Abstract
Cardiovascular disease (CVD) is a major cause of mortality in type 1 diabetes mellitus (T1D). However, evidence of its risks and management is often extrapolated from studies in type 2 diabetic (T2D) patients or the general population. This approach is unsatisfactory given that the underlying pathology, demographics and natural history of the disease differ between T1D and T2D. Furthermore, with a rising life expectancy, a greater number of T1D patients are exposed to the cardiovascular (CV) risk factors associated with an ageing population. The aim of this review is to examine the existing literature around CVD in T1D. We pay particular attention to CVD prevalence, how well we manage risk, potential biomarkers, and whether the studies included the older aged patients (defined as aged over 65). We also discuss approaches to the management of CV risk in the older aged. The available data suggest a significant CVD burden in patients with T1D and poor management of CV risk factors. This is underpinned by a poor evidence base for therapeutic management of CV risk specifically for patients with T1D, and in the most relevant population – the older aged patients. We would suggest that important areas remain to be addressed, particularly exploring the risks and benefits of therapeutic approaches to CVD management in the older aged.
Keywords
Introduction
Cardiovascular disease (CVD) is a major cause of mortality in type 1 diabetes mellitus (T1D) [Morrish et al. 2001]. The management of CVD in patients with T1D is, however, based on evidence that is at best sparse and often nonexistent. Frequently, management has been based on evidence extrapolated from studies in type 2 diabetes mellitus (T2D) or the general population [The National Collaborating Centre for Chronic Conditions, 2004]. This approach is unsatisfactory for a number of reasons. Firstly, there is emerging evidence that the pathogenesis of atherosclerosis in CVD differs between T1D and T2D and the nondiabetic population [Pajunen et al. 2000; Moreno et al. 2000]. Secondly, the age at which CVD becomes evident differs between T1D and T2D, compromising a reliance on therapies validated in older T2D patients. Thirdly, differences seen in the duration and natural history of CVD in patients with T1D and T2D raise the prospect of a need to initiate cardiovascular (CV) protective therapy earlier in T1D.
These considerations gain increasing importance in the context of a rising life expectancy in T1D [Miller et al. 2012; Lung et al. 2014]. As mortality from renal disease and acute metabolic complications fall (though not in all countries) [Pambianco et al. 2006], a greater number of T1D patients are exposed to the CV risk that associates with an ageing population. A recent cross-sectional survey undertaken by our group using data generated within a UK primary care setting identified a CVD prevalence of 40% in T1D patients aged over 65 years [Chapman et al. 2013].
This narrative review describes existing literature relating to CVD in T1D. Particular focus is applied to CVD prevalence, how well we manage risk, potential biomarkers in monitoring CVD, and the extent to which studies have included older aged participants (defined as aged over 65). We also discuss approaches to the management of CV risk in the older aged. Areas in which there is a paucity of available evidence are identified and a number of strategies suggested for improved research in to, and management of, T1D in older patients.
Methods
A literature search was undertaken using Medline (Ovid) and Embase (Ovid), with respective temporal limits of 1946 to March 2014 and 1974 to 1 April 2014. CVD, as defined by the British Heart Foundation, includes all diseases of the heart and circulation, including coronary artery disease (CAD), heart failure, congenital heart disease and stroke [British Heart Foundation, undated]. The search terms ‘cardiovascular disease’ (disease) and ‘type 1 diabetes’ (population) were used in the search. A total of 1765 papers were initially identified, 461 of which were duplicates.
The remaining 1304 manuscripts were subsequently screened by title. Review articles, conference abstracts and manuscripts with titles that were not relevant to CVD and T1D were removed. A total of 335 articles were subsequently reviewed and each was classified into 1 or more of 31 broad categories. We focused on 11 of these categories which were pertinent to the objective of this review (Table 1). A total of 99 papers from the 11 pertinent categories were included, in addition to 7 additional articles identified from the original search which were considered relevant and 30 papers identified through lateral searches. Flow chart 1 and Table 1 outline the search process of paper selection and the categories manuscripts were assigned to.
Inclusion and exclusion criteria and the broad categories.

CACTI, Coronary Artery Calcification in Type 1 Diabetes; CVD, cardiovascular disease; DCCT, Diabetes Control and Complications Trial; T1D, type 1 diabetes.

Search strategy flow chart with 99 studies included in this review paper.
Prevalence of CVD in T1D
For clarity, we have described prevalence for CVD disease, CVD mortality and CVD risk factors in separate sections. Incidence rate ratio (IRR) describes the incidence rate (incident cases over the follow-up length) of the study population as a proportion of the incidence rate of the controls [Sedgwick, 2010]. Hazard ratio (HR) compares the rate of death or event in the study population with that of the controls across the follow-up period [Sedgwick, 2011]. The standardized mortality ratio (SMR) is a comparison of the number of observed death in the study population with the number of expected deaths based on age specific rates in a standard population [Public Health England]. The cohorts of commonly referenced studies are abbreviated and listed in Table 2.
