Abstract
Hemophilia A and B are rare, X-linked bleeding disorders characterized by deficiencies in coagulation factor VIII (FVIII) and factor IX (FIX), respectively. Numerous advances have helped to reduce disease burden. However, hemophilia B is not as well studied as hemophilia A, likely reflecting its lower prevalence. Clinical management of hemophilia B has often relied on inference and extrapolation from hemophilia A. Despite being part of the same tenase complex, as enzyme (FIX) and cofactor (FVIII) when activated, with the main task being to activate factor X in the intrinsic pathway, the FIX and FVIII proteins display several molecular differences. These have the potential to impact the clinical phenotype of hemophilia, affect monitoring, and influence treatment options. Consequently, hemophilia B presents several outstanding challenges, requiring a greater degree of understanding and/or attention across a range of areas. Some of these challenges relate to the FIX molecule, with more knowledge needed in relation to: the biological/clinical impact of underlying genetic changes; hemostatic implications of the extravascular distribution of FIX; and FIX clearance. Other challenges relate to clinical management: determining the best ways to monitor the true biological activity of FIX; clarifying the relationship between FIX plasma levels and clinical outcomes when treating patients; inhibitors; affected girls and women; and appreciating the value of novel treatment approaches, while considering possible breakthrough bleeds, thrombosis, and monitoring. In addition, concerted effort is required to address global disparities, which can particularly affect hemophilia B. Identifying such challenges may help to facilitate research that will further existing knowledge, with better understanding being crucial for achieving health equity between hemophilia A and B.
Introduction
Hemophilia management continues to advance, with evolving treatments and therapeutic targets providing benefits for managing deficiencies of both factor VIII (FVIII; hemophilia A) and factor IX (FIX; hemophilia B). 1 However, with a smaller cohort of affected individuals than for hemophilia A, 2 and a knowledge base often extrapolated from this condition, 3 patients with hemophilia B have specific unmet needs, 4 as recently reviewed elsewhere. 5 Deficiencies in FVIII and FIX are both classically referred to as “hemophilia,” but in spite of many phenotypic similarities, they exhibit multiple genetic, biochemical, and pathophysiological differences that impact disease management.
This manuscript highlights outstanding challenges for improving care in hemophilia B, considered across nine areas in which a greater degree of understanding and/or attention is required, and defined based on a consensus discussion among the authors. Recognition of these challenges may help to facilitate action to improve outcomes for those with hemophilia B via a range of activities, including molecular research, clinical considerations, and action at the societal level. This will require input from scientists, clinicians, and global stakeholders, reflecting the multidisciplinary nature of truly comprehensive disease management. It is a very broad remit. Furthermore, these are not the only challenges in hemophilia B. However, identifying points such as these is a first step to enabling solutions, with the ultimate goal of achieving health equity. 6
Outstanding challenges
Nine areas with outstanding challenges for hemophilia B research are summarized in Figure 1 and considered in detail below.

Nine areas with outstanding challenges for optimal understanding and clinical management of patients with hemophilia B.
1. Genetics of FIX
Hemophilia B is a consequence of pathogenic variants in the F9 gene, which is located on the long arm of the X chromosome at Xq27.1, spans 33.5 kb of DNA, and has eight exons. 7 Reflecting the smaller size of the F9 gene than F8 (which spans around 180 kb of DNA and containing 26 exons 8 ), and its lack of predisposition to intronic rearrangement (unlike F8, 8 as considered below), hemophilia B comprises between 15% and 20% of all hemophilia cases. 2 The prevalence of hemophilia A and B has been reported as 17.1 and 3.8 cases per 100,000 males, respectively. 9 Females can also be affected, as considered in more detail below (see section “Girls and women with hemophilia B”).
