Abstract
Objective
To explore potential mechanisms and effects of gallincin on a mouse model of colitis induced by dextran sulfate sodium (DSS).
Methods
Network pharmacology analysis was used to predict the molecular mechanism of action of gallincin for treatment of colitis. Gallincin was administered orally to mice with DSS-induced colitis. Expression of tumor necrosis factor α (TNF-α), D-lactate, and interleukin-1β (IL-1β) and myeloperoxidase activity were assessed with real-time quantitative PCR and an enzyme-linked immunoassay, respectively. Expression of occludin, zonula occludens 1 (ZO-1), and phosphorylated extracellular signal-regulated protein kinase1/2 (p-ERK1/2) was analyzed with immunohistochemical staining and/or western blot assays.
Results
Using a network pharmacology approach, 12 mapping targets between gallincin and colitis were obtained, including ERK/mitogen-activated protein kinase. Further investigations in an experimental colitis mouse model showed that gallincin significantly ameliorated experimental colitis, reduced D-lactate levels, and remarkably increased occludin and ZO-1 expression, possibly in part by decreasing IL-1β, TNF-α, and p-ERK1/2 levels and inhibiting leukocyte penetration.
Conclusions
Gallincin regulated colonic barrier function and reduced colitis-associated inflammation, suggesting it is a promising drug for the treatment of ulcerative colitis.
Keywords
Introduction
Ulcerative colitis (UC), the most common type of human inflammatory bowel disease (IBD), is a refractory idiopathic disease that mainly affects the colorectal area. 1 Although the definitive etiology of UC remains unclear, numerous studies suggest that impaired colorectal barrier function plays a significant role in the pathogenesis of UC.2,3 In fact, previous studies have implicated proinflammatory cytokines in epithelial barrier dysfunction, which in turn enhances antigenic exposure to underlying immune cells.4,5 Therefore, blocking the vicious chain reaction of intestinal barrier dysfunction and excessive inflammation is of important clinical significance for UC.
Currently, approved drugs for UC mainly include chronic administration of 5-aminosalicylic acid drugs, glucocorticosteroids, immunosuppressants, and anti-tumor necrosis factor α (TNF-α) monoclonal antibodies.1,6 However, these conventional therapies produce variable response rates and are not always advisable as a maintenance therapy because of a variety of systemic adverse reactions and high cost.7,8 Interestingly, traditional Chinese medicine may be a promising complementary treatment for UC.7,9,10 Gallincin (methyl 3,4,5-trihydroxybenzoate), which is extracted from plants, has significant biological properties. Previous studies have shown that gallincin elicits anti-inflammatory effects by inhibiting activation of extracellular-signal regulated protein kinase (ERK). 11 In addition, gallincin has spasmolytic, anti-inflammatory, antidiarrheal, and antioxidant actions.12,13 However, it is still unknown whether intestinal barrier dysfunction can be treated by gallincin. Fortunately, network pharmacology can be used to identify potential mechanisms of traditional Chinese medicine.14,15 Thus, based on network pharmacology, this study was designed to explore whether gallincin protects against experimental UC by inhibiting inflammation and mucosal damage.
Methods
Prediction of targets of gallincin
First, we obtained the simplified molecular input line entry system (SMILES) data for gallincin from the PubChem database. Next, targets of gallincin were predicted using the Swiss Target Prediction Database (http://www.swisstargetprediction.ch/) and Binding Database (http://www.bindingdb.org/bind/index.jsp).16,17 The query keywords were “gallincin” and its synonyms. Homo sapiens was selected in the Swiss Target Prediction Database. Substructure and similarity (≥0.85) were used to identify targets of gallincin in the Binding Database. After removing duplicate targets, the predicted targets of gallincin were obtained.
Retrieval of targets of disease
Therapeutic targets for colitis were retrieved from the Therapeutic Targets Database (http://bidd.nus.edu.sg/group/cjttd/) and DrugBank (https://go.drugbank.com/).18,19 The query keywords were “colitis” or “ulcerative colitis”. Results associated with infectious colitis were removed from the Therapeutic Targets Database and DrugBank results. Retrieved results from the two databases were merged, and non-repetitive targets were selected.
