Abstract
The pathophysiology, diagnosis and treatment of female sexual interest in pre- and post-menopausal women present a complex arena for patients and physicians to navigate. Flibanserin was the first pharmacologic treatment, approved by the United States Food and Drug Administration in August 2015, for hypoactive sexual desire disorder (HSDD) in premenopausal women. Side effects, contraindications and lack of approval in postmenopausal women are all limitations, as are issues surrounding patient and physician knowledge and access. Testosterone, buspirone, sildenafil, bupropion, bremelanotide, as well as herbal medications (Herbal vX or Tribulus terrestris) have demonstrated some clinical benefit in women with sexual dysfunction disorders however, trials have significant design, dosing or generalizability limitations. Nonpharmaceutical cognitive behavioral therapy, mindfulness meditation, pelvic floor therapy, and clitoral stimulators are also interventions women may pursue. This manuscript will explore the clinical data regarding these therapeutic modalities so as to bring attention to this issue of female HSDD, to offer an overview of current research, and to incite providers to initiate discussion among themselves and their patients.
Keywords
Introduction
Sexuality and intimacy are common expressions of human identity and health. Theories developed by Alfred Kinsey and then further researched by William Masters and Virginia Johnson, at Indiana University, USA, characterized sexual response as a linear series of events: excitement, plateau, orgasm and resolution (Figure 1) [Masters and Johnson, 1966]. Further behavioral research developed more complex, nonlinear models that incorporate issues such as motivation, emotional intimacy and partner satisfaction (Figure 2) [Basson, 2000]. Neuroendocrine hormones are necessary, but are not the exclusive contributors to the female sexual response, function and pleasure, through modulation in dopamine, opioids, serotonin, prolactin and oxytocin [Bradshaw et al. 2008; Kingsberg et al. 2015]. Unfortunately, throughout a woman’s lifetime, there are a myriad of psychological, psychosocial and physical ailments that interfere with her ability to engage or maintain sexual satisfaction. The past decade has seen progress in the investigation and treatment of female sexual disorders. Distinct causes, to name a few, have been identified as hormonal imbalances, medication effects, chronic medical illness, mood disorders, interpersonal relationship issues, as well as specific sexual disorders [American College of Obstetricians and Gynecologists, 2011]. Various treatment modalities have been researched however, few have demonstrated clinical efficacy in rigorous trials. In August of 2015 the United States Food and Drug Administration (US FDA) approved flibanserin, a serotonin agonist/antagonist, for the treatment of hypoactive sexual desire disorder (HSDD), which has created a stir of excitement among providers and treatment-seeking patients [Sprout Pharmaceuticals Inc, 2015].
