Abstract
Background:
Patients with a Fontan circulation have altered cholesterol and lipoprotein values. We analysed small organic molecules in extended phopsholipid and acylcarnitine metabolic pathways (‘metabolomes’) in adult Fontan patients with a dominant left ventricle, seeking differences between profiles in baseline and Fontan circulations.
Methods:
In an observational matched cross-sectional study, we compared phosphatidylcholine (PC), sphingomyelin (SM), and acylcarnitine metabolomes (105 analytes; AbsoluteIDQ® p180 kit (Biocrates Life Sciences AG, Innsbruck, Austria) in 20 adult Fontan patients having a dominant left ventricle with those in 20 age- and sex-matched healthy controls.
Results:
Serum levels of total PC (q-value 0.01), total SM (q-value 0.0002) were significantly lower, and total acylcarnitines (q-value 0.02) were significantly higher in patients than in controls. After normalisation of data, serum levels of 12 PC and 1 SM Fontan patients were significantly lower (q-values <0.05), and concentrations of 3 acylcarnitines were significantly higher than those in controls (q-values <0.05).
Conclusion:
Metabolomic profiling can use small specimens to identify biomarker patterns that track derangement in multiple metabolic pathways. The striking alterations in the phospholipid and acylcarnitine metabolome that we found in Fontan patients may reflect altered cell signalling and metabolism as found in heart failure in biventricular patients, chronic low-level inflammation, and alteration of functional or structural properties of lymphatic or blood vessels.
Trial registration number:
ClinicalTrials.gov Identifier NCT03886935
Keywords
Key summary
What is new?
In adult Fontan patients with a dominant left ventricle the serum phospholipid and acylcarnitine profile is altered, possibly reflecting altered cell signalling and metabolism as found in heart failure, inflammation, or alteration of vascular properties.
What are the clinical implications?
Metabolic profiling with the help of ‘metabolomics’ offers promise in understanding Fontan (patho)physiology and may be of interest as a new diagnostic modality that can facilitate minimally intensive Fontan follow-up.
Introduction
Chronic heart failure is an important cause of morbidity and mortality in adults with congenital heart disease (CHD), in whom its prevalence is underappreciated. 1 The rarity of individual malformations and their often complex anatomy and physiology make the assessment of cardiac function in CHD difficult. 2 Heart failure (HF) symptoms may not always correlate with objective measures like systemic ventricular function or parameters of cardiopulmonary exercise testing, and the established biomarkers B-type natriuretic peptide (BNP) and N-terminal pro-BNP, used in diagnosis and management of HF due to acquired heart disease in a biventricular circulation, still are of controversial value in adults with CHD, as are high-sensitivity troponin, galectin-3, or growth differentiation factor 15, emerging biomarkers in adults with CHD.3 –7 A special subgroup are patients with univentricular circulation after a Fontan operation, which, without a subpulmonary ventricle, provides blood flow in series to the pulmonary and systemic circulatory systems. 8 Elevated systemic venous pressure to approximately 10–16 mm Hg in the Fontan circulation conveys blood passively into the pulmonary bed (total cavopulmonary connection), with low-shear and non-pulsatile blood flow. Only oxygenated blood passes to the single subsystemic ventricle. For Fontan patients especially, early and non-invasive biomarkers are required for the best haemodynamic monitoring and treatment, 9 as with increased numbers of increasingly older patients who have undergone Fontan surgery – in addition to specific cardiac concerns of Fontan pathophysiology such as systolic dysfunction or raised end-diastolic ventricular pressures – we face new situations that present an important combination of chronic Fontan-specific organ system alterations and aging-specific chronic co-morbidities. In addition to (subtle) ventricular dysfunction, the central feature determining end-organ damage is a high central venous pressure, ‘natural’ for these patients, which impedes lymphatic drainage into this high-pressure venous system. In addition to the development of chylothorax, plastic bronchitis, or protein-losing enteropathy (PLE), liver function and structure in particular are progressively affected.10 –12 Usual biomarkers of hepatobiliary injury are often abnormal in Fontan patients but do not reliably track the extent of hepatobiliary disease; the same holds for findings on imaging studies, including elastography. 10 The role of the liver as a central metabolising and synthesising organ implies that profiling metabolic patterns may offer insights into Fontan physiology.10,11 The last decade saw initial attempts to assess Fontan-patient metabolism with usual clinical-biochemistry testing. Besides hyperuricemia and abnormal glucose handling, alterations in cholesterol and lipoprotein metabolism were reported.13 –17 With that in mind, and extrapolating from findings in HF patients with a biventricular system, where the myocardial energy source switches from metabolism of fatty acids to the metabolism of glucose and ketone bodies, 18 and from findings on the important role as proinflammatory analytes that other lipids metabolised in the liver, especially phospholipids (PL), play in the setting of increased cardiovascular risk, 19 we inferred that patients with a Fontan circulation also would handle lipids anomalously. We thus elected to examine Fontan patients’ serum levels of a variety of PL and acylcarnitines (taking serum acylcarnitine levels as tracking intramitochondrial long-chain fatty acids in the form of their activated acyl-coenzyme-A-esters) and attempted in a homogeneous patient group with a dominant left ventricle (LV) to define patterns that reflect metabolic derangement and that offer promise of better understanding of Fontan physiology.