Abbreviations of the cohorts of commonly referenced studies.

CVD events are increased in T1D
There are a few established large T1D cohorts that provide valuable epidemiological data of CVD prevalence. Amongst these include the Pittsburgh Epidemiology of Diabetes Complication Study (EDC), the Finnish Diabetic Nephropathy Study (FinnDiane), the European Diabetes Prospective Complication Study (EURODIAB) and the Epidemiology of Diabetes Interventions and Complications study (EDIC, 1994) which is a long-term follow up of the Diabetes Control and Complications Trial cohort (DCCT, 1983–1993).
The EDC 1950–1980 cohort reported a CAD incidence density of 0.36 per 100 person-years (n = 906, baseline mean age 28, follow up censored in 2000) [Pambianco et al. 2006]. The FinnDiane followed 3110 T1D (baseline mean age 39) for a median of 5 years and found 269 (9%) patients had an incident CVD [Gordin et al. 2011]. At year 11 of the EDIC study, the CVD event rate was 0.38 and 0.80 per 100 patient-years in the intensive and conventional diabetes treatment group, respectively [follow up 17 years (mean), n = 593 and 589, mean age 45 at follow up] [Nathan et al. 2005].
Two large observational studies show higher rates of CVD in T1D compared with the general population. In the Scottish Registry Linkage Study (SRLS), data for T1D patients aged 20 and above were compared with the nondiabetic populations from the Scottish national surveys. This showed that the age adjusted IRR for first CVD event was 2.3 for men and 3.0 for women [Livingstone et al. 2012]. A separate study using the UK General Practice Research Database (GPRD) compared T1D patients with aged and sex-matched nondiabetic controls between 1992 and 1999. This study reported a HR for major CVD of 3.6 for T1D men and 7.7 for T1D women, with a mean age of 33 years in both groups [Soedamah-Muthu et al. 2006b].
The SRLS analysis found that the IRR of first CVD event for patients aged over 70 was 1.71 for men and 1.85 for women [Livingstone et al. 2012]. In the GPRD study, the major CVD HR for the age group 65–75 was 2.3 for men and 8.3 for women [Soedamah-Muthu et al. 2006b]. In both studies, the figures were lower than the younger age groups, likely reflecting the increasing risk of CVD with age in the general population.
The prevalence of CVD in T1D has also been reported in smaller observational studies within other worldwide populations. In a cohort of 209 Chinese with young-onset T1D (defined as diagnosis before age of 40; T1D participants’ mean age was 27.8 years) in Hong Kong, the incidence of CVD was 0.6 per 1000 person years [Luk et al. 2014]. In comparison, the crude incidence rate of first CVD event for T1D aged 20–39 in the SRLS was 2.73 (men) and 1.76 (women) per 1000 person years [Livingstone et al. 2012]. In a small sample of 100 Saudi Arabian T1D patients, 4% (4/100) developed CV complications [Ammari, 2004].
In summary, CVD prevalence appears to be higher in T1D than the general population, particularly in younger women but this effect was not so pronounced in the older aged group.
CVD mortality is increased in T1D
Two large population based observational studies reported the SMR for CVD in T1D. The Allegheny County childhood onset T1D registry (onset age <18 years) reported a SMR of 12.9 [Secrest et al. 2010] in a cohort of 1075 T1D patients diagnosed between 1965 and 1979, with a mean age of 42.9 years old. Data for childhood-onset T1D from the Norwegian Childhood Diabetes Registry (NCDR) (n = 1906, onset age <15 years, diagnosed between 1973 and 1982, follow up till 2002) reported SMRs of 11 for men and 10 for women with T1D [Skrivarhaug et al. 2006]. A smaller Swiss study assessed mortality of patients with T1D and T2D compared with the general Swiss population between 1974 and 2005. There were 225 Swiss T1D patients with a mean age of 43 years old. This study reported a CVD SMR of 6.6; CVD SMR did not differ significantly between T1D and T2D [Allemann et al. 2009]. Finally, a New Zealand paper of 995 insulin-treated diabetics (including T2D) showed a CVD SMR of 4.48 in T1D diagnosed before the age of 30; the lower SMR in this paper could be due to a dilution effect of T1D with an older age of onset. In fact in the same paper, the CVD SMR was halved in T1D with onset age >30 years compared with <30 years [Florkowski et al. 2003].
CVD related deaths have also been reported as IRR, HR and annual mortality rate. In the SRLS, the IRR for CVD mortality related to T1D was 3.4 and 3.5 for men and women, respectively. The HR for CVD deaths in T1D was 7.4 in the GPRD study [Soedamah-Muthu et al. 2006b]. The annual mortality rate for CVD was 1.4 per 1000 person-years (n = 2787, baseline mean age 33, 7 years follow up) [Soedamah-Muthu et al. 2008].