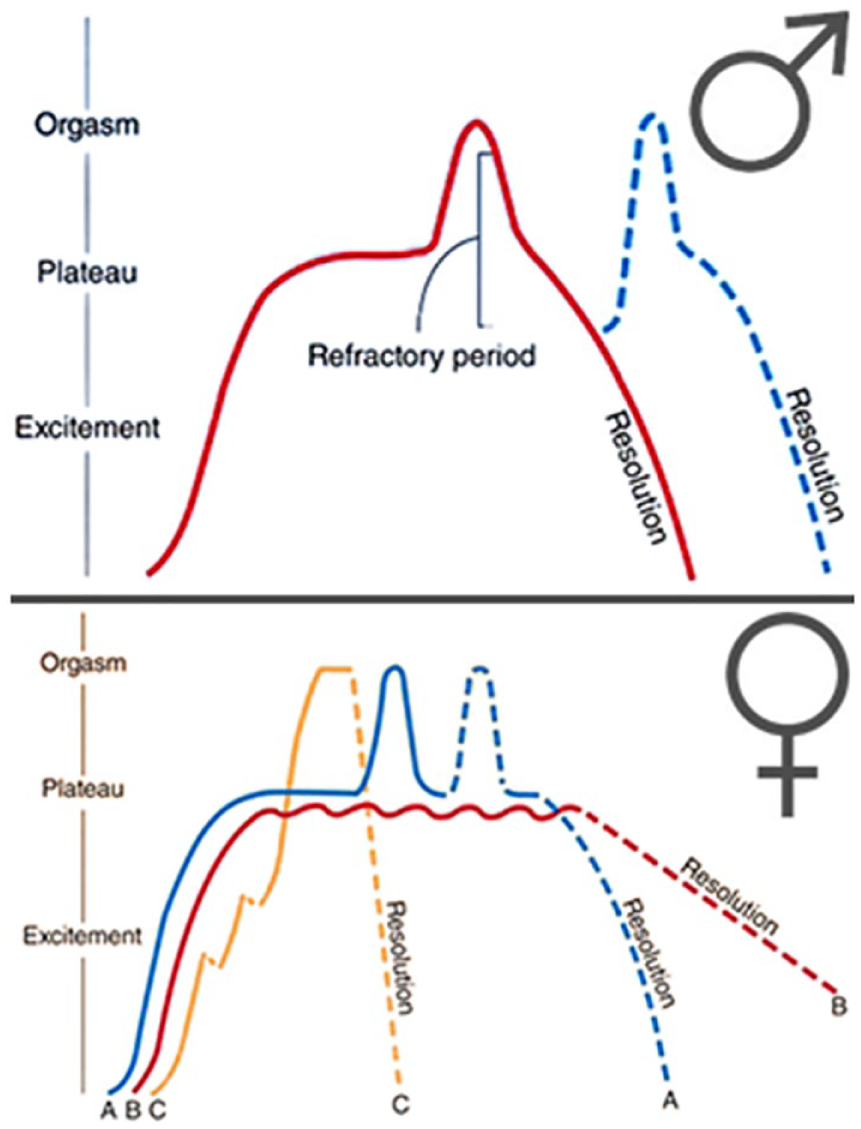
The female sexual response. Masters and Johnson, 1966.

Basson’s Non-Lineal Model. Association of Reproductive Health Professionals, 2008.
HSDD was previously defined by the fourth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) as ‘persistently or recurrently deficient (or absent) sexual fantasies and desire for sexual activity … (that) causes marked distress or interpersonal difficulty… (and) sexual dysfunction is not better accounted for by another Axis I disorder and is not due exclusively to the direct physiological effects of a substance or general medication condition’ [American Psychiatric Association, 2000]. Sexual arousal disorder is defined as ‘Persistent or recurrent inability to attain, or to maintain until the completion of the sexual activity, and adequate lubrication-swelling response of sexual excitement. The disturbance causes marked distress or interpersonal difficulty. The sexual dysfunction is not better accounted for by another Axis I disorder and is not due exclusively to the direct physiological effects of a substance or general medical condition’ [American Psychiatric Association, 2000]. In the DSM-5, HSDD and Sexual arousal disorder have been combined and renamed as female sexual interest/arousal disorder (FSIAD) [American Psychiatric Association, 2013]. As most of the published research involving the drug flibanserin (Addyi) utilizes the DSM-IV definition of HSDD and not the new combination definition of FSIAD, we will proceed to refer to HSDD for the duration of the article.
HSDD affects women across all age groups; its overall prevalence ranges from 5.4–13.6%. Prevalence is greater in women between the ages of 40–60 and in women who have undergone surgical menopause compared with natural menopause [American College of Obstetricians and Gynecologists, 2011]. A HSDD Registry of 1574 patients cited that 67.5% of the women were frequently or always distressed by their lack of sexual desire [Rosen et al. 2012]. Women were more likely to be married and living with their sexual partners, approximately 70% were happy in their relationships, and 60% were satisfied with partner communication. Less than half of these women sought professional care or assistance for their HSDD. Of those who did, 7.6% pre-menopausal and 23.7% post-menopausal women accepted hormonal treatment. Common reasons for declining to seek out care included: the false notion that diminished sexual desire is an inevitable part of aging, and that there are no treatments available for female sexual disorders [Rosen et al. 2012].