Materials and methods
Patients
At the Centre of Paediatric Cardiology and Congenital Heart Disease, Heart and Diabetes Centre North Rhine-Westphalia, Ruhr-University of Bochum, Germany, between September 2016 and March 2017, we prospectively examined adult Fontan patients with a dominant LV and one age- and sex-matched healthy biventricular control per case (observational matched cross-sectional study). All Fontan patients had undergone two-stage palliation (Norwood II and III) without aortic reconstruction. Age-and sex-matched healthy controls were recruited among staff of the Centre of Paediatric Cardiology and Congenital Heart Disease, Heart and Diabetes Centre North Rhine-Westphalia, Ruhr-University of Bochum, Germany.
Eligibility criteria for all subjects included written informed consent of participants, an age at testing of ⩾18 years, and 8 h fasting before blood sampling. Additional criteria for patients were that the left systemic ventricle be dominant and for controls both that the heart be biventricular without structural or functional abnormality and that they have no systemic, including cardiovascular, disease.
Exclusion criteria were: missing written informed consent; <8 h fasting before blood sampling; medication directly affecting metabolic state, such as cholesterol-lowering agents, or haemodynamic state, such as beta-blockers or sildenafil, with the exceptions of angiotensin converting enzyme inhibitors, diuretics, and anticoagulants; atrial or ventricular arrhythmia; coronary artery disease (history of myocardial infarction, myocardial revascularisation, percutaneous coronary intervention, or coronary artery bypass surgery); failure of the systemic ventricle as assessed by echocardiography; valvular heart disease with stenosis or with worse than mild atrioventricular or aortic regurgitation as assessed by echocardiography; recurrent effusions or PLE; any metabolic disease, such as diabetes mellitus; malignancy or other cachectic disease; liver or renal disease; inflammatory disease such as acute or chronic infection; a myeloproliferative disorder; pregnancy or lactation; multiple organ failure; malnourishment; mental handicap not allowing valid consent to participation in the study or precluding treadmill exercise; physical impairment precluding treadmill exercise; and uptake of oxygen at the anaerobic threshold (V̇O2AT) <20 ml/kg/min (patients) and <25 ml/kg/min (controls) on exercise capacity testing.20,21 The chart according to STROBE (Figure 1) gives details on the flow of patients through the present study, which is a prespecified subwork of the main study protocol [ClinicalTrials.gov identifier: NCT03886935].

Patient flow chart according to STROBE.
Since in Fontan patients loss of exercise capacity is a powerful predictor of mortality, we correlated metabolic results with routine laboratory parameters and exercise capacity parameters. 15 All patients underwent symptom-limited treadmill exercise capacity testing with expired-gas analysis. 21 A 12-lead electrocardiogram was used to determine heart rate, and oxygen uptake at rest (V̇O2 at rest; ml/kg/min), V̇O2AT (ml/kg/min), and maximum uptake of oxygen (V̇O2max; ml/kg/min) were measured.
Age, sex, weight, body mass index, vital parameters, cardiac risk factors, history of cardiac disease, and cardiac medications, with blood sampling for routine haematological and biochemical profiling, were assessed during an outpatient-clinic visit. Fasting patients underwent phlebotomy while recumbent. Echocardiography followed, and, after a defined snack rich in carbohydrates, exercise capacity was tested. Blood studies required samples 0.5 ml greater than those for routine assessments to permit determinations of concentrations of lipid-metabolism analytes. For serum preparation, the blood sample was directly drawn into a tube containing a clotting activator. To separate serum, the sample was centrifuged within 20 min (15°C, 10 min at 2500g). Serum aliquots were immediately frozen and stored at −80°C for further analyses (maximum storage time 4 months). Frozen samples were transported on dry ice to the analysing laboratory. Analyses were performed in batches of 10 samples.
Sample preparation and method
Before analysis, all serum samples were processed as described, with samples thawed on ice and centrifuged; the supernatant was subjected to further analyses. 22 The AbsoluteIDQ® p180 kit assay (Biocrates Life Sciences AG, Innsbruck, Austria) permitted targeted, fully automated quantification of 188 metabolites (142 lipids comprising PC, SM, and acylcarnitines) based on phenylisothiocyanate derivatisation in the presence of internal standards followed by flow injection analysis tandem mass spectrometry using a SCIEX 4000 QTRAP® instrument (SCIEX, Darmstadt, Germany) with electrospray ionisation.