CVD appears to be the predominant cause of death in adults with T1D. In the World Health Organization (WHO) multinational cohort, CVD accounted for 44% of T1D deaths [Morrish et al. 2001]. In a Danish study with 4821 T1D patients, CVD was the main cause of death [31% (125/402) and 30% (81/271) of all death for men and women, respectively] [Jørgensen et al. 2013]. This was also the case in the GPRD study [Soedamah-Muthu et al. 2006b].
Studies show that acute diabetic complications, such as ketosis and hypoglycaemia, are more likely to be the cause of death in the young, and CVD begins to predominate as patients become older. This was observed in a Japanese nationwide population-based cohort of 1385 T1D patients diagnosed between 1965 and 1979, where a lower mortality from acute diabetic complications and greater mortality from CVD was seen with increasing follow up. Here CVD was described as the main cause of death in those with more than 20 years’ disease duration [Morimoto et al. 2013]. Similarly in the SRLS, the most common cause of death was diabetes [41% (51/123)] for the under 40s but circulatory disease [38% (349/907)] for those aged over 40 [Livingstone et al. 2012]. This trend is supported by the Allegheny study where acute diabetic complication was the main cause of death (73%) within the first 10 years of diagnosis and CVD was the leading cause of death (40%) after 20 years of T1D [Secrest et al. 2010]. Finally, in the NCDR study, acute diabetic complication and violent death was the main cause of death for under 30s but CVD accounts for the most death [30% (11/37)] for over 30s [Skrivarhaug et al. 2006].
Focusing on ischemic heart disease (IHD), the Diabetes UK cohort (23,000 T1D patients followed up till 2000) reported a IHD mortality rate that was higher than the general population across all age groups: the overall SMR for IHD was 4.5 (men) and 8.8 (women). Within this cohort, the SMR for IHD was exceptionally high in young women: female T1D patients at ages 20–29 had a SMR of 44.8 [Laing et al. 2003]. This gender difference for mortality was also seen in Huxley and colleagues’ meta-analysis of 26 studies in T1D (n = 214114); the pooled women-to-men ratio of the SMR for fatal CVD and incident coronary heart disease was 1·86 and 2.54, respectively [Huxley et al. 2015].
Only the Diabetes UK cohort study showed data for older T1D, but this was for IHD: those aged 70–84 had IHD SMR of 2.2 for men and 5.3 for women. These figures are lower than the younger age bands and again likely reflect rising CVD risk with age in the general population, and the possibility that those with CVD are no longer alive to contribute to analyses.
One observational study from a tertiary centre in Australia found that there were more CVD death and risk factors in 354 young onset T2D (age of onset between 15 and 30 years) than the 470 young onset T1D observed [Constantino et al. 2013]. An 18-year observational study conducted in Finland involving 173 T1D and 834 T2D (aged 45–64 years at baseline) found that both types of diabetes had similar CVD mortality, although there was a 3–4 fold increase of risk in men and 10–13 fold increase for women. However, the impact of glycaemic control on CVD mortality was higher in T1D than in T2D: an increment of 1 unit (%) of glycated haemoglobin increased the risk of CV mortality by 52.5% [95% confidence interval (CI) 28.4–81.3] in T1D and 7.5% (95% CI 4.3–10.8) in T2D [Juutilainen et al. 2008].
CV risk factors are increased in T1D
Whilst diabetes itself is a risk factor for CVD, a majority of T1D patient will have at least one further risk factor. The proportion who do so ranges from 69% in 27,358 T1D patients aged 0.25–26 years in a cross-sectional study from a German surveillance database (DPV) to 89% of 177 T1D patients with end stage renal failure and a mean age of 37 in a Spanish study [Schwab et al. 2006; Rueda et al. 2009]. The percentage of T1D patients with 3 or more CVD risk factors ranged from 2% in the DPV study to 15% in a Norwegian Childhood Diabetes and Quality Project (NCDQ) cohort (2658 T1D patients with mean age of 13) [Schwab et al. 2006; Margeirsdottir et al. 2008].
Managing CVD risk
Guidelines for CVD risk management have been proposed by major diabetes associations [American Diabetic Association, 2014; The National Collaborating Centre for Chronic Conditions, 2004; European Society of Cardiology (ESC) et al. 2013]. The American Diabetes Association (ADA) guidelines are outlined in Table 3. These do not recommend routine screening for CVD in patients with diabetes and suggest this does not provide any greater benefit than screening for and actively managing CVD risk factors. The approach to managing CV risk in patients with T1D is consequently comparable with that of the nondiabetic population.
American Diabetes Association 2014 guidelines for type 1 diabetes management.