US FDA approved treatment for HSDD: flibanserin
Flibanserin was not initially developed for the treatment of HSDD; early clinical trials investigated its impact on major depressive disorder (MDD) [Deeks, 2015]. Flibanserin’s chemical structure is 2HBenzimidazol-2-one, 1,3-dihydro-1-[2-[4-[3-(trifluoromethyl)phenyl]-1-piperazinyl]ethyl] (Figure 3). Its chemical properties include: a half-life of approximately 11 hours, steady state was achieved at 3 days of dosing, Cmax of 419 ng/ml and mean AUC0-inf was 1543 ng × h/ml, with bioavailability of approximately 33% after oral dosing (Figure 4). In vitro flibanserin demonstrated high affinity for serotonin receptors (5-hydroxytryptamine or 5-HT), with agonist activity at 5-HT1A and antagonist activity at 5-HT2A. The majority of the substrate was bound to albumin. It was primarily metabolized by CYP3A4 and, to a less extent, by CYO2C19. Taking flibanserin with food was noted to increase the amount absorbed by slowing the rate of transit [Sprout Pharmaceuticals Inc, 2015]. In MDD clinical trials, women treated with flibanserin reported improvement on measures of sexual desire and drive [Deeks, 2015]. This in turn led to investigation of flibanserin’s effect on HSDD.

Structure of Flibanserin. Sprout Pharmaceuticals Inc., 2015.

Mean + SD Plasma Flibanserin Concentration-Time Profiles in Healthy Female Subjects Following a Single Oral Dose of 100mg of Flibanserin (Linear Scale). Sprout Pharmaceuticals Inc., 2015.
Approval for flibanserin as treatment for HSDD was submitted to the US FDA based on three randomized, double-blinded, placebo-controlled clinical trials in premenopausal women aged 19–55 years old (mean age 36) whose treatment spanned 24 weeks of enrollment. Participants in all trials received either flibanserin 100 mg once daily at bedtime (n = 1187) with 69% completion rate or placebo (n = 1188) with 78% completion rate. Primary study endpoints included number of satisfying sexual events (SSEs) and sexual desire; studies 1 and 2 had a different sexual desire endpoint than did study 3. The primary sexual desire endpoint for studies 1 and 2 was a change in baseline to week 24 based on the response to ‘indicate your most intense level of sexual desire’ scoring 0 (no desire) to 3 (strong desire). Participants recorded responses on a daily basis, which were then combined for a 28-day period to provide a monthly sexual desire score. The primary sexual desire endpoint for study 3 was the Female Sexual Function Index (FSFI Desire). There was a statistically significant increase in SSEs from baseline in all three clinical trials (mean SSE at baseline for studied 1, 2, and 3: 3, 2.6, 2.5 and following treatment: 3.9, 3.2, 3.5 respectively) with p values of <0.01 or greater (Table 1). Only study number 3 demonstrated a significant change in FSFI from 1.9 to 12.3 [Sprout Pharmaceuticals Inc, 2015].
Efficacy Results in Premenopausal HSDD Patients in Studies 1,2, and 3. Sprout Pharmaceuticals Inc., 2015.

Many adverse reactions experienced by enrolled patients in the flibanserin group were similar to those caused by traditional SSRIs: dizziness (11.4%), somnolence (11.2%), nausea (10.4%), fatigue (9.2%), insomnia (4.9%), and dry mouth (2.4%) [Sprout Pharmaceuticals Inc, 2015]. The more concerning side effects were noted to be hypotension, syncope, and somnolence particularly with alcohol or with moderate/strong CYP3A4 inhibitors such as antifungal medications or grapefruit juice. Patients are warned to discontinue flibanserin 2 days prior to taking another CYP3A4 inhibitor and to refrain from restarting flibanserin until 2 weeks after the inhibitor was discontinued [Deeks, 2015]. Side effects were noted in 0.2% of treated participants compared with 0.1% in the placebo group. Additionally, severe hypotension that required treatment was unacceptably high with the concomitant use of alcohol and was only tested in a subset of participants. Women who desire treatment with flibanserin must abstain from alcohol and other products that contain CYP3A4 inhibitors. US FDA approval was granted with strict precautions and the establishment of the Risk Evaluation and Mitigation Strategy (REMS) called the ADDYI REMS program, through which prescribers, outpatient pharmacies, and inpatient pharmacies are educated and certified to prescribe and distribute flibanserin. The purpose of this program is to inform providers and patients of the hypotension and syncope that occur when flibanserin is consumed with alcohol [Sprout Pharmaceuticals Inc, 2015]. The cost of the medication is an additional consideration. Lack or delay of preauthorization insurance coverage can bring the price to over US$10 a pill; as benefits have been demonstrated with nightly use for 28 weeks, treatment with flibanserin can become financially inaccessible to many women.
