Abstract
Background:
Our objective was to provide consensus recommendations on the optimal management of the immune-mediated inflammatory diseases (IMIDs) seen in patients with spondyloarthritis (SpA) using a multidisciplinary approach, and to develop a simple tool to help earlier recognition and referral of coexisting IMIDs in patients who already have one type of IMID.
Methods:
A total of 28 experts in the multidisciplinary management of the SpA-associated IMIDs assessed two questionnaires: one with statements focused on the multidisciplinary management of IMIDs, and a second questionnaire focused on questions useful for early recognition and referral. Panelists assessed the statements with a 9-point ordinal scale (1 = strongly disagree, 9 = strongly agree) using a modified Delphi methodology.
Results:
Consensus was reached on 72 out of the 82 statements (87.8%). Panelists agreed that the multidisciplinary approach to IMIDs is not sufficiently developed. The creation of multidisciplinary IMID units might be necessary. These units might focus primarily on patients with two or more coexisting IMIDs, or on IMIDs that are especially complex from a diagnostic or therapeutic point of view. Specialists who attend to patients with IMIDs should perform a screening for other coexisting IMIDs. A simple tool to help earlier recognition and referral of coexisting IMIDs is proposed.
Conclusions:
There is a need to improve care for patients with SpA-associated IMIDs. We provide expert recommendations to guide the adoption of a multidisciplinary approach for these cases, and a simple tool that may be useful for earlier recognition of coexisting IMIDs.
Keywords
Introduction
Immune-Mediated Inflammatory Diseases (IMIDs) are a group of conditions with unknown etiology but that share common inflammatory pathways with immune dysregulation and imbalance in inflammatory cytokines. 1 IMIDs encompass disorders as diverse as inflammatory bowel disease (IBD), rheumatoid arthritis (RA), ankylosing spondylitis (AS), psoriasis (PsO), psoriatic arthritis (PsA), and juvenile idiopathic arthritis (JIA) or uveitis, 2 among others. Although there is considerable variation in disease occurrence among different populations, overall, the estimated prevalence of IMIDs in Western countries is 5–7%. 3
Recommendations from many scientific societies and research groups emphasize the importance of a multidisciplinary and multispecialty approach in the management of IMIDs.4–10 However, the best model to deliver these multidisciplinary and multispecialty services is not fully defined.11,12 In addition, evidence shows that multiple IMIDs can coexist within the same patient. Early detection of coexisting IMIDs enables timely initiation of adequate treatment, and, in many cases, prevents irreversible progression. 4 In the setting of psoriatic disease, there are screening tools for the early diagnosis of PsA in patients with PsO. 13 In the field of Crohn’s disease, red flags have been developed to warn clinicians about the presence of this condition. 14 Nevertheless, there are many IMIDs that can coexist with a given IMID, and, in clinical practice, it is not common to have in hand all the validated screening tools for screening of IMIDs that clinicians are not familiar with.
Spondyloarthritis (SpA) is a group of inter-related chronic arthritides that share, among other features, a high prevalence of associated immune conditions such as PsO, uveitis, or IBD. It is therefore important that the specialists involved in the care of these patients establish appropriate communication channels for the detection and optimal management of these contingencies.
The objective of this project was to provide consensus recommendations on the optimal management of SpA-related IMIDs using a multidisciplinary approach. In addition, we tried to develop a simple, agreed tool to help earlier recognition and referral of coexisting IMIDs (specifically SpA, uveitis, PsO, and IBD) in patients who already have one of those IMIDs.