Network construction and pathway analysis
Targets of gallincin and colitis were used to construct an interaction network using Cytoscape software (https://cytoscape.org/, version 3.6.0). Pathway analysis was performed with the ClueGO/Clupedia applications within Cytoscape.20–22 “Homo sapiens” and “GO-Biological Process” were automatically selected. The p-value for statistical significance was kept at 0.01.
Animals
Male BALB/c mice (aged 6–8 weeks, weighing 18–23 g) were obtained from Tengxin Biotechnology (Chongqing, China). Mice were maintained in cages with fixed humidity (40%) and environmental temperature (25°C) and a 12:12-hour dark:light cycle. Water and standard mouse food were provided ad libitum. Animal care and experiments were approved by the Ethical Institution of Guizhou Provincial People's Hospital, Guizhou Medical University (No. 83, East Zhongshan Road, Guiyang 550002, Guizhou Province, China, date of approval: 17 March 2017).
Animal model and drug intervention
Experimental UC was generated by administration of 5% dextran sulfate sodium (DSS; molecular weight 36–50 kDa; MP Biomedicals, Aurora, CO, USA) via an oral route in water for 7 days, followed by 1 day of normal drinking water. Seven days after the induction of colitis, all mice were randomly divided into four groups of eight animals each. Group 1, defined as the control group, was not administered DSS or gallincin; Group 2 was administered saline solution and 5% DSS; Group 3 was administered 5% DSS and low-dose gallincin (7 mg/kg, once daily; Sigma, St. Louis, MO, USA); Group 4, which served as the high-dose gallincin group, received 5% DSS and high-dose gallincin (50 mg/kg, once daily). Drug dosage selection was based on preliminary experiments and the literature. 23 Mice were sacrificed under inhalation anesthesia after 5 days of gallincin intervention.
Assessment of colitis
The disease activity index (DAI) was assessed according to the experimental approach reported by Murthy et al. 24 Briefly, the DAI was determined by scoring changes in weight loss, stool occult blood or gross bleeding, and stool consistency. Hematoxylin and eosin staining was performed according to the conventional procedure used in our previous work. 25 Assessment according to the histological scoring method reported by Ten Hove et al. 26 was completed by two researchers in a double-blind manner. Histological scores were determined according to the degree of inflammation, depth of injury, regeneration, crypt injury, and extent of inflammation.
Enzyme-linked immunosorbent assay (ELISA)
Levels of TNF-α and interleukin-1β (IL-1β) in homogenized colonic tissue were determined with ELISA kits according to the manufacturer’s instructions (Elabscience, Wuhan, China). D-lactate concentrations were also measured with an ELISA kit (Guangshui Biological Technology, Shanghai, China).
Myeloperoxidase (MPO) activity
Leucocyte MPO activity was assessed with a commercially available kit according to the manufacturer’s instructions (CytoStore, Alberta, Canada).
Real-time quantitative PCR
The primer sequences used were as follows:
27
TNF-α: 5′-TCTCTTCAAGGGACAAGGCTG-3′, 5′-ATAG-CAAATCGGCTGACGGT-3′; IL-1β: 5′-TCGCTCAGGGTCACAAAGAAA-3′, 5′-CCATCAGAGGCAAGGA-GGAA-3′. The PCR conditions were as follows: pre-denaturation at 95°C for 10 minutes, denaturation at 95°C for 15 s, and annealing-extension at 60°C for 1 minute, for a total of 40 cycles. The relative TNF-α and IL-
Immunohistochemistry
Colonic specimens (3 µm thick) were successively treated with 10% formalin and paraffin. Next, sections were successively treated in series with ethanol (100%, 95%, 90%, 80%, and 70%), 1.5% hydrogen peroxide, and 3% bovine serum albumin. Then, sections were incubated with a zonula occludens 1 (ZO-1) antibody (1:500, Santa Cruz Biotechnology, Dallas, TX, USA), biotinylated secondary antibody (1:200, Dako, Glostrup, Denmark), diaminobenzidine tetrahydrochloride (Sigma), and 0.03% hydrogen peroxide. Negative controls underwent the same procedure but without the first antibody. Finally, the number of positive cells was evaluated by counting brown-stained cells in five random fields under a Leica microscope (Solms, Germany, 400×).