Concern about balance of clinical benefits and efficacy with daily flibanserin, versus the side effects, expenses and risks of treatment were given a critical review in a recent JAMA meta-analysis, which compiled data from three unpublished and five published clinical trials [Jaspers et al. 2016]. A total of 5914 women were included in the analysis. SSE, as an outcome, was noted to be increased by 0.49 [95% confidence interval (CI), 0.32–0.67] in pooled mean differences from baseline among treated women. Reported desire increase was noted to be 1.63 (95% CI, 0.32–0.67) and FSFI 0.27 (95% CI, 0.17–0.38). Risk of study discontinuation due to adverse events (AEs) was 2.19 (95% CI, 1.50–3.2) times for flibanserin versus placebo, as reported in four studies (p < 0.001). Dizziness, inability to work or undertake activities, syncope, hypotension, or somnolence were all AEs. Serious AEs did not differ between the two groups. Authors cited significant concern regarding the clinical relevance of 0.5 SSEs per 4 weeks as a clinically-insignificant margin of improvement with concomitantly reported AEs, which were not consistently reported across all trials. Furthermore, the generalizability of treatment effect was brought into concern given that participants were predominately married, heterosexual, overweight, premenopausal, white women who reported a baseline of 2.5 sexual exposures, absence of other concomitant sexual health disorders, and were willing to engage in sexual encounters for the duration of the clinical trial. Authors cited that biopsychosocial models of intervention that address the ‘medical, psychiatric, psychological, couple-relationship, and sociocultural domains’ should be explored as primary therapy [Jaspers et al. 2016].
Non-US FDA approved treatment for HSDD: testosterone, sildenafil, buspirone, bupropion, bremelanotide
Currently the US FDA approves no alternative pharmacologic treatments for premenopausal women with HSDD. Clinical trials have demonstrated improved SSEs or scores on sexual function indexes with the following medications: testosterone, testosterone and sildenafil, testosterone and buspirone, bupropion, and bremelanotide. Investigations exist into testosterone alone or with sildenafil or buspirone, buspirone alone, bupropion, bupropion with trazodone, and bremelanotide [Kingsberg et al. 2015]. Continuous therapy with testosterone 300 mcg/day patches (n 279) over 24 weeks was compared with placebo (n 283) among surgically menopausal women with HSDD, who were also taking estrogen therapy. Women were randomized and patches were changed twice a week. SSEs increased from 0.98 episodes per month to 2.10 episodes per month (p = 0.0003), sexual desire scores improved (p = 0.0006) and personal distress scores decreased (p = 0.05) in the treatment group compared with placebo [Simon et al. 2005]. A similar study in physiologically menopausal women with HSDD on estrogen demonstrated an increase of 2.1 SSEs with transdermal testosterone compared with 0.5 SSEs with placebo (p = 0.0001) [Shifren et al. 2006]. In a separate trial 814 physiologic and surgically post-menopausal women (not receiving estrogen) were randomized to two doses of transdermal testosterone (n = 267 150 mcg/day, n = 270 300 mcg/day) or placebo (n = 277). After 24-weeks the 150 mcg/day group had 1.2 more SSEs over 4 weeks (p = 0.11) the 300 mcg/day reported 2.1 more SSEs and the placebo group reported 0.7 more SSEs (p < 0.001). Both doses of testosterone showed statistically significant improvement over placebo with regard to distress and desire indexes [Davis and Braunstein, 2012]. Testosterone has also been tested in numer-ous other formulations (i.e. oral, sublingual, intramuscular, transdermal and topical forms) with similar or inferior results [Nappi et al. 2010]. Adverse effects include hirsutism, acne, and rarely virilization, with an overall favorable toxicity and safety profile. There are theoretic concerns regarding behavior changes, increase risk for breast or endometrial cancer, hepatotoxicity and bone or cardiovascular health however, clinical trials and scientific models have consistently refuted these risks in the doses used to treat female sexual health disorders [Al-Imari and Wolfman, 2012]. Therapy beyond 6 months has not been examined for the treatment of HSDD. However, higher doses and duration of hormonal regimens have been utilized in groups of transgender men to achieve virilization [Kingsberg et al. 2015].
