intestinal immune homeostasis. Many studies suggest a causal link between defects in the resolution of intestinal inflammation and altered monocyte-macrophage differentiation in patients with IBDs, and macrophages have been considered as a novel potential target to develop new treatment approaches. 27 Therefore, although ABIN1 is expressed in many types of immune cells, we specifically investigated overexpression of ABIN1 in macrophages in the present study.
We next explored the roles of ABIN1 in the expression of inflammatory cytokine in BMDMs induced by LPS. LPS, a well-characterized polysaccharide, is known to induce an inflammatory response by interaction with toll-like receptors expressed on macrophage surfaces. 28 We measured inflammatory cytokines, including IL-6 and IL-23α. IL-6 plays an important role in the transition from acute inflammatory status and chronic inflammatory status in colitis. 29 High expression of IL-23 is a hallmark of the development of colitis and colon cancer. 30 IL-23 consists of two subunits called p40 (IL-12) and p19 (IL-23α). 30 NOS2 was also determined in the present study as NOS2 is responsible for the production of nitric oxide, and, thereby, contributes to anti-microbial activities. 31 However, initially, we did not observe any significant difference in IL-6, IL-23α, and Nos2 mRNA levels between ABIN1MTg and ABIN1WT derived BMDMs induced by LPS. Previous studies have demonstrated that ABIN1-mediated anti-inflammatory mechanisms are A20/TNFAIP3 dependent. ABIN1 has been reported to be an adaptor protein that links A20/TNFAIP3 to NF-κB mediators.32,33 Therefore, we speculated that ABIN1 overexpression could not regulate the expression of inflammatory cytokines in the absence of A20/TNFAIP3. To confirm our speculation, we pre-treated the BMDMs with TNFα, which could increase the levels of endogenous A20/TNFAIP3. 34 Interestingly, the results showed that the mRNA levels of IL-6, IL-23α, and Nos2 in the BMDMs were significantly decreased in the AbinMTg mice as compared with those in the AbinWT mice. Besides, protein levels of IL-6 and IL-23 were also decreased in AbinMTg-derived BMDMs. It is known that ABIN1 is an inhibitor for NF-κB but not for MAPK family proteins. 35 Therefore, in addition to investigating the expression of inflammatory cytokines and mediators, we also determined the phosphorylation status of NF-κB signaling pathway and MAPK family proteins. As expected, we observed that phosphorylation of the NF-κB signaling pathway was affected by ABIN1 overexpression in the presence of TNFα. These results confirmed that the roles of ABIN1 in the BMDM inflammation are associated with A20/TNFAIP3 (Figure 6).

Schematic figure for the cell signaling cascades underlying mechanism of ABIN1 in LPS-mediated BMDM activation.
We also investigated the roles of ABIN1 in the DSS-induced mouse colitis model, a well-characterized animal model used to study colitis. 21 As discussed, colitis is characterized by the loss of body weight and inflammatory responses in the inner lining of the colon. 21 In the present study, we observed that ABIN1 overexpression ameliorated the symptoms of colitis, including the recover of body weight loss, to reduce leukocyte infiltration in colon tissue, and prolong survival time in DSS-induced colitis. In addition, colon length is measured as a marker of tissue integrity, determined using a measuring scale and analyzed for intestinal damage. 36 Low tissue damage led to less shortening of colon length than in the WT group. In the present study, consistent with lower production of pro-inflammatory cytokines, ABIN1 overexpression also protected against inflammation-induced colon damage. Previous studies have demonstrated that knock-in mice expressing an inactive form of ABIN1 develop lupus-like autoimmune disease in vivo and activation of NF-κB is enhanced in immune cells after toll-like receptor stimulation in vitro. 37 Our results also suggest that targeting ABIN1 may also affect other autoimmune diseases such as lupus.
Finally, to confirm the anti-inflammatory mechanisms of ABIN1 that are A20/TNFAIP3 dependent, we constructed A20/TNFAIP3 conditional knockout mice by crossing ABIN1MTg mice with A20MKO mice. We observed the mRNA levels of IL-6, IL-23α, and Nos2 were significantly decreased in the ABIN1MTg A20WT mice. These phenomena were not observed in ABIN1MTg A20MKO and ABIN1WT A20MKO mice. Moreover, we observed that the survival time of DSS-induced ABIN1MTg A20MKO mice was prolonged as compared with that of ABIN1WT A20MKO mice. These results support the view that ABIN1 affects inflammatory responses by regulating A20/TNFAIP3 activity. On the other hand, one limitation is that only the DSS-induced ABIN1MTg A20MKO mouse model was applied in the current study. To support the current conclusion, additional colitis models are warranted in further studies.
Conclusion
ABIN1 overexpression impaired expression of inflammatory cytokines, including IL-6, IL-23α, and Nos2, in BMDMs, and ameliorated symptoms of DSS-induced colitis. ABIN1 affects the production of inflammatory cytokines in BMDMs in part by regulation of NF-κB signaling proteins. In addition, the effects of ABIN1 on symptoms of DSS-induced colitis were associated with A20/TNFAIP3. These results suggest that ABIN1 might be a target for the treatment of colitis.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 2019 Provincial and ministerial co construction projects of medical science and technology in Henan Province (SB201901047).