Western blot analysis
To examine protein expression in colonic tissue, extracts were prepared from colonic tissue, and occludin, ZO-1, and phosphorylated ERK1/2 (p-ERK1/2) levels in the intestinal tract were analyzed by western blot according to our previous study 25 and standard methods 28 . The protein concentration was adjusted to 5 µg/µL. Protein samples were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to polyvinylidene fluoride membranes, which were then blocked with blocking buffer containing 10% skim milk. The membranes were then incubated with rabbit anti-mouse antibodies against occludin (1:100), ZO-1 (1:1000), p-ERK1/2 (1:100), and β-actin (1:150) (Santa Cruz Biotechnology). Next, the membranes were incubated with a biotin-labeled secondary antibody (goat anti-rabbit IgG, 1:2000, Beijing Zhongshan Jinqiao Company, China). The immune complexes were detected with an ECL system (Amersham Pharmacia Biotech Inc, Arlington, USA), and the bands were measured by a Fluorchem imaging system for quantitative densitometric analysis (Alpha Innotech Corp, San Leandro, CA, USA).
Statistical method
All experimental data are presented as the mean ± standard deviation and were analyzed by analysis of variance or Student's t-test. A value of p < 0.05 was considered statistically significant.
Results
Targets of gallincin or colitis
Approximately 146 predicted targets of gallincin (for the chemical structure, see Figure 1) were screened by the Swiss Target Prediction Database and Binding Database (Figure 2). A total of 121 targets of colitis were acquired from the Therapeutic Targets Database and DrugBank (Figure 3).
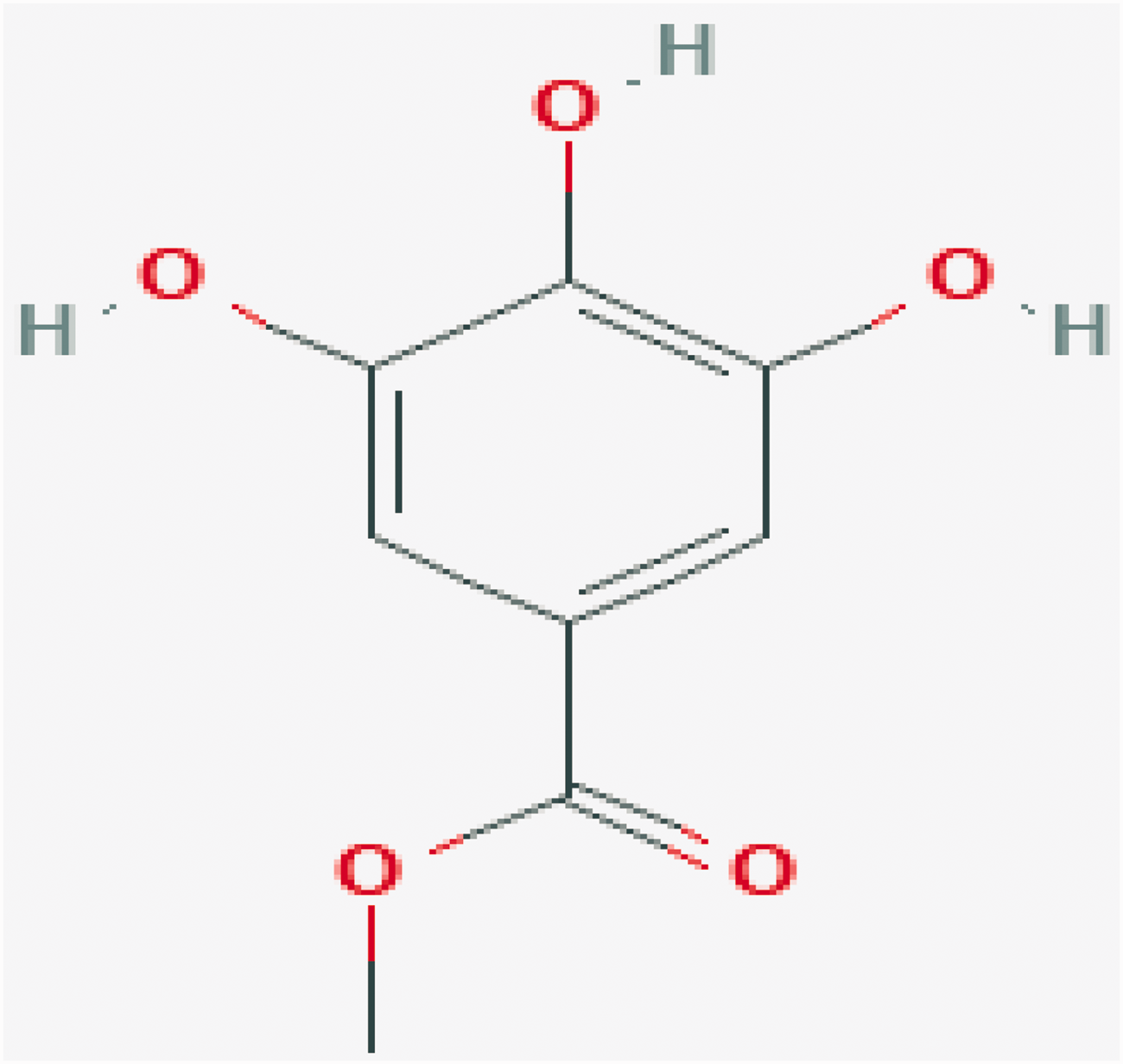
Chemical structure of gallincin.

Gallincin-target interaction network.

Colitis-target interaction network.
Collective targets of gallincin and colitis
Collective targets of gallincin and colitis are shown in Figure 4a and Figure 4b. Twelve mapping targets of colitis and gallincin were obtained (Figure 4a). The resulting protein interaction network, including 269 nodes and 267 edges, is shown in Figure 4b.

Collective targets of gallincin and colitis. (a) Venn diagram of the collective targets. (b) Merged protein–protein interaction network for gallincin and colitis.
Pathway analysis
The pathway analysis results are shown in Figure 5. Statistically significant pathways identified included “ammonium ion”, “immune”, and “regulation of synaptic transmission”, in addition to many other biological processes.
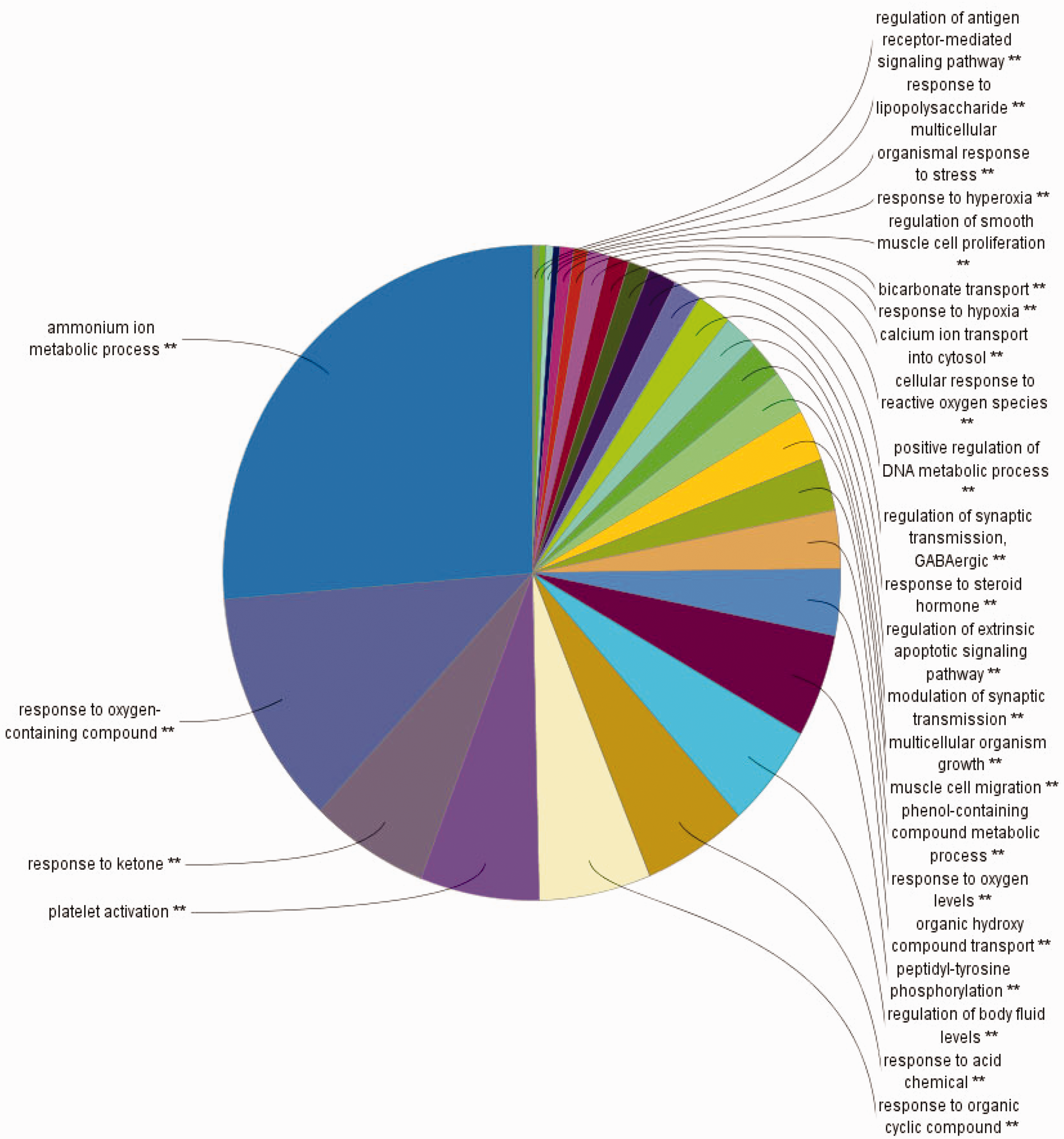
ClueGO analysis of the candidate targets.
Regulation of inflammation by gallincin