The SCIEX 4000 QTRAP instrument was calibrated periodically after each cleaning cycle, at least once a year, using a chemical standards kit with low/high concentrations of polypropylene glycol polymers (SCIEX, MS Chemical Kit 1, Low-High Conc. PPGs, part number 4406127). The solutions were measured according to the kit instructions and mass calibration and resolution were manually adjusted to meet the specifications of the manufacturer. Measurements were validated by meeting criteria specified by the manufacturer.
Statistics
To exclude metabolites below the limit of detection (LOD), the raw data (µmol/l) were cleaned applying a modified 80% rule; thus, for statistical analysis, at least 80% valid values above LOD needed to be available per analyte in the samples for each group. This reduced the dataset to 143 analytes, including 97 phosphatidylcholines (PC) and sphingomyelins (SM) and 23 acylcarnitines. Remaining values below LOD were imputed applying a logspline method with values between LOD and LOD/2. After log2 transformation of metabolomics data as well as of ‘routine analytes’ and clinical data, the dataset was used for multivariate (hierarchical cluster analysis) and univariate statistical analyses. Student’s t-tests with a Benjamini–Hochberg correction identified significant metabolite (and clinical routine-parameter) differences between patients and controls and p values were calculated, with values <0.05 considered statistically significant. Correlations between the concentration values of the metabolites (and metabolite sums and ratios) with the clinical variables of the participants were evaluated by calculating the Pearson’s correlation coefficient r, with r a measure of linear correlation that can have values between −1 and 1, where 1 indicates total positive linear correlation, 0 indicates no linear correlation, and −1 indicates total negative linear correlation. In general, r values >0.5 (< −0.5) indicate a moderate correlation and r values >0.8 (or < −0.8) indicate a strong correlation. 23 To interpret the correlation, the significance of the correlation was calculated from r and the degrees of freedom (a variable dependent on the sample number). The resulting p value was adjusted for multiple testing, or false discovery rate (FDR), according to Benjamini and Hochberg. 24 Correlations with FDR-adjusted p-values <0.05 were considered statistically significant. To differentiate between general lipid mass changes and species-specific alterations the data were normalised in a class- and samplewise manner and were subjected to statistical analysis (t-tests with Benjamini–Hochberg correction, p < 0.05). Values below LOD were not considered for normalisation and were removed before hypothesis testing. For calculation of the ratio between total PC and SM LOD values were treated similarly. Statistical analysis was performed using R version 3.5.1 or 3.6.1 (R Core Team, 2019). 25
Ethics
The study conformed to the principles outlined in the Declaration of Helsinki and was approved by the local ethics committees of the Medical University of Innsbruck, Austria (AN2015-0303 357.43), and of the Heart and Diabetes Centre North Rhine-Westphalia, Ruhr-University of Bochum, Germany (AZ 52/2016).
Results
Of the 398 Fontan patients registered at the Centre of Paediatric Cardiology and Congenital Heart Disease, Heart and Diabetes Centre North Rhine-Westphalia, Ruhr-University of Bochum, Germany, 176 patients were potentially eligible for our study by morphology (dominant LV). Applying the criterion of ⩾ 18 years of age, 71 patients were eligible. After all exclusion criteria were applied, 20 Fontan patients and 20 age- and sex-matched controls were enrolled (Figure 1). Data were complete for all of them. Table 1 lists clinical and echocardiographic parameters of patients and controls.
Participants’ clinical characteristics (mean values ± standard deviation).

ACE, angiotensin-converting-enzyme; exam, examination; LPA, left pulmonary artery; PA, pulmonary atresia; pts, patients; PS, pulmonary stenosis; TA, tricuspid atresia; TCPC, total cavopulmonary connection; VSD, ventricular septal defect; vv, veno-venous. Patients with celiac disease were on gluten-free diets.
Exercise capacity testing
In Fontan patients, V̇O2 at the anaerobic threshold and at maximum was significantly lower than in controls (Table 2).
Participants’ exercise capacity testing results.

Values are given as mean ± standard deviation. AT, anaerobic threshold; BP, blood pressure; RR, Riva Rocci; SpO2, pulsoxymetric oxygen saturation; V̇O2, oxygen uptake.
Routine analytes
Haemoglobin and haematocrit, partial thromboplastin time, international normalised ratio (INR), triglyceride, gamma glutamyl transferase activity, alanine aminotransferase activity, total bilirubin, creatinine, urea, and uric acid values were significantly higher, and high density lipoprotein-cholesterol (HDL-C) values and thrombocyte counts were significantly lower in patients than in controls (Table 3).
Values, routine analytes.

Values are given as mean ± standard deviation. ALT, alanine aminotransferase activity; AST, aspartate aminotransferase activity; C, cholesterol; CRP, C-reactive protein; gGT, gamma glutamyl transferase activity; GLDH, glutamate dehydrogenase activity; HDL, high density lipoprotein; INR, international normalised ratio; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; PTT, partial thromboplastin time; *↑, statistically significant higher serum concentration in Fontan patients than in controls; *↓, statistically significant lower serum concentration in Fontan patients than in controls.
