Abstract
Purpose
Sarcopenia is a major health problem in community-dwelling elderly individuals. Hypertension is postulated to aggravate sarcopenia. The present study was performed to estimate the prevalence of and factors associated with sarcopenia among elderly individuals with hypertension.
Methods
This study involved 165 Chinese individuals with hypertension aged ≥60 years who were evaluated for sarcopenia using the Asian Working Group for Sarcopenia criteria. Data on their sociodemographic information, physical illnesses, and clinical and functional status were collected.
Results
The overall prevalence of sarcopenia among elderly individuals with hypertension was 20.2%. The factors significantly associated with sarcopenia were an age of ≥70 years (adjusted odds ratio (OR), 3.01; 95% confidence interval (CI), 1.17–5.39), diabetes (OR, 4.45; 95% CI, 1.32–11.16), osteoporosis (OR, 2.52; 95% CI, 1.13–5.37), drinking (OR, 3.28; 95% CI, 1.26–7.85), and a body mass index of 24.0 to 27.9 kg/m2 (OR, 0.74; 95% CI, 0.59–0.91).
Conclusions
This study revealed a very high prevalence of sarcopenia among elderly individuals with hypertension (20.2%). Sarcopenia may be associated with advanced age, drinking, diabetes, the body mass index, and osteoporosis. The longitudinal relationship between clinic visits and sarcopenia should be further evaluated.
Background
Sarcopenia, a newly recognized geriatric syndrome, is the age-related loss of muscle mass and muscle function. 1 In 2014, the Asian Working Group on Sarcopenia (AWGS) specified cut-off points for the diagnostic variables in Asians. 2 Based on these criteria, the prevalence of sarcopenia was estimated to be 11.3% in women and 9.7% in men among community-dwelling Chinese adults aged ≥60 years. 3 Previous studies have confirmed the association between sarcopenia and adverse health outcomes, such as falls, disability, hospital admission, long-term care placement, poorer quality of life, and mortality. 2 These findings denote the importance of sarcopenia in the health care of older people.
Hypertension is a major public health problem in developing countries. According to the latest data, the prevalence of hypertension in China has reached 23.2%. 4 Hypertension is inextricably linked to insulin resistance, which exacerbates the loss of muscle strength. 5 Insulin resistance contributes to hypertension through several mechanisms, among which are enhancement of tissue angiotensin II and aldosterone activities, increased sympathetic nervous system activity, oxidative stress, 6 and a new phenomenon known as “endothelial insulin resistance.” The suppression of reactive oxygen species-dependent pathways in the endothelium has been shown to restore insulin delivery to peripheral organs by preserving nitric oxide availability. 7 According to this view, insulin resistance is the basis of hypertension. 8 Moreover, there is increasing evidence of an association between insulin resistance and sarcopenia. Intracellular insulin signaling cascades activate the mTOR pathway and inhibit autophagy, including lysosomal degradation of proteins and organelles in muscle. Insulin resistance may interfere with this signaling mechanism and accelerate muscle loss. These abnormalities exacerbate the loss of muscle strength. 5
Although the above studies have confirmed a link between hypertension and sarcopenia through the intermediary of insulin resistance, differences among populations have effects on the prevalence of sarcopenia. The epidemiology of sarcopenia in Chinese elderly individuals with hypertension remains to be studied. Thus, the present study was performed to estimate the prevalence of and factors associated with sarcopenia among Chinese elderly individuals with hypertension.