ACE, angiotensin converting enzyme; ARB, angiotensin II receptor blocker; HbA1c, glycated haemoglobin; HDL, high density lipoprotein; LDL, low density lipoprotein.
Assessment therefore consists of measuring clinical risk factors, calculating risk from appropriate risk engines and actively enquiring about symptoms of CVD. Current risk engines for CVD risk in diabetes are largely based on data from studies of the general population. These risk engines include PROCAM (a cohort of working people) (http://www.chd-taskforce.com/procam_interactive.html) and QRISK®2 (a primary care population) (http://www.qrisk.org/) [Assmann et al. 2002; Hippisley-Cox et al. 2008]. The latter has the option to include T1D.
Achieving target glycated haemoglobin (HbA1c) in T1D
Target HbA1c achievement is generally low (Table 4). The percentage of T1D patients achieving HbA1c < 7% ranged from 13% (SRLS) to 26% in a multicentre outpatient based T1D study in Castilla-La Mancha, Spain (CLM; n = 1465, mean age 39) [Livingstone et al. 2012; Wallymahmed et al. 2005; Sastre et al. 2012]. A recent large observational study using regional and national T1D registries across 19 countries (n = 324,501) showed that only 28% of the people in the whole dataset had HbA1c < 7.5% [McKnight et al. 2014].The overall prevalence of poor glycaemic control has been determined for paediatric patients in a number of cross-sectional studies. This is reported to range from 60.6% (HbA1c > 7.5%) in a paediatric department in Austria (UHVGPD study; n = 264, mean age 13) to 91% (HbA1c > 7%) of a small cohort of Caribbean youth with T1D in Jamaican major referral hospitals (JMRH; n = 36, mean age 18), respectively [Steigleder-Schweiger et al. 2012; Margeirsdottir et al. 2008; Tulloch-Reid et al. 2009].
Achieving CVD risk targets in T1D.

BMI, body mass index; CVD, cardiovascular disease; DBP, diastolic blood pressure; HbA1c, glycated haemoglobin; HDL, high density lipoprotein; IQR, interquartile range; LDL, low density lipoprotein; n/a, not available; NCEP-ATP III, National Cholesterol Education Program-Adult Treatment Panel III; PA, physical activity; SBP, systolic blood pressure; SD, standard deviation; TC, total cholesterol; TG, triglycerides.
In the older aged patient, the SRLS observed that the median HbA1c level for those aged over 60 was 8.1% (male) and 8.3% (female) [Livingstone et al. 2012].
Achieving target blood pressure in T1D
Estimates for the prevalence of hypertension vary between studies (Table 4). In paediatric T1D, the prevalence ranged from 7% in the NCDQ cohort (0.3% on antihypertensives) to 8% in the DPV study (2% on antihypertensives) and 21% in a Lithuanian T1D register database study (LDRDS; n = 539) [Margeirsdottir et al. 2008; Schwab et al. 2006; Dobrovolskienė et al. 2013]. For adults, this ranged from 8% in a group of Iranian T1D patients at the IEMR (n = 219; mean age 23), 13% in a UK consultant led T1D clinic (LAHDCA, n = 218, mean age 34) and 23% in the CLM study to 37% in over 40s in the SRLS [Kalantari et al. 2007; Wallymahmed et al. 2005; Sastre et al. 2012; Livingstone et al. 2012]. In the EDIC year 11 study, 38% and 41% of patients from the intensive treatment and conventional treatment group were hypertensive (>140/90 mmHg) [Nathan et al. 2005]. At baseline, 55% of the deceased versus 22% survivors in the EURODIAB study were hypertensive (>140/90 mmHg); 36 (35%) and 225 (8%) were on antihypertensives, respectively [Soedamah-Muthu et al. 2008].
In the SRLS, a greater proportion of older aged patients were prescribed antihypertensive medications (80% and 79% in over 60s compared with 50% and 44% in those aged 40–59, men and women, respectively). Despite this, median blood pressure remained elevated in the older aged population when compared with younger persons. [Livingstone et al. 2012].
Achieving target lipids in T1D
Four studies have measured overall dyslipidaemia, two of which observed paediatric T1D patients. The CLM study of adult T1D patients found 35% had dyslipidaemia [Sastre et al. 2012]. In the EDIC year 11 study, 52% and 48% of patients from the intensive treatment and conventional treatment group had hyperlipidaemia; 34% and 33% were on a statin, respectively [Nathan et al. 2005]. From the DPV, 29% of paediatric T1D patients had dyslipidaemia, of whom only 0.4% were prescribed lipid lowering therapy [Schwab et al. 2006]. In a small US observational study, 50% of paediatric T1D patients were diagnosed with dyslipidaemia [Reh et al. 2011].