Metabolomic examination of serum
Serum concentrations of 14 lyso-PC (12 above LOD), 76 PC (71 above LOD), and 15 SM (14 above LOD) were determined. Among the 97 analytes above LOD, serum concentrations of 60 (62 %), including analytes in all three subgroups and fatty acids with mostly higher carbon numbers, were significantly lower in Fontan patients than in controls (q-values <0.05) (Figure 2, Figure 3). Values for no single analyte were higher in patients than in controls. Serum concentrations of 40 acylcarnitines were determined; 23 with values above LOD, 3 were significantly higher in patients than in controls (C0, C18:2, C3; q-values < 0.05). Summed by subgroups – all PC, all lyoPC, all SM, and all acylcarnitines – all lipids were significantly decreased in patients. Table 4 shows concentrations and statistical results are shown for all metabolites determined.

Heat map of phospholipid serum concentrations.

Serum concentrations of phospholipid subgroups.
Metabolite concentrations.
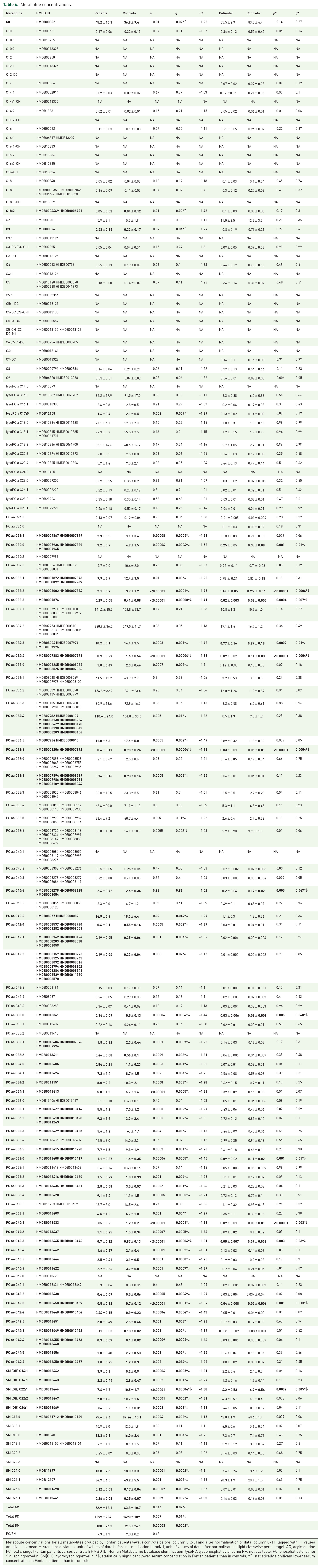
Metabolite concentrations for all metabolites grouped by Fontan patients versus controls before (column 3 to 7) and after normalisation of data (column 8–11, tagged with °). Values are given as mean ± standard deviation, unit of values of data before normalisation (µmol/l), unit of values of data after normalisation (lipid classwise percentage). AC, acylcarnitine FC, fold change (Fontan patients versus controls); HMBD ID, Human Metabolome Database identification; lysoPC, lysophosphatidylcholine; NA, not available; PC, phosphatidylcholine; SM, sphingomyelin; SM(OH), hydroxysphingomyelin; *↓, statistically significant lower serum concentration in Fontan patients than in controls;
Normalisation of metabolomics data by lipid class reduced the 60 (lyso)PC and SM with significant LODs to just 13 (PC aa C30:0, C32:2, C32:3, C34:3, C34:4, C36:6, C40:4; PC ae C30:0, C38:0, C40:1, C40:3, C42:3; SM (OH) C22:1). All these are low in relative abundance (<5% of total lipid mass). With normalisation all the 3 acylcarnitines with significant LODs lost significance.
Correlations of routine biochemical and clinical parameters with metabolomic parameters
Among routine biochemical analytes, the variables INR, alanine aminotransferase activity, haemoglobin, and haematocrit displayed significantly negative correlations with metabolic parameters. Significantly positive correlations existed between metabolomic parameters and minimum and maximum oxygen saturations. No other correlations could be established, especially none with respect to platelet count, total protein, albumin, CRP, NT-proBNP, and any exercise capacity parameter, including peak V̇O2 (Online Supplemental Table 1).
Discussion
To the best of our knowledge, ours is the first clinical metabolomics study focussing on Fontan patients’ serum PL and acylcarnitine levels. Its main result is the observation that PC, SM, and acylcarnitine values differed significantly between Fontan patients and controls: Fontan patients had lower total PC and SM values and higher total acylcarnitine values.