The combination of testosterone with a neuromodulator has been used in combination in the hopes to achieve higher cerebral response to sexual cues with improved balance of excitatory or inhibitory pathways. The addition of sildenafil, a phosphodiesterase type 5 inhibitor, modulates the excitatory pathway whereas the addition of buspirone, a 5-HTA1 partial agonist, modulates the inhibitory pathway. A randomized, double-blinded, placebo-controlled, crossover trial investigated 4-week treatment course with three different therapeutic regiments. A total of 56 women were randomized to sublingual testosterone followed by oral buspirone in 2.5 h, sublingual testosterone and sildenafil, or placebo. Participants took the first medication combination 4 h prior to sexual encounters and waited 48 h prior to repeat dosing. Participants had significantly higher vaginal pulse amplitude measures and subjective sexual desire with testosterone/buspirone compared with placebo (p = 0.006, p = 0.014) [van Rooji et al. 2012]. Testosterone with sildenafil resulted in significant improvement in sexual desire or arousal when compared with placebo (p = 0.017) [Poels et al. 2013]. AEs for both sildenafil and buspirone were reported as flushing, headache, lightheadedness, and dizziness; none of which caused patients to withdraw from the study [van Rooji et al. 2012]. Limitations of this investigation include duration of therapy, timing of administration and repeat dosing, and lack of data regarding SSEs.
Bupropion is a norepinephrine and dopamine reuptake inhibitor, thereby increasing the neuromodulators in the excitatory pathway [Kingsberg et al. 2015]. Clinical trials have concluded that it significantly improves sexual satisfaction, sexual function, and sexual desire when compared with placebo. In a randomized, double-blind, placebo-controlled trial, 232 pre-menopausal women with the diagnosis of HSDD who were actively seeking treatment were randomized to either receiving bupropion sustained release 150 mg or a placebo for 12 consecutive weeks. Participant response to the treatment was evaluated with the Brief Index of Sexually Functioning for Women (BISF-W) and Personal Distress Scale (PDS). At 12-weeks BISF-W scores increased from 15.8 to 33.9 for women receiving bupropion and 15.5–16.9 for women receiving placebo (p = 0.001); sexual thoughts and desires doubled in patients undergoing treatment (p = 0.001). PDS scores decreased by 29.4% in the treatment group versus 4.7% placebo (p = 0.01); 78.1% of treatment patients were satisfied compared with 3.7% of the placebo patients (p = 0.001). Documented AEs included headache, insomnia, dry mouth, nausea, muscle aches, decreased appetite, constipation, stomach ache, fatigue, tinnitus, diarrhea, and somnolence [Safarinejad et al. 2010]. The US FDA label for the treatment of depression includes: agitation, insomnia, decrease appetite and weight loss, hypertension, hepatic and renal impairment, as well as worsening depression, psychosis or mania [GlaxoSmithKline, 2016].