Methods
In this project, a consensus method (modified Delphi) was used following recommendations from RAND/UCLA.15,16 A scientific committee, consisting of five specialists (from rheumatology, ophthalmology, gastroenterology, and dermatology) with vast experience managing multidisciplinary units, met to develop a questionnaire with statements focused on controversial or unanswered questions about the multidisciplinary management of IMIDs. A second questionnaire specifically focused on questions useful for the screening of IMIDs in patients who already had an IMID was developed, emphasizing the screening for SpA, uveitis, PsO, and IBD. In a first step, the steering committee met to design the questions to be addressed with reference to participants, interventions, comparisons, outcomes, and study design (PICOS). To answer the questions generated in this first meeting, and to be able to raise the questions for a subsequent DELPHI process, a literature review was conducted on the international databases (MedLine/PubMed, Google Scholar, Cochrane library, and ISI-WOK) to identify early screening tools as well as multidisciplinary management approach studies referring to patients with PsO, PsA, SpA, uveitis, and IBD. Clinical trials, observational studies, and literature reviews related to the aforementioned aspects published in English or Spanish until December 2018 were selected.
The questionnaires designed after this first step were submitted online in two rounds (during January and February 2019, respectively) to a panel of Spanish specialists with recognized experience in the multidisciplinary management of IMIDs related to four specialties: ophthalmology, dermatology, rheumatology, and gastroenterology. The selection of the panelists was based on their expertise in the management of IMIDs and according to a geographical dispersion representative of the whole of Spain.
Panelists evaluated the statements with a 9-point Likert ordinal scale (1 = strongly disagree, 9 = strongly agree). Responses were grouped into three categories: 1–3 = disagree; 4–6 = neither agree nor disagree; and 7–9 = agree. Consensus on a statement was reached when the median of the responses was included in the 7–9 category (consensus on agreement) or in the 1–3 category (consensus on disagreement), and less than one-third of the panelists voted outside these categories. Statements on which panelists did not reach consensus in the first round were re-evaluated during a second round of voting. Between rounds, panelists were informed of the responses from the first analysis.
Results are shown in tables as median and interquartile range (IQR) of the answers and degree of agreement, which was defined as the percentage of panelists who voted within the category that included the median of the answers (1–3, 4–6, or 7–9). Taking into account the answers to the questionnaire, the scientific committee developed a table of conclusions and recommendations, and a figure outlining a tool that can be used for the screening of coexisting IMIDs in patients with one IMID.
The Ethics Committee of the Hospital Universitario Central de Asturias (Oviedo-Spain) waived the need for ethics approval and the need to obtain consent for the collection, analysis and publication of the anonymized data obtained retrospectively for this noninterventional study.
Results
One questionnaire consisted of 51 items divided into three blocks (Tables 1–3), and was submitted to 28 specialists: 7 ophthalmologists, 7 dermatologists, 7 rheumatologists, and 7 gastroenterologists. In addition, there was a second questionnaire with 31 items suggesting questions that could be useful for screening for coexisting IMIDs in patients with one IMID (Figure 1 and Table S1).
Results block I. State of the question and general concepts.

IMID, immune-mediated inflammatory disease; IQR, interquartile range.
Results block II. Implementation of IMID units.

ICT, information and communication technology; IMID, immune-mediated inflammatory disease; IQR, interquartile range.
Results of block III. Screening of IMID in daily practice.