Materials and methods
Patients
The study sample comprised consecutive patients aged ≥60 years with hypertension who visited the Tianjin Port Hospital in China from January 2019 to December 31, 2020. All 165 patients were invited to participate in a comprehensive geriatric assessment. We excluded those with known risks that hindered or compounded sarcopenia assessment, such as severe hip or knee osteoarthritis, physical disabilities that affected handgrip strength and/or walking, and use of electronic implants such as pacemakers. We also excluded patients unable to stand for measurement of body composition, weight, and height and with any form of other disability that rendered them incapable of providing informed written consent, such as cognitive impairment. The final study population comprised 158 patients after excluding 7 patients. The participants were fully informed of the nature of the research and provided written informed consent to participate. This research was approved by the Ethics Committee at Tianjin Medical University (register number 91075317, approval date December 2018). The reporting of this study conforms to the STROBE guidelines. 9
Clinical evaluation
Sociodemographic variables were assessed, including sex, age, marital status, living status, fall history, and educational level. Information on the smoking status (“not currently smoking” and “currently smoking >10 cigarettes a week”) and drinking status (“not currently drinking” and “currently drinking >3 times a week, and each time >6 units”) was obtained from the questionnaire. The information regarding the history of physical illness was evaluated on the basis of the participants’ response (yes or no) to questions, the physician’s diagnosis or a diagnosis that had been made in the past, and whether the corresponding treatment was or was not being applied at present. Physical activity was assessed with the short form of the International Physical Activity Questionnaire (IPAQ). Responses were converted to metabolic equivalent task (MET) minutes per week according to the IPAQ scoring protocol as follows: total minutes during the last 7 days spent on vigorous activity, moderate-intensity activity, and walking were multiplied by 8.0, 4.0, and 3.3, respectively, to create a MET score for each activity level. The MET scores across the three subcomponents were summed to indicate the overall physical activity. 10 Anthropometric assessments were performed to measure the patients’ weight, height, body mass index (BMI), waist circumference, and hip circumference. Waist circumference was measured at the approximate midpoint between the lower margin of the last palpable rib and the top of the iliac crest, whereas hip circumference was measured around the widest portion of the pelvis. 11 The bone mineral density was measured at the calcaneus by quantitative ultrasound (OsteoPro UBD2002A; B.M.Tech Worldwide Co. Ltd., Seoul, Korea) using T-scores based on the WHO criteria. These were obtained from automated equipment. A T score of ≤2.5 was defined as osteoporosis. 12
Assessment of sarcopenia
According to the diagnostic criteria recommended by the AWGS, 13 low muscle mass was defined as an appendicular skeletal muscle mass (ASM) index of <7.0 kg/m2 for men and <5.7 kg/m2 for women. Muscle mass was measured using direct segmental multifrequency bioelectrical impedance analysis (InBody S10; Biospace, Seoul, Korea). ASM was calculated as the sum of the skeletal muscle in the arms and legs.
Low muscle strength was defined as handgrip strength of <28 kg for men and <18 kg for women. 13 Muscle strength was assessed by the grip strength, which was measured using a dynamometer (GRIP-D; Takei Scientific Instruments Co., Ltd., Niigata, Japan). The patients were asked to exert maximum effort during three measurements in both hands, and the result from the strongest hand was used for analysis in accordance with the protocol of the American Society of Hand Therapists. 14
Low physical performance was defined as a gait speed of <0.8 m/s. 13 Usual walking speed (m/s) on a 6-m course was used as an objective measure of physical performance.
Hypertension definition
Hypertension was defined by self-reporting of a physician diagnosis or the use of antihypertensive medication. In undiagnosed participants, the clinician diagnosed hypertension as a consistent blood pressure of >130 and/or >80 mmHg. 15
Statistical analysis
For numerical variables, data are presented as mean, standard deviation, and median, and classification variables are reported as percentage. The Mann–Whitney U-test was used to analyze the participants’ characteristics because the independent variables were not normally distributed. We used Pearson’s chi-square test and Fisher’s exact test to investigate the association between categorical variables. Logistic regression analysis was used to analyze the factors associated with sarcopenia. Significant variables via a backward selection approach were entered into the multivariable logistic regression to determine the factors associated with sarcopenia. A difference was defined as statistically significant at p < 0.05. All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 19.0 (IBM Corp., Armonk, NY, USA).
Results
A total of 165 patients participated in the study, and 158 were included in the analysis according to the AWGS (Figure 1) after application of the exclusion criteria. The mean age of the study population was 68.8 ± 6.5 years (age range, 60–93 years). The overall prevalence of sarcopenia among elderly individuals with hypertension was 20.2%. Among these individuals, 10 (6.3%) had slow gait speed, 22 (13.9%) had low muscle strength (Figure 1), and 11 (6.9%) had both slow gait speed and low muscle strength.

Sarcopenia status of study participants according to the Asian Working Group on Sarcopenia (AWGS).
The demographic characteristics of the study population are summarized in Table 1. Patients with sarcopenia were older than those without sarcopenia. Compared with patients without sarcopenia, patients with concurrent hypertension and sarcopenia had a higher incidence of cancer, osteoporosis, and fall history and a higher rate of widowhood (p < 0.05); they also had a significantly lower ASM, handgrip strength, walking speed, and IPAQ score (p < 0.05).
Characteristics of the study population.

Data are presented as n (%) or mean ± standard deviation.