In hemophilia, the pathologic gene variant influences the clinical manifestation of disease. 8 Larger gene defects tend to result in a more severe clinical phenotype. 10 Detailed comparison of the FVIII and FIX molecules, and in-depth consideration of the genetic variants responsible for hemophilia A and B, respectively, is beyond the scope of the current manuscript and available elsewhere.8,11 Nevertheless, it is appropriate to note that almost 1700 genetic variants in the F9 gene have been identified, 12 and information in numerous databases provides details of both F9 and F8 variants.13–16 A summary comparison is presented in Table 1. For some variants, the genotype–phenotype correlation is stronger in hemophilia B than in hemophilia A. Many of those with hemophilia B harbor single-point variants in the F9 gene.11,12 Severe forms of the disease (as defined by the factor levels < 1%) occur at a lower frequency in hemophilia B compared with hemophilia A. 7 This reflects common intronic inversions that arise due to sequence homology in the F8 gene, 8 a situation that is not paralleled in hemophilia B: as F8 is much larger and more complex than F9, 8 and affected by repeats, it is predisposed to rearrangement. 17 The nature of the underlying variant may also influence the susceptibility toward the development of neutralizing inhibitors11,18 (see section “FIX inhibitors”). However, bleeding phenotype is not determined solely by the genetic variant; patients with hemophilia B who have the same variant can differ in their bleeding tendency.19–21 It has been speculated that missense variants may result in milder bleeding in hemophilia B compared with hemophilia A. 10 In addition, a higher proportion of missense variants in hemophilia B versus A, whereby FIX/FVIII molecules are produced but remain dysfunctional, may account for higher levels of cross-reactive material (CRM) positivity, 22 in which FIX/FVIII antigen is apparent (reported for < 5% in patients with severe hemophilia A compared with > 50% in severe hemophilia B 23 ).
The percentages show the number of different variants identified for various gene changes as a proportion of the total number of different variants identified. They do not show percentages of patients with each type of variant.
50 base pair cut-off for large and small structural changes.
The biological relevance/clinical impact of the different spectrum of genetic changes seen in hemophilia B compared with those observed in hemophilia A remains to be fully determined, including with respect to CRM status, the risk of complications, and impact on new genetically based therapeutic modalities.
2. Extravascular distribution of FIX
The plasma concentrations of FIX and FVIII are 90 nM and 0.4 nM, respectively. 24 FVIII resides intravascularly, circulating with its chaperone von Willebrand factor (VWF). 25 In contrast, the relatively small size of the FIX molecule allows a substantial proportion of FIX to distribute to the extravascular space, at least in part mediated via binding to type IV collagen in the subendothelial basement membrane 26 (Figure 2). This may increase the apparent volume of distribution of FIX, with possible pharmacodynamic implications for plasma half-life. Initially, a half-life of 18–24 h was ascribed to wild-type FIX, but more recent studies have shown that extravascular FIX may redistribute into the plasma compartment resulting in a longer secondary half-life than previously accepted. 27 With ongoing research in this area, recent data have shown that the protease domain heparin-binding exosite can also regulate extravascular binding, particularly in the liver. 28
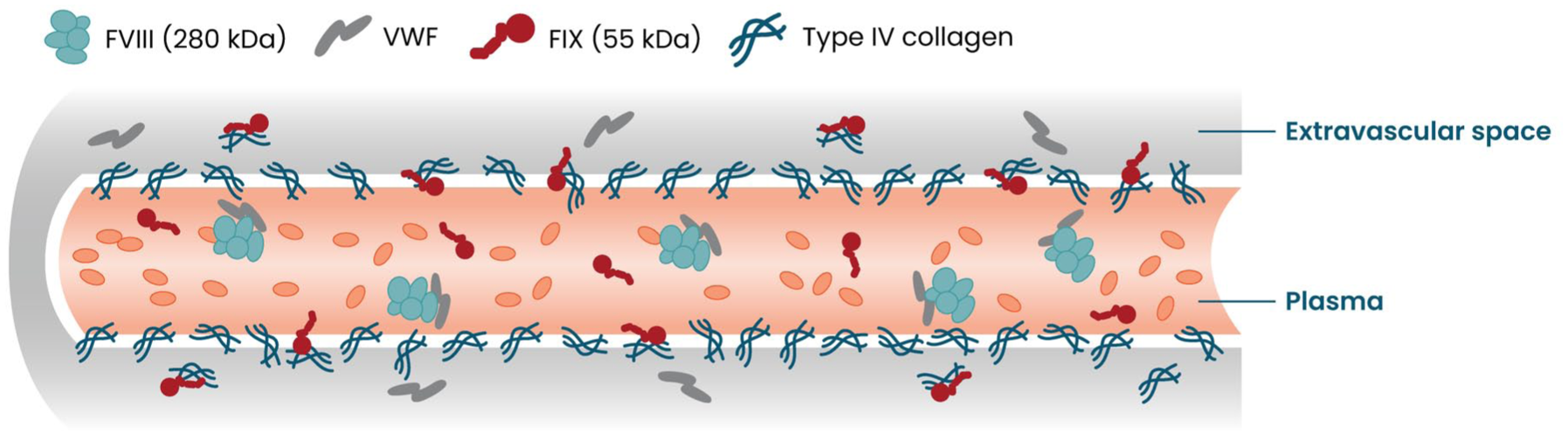
Distribution of factor VIII (FVIII) and factor IX (FIX).
