Abstract
Background:
Outcomes for primary central nervous system lymphoma (PCNSL) have been improved with high-dose methotrexate (HD-MTX)/Thiotepa-based chemotherapy followed by autologous stem cell transplantation. In limited-resource settings, HD-MTX/Ifosfamide plus whole brain radiotherapy (WBRT) has become the local standard of care.
Objectives:
This study investigated the real-world effectiveness, neurocognitive functions, and health-related quality of life (HRQoL) of HD-MTX/Ifosfamide in newly diagnosed PCNSL patients.
Design:
A single-center retrospective and prospective study.
Methods:
Newly diagnosed PCNSL patients treated with HD-MTX (±Ifosfamide) between 2011 and 2024 were analyzed for treatment effectiveness using binary logistic regression and a Cox regression model. The age range- and education-matched PCNSL and non-central nervous system (CNS) lymphoma patients in first remission were assessed using standardized cognitive tests and European Organization for Research and Treatment of Cancer (EORTC) QLQ-C30/BN20 questionnaires. The categorical data and continuous data were calculated with the Chi-square test or Fisher’s exact test and analysis of variance, respectively.
Results:
Among 94 PCNSL patients, 56 patients received HD-MTX/Ifosfamide (median age 56 (range 16–69) years; 68% Eastern Cooperative Oncology Group (ECOG) 0–1) and 38 patients received HD-MTX monotherapy (median age 66 (range 51–82) years; 63% ECOG ⩾2). HD-MTX/Ifosfamide demonstrated a significantly longer event-free survival (39 months vs 8 months, p = 0.021) than HD-MTX monotherapy. Poor performance status (ECOG ⩾2) was associated with inferior response (54.8% vs 78.8%, p = 0.013) and overall survival (hazard ratio 2.4 (95% confidence interval 1.57–4.56), p = 0.007). Patients who received WBRT consolidation had a superior 2-year progression-free survival (74.5% vs 35.6%, p < 0.001). Comparing neurocognitive tests in 20 PCNSL and 20 non-CNS lymphoma survivors showed no difference in overall scores, but trends toward lower attention and executive function scores in the PCNSL group. Most (16/20) PCNSL survivors received WBRT. Compared to non-CNS lymphoma survivors, PCNSL survivors reported significantly lower HRQoL, particularly in physical functioning, which might be attributed to residual neurological deficits.
Conclusion:
This study supports HD-MTX/Ifosfamide as an effective, well-tolerated regimen for younger, fit PCNSL patients. WBRT remains a valuable consolidation therapy to prevent recurrence without a pronounced decline in cognitive function. However, PCNSL survivors may experience subtle declines in attention, executive function, and HRQoL.
Keywords
Introduction
Primary central nervous lymphoma (PCNSL) is a rare and aggressive lymphoma subtype solely located in the central nervous system (CNS). 1 Diffuse large B-cell lymphoma (DLBCL) is a major histopathology classified as primary DLBCL of the CNS.2,3 Among immunocompetent populations, the incidence of PCNSL increases with advancing age, with a median age at diagnosis ranging from 56 to 65 years.2,4 Despite the improvement in PCNSL treatment over the past few decades, prognosis remains poor, particularly in patients older than 60 years.4,5 Several evidence-based practice guidelines recommend high-dose methotrexate (HD-MTX) based regimens for first-line induction treatment followed by consolidation therapy with autologous stem cell transplantation (ASCT) or reduced-dose whole brain radiotherapy (WBRT) to fit patients.6–11 The thiotepa-based conditioning regimen is a standard high-dose chemotherapy regimen for ASCT. In addition to treatment guidelines, meta-analyses have demonstrated that HD-MTX-based triplet combination regimens, MTX/thiotepa/cytarabine, MTX/cytarabine/rituximab, MTX/procarbazine/vincristine, and MTX/temozolomide/rituximab, significantly improved response rate compared to HD-MTX monotherapy, especially for young and fit patients.12–14 The role of rituximab for PCNSL treatment remains controversial due to its potential efficacy, depending on different chemotherapy regimens. Recent meta-analyses have shown rituximab to improve complete response (CR) rates and survival.15–18
In Thailand, most patients cannot afford thiotepa, rituximab, and temozolomide as they are expensive and non-reimbursable under the national health coverage. Procarbazine and teniposide are also not available. Therefore, the treatment of PCNSL in Thailand primarily involves HD-MTX–based chemotherapy, with or without WBRT, depending on the protocol of each center.19–21 This impacts Thai PCNSL treatment guidelines for both frontline treatment and thiotepa-based conditioning regimens followed by ASCT for consolidation therapy. Since the 2010 publication of Thiel et al.’s phase III study, our center has introduced the HD-MTX/Ifosfamide regimen for young and fit PCNSL patients and HD-MTX monotherapy for older and fit PCNSL patients, while avoiding WBRT for patients in CR. 22 We have reported experiences with these regimens in a cohort of patients and discovered inferior progression-free survival (PFS) in CR patients without WBRT consolidation. 19 Results from our study underscored the important role of consolidation therapy in CR patients to prevent early relapse, for which treatment options are extremely limited in Thailand. Although several studies showed a comparable efficacy of WBRT and ASCT consolidation, there is a concern of neurocognitive decline associated with WBRT.23–27 Since ASCT is unavailable for most Thai patients, we provided 23.4–30.6 Gy of WBRT for consolidation therapy as an option for CR patients who accepted the risk of WBRT neurotoxicity to lower the risk of relapse.