Prevalence of raised total cholesterol (>4.8 mmol/l) ranged from 22.3% in the LDRDS paediatric cohort to 55% of the LAHDCA cohort [Dobrovolskienė et al. 2013; Kalantari et al. 2007; Wallymahmed et al. 2005]. In a cohort of T1D patients aged over 60 years, the SRLS identified a median cholesterol level of 4.0 and 4.4 mmol/l in men and women, respectively, which was slightly lower than that of younger patients (4.4 and 4.8 mmol/l in men and women, respectively, aged 40–59) [Livingstone et al. 2012].
In the paediatric cohort, prevalence of high low density lipoprotein (LDL) (>2.5 mmol/l) ranged from 14.7% in the LDRDS to 67% in the small JMRH cohort [Dobrovolskienė et al. 2013; Tulloch-Reid et al. 2009; Margeirsdottir et al. 2008; Kalantari et al. 2007]. Amor and colleagues identified an improvement in the prevalence of target LDL (<2.5 mmol/l) from 26.3% in 1999 to 65.9% in 2009 in patients undergoing assessment for kidney–pancreas transplant [Amor et al. 2011].
The existing literature relating to high density lipoprotein (HDL) in patients with T1D is poor. The proportion of patients with HDL <1.1 mmol/l or <35 mg/dl (undesirable) ranged from 3.3% in a Colorado cohort studied by Maahs and colleagues [Maahs et al. 2007] and 4.1% in the LDRDS paediatric cohort [Dobrovolskienė et al. 2013] to 7% in the NCDQ cohort [Margeirsdottir et al. 2008] versus 23% in the IEMR cohort to 33% in the JMRH cohort [Kalantari et al. 2007; Tulloch-Reid et al. 2009]. Over 60s in the SRLS had median HDL of 1.4 mmol/l (men) and 1.7 mmol/l (women), which was similar to the younger age group [Livingstone et al. 2012].
Prevalence for high triglycerides (TG) ranged from 18% in the LDRDS and IEMR cohort to 23% in the UHVGPD cohort [Dobrovolskienė et al. 2013; Kalantari et al. 2007; Steigleder-Schweiger et al. 2012]. In the SRLS over 60s cohort, the median triglyceride levels were 1.2 mmol/l (male) and 1.1 mmol/l (female), which was again similar to the younger age group [Livingstone et al. 2012].
The use of statins varied across age groups. In the NCDQ cohort, 0.2% T1D adolescents were prescribed statin therapy, contrasting with 6.4% (23/360) of paediatric T1D patients in Colorado, USA studied by Maahs and colleagues [Margeirsdottir et al. 2008; Maahs et al. 2007]. Within the SRLS, 41% of T1D patients were prescribed statins, although this figure was higher for older patients aged over 60 at 73% for males and 74% for females [Livingstone et al. 2012]. Data are summarized in Table 4.
Smoking status in T1D
The prevalence of smoking in T1D adult patients ranged from 7% in the IEMR study to 35% in the DPV study (Table 4) [Kalantari et al. 2007; Livingstone et al. 2012; Sastre et al. 2012; Soedamah-Muthu et al. 2008; Schwab et al. 2006]. In the FinnDiane cohort, 60% and 40% of T1D patients with incident CVD events and no CVD events smoked [Gordin et al. 2011]. A total of 14% and 11% of those who were in the intensive treatment and conventional treatment groups, respectively, were a current smoker at year 11 of the EDIC study [Nathan et al. 2005]. In the paediatric cohort, the NCDQ cohort identified that 3% of those ⩾12 years old reported smoking [Margeirsdottir et al. 2008]. For older aged patients, 19% male and 15% female T1D in the SRLS smoked, this was lower than the younger patients [Livingstone et al. 2012].
Achieving a healthy diet in T1D
In studies to date, there appear to be an overconsumption of fat and poor fibre consumption by patients with T1D (Table 4). In the NCDQ cohort, almost all study subjects had higher fat intake and lower fibre intake than recommended [Margeirsdottir et al. 2008]. Similar dietary pattern was observed in children aged under seven with T1D (n=24) in a small Swedish study [Sundberg et al. 2014]. In the EURODIAB study, European T1D patients consumed a high atherogenic diet, and very few patients achieved the recommended intake of dietary fibre (2%) and saturated fat (13%) [Soedamah-Muthu et al. 2013]. When comparing dietary patterns geographically, fibre intake was lowest in eastern Europe and highest in southern Europe [Toeller, 2002]. No study specifically observed the older age group.
Achieving target physical activity levels in T1D
Whilst studies have yet formally and objectively measured exercise and physical activity in T1D, a number of studies have analysed this subjectively (Table 4). The EURODIAB study quantified exercise through the use of questionnaires sent to over 2000 patients and showed that about a third undertook no or only mild physical activity [Tielemans et al. 2013]. Similarly, the Finnish Diabetic Neuropathy Study (FinnDiane) showed that 23% of people with T1DM were sedentary with a further 21% doing less than 1 session of exercise per week [Wadén et al. 2008]. A significant proportion of patients with T1D may therefore be considered physically inactive.