Following the advent of recombinant FIX (rFIX), it rapidly became apparent that initial recovery following infusion was 25%–30% lower than for plasma-derived FIX (pdFIX), whereas the half-life was unaffected. 29 Reduced sulfation of Tyr155 and phosphorylation of Ser158 in rFIX compared with pdFIX, may contribute to the reduced recovery.29,30 This is surprising, as current data suggest that the Gla-domain mediates binding to collagen IV and thus is responsible for FIX extravasation. 31 To understand the regulation of FIX biodistribution, it is therefore relevant to further explore the role of the activation peptide in general, and residues Tyr155/Ser158 in particular. Does the activation peptide play a separate role in the extravasation of FIX by binding to another, unidentified matrix protein, or is linkage with the Gla-domain involved? Analysis of histological tissue sections stained for FIX suggests that there might be two distinct extravascular pools: one located close to the lumen of the vessel and one in the vessel wall. 32 Are both pools available for redistribution into the circulation, or is the pool in the vessel wall no longer available, serving another yet unidentified function? Data from murine models have provided valuable insight relating to extravascular FIX, 33 and continue to do so. 34 For example, results from FIX-treated hemophilia B mice have shown FIX plasma levels to decrease to < 1% within 3 days of infusion, but hemostatic protection is still apparent 7 days after treatment. 33 Such data suggest that beyond plasma levels, the hemostatic potential of FIX in the extravascular space may have implications for therapeutic efficacy. However, more information is needed. Furthermore, little is known about collagen binding of different genetic variants of FIX in CRM-positive patients, whose altered FIX might still bind to collagen and compete with exogenous FIX. To better understand the role of extravascular FIX, these issues are relevant and need further research.
In addition, the extravascular distribution of various extended half-life (EHL) FIX products appears to differ35–37 and remains to be fully understood. What is the precise nature of the extravascular distribution and binding of different products? Increased knowledge may help to optimize treatment guidance.
3. Clearance of FIX
Little is known about clearance of FIX. Whereas studies over the last two decades have provided insight into the molecular aspects of FVIII clearance, identifying cell types and clearance receptors mediating FVIII catabolism, 38 no such studies have been performed regarding FIX or EHL FIX products. While earlier studies suggested possible involvement of human neutrophil elastase and plasmin in regulating FIX activity,39,40 no detailed follow-up of these findings/definitive conclusions have been reported since these were published over 20 years ago. Apart from the possibility that de-sialylated FIX may be catabolized via the Ashwell receptor, 41 no other clearance receptors have been linked to the clearance of FIX. Elucidating the molecular basis of FIX clearance (i.e., identifying specific clearance receptors for FIX and residues within FIX that are critical for binding to these) could be fundamental for designing new FIX variants with reduced clearance and could be applied to replacement and/or gene therapy. In addition, increased knowledge could contribute to a better understanding of the pathogenesis of hemophilia B; specific variants might be associated with increased binding to clearance receptors, similar to findings for von Willebrand disease. 42
4. FIX monitoring
As comprehensively reviewed elsewhere,43,44 FIX is a proenzyme, activated via two different pathways, one involving the tissue factor/activated factor VII (FVII) complex and another involving activated factor XI. Following its activation, activated FIX (FIXa) joins the tenase complex, in which it is responsible for the proteolytic activation of factor X (FX). Measuring FIX activity is, therefore, key to classifying the severity of hemophilia B and may help to guide treatment, although, as mentioned elsewhere, there may be confounding influences here, including the extravascular distribution of FIX.