ASM, appendicular skeletal muscle mass; BMI, body mass index; IPAQ, International Physical Activity Questionnaire; MET/week, metabolic equivalent task minutes per week.
The factors associated with sarcopenia are summarized in Table 2. Univariate logistic regression analyses showed that the following factors were associated with a greater risk of sarcopenia: age of ≥70 years (odds ratio (OR), 3.14; 95% confidence interval (CI), 1.34–6.69; p < 0.001), a widowed status (OR, 3.70; 95% CI, 1.05–13.03; p = 0.041), a drinking habit (OR, 4.29; 95% CI, 1.90–9.66; p < 0.001), diabetes (OR, 5.65; 95% CI, 2.38–13.39; p < 0.001), osteoporosis (OR, 3.06; 95% CI, 1.32–7.08; p = 0.009), and hyperlipidemia (OR, 2.19; 95% CI, 1.01–4.95; p = 0.040). Sarcopenia was inversely associated with a BMI of 24.0 to 27.9 kg/m2 (OR, 0.69; 95% CI, 0.55–0.87; p = 0.002). However, multivariable logistic regression analyses (Table 3) revealed that the OR and 95% CI in the adjusted model for the factors significantly associated with sarcopenia were 3.01 (1.17–5.39) for age of ≥70 years, 4.45 (1.32–11.16) for diabetes, 2.52 (1.13–5.37) for osteoporosis, 3.28 (1.26–7.85) for drinking, and 0.74 (0.59–0.91) for a BMI of 24.0 to 27.9 kg/m2.
Factors associated with sarcopenia.
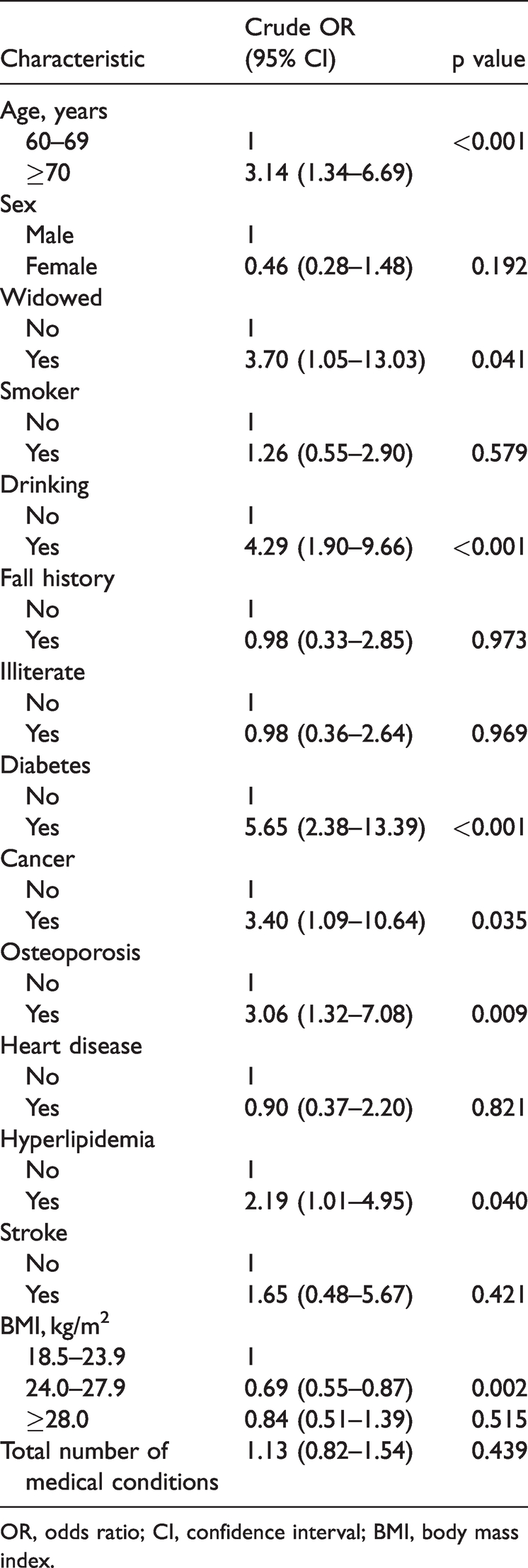
OR, odds ratio; CI, confidence interval; BMI, body mass index.
Factors associated with sarcopenia using backward stepwise logistic regression.

OR, odds ratio; CI, confidence interval; BMI, body mass index.