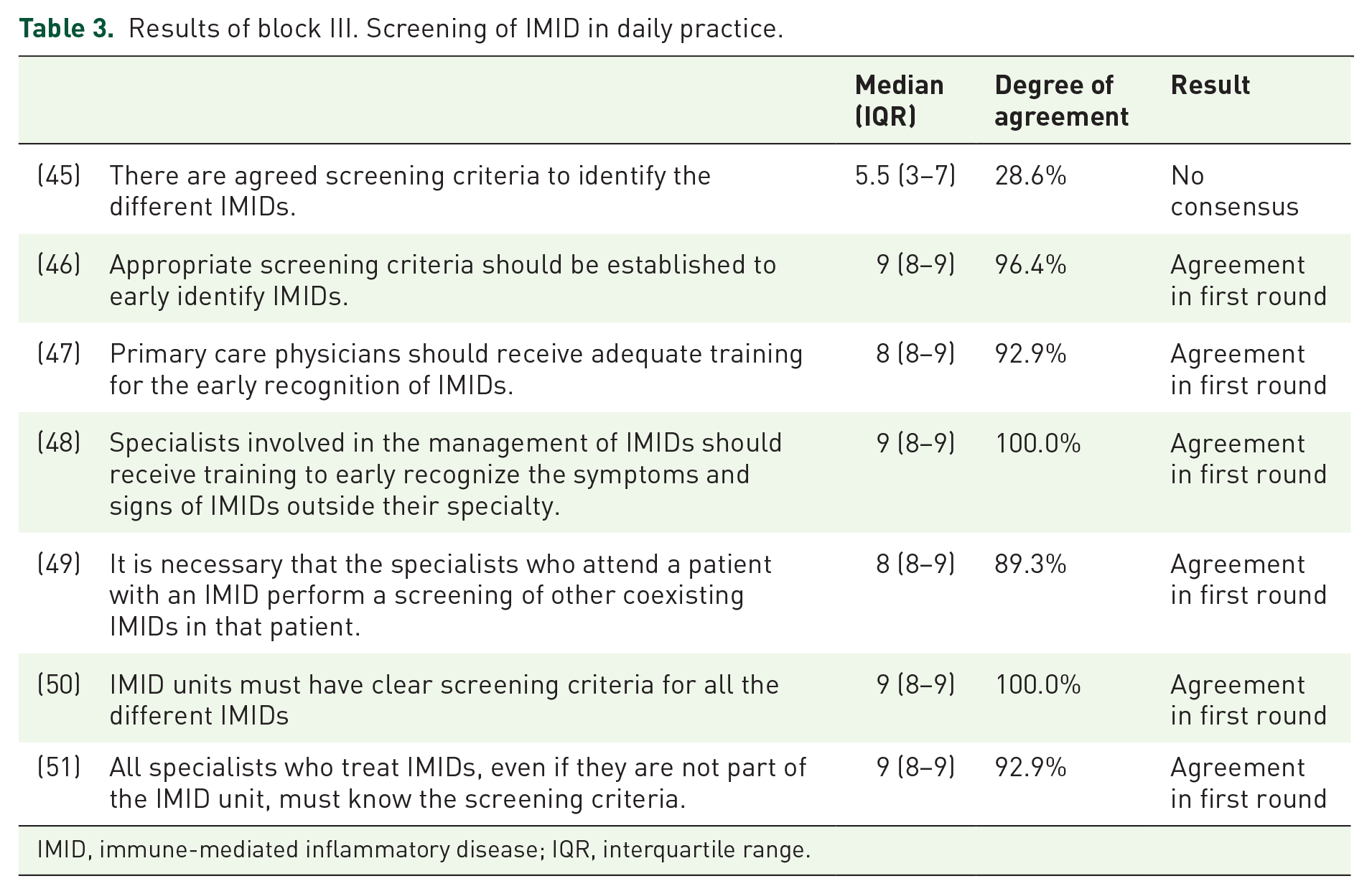
IMID, immune-mediated inflammatory disease; IQR, interquartile range.