A general decrease in total PL mass in Fontan patients accords with the reduction that we observed in HDL-C, a major lipid constituent of serum. To differentiate effects on lipid mass in general from particular changes in individual lipid species, we normalised our metabolomics data by lipid class. Following normalisation, statistical significance in LOD was lost for > 80% of analyte comparisons, with significance persisting for some low-abundance lipid species (13 PL, <5% of lipid mass). This indicates that in Fontan patients the imbalance in the metabolism of polar lipids is not limited to a subset of distinct lipids, constituting a lipid profile, but instead quantitatively affects all classes of PL and acylcarnitines.
PC and SM are PL; SM also is a sphingolipid (a phosphosphingolipid). PL typically are major participants in structural-lipid metabolism, in concert with cholesterol and steroids forming the extracellular lipid bilayer of cells and located in all tissues. 26 While cholesterol and steroids are stabilising and stiffening components, PL confer fluidity upon polar-bilayer cell membranes. They also are important components of HDL. 27 In addition to their function in structural metabolism, PL are importantly involved in signalling and they can affect synthesis of prostaglandins and leukotrienes and thus inflammation. 28 Moreover, like fatty acids, PL are involved in energy metabolism. Of the 13 PL that we still found to be decreased in Fontan patients’ serum after normalisation of data, 12 are PC. Aside from pregnancy and obesity, no distinct conditions and disorders, especially no cardiovascular disorders, are associated with altered concentrations of these analytes.
A further PL for which we still found decreased serum concentrations in Fontan patients after normalisation of data is SM (phosphosphingolipid). On hydrolysis into ceramides, SM like the one that we identified are generally involved in necroptosis (as long chain fatty acids are), a type of regulated cell death characterised by plasma membrane rupture and inflammatory response. 29 Tissues are enriched in HDL-SM in Niemann–Pick disease, which is caused by an acid sphingomyelinase deficiency due to mutations in SMPD1, 30 and SM-depleted HDL was observed in patients with diabetes. 31 Furthermore, SM is substantially involved in the development and progression of the atherosclerotic plaques of coronary artery disease (CAD): human atherosclerotic lesions are enriched in SM and ceramide species, 32 and elevated SM serum levels are independent predictors of CAD in humans.33,34
An increase in acylcarnitine blood concentration can mirror an imbalance of mitochondrial fatty acid degradation, as found in patients with pathogenic mutations in genes encoding enzymes involved in long-chain fatty acid oxydation. 35 Interestingly, one important feature of mitochondrial disorders can be the development of a cardiomyopathy. Of further note is that, like SM, long-chain fatty acids are involved in necroptosis, with increased acylcarnitine cell concentrations. 29 Possibly underlying the changes that we saw are shifts in cellular signalling associated with HF, chronic low-level inflammation, and alteration of functional or structural properties of lymphatic or blood vessels.
Altered cell signalling and metabolism as indicators of subtle HF
That decreased PL concentrations of PL in our Fontan patients accompany increased acylcarnitine concentrations agrees with our second hypothesis that in addition to structural homeostasis, energy metabolism also might be perturbed, marking early, subtle changes in (myocardial) cell metabolism. The normal heart primarily utilises free fatty acids as a source of energy and switches to glucose metabolism during stress. In biventricular patients with HF, changes in myocardial mitochondrial function result in preferential use of glucose and ketone bodies. When the substrate shifts as HF progresses is still unknown. 36
PL, especially SM, with their location mainly in the (cell) membrane, serve as signalling molecules in cell metabolism. Sphingolipid content of myocardial tissues is decreased in cardiomyopathic hamster models at the onset of HF.37,38 Even subtle stages of HF may be indicated, as emphasised by the fact that Fontan patients in our study ‘clinically’ had sustained haemodynamics with a mean V̇O2AT of 25 ml/kg/min and V̇O2max of 29 ml/kg/min, which correlates well with normal values for right-ventricle Fontan patients. 39
Findings in atrial-fibrillation patients with a biventricular system, in whom blood PL levels rose between closure of left atrial appendage (LAA) and 6 months thereafter, support the assumption that decreased PL levels in our patients indicate subtle stages of HF with slightly raised end-diastolic pressures, hinting at altered diastolic and sustained systolic ventricular function. 40 In these atrial-fibrillation patients and in our patients, values for PL with fatty acids of higher carbon numbers were altered. However, whilst in the atrial-fibrillation patients analyte concentrations were higher than in controls, we found lower analyte concentrations in patients than in controls. This calls attention to the fact that LAA closure leads to a loss of LAA metabolic and endocrinologic functions. In our patients, who had all undergone extracardiac tunnel surgery, these LAA functions should be relatively intact. Typical of the aging Fontan circulation (even of the so-called ‘intact’ Fontan circulation) is the development of raised end-diastolic ventricular pressures with sustained systolic function, a feature still difficult to assess by conventional echocardiographic measures of diastolic function. 41 Many of our patients (all adult) thus may have experienced slightly raised atrial pressures, which might explain the studies opposed findings with respect to how analyte values shifted. Haemodynamic measurements of end-diastolic ventricular and atrial pressures might buttress this inference. Moreover, even if our patients did not exhibit differences from controls in NT-proBNP levels, and even if this established marker for HF could not be correlated with the lipidome alterations that we found, measurements in our patients of cardiovascular biomarkers that are established in the normal population and emerging in adults with CHD, such as high-sensitivity troponin, galectin-3, or growth differentiation factor 15, would be interesting.6,7,42