There are several ways to measure FIX activity. One-stage clotting assays have traditionally been used for the diagnosis of hemophilia B, although chromogenic assays have recently become more broadly available. 45 When used for diagnosis, the two methods generally provide comparable results, and assay discrepancy in hemophilia B is infrequent. 46 Nevertheless, physicians should be aware of possible assay discrepancies in relation to hemophilia B, particularly in relation to non-severe disease. 47 Variants in the FIX serine protease domain and at the cleavage site of the activation peptide may affect assay performance.48,49 In addition, there are intrinsic differences between rFIX and pdFIX, as well as between standard half-life (SHL) and EHL FIX products.47,50,51 Recent data suggest that using both types of assays, together with genotyping, may help to optimize the clinical management of patients with hemophilia B. 48
The potentially beneficial hemostatic effects of prophylaxis contributing to the extravascular reservoir of FIX 34 may further complicate the situation. This reservoir remains undetected when measuring plasma levels of FIX, independent of whether traditional FIX activity assays (one-stage or chromogenic activity assays) or global assays (thrombin generation assays or rotational thromboelastometry) are being used. The extent to which this extravascular reservoir contributes to the hemostatic protection by FIX is currently unknown, and no assay is routinely available to measure either the affinity of FIX for collagen IV or its level of binding.
Assay discrepancies, inherent to FIX-Padua involved in this approach, 52 are apparent in both one-stage clotting assays (associated with the activating agent/phospholipids) and chromogenic assays (where FX influences relative potency). 53 Best practices for measuring FIX after gene therapy remain to be established.
5. FIX activity required for (optimal) bleed protection
For hemophilia A, there is growing evidence regarding the FVIII plasma levels required for specific situations, for example, prophylactic prevention of spontaneous joint bleeds or maintaining hemostasis during physical activity.2,54 For hemophilia B, the association between FIX plasma levels and clinical outcomes is less clearly elucidated, with extravascular FIX adding complexity. Although extravascular FIX may contribute to the hemostatic response, 34 the extent of, and conditions required for this are unclear. More information on the relationship between plasma levels and outcomes for both SHL and EHL FIX products will help to optimize treatment. Moreover, it is not clear how the plasma trough levels required for effective prevention of bleeding compare for different FIX products, or the extent to which extravascular distribution may influence product efficacy. Knowledge of inter-individual variability in concentrate recovery and clearance kinetics is limited.
It should also be appreciated that although patients with hemophilia B have deficient FIX, levels of FVIII and VWF still increase in response to trauma, surgery, and stress (both physical and psychological), while increases are also associated with chronic conditions (malignancy, inflammatory diseases, and chronic infection), as well as pregnancy. 55 The clinical implication of this is unknown. In addition, more information is required in relation to thromboprophylaxis, since this may become increasingly important in the aging hemophilia population.
6. FIX inhibitors
Most of the “knowledge” about inhibitors in hemophilia B has been extrapolated from hemophilia A. A major problem is that hemophilia B is less common, the overall lifetime risk of inhibitors occurring in these patients is only around 1%–5%, and inhibitors develop predominantly in those with severe hemophilia B (up to 10%–15%).2,18,56 In contrast to hemophilia A, in hemophilia B, inhibitors very rarely occur in patients with non-severe disease. 57
Factors affecting risk of inhibitor development in hemophilia B remain poorly understood. Considering the higher inhibitor rate in severe hemophilia A (cumulative incidence of around one-third in previously untreated patients 2 ), it is notable that a large proportion of patients with severe hemophilia B carry single-point variants and are CRM-positive, which may reduce inhibitor risk. 23 In addition, it has previously been speculated that the similarity of FIX to other vitamin K-dependent factors (factor II, FVII, and FX) may also impact its immunogenicity, 18 although structural homology is not a guarantee for a similar immunogenic profile, based on the similarity of FVIII with factor V and ceruloplasmin. 58
Improved understanding of FIX inhibitor development is important for future gene therapy/genetically targeted approaches for subjects with a previous history of inhibitors and/or unknown tolerance (children).