Bremelanotide is a melanocortin 3 and 4 receptor agonist, with effects similar to those of bupropion. Study results from 327 pre-menopausal women randomized to receiving bremelanotide 0.75 mg/1.25 mg/1.75 mg or placebo subcutaneously 45 min prior to sexual activity revealed an increase in SSEs in the 1.25/1.75 mg pooled dose compared with placebo (p = 0.0180). Side effects reported by participants were nausea, flushing, headache, injection-site pain, upper respiratory tract infections, and injection-site pruritus [Clayton et al. 2016]. An earlier intranasal formulation caused concerning elevated blood pressures, but the subcutaneous dose has much milder effects such as nausea, flushing, and headache [Kingsberg et al. 2015].
Nitric Oxide (NO), an important component of male erectile function has been identified in female clitoral tissue [Oliver et al. 2014]. It functions in a similar manner via the NO/cyclic guanosine monophosphate pathway to increase genital blood flow and relax smooth muscles in the pelvis [Musicki et al. 2009]. Amplification of NO via a phosphodiesterase type 5 inhibitor, such as sildenafil, has been shown to improve arousal (p < 0.001), orgasm (p < 0.001), and enjoyment (p < 0.001) in sexually asymptomatic women [Caruso et al. 2003]. But, for women with HSDD, sildenafil has shown no sexual benefit compared with placebo [Berman et al. 2003]. There are very few clinical trials involving sildenafil for women with the diagnosis of HSDD and even fewer clinical trials directly utilizing NO in human patients.
Herbal supplementation and nonpharmacological therapies
With regard to medication therapy, there is the option of herbal supplementation for the treatment of HSDD. Production quality, active ingredients, dosage concentrations, AEs and safety are not regulated by the US FDA or any regulatory third party. Many herbal supplements labeled as libido enhancers were tested in male animals, and are advertised as predominantly beneficial for men [Kotta et al. 2013]. Supplements that have been tested in women include Herbal vX (muira puama and gingko biloba), ArginMax (L-arginine, ginseng, ginkgo, damiana), and Tribulus terrestris [Kotta et al. 2013]. Clinical trials have shown improved sexual desire and satisfaction. A total of 202 women were surveyed before starting Herbal vX and again 4 weeks after treatment with improvement in sexual desire, intercourse, fantasy, and satisfaction in 65% of participants. There was no control group in this trial [Waynberg and Brewer, 2001]. A randomized, double-blind, placebo-controlled study of Tribulus terrestris among 60 women for 4 weeks showed improvement in the treatment group (n = 30) in FSFI (p < 0.001), desire (p < 0.001), lubrication (p < 0.001), arousal (p < 0.037), satisfaction (p < 0.001), and pain (p < 0.041) [Akhtari et al. 2014]. ArginMax was also studied in a double-blind, placebo-controlled trial; 108 women were randomized to either receive placebo (n = 53) or ArginMax (n = 55). After 4 weeks of treatment improved desire (p = 0.03) and satisfaction were noted (p = 0.01) [Ito et al. 2006].
Women with HSDD can engage in treatment with nonpharmacologic therapies such as: sex therapy, vaginal dilators or, if they have elements of sexual arousal/orgasmic disorders, with the EROS device. Sex therapy is led by certified sex therapists trained in the treatment of patients with sexual disorders and concerns. Therapy may involve the development and acknowledgement of realistic sexual goals, exploration of sexual fantasies, identification of specific stimuli and hindrances affecting desire and sexual satisfaction, cueing exercises that assist patients in recalling previously sexually satisfying experiences and carrying over that sensation into the current sexual experience. It is a dialogue between the therapist, the patient and the patient’s sexual partner, with the goal of engaging in sensate focus exercises in which patients and partners pleasure one another through strokes and caresses, practicing mindfulness (i.e. incorporating all five senses into the sexual act and not being distracted or deterred from it), alternate sexual expression or acts, and identifying and correcting sexual boredom [Association of Reproductive Health Professionals, 2010b]. Several studies have been performed evaluating the effects of cognitive behavioral therapy and mindfulness meditation training as independent treatment modalities for women with sexual desire, arousal or distress disorders. The trial results indicate that there are statistically significant improvements in various sexual desire scale scores for those randomized to treatment compared with placebo [Pyke and Clayton, 2015]. Unfortunately, the studies have limitations such as unpublished data, study dropout rates, lack of control groups or randomization, lack of standardized scales, or small sample sizes [Pyke and Clayton, 2015]. This should not discourage providers from offering patients such therapies and initiating discussions regarding sexual health; provider tool kits are available on the Association of Reproductive Health Professionals website [Association of Reproductive Health Professionals, 2010c].