Weight, body mass index (BMI) and the ‘metabolic syndrome’ in T1D
A total of 13.4% of the children in the LDRDS and 22% of the JMRH adolescent cohort have been described as overweight [Dobrovolskienė et al. 2013; Tulloch-Reid et al. 2009]. The percentage of T1D patients classified as obese ranged from 4% of the NCDQ paediatric cohort to 15% of the CLM cohort [Margeirsdottir et al. 2008; Tulloch-Reid et al. 2009; Sastre et al. 2012]. A total of 20% of the UHVGPD cohort to 25% of young adults in the DPV study had a BMI > 90th centile [Steigleder-Schweiger et al. 2012; Schwab et al. 2006]. In the EURODIAB cohort, the baseline BMI for the deceased and the survivors were virtually the same; in those who survived the mean BMI was 24 [Soedamah-Muthu et al. 2008].
The Paediatric Diabetes Consortium’s study of 520 T1D youth (mean age 10, median BMI percentile 50%) found that the largest increase in BMI was in the first 3 months post diagnosis of T1D and thereafter remained stable at 12 months, thus reflecting gain of weight lost before diagnosis [Gregg et al. 2015]. The DCCT group examined the temporal trends of overweight/obesity across 4 cohorts representing different time point over a decade (1999, 2002, 2006, 2009; n = 507, mean age 12.0–12.8, mean BMI percentile 70–72) and found that the prevalence of overweight/obesity was similar, ranging from 27% to 36% [Baskaran et al. 2015]. However, at a cohort level, the Pittsburgh EDC group followed 589 T1D from 1986 to 1988 for 18 years and found that the prevalence of overweight and obesity increased by 47% and 7 fold, respectively (mean age 29, baseline prevalence of overweight and obesity 29% and 3%). Simultaneously, the use of intensive insulin regime increased from 7% to 82% and was quoted as a predictor of weight change [Conway et al. 2010].Similarly, a 10-year observational study at an Ethiopian hospital diabetes clinic reported that BMI increased from 16 to 18, although this remains in the underweight category (2000–2009, n = 778, mean age 29) [Abebe et al. 2013].
For the over 60s T1D patients, the median BMI was 27 in the SRLS; this was similar to the younger age groups [Livingstone et al. 2012] (see Table 4).
A Spanish hospital study showed that 32% of T1D outpatients had metabolic syndrome [Chillarón et al. 2010]. From the FinnDiane study, the prevalence of metabolic syndrome (by WHO definitions) at baseline was 44% [Thorn et al. 2009]. There were no data targeting the older aged group.
Whilst we accept that striving for prespecified targets may be inappropriate for some patients, these studies suggest that risk factors for CVD are suboptimally controlled in patients with T1D. We have previously shown in a UK single city multihospital study that targets of CVD risk factors were suboptimally recorded and only 0.7% of patients were achieving all minimal dataset target (total cholesterol, smoking, HbA1c) [Syed et al. 2007].
Biomarkers for CVD in T1D
Whilst the approach of managing CV risk through clinical assessment is simple, relatively straightforward and can be used in the clinical situation, there are a number of potential biomarkers for CVD in T1D that may prove to be useful. These are outlined in Table 5.
Potential biomarkers for CVD risk in T1D that has been investigated.
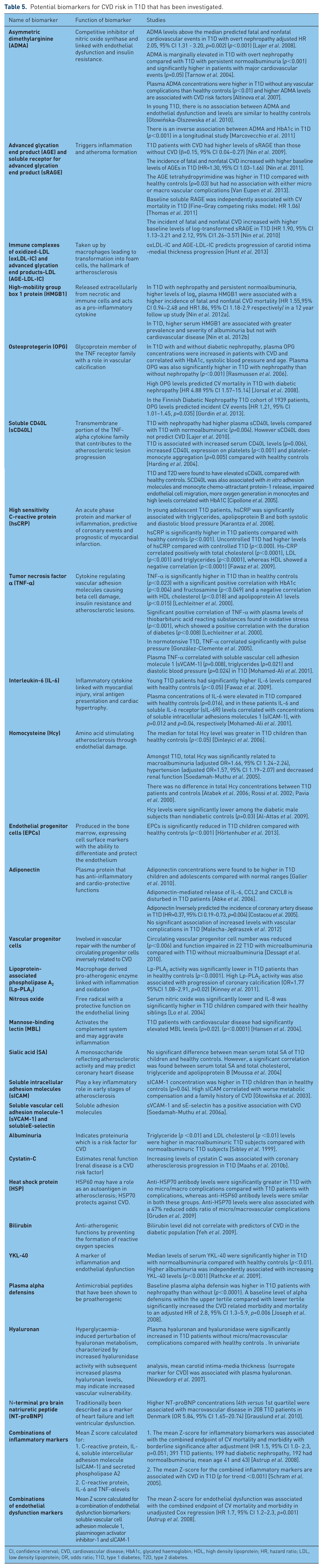
CI, confidence interval; CVD, cardiovascular disease; HbA1c, glycated haemoglobin; HDL, high density lipoprotein; HR, hazard ratio; LDL, low density lipoprotein; OR, odds ratio; T1D, type 1 diabetes; T2D, type 2 diabetes.