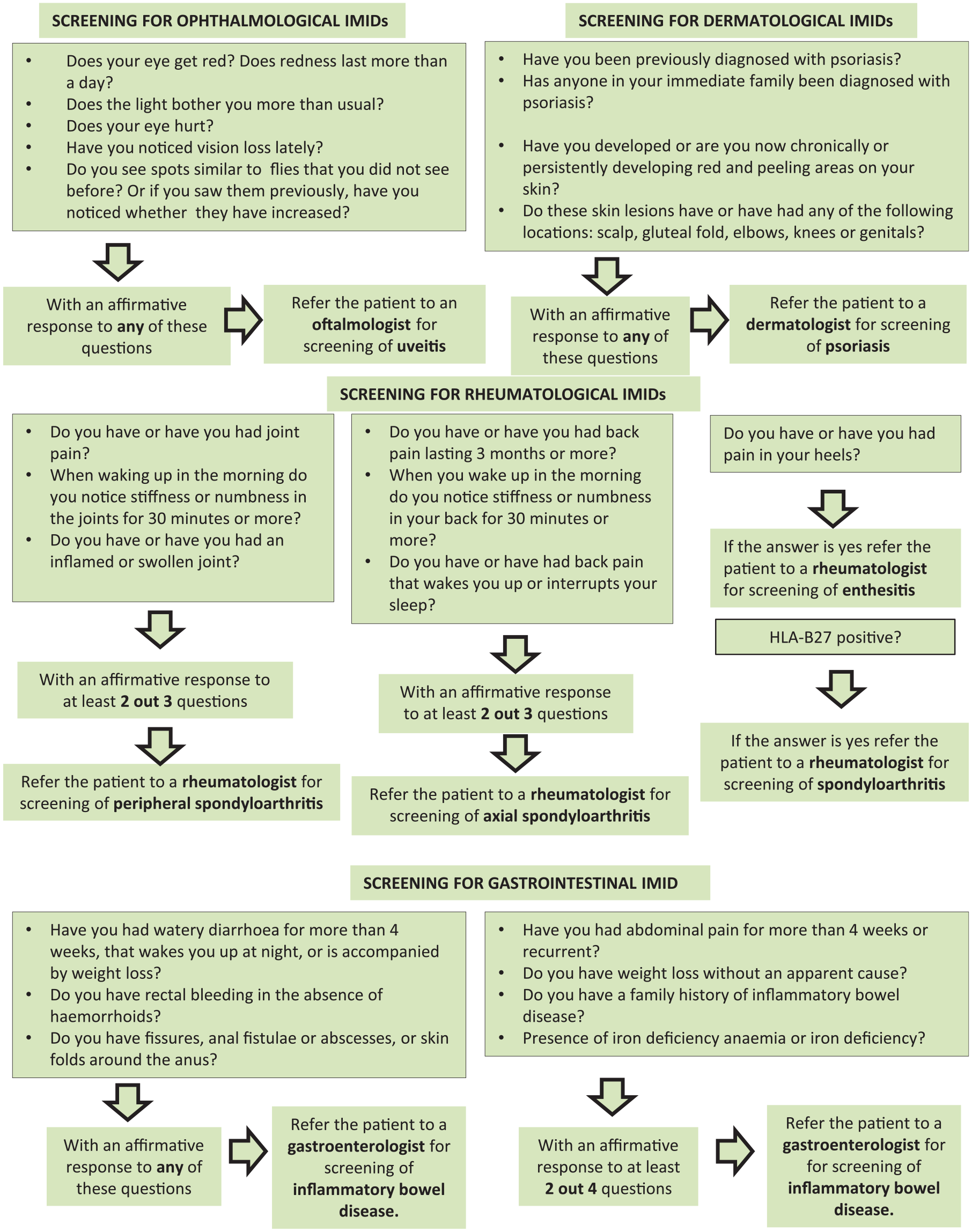
Flowchart of questionnaire development for early detection of IMIDs.
In the first and second rounds of evaluation, 28 out of 28 panelists (100.0%) responded to the first questionnaire. Similarly, all specialists (7 for each specialty) responded to the second questionnaire in the first and second rounds but only to the questions referred to their specialty. After two rounds of voting, a consensus was reached in 41 out of 53 items of the first questionnaire (77.3%): 37 items in the first round and 4 in the second round. Analogously, after two rounds of voting, consensus was reached in 31 out of 31 questions included in the second questionnaire (100.0%): 27 in the first round and 4 in the second. Considering both questionnaires, consensus was reached in 72 out of 82 statements evaluated (87.8%). All of them were consensus on agreement.
Table 1 summarizes conclusions and recommendations drawn from the responses to the first questionnaire. Based on the second questionnaire, Figure 1 illustrates the agreed questions about screening for coexisting IMIDs in a design that can facilitate its use in clinical practice. The degree of agreement on these questions is shown in Table S1.