As highlighted in a previously published comprehensive review of FIX inhibitors, their uncommon nature has hampered the generation of evidence on which to base management of affected patients. 59 Inhibitors in hemophilia B are particularly challenging and may be associated with a propensity to develop severe anaphylactic reactions. 23 Understanding why such reactions occur will help predict their occurrence. Contributing influences might include a requirement for large amounts of replacement therapy, immune tolerance induction (ITI), and rapid extravascular distribution of FIX. 60 While the underlying genetic pathology in hemophilia B may influence the development of inhibitors, in that these are more likely to occur in individuals affected by a variant resulting in an absence of endogenous FIX, 18 the possibility of additional genetic influences, such as co-deletion of genes, remain to be fully explored. The risk of this complication prompts administration of FIX for the first 20 exposures in clinics equipped to manage any sequelae. 2
Patients with hemophilia B and inhibitors can also develop nephrotic syndrome. 61 The causative mechanisms of this are unknown. However, it is more common in those with allergic reactions and may develop during ITI. 2 A better understanding of how and why nephrotic syndrome may develop is of major importance, since many patients with hemophilia B developing inhibitors will not be offered ITI due to fear of this complication. In addition, and for unknown reasons, ITI seems generally less successful in patients with hemophilia B than hemophilia A (success rates of approximately 30% vs 60%–70% 10 ), 23 more often requiring combined use of FIX and immunosuppression.
7. Girls and women with hemophilia B
Although hemophilia is an X-linked disorder, females can be affected.62,63 The “carrier” terminology understates bleeding symptoms, and updated nomenclature has been proposed to better reflect effects on females, 64 including symptomatic and asymptomatic hemophilia in individuals with FVIII/FIX plasma levels ⩾40 IU/dL. Low levels of FIX in females can be homozygous, compound heterozygous, hemizygous, or heterozygous with regard to hemophilia alleles. 62 Lyonization, random skewed X chromosome inactivation, has an effect on how hemophilia manifests. 65
Although the carriership prevalence remains to be definitively determined, around three to five carriers have been estimated for every male with hemophilia.66–68 World Federation of Hemophilia (WFH) data show the proportions of females with hemophilia A and B to be 4% and 6% of the total hemophilia populations, respectively. 69 There may be proportionally more females with hemophilia B. However, the distribution of severity is similar among females with hemophilia A and B (based on data from high-income countries). 69 Overall, the data show fewer diagnosed patients than expected. 69 This lack of awareness may be particularly acute for girls and women with hemophilia B, with a paucity of demographic data failing to accurately reflect the prevalence and severity of FIX deficiency in this population, and having implications for health equity.
Following on from this, there are also limited data on the hemostatic efficacy and safety of the different treatment options for girls and women with hemophilia B-specific bleeding symptoms, including in relation to pregnancy and delivery, and, importantly, with regard to vitamin K deficiency.
8. New treatment options
Detailed consideration of the expanding range of treatment options is beyond the scope of this review but considered in detail elsewhere, including with regard to how these can help to facilitate personalized therapy.70,71
Unlike FVIII, which is a procofactor, FIX is a zymogen, and this may limit approaches for the development of new therapies. There are no bispecific antibodies that mimic the activity of FIXa (akin to emicizumab that mimics activated FVIII [FVIIIa] cofactor activity72,73). However, recent data suggest that emicizumab could be used in some cases of hemophilia B, in which variants in the FIX protein impair the interaction of FIXa with FVIIIa. 74
Various other novel treatment approaches, including targeting tissue factor pathway inhibitor, antithrombin, or activated protein C, 75 have been investigated. As rebalancing therapies, these aim to alleviate bleeding by promoting a more pro-coagulant state in hemophilia. 76 While such agents may be useful for prophylaxis, breakthrough bleeds will require alternative treatment with factor therapy, and thrombosis may be a concern. 75 Therapeutic monitoring will pose new challenges. 75 Nevertheless, the potential role of rebalancing agents should be better determined with regard to the treatment of patients with hemophilia B, and an alternative in patients with inhibitors.
While the majority of variants in the F9 gene impair FIX activity, the Padua, Shanghai, and Shanghai II single-point variants are associated with gain of function and thrombophilia.77–79 Gene therapy for hemophilia B involving FIX Padua is now available in some countries.80–82
Initial approaches to deliver gene therapy have used adeno-associated virus-based liver-directed technology.83,84 Different methodologies, for example, use of lentiviral vectors or clustered regularly interspaced short palindromic repeat (CRISPR) technology, are being studied.83,85,86 Gene editing could represent an attractive alternative to currently available gene therapy, and the role of complementary approaches should be evaluated.
Table 2 summarizes the various approaches to treatment, comparing applicability in hemophilia B with hemophilia A.
Approaches to hemophilia treatment, comparing applicability in hemophilia B with hemophilia A.