Management of CV risk in the older aged patient with T1D
There is a paucity of literature concerning effective strategies for the management of CV risk in the older aged patient with T1D. It appears on the basis of current evidence, however, that strategies should include tight control of both diabetes specific factors, such as blood glucose regulation, and the more general modifiable CV risk factors. The relative benefit afforded by targeting each of these risk factors remains unclear, with no single dominant factor predicting CV morbidity in patients with T1D and evidence to implicate the metabolic syndrome in its pathogenesis [Mäkinen et al. 2009; Thorn et al. 2009].
Interestingly, the Joslin 50-year medallist study provides evidence to suggest that there is a limit to the extent to which risk management strategies are effective in ageing patients with T1D [Sun et al. 2011]. Substantiating this, the authors provide evidence for a greater prevalence of CVD amongst patients with lower systolic blood pressure, mean arterial pressure, heart rate, total cholesterol and LDL, likely reflecting the use of pharmacological agents amongst these patients. Despite this, there is a clear link in the study between deranged lipids and CV risk, emphasizing a need for effective lipid management in aged patients with T1D. There is, in addition, further evidence within the Golden Years Cohort for genetically determined elevated HDL-cholesterol affording protection from large vessel disease in long-lived subsets of patients with T1D [Bain et al. 2003].
Taken together, these analyses of long-lived patients appear to suggest that HDL control may afford a significant therapeutic target for preventing CVD in ageing patients with T1D. There is, nevertheless, wider evidence amongst nonaged populations for a multifactorial approach to CV risk reduction in patients with T1D. Given that a number of these studies report extended follow up, albeit in younger patients than focused on in this review, their results are likely generalizable to an aged cohort.
Wallymahmed and colleagues provide evidence to suggest that lifestyle modifications may improve CV health within their randomized controlled trial comparing nurse-led CV risk factor intervention to routine care in patients with a mean age of 34.6 years [Wallymahmed et al. 2011]. In identifying positive impacts stemming from nurse-led intervention they do, however, note that much of the improvement seen was likely secondary to greater use of lipid-lowering or antihypertensive agents. It is additionally difficult to translate many of the lifestyle findings relating to young patients with T1D to their older aged counterparts. Chen and colleagues previously identified low physical activity to equate to decreased heart rate variability in children with T1D, for example, suggesting that strategies to improve exercise are important for preventing CVD [Chen et al. 2008].It is not clear whether this is possible in an aged cohort with multiple comorbidities, many of which are likely to be musculoskeletal.
The putative impact of pharmacological agents in older aged patients is again unclear. This is further compounded by the lack of trials evaluating the impact of optimal blood pressure control or use of antihypertensive medications on CVD in T1D. The major clinical trials (UKPDS, HOT, ADVANCE) were conducted in the T2D cohort [UKPDS group 1998; Hansson et al. 1998; Patel et al. 2007]. Nevertheless, observations from the EDC cohort showed that higher blood pressure was associated with higher relative risks of CAD [Orchard et al. 2001]. Amongst the general population, there is an additional recognition that effective management of widened pulse pressure in older aged persons reduces CVD. Created by a concomitant rise in systolic blood pressure and fall in diastolic blood pressure, increases in pulse pressure are recognized to occur earlier in patients with T1D, indicating accelerated arterial stiffness and ageing [Rönnback et al. 2004; Gordin et al. 2012]. The additional recognition that pulse pressure predicts a first ever CVD event in patients with T1D, identified in a cohort with a mean age of 37 years, seems to support the need for effective blood pressure control in older aged patients [Gordin et al. 2011].
There were a few small studies from the literature search that evaluated nonconventional pharmacological treatment in T1D. Cavallo and colleagues evaluated the use of melatonin in lowering nocturnal diastolic blood pressure in 11 T1D and 10 healthy controls using a randomized placebo-controlled double-blind crossover study design and found a significant but marginal reduction in nocturnal diastolic blood pressure (17.8 mmHg versus 16.0 mmHg) [Cavallo et al. 2004]. Djurhuus and colleagues found that magnesium repletion lowered atherogenic lipid fraction in 10 magnesium depleted T1D patient, there was no randomization or control group [Djurhuus et al. 2001]. These studies did not target the older aged T1D patients, have small sample sizes and lack long-term data to support the efficacy in improving CVD risk or mortality.