Discussion
In this multidisciplinary Delphi consensus, we propose practical and simple screening questions for the early identification of coexisting IMIDs in patients already affected by one of these inflammatory conditions. In addition, we provide some consensual recommendations to guide the adoption of a multidisciplinary approach for the management of IMIDs, including IBD, SpA, PsO, and uveitis.
Although each of these inflammatory diseases has unique epidemiology, the prevalence of IMIDs as a whole is about 5–7% in Western countries.2,3 Despite this remarkable prevalence, the results of our study seem to suggest that healthcare providers and managers might not be fully familiar with this concept. This lack of knowledge may pose the risk of underestimating the significant clinical and economic burden of the IMIDs. 17 Furthermore, many members of the panel have the perception that the care of patients with one or more IMIDs might not be entirely adequate in our health system. The panel also agreed that there is a need to further develop a multidisciplinary approach to the care of patients with these conditions.
Multidisciplinary care of patients with IMIDs provided by a team composed of several healthcare professionals is considered a valuable approach and is widely recommended. 4 For instance, the updated 2015 EULAR (European League Against Rheumatism) recommendations for the management of PsA state that this condition may require multidisciplinary treatment, and that, in the presence of clinically significant skin involvement, a rheumatologist and a dermatologist should collaborate in diagnosis and management. 18 Accordingly, GRAPPA (Group for Research and Assessment of Psoriasis and Psoriatic Arthritis) recommendations consider that multidisciplinary and multispecialty assessment and management of PsA will be most beneficial for individual patients. 6 Regarding other IMIDs, and in line with these principles, the updated 2016 ASAS (Assessment of SpondyloArthritis international Society)/EULAR management recommendations for axial SpA states that this is a potentially severe disease with diverse manifestations, usually requiring multidisciplinary management coordinated by a rheumatologist. 9 Other groups also consider that the management of musculoskeletal and extra-articular manifestations of SpA and PsA should be coordinated, as needed, between a rheumatologist and other specialists, such as dermatologists, gastroenterologists, and ophthalmologists. 19 Regarding other IMIDs, psoriasis guidelines, such as the National Institute for Health and Care Excellence (NICE) clinical guideline on the assessment and management of psoriasis, also stress the importance of interdisciplinary teamwork when both skin and joints are significantly affected. 10 Finally, with regard to IBD, NICE recommends that services provide age-appropriate support from a multidisciplinary team for people with IBD, and their family members or caregivers. The team should have access to essential supporting services with an interest in IBD, including a rheumatologist, an ophthalmologist, and a dermatologist, among others. 20
The panel agreed that this multidisciplinary approach, or the implementation of multidisciplinary units for the care of patients with IMIDs, might provide many benefits such as reducing the cost of care, improving the quality of care, allowing for better management of the drugs, or decreasing the delay in the diagnosis of coexisting IMIDs. These perceptions are in accord with recent studies indicating that PsO and PsA multidisciplinary units may facilitate early diagnosis of these conditions, offer a more comprehensive treatment approach and timely treatment initiation, and increase patient satisfaction and quality of life.21–23 Similarly, patients with IBD exposed to a multidisciplinary IBD clinic may have lower rates of IBD-related hospitalization, lower odds of corticosteroid dependence, and lower risk of IBD-related surgeries as compared with patients under standard practice. 24
In addition to the clinical impact of IMIDs on patients in terms of physical suffering, impaired function, and diminished quality of life, these conditions have a significant economic impact on both the individual patient and society as a whole.25,26 In IBD, coordinated multidisciplinary teams might decrease health care utilization, number of surgeries, and chronic steroid use, and, as a result, they may reduce costs. 27 Data about the cost-effectiveness of different multidisciplinary care models for PsA is scarce. 28 There is a need to develop rigorous research on the cost-effectiveness of different multidisciplinary approach models or units, yet the direct and indirect economic impact of this approach in terms of health outcomes is difficult to quantify. 28
Despite the general agreement, in the literature and in our consensus, on the benefits of multidisciplinary approaches, the best multidisciplinary model of care remains to be defined.11,12 For instance, in the management of PsA, different types of models have been described, including face-to-face, parallel, or preferential circuit models.12,29 Our panel considered that joint face-to-face consultations (two or more specialists attend to the patient in the same room) and parallel consultations (two or more specialists attend the patient sequentially in different rooms on the same day) are adequate models to provide multidisciplinary health services. There was also agreement on the use of preferential circuits (two or more specialists attend to the patient in a relatively short time, e.g. in a maximum period of 2 weeks), but this consensus was reached after the second round. Conversely, there was no consensus on the model that suggests the implementations of telematic consultations [two or more specialists deal with a specific case but not face-to-face, e.g. through some information and communication technology (ICT)].