AAV, adeno-associated virus; Cas9, clustered regularly interspaced short palindromic repeat associated protein; CRISPR, clustered regularly interspaced short palindromic repeat; EHL, extended half-life; EMA, European Medicines Agency; Fc, fragment crystallizable; FVIII, factor VIII; FIX, factor IX; FIXa, activated factor IX; FX, factor X; FXa, activated factor X; gRNA, guide ribonucleic acid; HSC, hematopoietic stem cell; IgG1, immunoglobulin 1; LNP, lipid nanoparticle; mRNA, messenger ribonucleic acid; PEG, polyethylene glycol; VWF, von Willebrand factor.
9. Global disparities and inequities
Global challenges in hemophilia management may particularly affect those with hemophilia B. As previously mentioned, much of the knowledge about hemophilia B has been extrapolated from hemophilia A, which may in itself be regarded as a source of inequity. Inequities are likely to be exacerbated in resource-constrained countries, negatively impacting rates of diagnosis and access to treatment for those with hemophilia B.
The WFH is working to address global disparities and inequities, with the “Treatment for All” vision aiming to provide care for everyone affected by hemophilia, and the “Theory of Change” initiative aimed at facilitating global stakeholder collaboration. 107 The WFH Humanitarian Aid Program has promoted partnerships between commercial and not-for-profit organizations helping to advance health equity. 108
Nevertheless, it has been estimated that despite therapeutic advances in hemophilia, the majority of the resulting products are likely to remain out of reach for more than 70% of the global population of hemophilia patients. 109 As a historical treatment for hemophilia A, 110 with limitations including infection risk and relatively low FVIII content, 111 cryoprecipitate remains on the World Health Organization Model List of Essential Medicines. 112 However, as an example of attempting to promote global access to new treatments, partnerships and collaboration are under consideration with regard to gene therapy, and this has been trialed in patients living in resource-constrained regions.113,114 Globally, concerted efforts and increased focus are required to improve the situation for the majority of those with hemophilia B, both male and female, including for individuals in whom the condition currently remains undiagnosed.
Discussion and conclusion
Despite being part of the same tenase complex, as enzyme (FIX) and cofactor (FVIII) when activated, with the main task being to activate FX in the intrinsic pathway, the FIX and FVIII proteins display several molecular differences. These have the potential to impact the clinical phenotype of hemophilia, affect monitoring, and influence treatment options. As a consequence of its lower incidence and prevalence, most of the assumed knowledge regarding hemophilia B has been transferred from data on hemophilia A.
To some extent, hemophilia B presents a paradox. Although occurring less frequently than hemophilia A, the proportion of the hemophilia B population who are female may be higher than for hemophilia A. Hemophilia B is the less “visible” of the two conditions, with numerous features that confer differences compared with hemophilia A. Not only might the smaller size and lack of predisposition to intronic rearrangement, of the F9 gene than F8 result in a lower frequency of disease, the smaller size of the resulting protein may influence its distribution, with the extravascular presence of FIX having possible therapeutic implications. Lacking a chaperone, the stability of the FIX molecule initially lent itself more easily to EHL factor therapy. However, patients who develop inhibitors, very rarely those with non-severe disease, may be affected by anaphylaxis, and are more challenging to tolerize than their hemophilia A counterparts. More knowledge is required.
Better basic understanding of relevant differences between hemophilia A and B, addressing areas with outstanding challenges as described in this article, for which further research may be required, will be crucial for future health equity in patients with these conditions. Further knowledge of differences in the profiles of the underlying genetic variants, as well as the size and nature of the mature protein, will be of both immunological and functional importance. In addition, FVIII, together with its VWF chaperone protein, is an acute phase reactant, whereas FIX is not, which may potentially impact clinical manifestations of hemophilia in both males and, importantly, also in females with non-severe forms of the disease.
Altogether, as a consequence of being regarded as the “minor heterozygous twin,” more research focused on hemophilia B will be required to achieve future health equity, as well as to fully explore and use the benefits the specific characteristics FIX may offer with regard to the development of new treatments.
Footnotes
Acknowledgements
Medical writing and editorial support, funded by Sobi, were provided by Andy Lockley, PhD, Bioscript Group, Macclesfield, UK. Sobi and Sanofi received the manuscript for courtesy review; however, they did not influence the content. Editorial assistance and support with the submission of this article was provided by Liz Beatty of Bioscript Group and supported by Sobi. The authors have authorized this support and approved the inclusion of all conflicting interests and funding disclosures.