Chronic low-level inflammation
Involvement of PL in arachidonic acid metabolism and thus in the production of pro-inflammatory metabolites like leukotrienes agrees with our second hypothesis that our results indicate inflammation in our patients. In addition, sphingomyelinase activation of the SM-ceramide pathway is thought to induce inflammation, adding to oxidative stress, and to induce cell death, resulting in atherosclerosis, aging, and cardiovascular events.29,32 An activated pathway can be characterised by reduced levels of SM, as seen in our patients. 43 Even if acute inflammation is not to be expected in our patients 18 years after Fontan repair and independent of surgery in general, as supported by the normal CRP levels in patients and controls and by the lack of correlation with lipidomic variables, chronic low-level inflammation might still be present. High-sensitivity C-reactive protein (hsCRP) is a potential biomarker in adult patients with various forms of CHF 44 ; assays measuring hsCRP can differentiate concentrations within the normal range and can thus in the normal population indicate chronic low-level inflammation, which is known to reflect atherosclerotic cardiovascular disease. Higher hsCRP levels, even within the normal range, are associated with an increased risk for cardiovascular events. Adults with various forms of CHD (including single-ventricle Fontan circulation) with elevated hsCRP not only have a worse functional status and exercise capacity as measured by peak V̇O2, but also have a greater risk for death or non-elective cardiovascular hospitalisation. 44 The CHD cohort showed no association between hsCRP and history of coronary artery disease or cerebrovascular events, but increased hsCRP levels still indicate chronic low-level inflammation due to non-atherosclerotic causes and higher values might predict adverse outcomes. 44 These findings support our hypothesis that the altered levels of PL observed could be due to inflammation, even if in our study patients (and controls) ‘routine’ CRP levels could not be correlated with lipidomic variables and even if CRP levels did not differ between Fontan and control cohorts. One reason for a chronic inflammatory status in Fontan patients might be the combination of (intestinal) inflammation and altered mesenteric haemodynamics, both affecting or even provoking PLE.45,46 In combination, subtle impairment of myocardial (diastolic) function, inflammation in biventricular patients with chronic congestive HF and a chronic low cardiac output, and altered PL levels (with induction of endothelial-cell oxidative stress) in patients with CAD underscore the possibility that chronic low-level inflammation exists in our Fontan patients even without overt HF and without overt PLE.47,48 Of note is that in addition to decreases in HDL-C values and different structural and functional HDL properties in patients with an acute-phase response 3 days after cardiopulmonary bypass surgery in comparison with healthy controls, 49 patients with rheumatoid arthritis and thus with chronic inflammation also have low values for total cholesterol, low density lipoprotein cholesterol, and HDL-C, similar to the significant decrease in HDL-C values that we found in Fontan patients and similar to low cholesterol metabolism profiles in Fontan patients.15 –17,50 Such alterations in cholesterol profile exist even 5 years before rheumatoid arthritis manifests clinically, and a similar cholesterol profile is associated with an increased risk of cardiovascular disease. 50 We propose that inflammation affects cholesterol and PL metabolism at different stages of liver and/or vessel disease. If so, the combination of cyanosis with a chronic inflammatory state might exacerbate changes like those that we found: adults with cyanotic CHD have hypocholesterolaemia, although their extended lipid status has not been further examined. 51
Alteration of structural or functional lymphatic-vessel or blood-vessel properties
As with subtle alteration of myocardial function and a chronic inflammatory state, decreased serum concentrations of PL may also indicate alteration of structural or functional vascular properties in our Fontan patients.
This might involve lymphatic vessels. Various disorders such as HF and inflammatory diseases disrupt the lymphatic vascular system. 12 Assuming that the Fontan circulation imposes a state of (subtle) chronic HF associated with chronic low-level systemic inflammation, increased vascular permeability, and increased production of interstitial fluid, the chronic challenge to the lymphatic system is evident, with poor drainage especially into the Fontan-specific high-pressure systemic venous system. 52 Besides lymphangiectasia, lymphatic collaterals form and tortuosity and dilation of the thoracic duct occur in Fontan patients; these interestingly have also been reported in children before Fontan surgery, but might be exacerbated by the Fontan circulation. 52 Lymphatic dysfunction can contribute to deterioration of organ function in Fontan patients, with lymphoedema, plastic bronchitis, PLE, and liver dysfunction. The two latter disorders can be seen as major reasons for the alterations in PL, acylcarnitine, and HDL-C levels that we found; even if our patients exhibited no overt signs of PLE and even if lipidomic variables did not correlate with serum total protein or albumin levels, enteral lipid uptake might be altered, in combination with altered hepatic metabolism of lipids and of lipoproteins, which are important carriers of PL. 15 Whether this mirrors the ‘normal’ Fontan situation or is already a subtle indication that the Fontan circulatory system is impaired remains to be seen.