Gallincin markedly alleviated DSS-induced colitis in mice (p < 0.05). Histological injury scores and the DAI are shown in Table 1. Nevertheless, there were no statistically significant differences between the low-dose and high-dose gallincin groups with regard to the DAI or histological injury scores.
Effects of gallincin on clinical indices, histological injury scores, cytokines, and D-lactate expression.

Results are expressed as the mean ± SEM.
*p < 0.05 (versus normal control group and gallincin group).
DAI, disease activity index; MPO, myeloperoxidase; DSS, dextran sulfate sodium; TNF, tumor necrosis factor; IL, interleukin.
Gallincin inhibited TNF-α and IL-1β mRNA expression, MPO activity, and IL-1β and TNF-α protein expression (p < 0.05). However, no significant differences were observed between the low-dose and high-dose gallincin groups. Data are shown in Table 1.
Effects of gallincin on intestinal barrier function
As shown in Table 1, concentrations of serum D-lactate, which is considered a marker of increased intestinal permeability, were significantly reduced by gallincin (p < 0.05).
Tight junction (TJ) proteins, such as ZO-1 and occludin, are the main regulators of intestinal epithelial barrier function. In our experiment, the gallincin-treated groups exhibited significantly upregulated ZO-1 and occludin protein expression compared with the DSS group (p < 0.05, Figures 6–8).