Discussion
In this study, we found a very high prevalence of sarcopenia among elderly individuals with hypertension (20.2%). The detailed epidemiology of sarcopenia in elderly individuals with hypertension remains to be studied. A recent systematic review estimated an overall prevalence of sarcopenia of 10% in adults aged ≥60 years. 16 Current research confirms that sarcopenia is associated with hypertension. 17,18 The possible mechanism is that hypertension can lead to the production of related catabolic cytokines. 19 Chronic inflammation and its associated catabolic factors remain the most widely accepted mechanism of sarcopenia. 20,21 This might partly explain why the prevalence of sarcopenia was higher in this study. Another reason may be the COVID-19 pandemic, which has led the government to take measures such as isolation and social alienation, extending people’s time spent at home. This has resulted in reductions in physical activity and changes in dietary intake that have the potential to accelerate sarcopenia. 22 Furthermore, quarantine and social isolation are known to result in increased levels of stress and anxiety, the consequences of which may be increased markers of atrophy and elevated loss of muscle mass. 23
The results of this study showed that age was associated with a higher risk of sarcopenia, which is compatible with the findings from previous studies. 24 –26 Muscle mass and function decline with age 27 ; this is considered a normal phenomenon, but it can rapidly progress in the setting of chronic conditions such as hypertension. 28 The possible reason is that high-energy skeletal muscle tissue relies upon mitochondria for proper energy production and contractile function, and the quantity of mitochondria decreases with advancing age. 29
This study also showed an association between diabetes and sarcopenia. The mechanism leading to decreased muscle mass and function in patients with diabetes remains unclear. Insulin resistance causes loss of muscle tissue, which in turn decreases insulin sensitivity if the glucose supply remains constant; this may lead to a vicious circle and the onset of sarcopenia. 30 Additionally, as mentioned in the Introduction, hypertension leads to insulin resistance, which might explain why the combination of hypertension and sarcopenia is associated with diabetes.
Our findings are consistent with the results of previous studies showing a strong link between sarcopenia and osteoporosis. 31,32 Biochemically, osteocalcin, which is secreted solely by osteoblasts, stimulates muscle tissue, resulting in increased myoblast proliferation. 33 However, myostatin deficiency may lead to increased bone mass. 34 In our study population, hypertension was also a risk factor for both sarcopenia and osteoporosis, 21,35 and these three diseases may form a vicious circle.
The results of this study suggest that sarcopenia is negatively correlated with the BMI. This finding is supported by the results of previous studies showing that an increased BMI is a protective factor against sarcopenia in older Chinese individuals. 36,37 Individuals with higher fat mass may have higher protein intake, which is a protective factor against sarcopenia. Sarcopenia was also significantly associated with daily drinking in our study. This is consistent with previous research results 37,38 showing that alcohol might damage muscle by disrupting muscle protein synthesis. 38
Some limitations of this study should be mentioned. First, because of the cross-sectional design of this study, the causal and chronological relationship of the associated factors with sarcopenia could not be established. Second, the potential recall bias and the data reliability and accuracy could not be objectively ascertained in the self-reported variables. Third, we did not measure other variables of concern in sarcopenia (e.g., fragility fractures and pharmacological therapy). At present, this study represents preliminary descriptive research for identification of factors that may be associated with sarcopenia. We will add further relevant data in our subsequent study. Fourth, the data from our single study site are not reflective of the nationwide prevalence of sarcopenia in China.
Conclusion
We examined the prevalence of sarcopenia and identified several associated factors in elderly individuals with hypertension in Tianjin, China. Sarcopenia may be associated with advanced age, drinking, diabetes, the BMI, and osteoporosis. The longitudinal relationship between clinic visits and sarcopenia should be further evaluated.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221110490 - Supplemental material for Prevalence of and factors associated with sarcopenia among elderly individuals with hypertension
Supplemental material, sj-pdf-1-imr-10.1177_03000605221110490 for Prevalence of and factors associated with sarcopenia among elderly individuals with hypertension by Enwang Xing and Chunxiao Wan in Journal of International Medical Research
Footnotes
Acknowledgement
The authors thank Jiazhong Wang from the Department of Rehabilitation Medicine, Tianjin Medical University, for providing organization.
Author contributions
Chunxiao Wan designed the study. Enwang Xing performed the data collection and collated and completed the article.
Declaration of conflicting interests
No potential conflict of interest was reported by the authors.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Tianjin Key Medical Discipline (Specialty) Construction Project (2019-12-001).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