This could refer to the alteration of structural or functional properties of blood vessels. Two facts reinforce this assumption. First, SM is important in the development and progression of the atherosclerotic plaques of CAD.32 –34 Second, stiffening of vessels is seen in patients with non-alcoholic fatty liver disease or with hypoplastic left heart syndrome, in the latter group an observation discussed as evidence of ventriculo-vascular or rheo-vascular coupling.53 –55 The Fontan patient with a right systemic ventricle who has undergone extensive aortic surgery is especially likely to exhibit aortic stiffness <5 years after Fontan completion. 54 Young Fontan patients with a dominant LV, like our patient group, show arterial stiffening, and this increased-stiffening process can be seen especially in the ascending aorta. 55 Non-invasive work in a heterogeneous group of young adult Fontan patients (80% showing dominant LV morphology) supports these findings, with elevated global arterial stiffness detected on peripheral-artery sphygmomanometry. 56 Positive correlations also existed between increased arterial stiffness and inflammation, which might be explained by disturbed vitamin D/parathormone interactions frequent in this patient group.56,57 Vascular stiffening also occurs in cerebral arteries in young Fontan patients, suggesting altered (arterial) vascular properties in general or at least in vessels neighbouring the heart. 58 Local assessment of arterial stiffness would be desirable.59,60 Might these alterations, which we interpret as altered structural or functional blood vascular properties and regard as adaptation to Fontan physiology, be simultaneously adverse with regard to elevated cardiovascular risk as known in the biventricular patient and favourable in the setting of Fontan physiology? Might altered structural and functional properties of blood vessels, especially vascular stiffening, maintain arterial pulsatility sufficient to support end-organ perfusion and supply a driving force for pulmonary circulation? In histopathologic investigations of Fontan patients with a mean age of 30 years, mural remodelling has been demonstrated in the pulmonary artery and in the inferior and superior vena cava. 61 Moreover, in the course of their signalling function, PL regulate angiogenesis, possibly linking their concentrations in serum with altered collateral-vessel formation. 37 A decrease in PL concentrations as measured might mean that PL are present in another form, an activated or oxidised one, which in our assay was not determined. 37 Further to assess this hypothesis it would be useful in Fontan patients to determine the forms and concentrations of PL in vascular walls and to assess the degree of collateral flow.
Summary
The metabolism of PL and acylcarnitines is distorted in adult Fontan patients with a dominant LV. Three mechanisms may act alone or in combination in altering PL and/or acylcarnitine metabolism: a) altered cell signalling and metabolism as indicators of subtle HF; b) chronic low-level inflammation; d) alteration of functional or structural properties of lymphatic or blood vessels. Our results do not identify single metabolites that may permit differentiation between disease groups and controls, but differences in the content of an entire substance class such as – in our study – serum PL and acylcarnitine content may suggest extended metabolic alterations in LV Fontan patients. These findings may, extended and confirmed, permit better understanding of Fontan physiology, which should be regarded as a complex state involving diverse pathways and (patho)physiological processes.
Limitations
In patients with a Fontan circulation, the underlying (embryologic) cardiovascular malformation, extent of surgical reconstruction of the neoaorta, or loading conditions (also in the case of relevant collateral flow) can differ significantly. To limit variability, we focused on Fontan patients with a dominant LV who underwent Norwood II and III surgery. Our patient group thus is small. This may limit the extent to which our findings can be generalised. So may our choice of a targeted metabolomic approach, as metabolites not included in the commercial kit that we used might be important. That we analysed serum samples precludes direct comparisons between our results and those of studies that used tissue samples. That PL and acylcarnitines are reliably detectable in healthy human serum and plasma lends our data credence. 27 Still, having used not vascular (or myocardial) tissue but serum, we cannot rule out certain system-driven aspects of pathogenesis – unknown confounders that affect metabolic profiles might be the true basis for the observed differences. We strove to lessen the likelihood of such errors by following a strict inclusion and exclusion protocol, especially with regard to (known) comorbidities or medication. Although only fasting serum was tested, differences in body composition or lifestyle might still have influenced our results to an unknown degree. The opportunity to correlate our findings with those on invasive assessment would have been advantageous, especially for the general possible explanation of our findings that they might reflect (slightly) raised end-diastolic ventricular pressure, which our conventional echocardiographic measurements were not able to reveal. Furthermore, to distinguish Fontan patients with preserved haemodynamic status (as suggested by clinical findings, echocardiography results, and routine laboratory biomarker values) from those with impaired haemodynamic status, we might not have focused only on the configuration of the systemic ventricle, oxygen saturation, exercise capacity, and valve regurgitation, but also ideally on haemodynamic parameters like fenestration due to elevated pulmonary vascular resistance and high central venous pressure, as well as on the presence of substantial collateral flow, dilatation of the tunnel, diaphragmatic paresis, or cardiac index. Without data from angiography, our hypothesis that our findings indicate angiogenesis/collateral flow must be considered carefully. Further influencing our findings might be the intake of phenprocoumon, which 80% of our patients received. As an oral anticoagulant inhibitor of vitamin K oxidoreductase, it interacts with hepatic metabolism and can even induce hepatitis. An effect on our results cannot be excluded, a possibility that should be examined in a larger cohort of patients with more patients not taking phenprocoumon.