The EROS clitoral therapy device was approved to treat female sexual arousal and orgasmic disorders by the US FDA in April 2000, for women who experience reduced sensation, lubrication, and ability to achieve orgasm (Figure 5). It can be dispensed with a prescription and the device is marketed as Viviant outside of the US without a prescription. The device consists of a battery operated pump with a disposable cup that is placed over the clitoris to apply gentle suction and achieve increased regional blood flow. The vacuum level will not exceed 9.8 inHg. The device was tested on 25 women, 15 with sexual arousal disorder (6 pre-menopausal and 9 post) and 10 without (8 pre-menopausal and 2 post). The device was applied for 5–15 min and Female Intervention Efficacy Index question 4 was the primary outcome of interest (addressing lubrication, sensation, orgasm, and satisfaction). No AEs were noted and all groups reported improvement, with the most improvement noted by the participants with sexual arousal disorder [Urometrics, Inc., 2000].

Eros Device. Zanders M, 2016.
Conclusion
The number of women with HSDD in the US ranges from 5.4–13.6% [American College of Obstetricians and Gynecologists, 2011]. This may not be representative of the burden of disease seen by practitioners in the general population due to limitations in patient disclosure and providers’ ascertainment of dysfunction [Association of Reproductive Health Professionals, 2010a]. Effective techniques to medically and socially address stigma and bias are crucial to the effective identification and therapeutic treatment of women with sexual dysfunction, including HSDD. There is broad consensus that there is inadequate effective teraputic interventions for women with sexual dysfunction. A lack of resources, skills and comfort with treatment are all cited among providers and patients [Association of Reproductive Health Professionals, 2010a]. The USA FDA’s approval of flibanserin represents the first US-approved pharmacologic treatment for premenopausal women with HSDD. Appropriate concerns regarding side effect profiles, generalizability of treatment efficacy with other groups such as non-white or unmarried women, lack of data regarding impact with other concomitant diseases and the lack of approval for post-menopausal women are appropriate critiques. Additionally, there is concern that approximately 1 to 0.5 additional SSEs per month may not represent clinical efficacy that is impactful to women who are living with HSDD.
Investigations into alternative small molecules, such as steroids or proteins, have also show some promise however; many of these regimens require complex repeat dosing and or daily adherence without any long term data or products in the final phases of development. Additionally, to date the pharmaceutical industry has not presented multiple new drug applications for the treatment of sexual dysfunction. Clinical trials in herbal therapies span short duration intervals and are not widely published in the scientific literature. The need to acknowledge and engage the treatment of concomitant interpersonal and relationship, physical or mental health disorders such as depression and anxiety are crucial to the therapeutic care of women with sexual function disorders [Association of Reproductive Health Professionals, 2010a]. Shortcomings of research to date include the focus on limited populations of women who were predominately: married, white, affluent or reside in resource rich countries such as the US, Canada, and European nations. The treatment of women with HSDD and sexual dysfunction will benefit from rigorous randomized and placebo-controlled scientific trials that incorporate larger, more diverse samples, for longer treatment durations. The ADDYI REMS program is an effort to continue ongoing data collection regarding clinical utilization of the product in the US. The ability for patients to access this treatment may be impacted by issues including provider knowledge and bias, as well as, patient access. Consistent with the autonomous and ethical provision of medical care, providers should counsel women on the side effect profile, adherence requirements, and contraindications for use (i.e. alcohol consumption and interacting medications). In the treatment of HSDD, flibanserin is one option that a woman may choose to embrace while addressing how to attain sexual intimacy, fulfillment, health and quality of life.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