Our consensus provides some insights into the types of patients that should be attended to in theoretical IMID multidisciplinary units and in the organization of the service (Table 4). Interestingly, the panel agreed that these units should be focused on patients with two or more coexisting IMIDs or those that are especially complex from a diagnostic or therapeutic point of view. The results suggest that patients with only one IMID without diagnostic or therapeutic complexity might benefit from standard care.
Conclusions and recommendations.

IMID, immune-mediated inflammatory disease.
The coexistence of IMIDs within the same patient is common since they have a common genetic background and share pathogenic mechanisms. 4 For instance, in a meta-analysis of 143 studies in patients with AS (n = 44,372) the prevalence of uveitis, PsO, and IBD was 25.8%, 9.3%, and 6.8%, respectively. 30 Another meta-analysis of 71 studies reporting on the prevalence of sacroiliitis, AS, arthritis, enthesitis, and dactylitis in IBD patients found that the prevalence was 13% for arthritis, 10% for sacroiliitis, and 3% for spondylitis. 31 In the case of uveitis, a large percentage of patients with clinically significant anterior uveitis have an undiagnosed SpA (⩾50%), and this percentage is even higher if the HLA-B27 haplotype is positive. 32 Any delay, even if only a few months, in the diagnosis of coexisting IMIDs (e.g. the diagnosis of PsA in patients with PsO), could lead to an irreversible progression of joint damage and functional disability. 4 It has been suggested that one of the goals of the multidisciplinary approach is the early diagnosis of other IMIDs that may coexist with the condition that has been diagnosed first. 4 Our consensus developed an agreed questionnaire that may be helpful in the early recognition of coexisting IMIDs in patients with one IMID, in particular focused on screening for SpA, uveitis, PsO, and IBD. This simple questionnaire was agreed upon by specialists related to the different conditions (uveitis, PsO, PsA, or IBD) and not by the entire panel. When developing and evaluating the questionnaire, some issues arose concerning whether to include more details into the questions, such as the age of the patients, whether the onset of symptoms is chronic or acute, or the inclusion of additional symptoms. We are aware that we could go into more detail in many questions, but we rather prefer to keep the questionnaire simple with fairly broad questions, losing specificity, but perhaps increasing sensitivity. The tool we are proposing needs to be validated and does not preclude the use of other screening tools.13,14,33,34 On the contrary, it can be used in addition to these other tools.
Despite the apparent benefits of a multidisciplinary approach to the management of IMIDs, many questions remain. For example, the benefit of these approaches has not been formally demonstrated from a cost-effectiveness point of view. 35 In a recent study, a tight control strategy to treat PsA was an effective intervention in the treatment pathway; however, the study did not find tight control to be cost-effective in most analyses. Authors suggested that lower drug prices, targeting polyarthritis patients, or reducing the frequency of rheumatology visits might improve value for money metrics in future studies. 36 On the other hand, although IMID detection strategies may be adequate to detect the early stages of these diseases (subclinical and prodromal phases), formal proof of this is lacking. This is a key issue as we know that some therapeutic interventions in the subclinical and prodromal phases of some of these diseases are effective and could change the natural history of these conditions. 37
In recent years, combined clinics are becoming a novel model of care for patients with coexisting IMIDs. They potentially improve outcomes, patient and physician satisfaction, and efficiency. Nonetheless, as more of these clinics are established, we will need to further understand their impact on outcomes and care processes. 11
The limitations of this article are those of the Delphi consensus, particularly the impossibility of including the individual opinions of the panelists or more details into the proposed items. Panel selection is another limitation of the Delphi methodology, although we consider that the expertise of the panel is contrasted, and that their opinion represents the majority opinion of other experts in the field. The possible influence of the scientific committee in the consensus is limited since they did not participate in the Delphi vote.