Finally, to evaluate our hypothesis that chronic low-level inflammation contributes to the alterations in the lipidome that we found, measuring hsCRP levels would be elegant. To demonstrate higher hsCRP levels between in Fontan patients than in controls would support that hypothesis; such a study might even expose correlations with the lipidome that ‘routine’ CRP levels could not unmask. 44
Outlook
To use non-invasive haemodynamic MRI studies, assessing end-diastolic ventricular pressures and atrial pressures, flow patterns, and calculating collateral flow, in evaluating our hypotheses regarding raised end-diastolic ventricular pressures, inflammation, and functional or structural alteration of vascular systems is an interesting prospect. To compare and to contrast lipidome-constituent levels in subgroups of Fontan patients stratified by underlying malformation, morphologic features of the systemic ventricle, type of aortic surgery, comorbidities, (semi-)invasive haemodynamic parameters, and particularly by means of evaluation of hepatic function is an immediate aim, as is, in the longer term, follow-up to investigate how rapidly metabolomic changes develop in patients with Fontan circulation. 20 Further, to correlate our results with proteomic studies, especially those that dissect inflammation, will be very satisfying, as will be both histopathologic studies to assess vessel alterations and genomic investigations.
Conclusion
Requiring only minute specimens for simultaneous quantitation of participants in multiple metabolic pathways, lipidomic profiling was useful for assessment of metabolic derangement in LV Fontan patients, in whom we found striking PL and acylcarnitine level alterations that may reflect altered cell signalling and metabolism as found in HF in biventricular patients; inflammation; or alteration of structural or functional properties of the lymphatic and blood vessel system.
Supplemental Material
Supplemental_Table – Supplemental material for Targeted metabolomic analysis of serum phospholipid and acylcarnitine in the adult Fontan patient with a dominant left ventricle
Supplemental material, Supplemental_Table for Targeted metabolomic analysis of serum phospholipid and acylcarnitine in the adult Fontan patient with a dominant left ventricle by Miriam Michel, Karl-Otto Dubowy, Manuela Zlamy, Daniela Karall, Mark Gordian Adam, Andreas Entenmann, Markus Andreas Keller, Jakob Koch, Irena Odri Komazec, Ralf Geiger, Christina Salvador, Christian Niederwanger, Udo Müller, Sabine Scholl-Bürgi and Kai Thorsten Laser in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors gratefully thank the laboratory team of the Erich and Hanna Klessmann Institut, Bad Oeynhausen, Germany, and the team of the Kinderlabor Innsbruck, Austria (Claudia Ertl, Ulrike Eichinger-Öttl, Christine Kluckner), for supporting our work by giving us the facilities to preanalyse and to store the probes, Schwester Dagmar of the laboratory for exercise capacity testing at the Center of Pediatric Cardiology and Congenital Heart Disease, Heart and Diabetes Center North Rhine-Westphalia, Bad Oeynhausen, Germany, for supporting us in exercise capacity testing and in blood drawing, and A.S. Knisely and E. Michel for comments on manuscript drafts.
Author contributions
MM designed the study, examined patients, prepared samples, analysed and interpreted results and wrote the manuscript; KOD supervised lung function and exercise testing; KOD and KTL helped classify the patients; MGA and UM performed biochemical assays and helped with statistical analysis of metabolomic-examination results; SSB, KTL, KOD, MAK, and JK assisted in analysis and interpretation of results; AE, UM and JK provided the figures; all authors proofread the manuscript and read and approved the final version of the manuscript.
Funding
This work is part of the project Metabolic aspects in Fontan patients, which is financed by a grant from the Tiroler Wissenschaftsförderung (grant number UNI-0404-2126).
Conflict of interest statement
MM, KOD, MZ, DK, MGA, AE, MK, JK, IOK, RG, CS, CN, UM, SS-B, and KTL declare that they have no financial or non-financial competing interests with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