Poor glycaemic control is predictive of CVD events in patients with T1D, as highlighted by the FinnDiane prospective multicentre study that demonstrated a strong association between HbA1c variability and CVD events [Wadén et al. 2009]. The relationship between glycaemic control and CV health is, however, complex. In their 2010 analysis of 652 patients with T1D followed up over a period of 6 years, Maahs and colleagues identified that whilst good HbA1c control affords changes in fasting lipids, dyslipidaemia medications are nevertheless still required even in patients with well controlled diabetes in order to optimize CV health [Maahs et al. 2010a]. There is also some evidence to suggest that attempting to control blood glucose within too regimented a range might lead to adverse effects, though this is contested. Gruden and colleagues, for instance, argue that their analysis of 2181 T1D patients taken from the EURODIAB Prospective Complications Study suggests that severe hypoglycaemia does not increase the risk of CVD [Gruden et al. 2012]. Similarly, Eeg-Olofsson and colleagues highlight in their observational study of 7454 patients that, whilst CV risks increase with HbA1c levels, there is no J-shaped curve to indicate an increase risk resulting from hypoglycaemia [Eeg-Olofsson et al. 2010]. This linear relationship between HbA1c and CV health is further supported by a number of other authors reporting both observational studies and a meta-analysis [Wadén et al. 2009; Shankar et al. 2007; Selvin et al. 2004]. Somewhat conflictingly, an analysis published by Giménez and colleagues reported the opposite, with repeated severe hypoglycaemia increasing CV risk [Giménez et al. 2012]. This latter study is however a retrospective study and smaller than the EURODIAB studies.
Strategies to ameliorate the potential for CVD in older patients may additionally focus on oxidative stress, exposure to which arguably increases significantly with age. Costacou and colleagues have, for instance, identified that the anti-oxidant alpha-tocopherol provides protection against CAD in patients with T1D [Costacou et al. 2006]. Whilst this research was undertaken in a population with a mean age of 28 years, results were taken at a follow up of 10 years, suggesting an extended advantage to targeted antioxidant therapy which might extend into older age.
In their respective analyses of patients undergoing pancreas transplant alone, both Boggi and colleagues and Larsen and colleagues highlighted a number of improvements to independent CV risk factors, in addition to evidence to suggest directly improved left ventricular ejection fraction resulting from pancreatic transplantation [Boggi et al. 2012; Larsen et al. 2004]. Furthermore, combined pancreas and kidney transplantation for patients with T1D and end stage kidney disease (ESKD) has been associated with significantly lower mean arterial pressure, lower pulse pressure, lower LDL cholesterol and fewer required lipid-moderating medications which is likely secondary to a resultant lower atherosclerotic risk profile [Luan et al. 2007; Fiorina et al. 2001].
There are, finally, numerous reports within the literature concerning the proinflammatory state considered to accompany ageing. Inflammation in this context relates to a chronic overresponse that results in the accrual of cytokines and immune cells predisposing to atherosclerotic disease.
Whilst there is no direct evidence to link this state, often referred to under the umbrella term of ‘inflammaging’, to adverse events amongst aged patients with T1D, it is arguably implicated by work conducted within younger patient populations. González-Clemente and colleagues have, for instance, identified an association between interleukin (IL) 6 levels and lower heart rate variability, implying adverse outcomes stemming from raised cytokine levels [González-Clemente et al. 2007]. Although an untested hypothesis, it is possible that strategies to moderate inflammation amongst ageing patients may positively impact on CV morbidity and mortality.
Discussion
The available data suggest a significant CV burden in patients with T1D and poor management of CV risk factors. This is underpinned by a poor evidence base for therapeutic management of CV risk specifically for patients with T1D and in the most relevant population – the older aged patients. Whilst recent years have seen a decrease in CVD related mortality in patients with T1D [Miller et al. 2012], it still remains the leading cause of mortality and therefore significant further effort is required.
We would suggest that important areas remain to be addressed, particularly exploring the risks and benefits of therapeutic approaches to CVD management in the older aged. Thought will be required around the design of these studies. Clinical CVD outcomes (myocardial infarction, heart failure) may appear sooner than in a younger population because the older aged patients are more at risk and therefore studies could potentially be shorter and/or smaller. We also see greater risk of side effects associated with polypharmacy in the older aged patients and so dropout rates may be higher. The use of surrogate endpoints such as carotid intima thickness and cardiac magnetic resonance imaging may provide useful information more quickly in the interim.
An important and urgent question relates to the benefits of blood pressure, lipid and glucose control in patients with T1D and at what age these benefits become significant. This is particularly relevant in the older aged patients where we risk committing them to many years of therapy against the risk of side effects and potentially minimal benefit.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