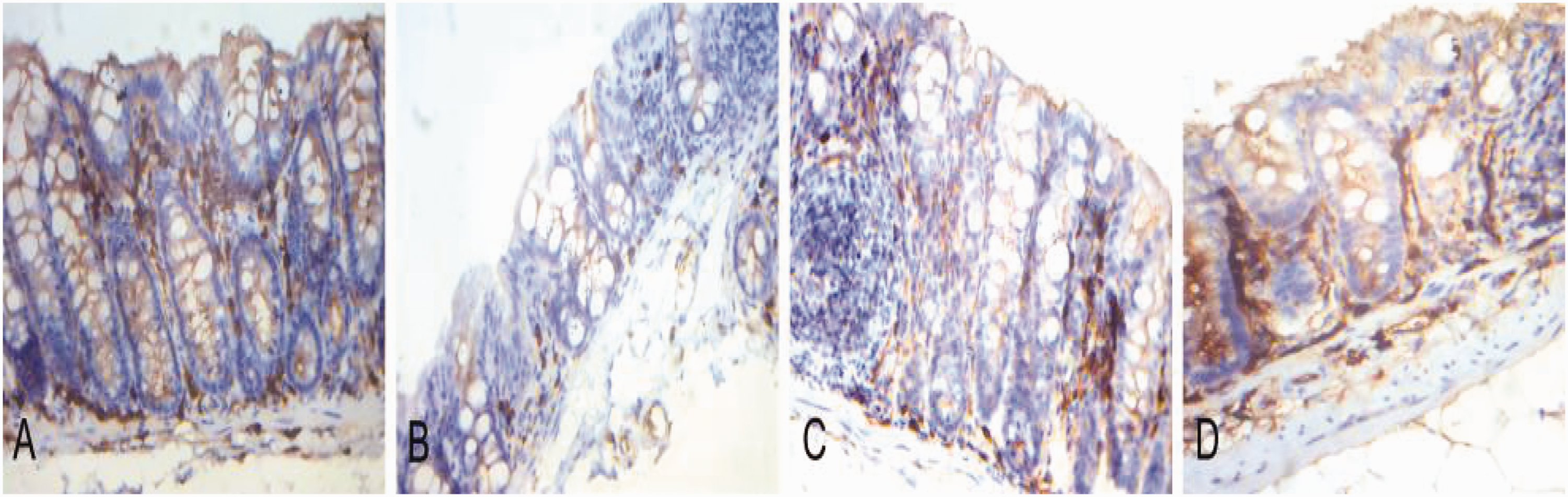
Effects of gallincin on zonula occludens 1 (ZO-1)-positive cells. Immunohistochemical staining of ZO-1 in colonic tissues (magnification 400×). (a) Normal control group. (b) Dextran sulfate sodium (DSS) group. (c) Low-dose gallincin group. (d) High-dose gallincin group. Compared with that of the normal control group, the colons of DSS-treated mice showed significant reduction of ZO-1 expression, which was increased significantly with gallincin treatment (p < 0.05). Most of the ZO-1-positive cells displayed cytoplasmic and cytomembrane staining.

Effects of gallincin on zonula occludens 1 (ZO-1) protein expression. (a) Western blot results display protein expression of ZO-1 in the colonic tissues. (b) Densitometric analysis of ZO-1 expression. Dextran sulfate sodium treatment decreased ZO-1 expression, which was increased significantly with gallincin treatment. Data are expressed as
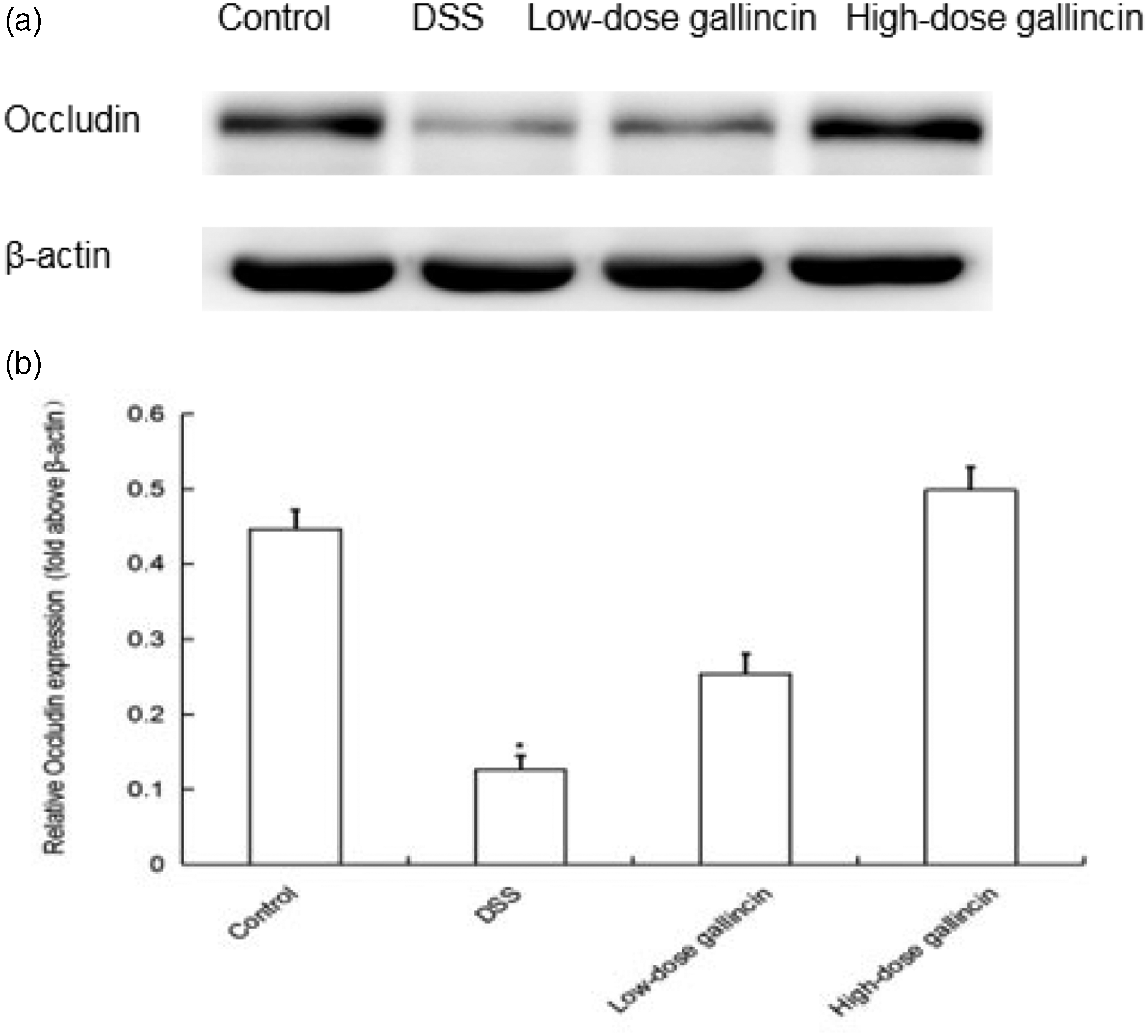
Effects of gallincin on occludin protein expression. (a) Western blot results display protein expression of occludin in the colonic tissues. (b) Densitometric analysis of occludin expression. Dextran sulfate sodium treatment reduced occludin expression, which was increased significantly with gallincin treatment. Data are expressed as
Action pathway of gallincin
Expression of p-ERK1/2 in the DSS group was significantly increased compared with the control group but was suppressed by gallincin, especially low-dose gallincin (p < 0.05, Figure 9).

Effects of gallincin on phosphorylated extracellular-signal regulated protein kinase1/2 (p-ERK1/2) protein expression. (a) Western blot results display protein expression of p-ERK1/2 in colonic tissues. (b) Densitometric analysis of p-ERK1/2 expression. Dextran sulfate sodium treatment markedly increased p-ERK1/2 expression, which was decreased with gallincin treatment. Data are expressed as
Discussion
Our results demonstrated impaired barrier function in mice following DSS-induced colitis, as well as marked inhibition of inflammation by gallincin in vivo. Importantly, our findings indicate that gallincin restored colonic barrier function and ameliorated murine experimental colitis induced by DSS. The therapeutic effect of gallincin might be associated with inhibition of TNF-α and IL-1β production and reduced leukocyte infiltration. These results suggest that gallincin could serve as an adjuvant or prophylactic agent to protect colorectal barrier function against mucosal inflammation. However, one limitation of this study was that examination of differences between the normal group and the gallincin group was not the goal of our experiments. Therefore, comprehensive experimental verification of these differences, including cell experiments and rescue experiments, will be necessary in future studies.