In summary, IMIDs are a group of highly disabling chronic conditions that share inflammatory pathways and have severe clinical and economic consequences. The success of IMID management is reliant upon coordination and close collaboration among different health providers. Optimal management of IMIDs using a multidisciplinary approach is necessary, but remains a major challenge. We provide some recommendations to guide the adoption of this multidisciplinary approach, and a simple tool that may be useful for earlier recognition and referral of coexisting IMIDs in patients with one IMID.
Supplemental Material
Supplementary_table_1_2 – Supplemental material for Improving the standard of care for patients with spondyloarthritis-related immune inflammatory conditions: results of a Delphi study and proposal for early detection
Supplemental material, Supplementary_table_1_2 for Improving the standard of care for patients with spondyloarthritis-related immune inflammatory conditions: results of a Delphi study and proposal for early detection by Pablo Coto, Sabino Riestra, Paloma Rozas, Ana Señaris and Rubén Queiro in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors would like to acknowledge the contribution of the participating panellists, who voted the statements following the Delphi methodology: Alfredo Adán Civera (Ophthalmology), Guillermo Alcaín Martínez (Gastroenterology), Sara Alonso Castro (Rheumatology), Carolina Álvarez Castro (Rheumatology), Susana Armesto (Dermatology), Francesc Casellas (Gastroenterology), Pablo de la Cueva (Dermatology), Rita de la Cruz Kuhnelb (Ophthalmology), Ana Echarri Piudo (Gastroenterology), Noemí Eiris (Dermatology), Marta Feito (Dermatology), Luis Fernández-Vega Sanz (Ophthalmology), Alex Fonollosa Calduch (Ophthalmology), Santiago García López (Gastroenterology), Rosa Izu (Dermatology), Beatriz Joven Ibáñez (Rheumatology), Marc Julià (Dermatology), Ana Laiz Alonso (Rheumatology), Roberto Martínez Rodríguez (Ophthalmology), Jesús Merayo Lloves (Ophthalmology), Carlos Montilla Morales (Rheumatology), Isla Morante Bolado (Rheumatology), Fernando Muñoz Núñez (Gastroenterology), Jose A. Pinto Tasende (Rheumatology), Montserrat Rivero Tirado (Gastroenterology), Cristina Rodríguez Gutiérrez (Gastroenterology), Laura Salgado (Dermatology), Antonio Sampedro López (Ophthalmology).
The authors would like to acknowledge Pablo Rivas, who provided medical writing support on behalf of Springer Healthcare
Funding
The author(s) received no financial support for the research, authorship, and publication of this article.
Conflict of interest statement
Pablo Coto has received honoraria, research grants, participated in advisory boards or acted as a speaker to Abbvie, Lilly, Novartis, Celgene, UCB, Janssen.
Sabino Riestra has served as a speaker, consultant and advisory member for or has received research funding from MSD, Abbvie, Pfizer, Takeda, Janssen, Kern Pharma, Tillots Pharma, and Ferring.
Rubén Queiro has received payments as panelist, lecturer, member of advisory boards and as researcher from Abbvie, MSD, Pfizer, Lilly, Novartis, Janssen, UCB, and Celgene. He has also received unrestricted research funds from Abbvie and Novartis.
Paloma Rozas and Ana Señarís declare that they have no conflict of interest.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