In our study, the UC model was successfully constructed, and impairment of intestinal barrier function was observed. In fact, this is not the first study to show colorectal barrier dysfunction in experimental colitis, as intestinal barrier damage has also been observed in other colitis models.29–32 Schmitz et al. 33 reported that the inflamed intestinal mucosa observed with UC exhibited impaired barrier function; altered TJ structures contributed to this barrier defect. Disruption of the TJ structure has been recognized as a potential cause or result of disease. 34 In addition, genetic studies have indicated the importance of innate barrier function in the pathogenesis or etiology of IBD. 2 These results support the potential of barrier-restoring agents to treat disease or prevent disease recurrence. 34
At present, the mechanism of impaired barrier function is unclear. Some articles have reported that proinflammatory factors, such as TNF-α and IL-1β, impair intestinal mucosal barrier function. 4 However, increased colonic mucosal penetrability increases the likelihood of antigen exposure to intestinal immune cells, which can trigger mucosal inflammation. These results suggest that inflammation and colonic barrier function are part of a complex regulatory network and can activate or inhibit one another by multiple modalities. Indeed, Poritz et al. 29 reported that TJ changes can precede colitis, instead of being its consequence. Similarly, in the present study, we observed that serum concentrations of D-lactate, which was used as a marker for increased intestinal permeability, were increased at an early stage of DSS-induced colitis.
The intercellular TJ is the primary cellular determinant of the epithelial barricade. 34 Dysregulation of TJ proteins is involved in the pathogenesis of UC.35,36 Occludin and ZO-1, which are two important epithelial TJ proteins, appear to be downregulated in UC and chronic pouchitis. 35 Similarly, we observed remarkable decreases of ZO-1 and occludin expression in mice with DSS-induced colitis. Importantly, we also found that gallincin could maintain intestinal epithelial integrity by increasing ZO-1 and occludin expression, thereby diminishing the recurrence of or attenuating intestinal inflammation.
As described above, our results showed that gallincin inhibited inflammation and mucosal damage in mouse models. Anzoise et al. 12 also observed that gallincin decreased levels of TNF-α, IL-6, and cyclooxygenase 2 in rats with experimental colitis. Another report from Chae et al. 11 also showed that gallincin inhibited lipopolysaccharide-induced inflammatory cytokines by suppressing phosphorylation of ERK1/2. Based on these results, it is reasonable to expect that gallincin can protect the colonic barrier from damage, at least in part, via inhibition of excessive inflammation. In addition, mapping of target genes between drugs and diseases, including mitogen-activated protein kinase (MAPK), was identified with our network pharmacology method. Pathway analysis of target genes also indicated their involvement in the regulation of the inflammation and transmission processes. Subsequently, our animal experiment verified that gallincin inhibited ERK signaling, which is one of the best-characterized members of the MAPK family. There are similar reports of p-ERK1/2 inhibition by gallincin.11,23 Moreover, the involvement of ERK in colitis-associated T regulatory cell and Th17 cell development has been recognized. 37 Therefore, it is possible that gallincin alleviates colitis at least in part by inhibiting ERK activation.
Conclusion
Based on network pharmacology analysis and experimental verification, gallincin may be a promising complementary or alternative therapeutic agent for UC. However, further studies are required to explore the long-term safety and exact mechanism of action of gallincin.
Footnotes
Acknowledgements
We thank the Chinese Medical Association and Takeda Science Foundation for providing scholarship support.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was financially supported by the Cultivation Fund of the National Natural Science Foundation (Grant No. qiankehe2018-5764-11), Beijing Medical and Health Foundation (No. YWJKJJHKYJJ-B184053), and the Doctor Foundation of Guizhou Provincial People's Hospital (No. GZSYBS(2017)09).