This study aimed to investigate the effectiveness and acute/delayed toxicities of HD-MTX/Ifosfamide and HD-MTX regimens, some of which included WBRT, in newly diagnosed PCNSL patients. For the delayed toxicity assessment, we comprehensively evaluated neurocognitive functions and health-related quality of life (HRQoL), comparing survivors of PCNSL to non-CNS lymphomas.
Methods
This single-center study held at King Chulalongkorn Memorial Hospital comprised two parts. The first part analyzed a retrospective (2011–2023) and prospective (2023–2024) cohort of newly diagnosed PCNSL patients to investigate treatment outcomes. The second part, which was conducted between 2023 and 2024, is a prospective enrollment of PCNSL and non-CNS lymphoma survivors who were in CR for assessment of the cognitive functions and HRQoL. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 28
For the treatment effectiveness analysis, newly diagnosed PCNSL (DLBCL) patients who were treated with high-dose MTX (3–4 g/m2) with or without ifosfamide between 2011 and 2024 were enrolled. 22 The diagnosis required the presence of CD20-positive large cells in CNS tissue biopsy without evidence of systemic lymphoma. Patients with primary vitreoretinal lymphoma and immunocompromised conditions such as human immunodeficiency virus (HIV)-associated lymphoma or post-transplantation were excluded. The WBRT was performed with 3D conformal radiotherapy, and the boosting tumor used intensity modulated radiation therapy.
For the cognitive functions and HRQoL analyses, both PCNSL and non-CNS lymphoma survivors required evidence of lymphoma in the first CR during the assessment period (year 2023–2024). The PCNSL group was comprised of recruited patients who were treated with an HD-MTX–based regimen between 2016 and 2024, with or without WBRT. The non-CNS lymphoma comparison group enrolled any lymphoma patients without CNS involvement that did not receive CNS therapy/prophylaxis (HD-MTX or intrathecal chemotherapy or brain radiation). Both groups were matched for similar age ranges (<50, 50–59, 60–69, or ⩾70 years old) and education levels (less than a bachelor’s degree or bachelor’s degree and above).
Objectives and definitions
The primary outcome was to identify factors associated with event-free survival (EFS) of PCNSL patients. The secondary outcomes included response rate, overall survival (OS), and PFS, including the factors associated with these outcomes. In addition, the secondary outcomes also focused on post-remission cognitive functions and HRQoL of PCNSL survivors compared to non-CNS lymphoma survivors. Clinical information and treatment outcomes were extracted from the medical records. Prognostic scoring used the International Extranodal Lymphoma Study Group (IELSG) criteria. 29 Tumor criteria of CR, partial response (PR), stable disease (SD), and progressive disease (PD) were determined following International Primary CNS Lymphoma Collaborative Group standards. 30 EFS was defined as the date of diagnosis until the date of event occurrence. The events of EFS were defined as disease relapse/progression, death, or absence of CR at the end of protocol treatment, which also includes early chemotherapy discontinuation due to toxicity before CR achievement. PFS was defined as the date of CR/PR achievement until the date of disease relapse/progression or death from any cause. OS was defined as the time from the diagnostic date until death from any cause.
Neuropsychological function and HRQoL assessments
Neuropsychological function evaluation was conducted by a psychologist (N.R.) at a single time point. The Montreal Cognitive Assessment (MoCA), which has been validated for use with Thai patients, was selected as a cognitive screening test. 31 According to the IELSG-32 phase II trial, 32 WBRT significantly impacts attention and executive functions. Due to the time-consuming nature of a full neuropsychological evaluation, the use of standardized tests for attention and executive functions was employed in this study. 33 The digit span test and symbol search test were selected for assessment of attention and visuospatial attention, respectively. Executive function was evaluated using the Wisconsin card sorting test-short form (WCST-64). Raw test scores were converted to standard scores adjusted by age and education. For HRQoL analysis, the European Organization for Research and Treatment of Cancer (EORTC) QOL Questionnaire-30 (QLQ-C30) and EORTC QLQ-BN20 were selected.
Statistical analysis
The statistical analysis was performed using Statistical Package for the Social Sciences (SPSS) 22.0 (SPSS Inc., Chicago, IL, USA) for investigating the treatment outcome cohort. The prospective cognitive function and HRQoL evaluation cohort was analyzed using Stata SE version 18.5 (StataCorp, TX, ISA). The descriptive data were presented as means and percentages. The categorical data and continuous data were calculated with the Chi-square test or Fisher’s exact test and analysis of variance, respectively. Binary logistic regression was used for treatment response analysis. Survival outcomes were analyzed using Kaplan–Meier curves with the log-rank test. The Cox regression model was applied for univariate and multivariate analysis. Factors with a p < 0.1 from the univariate analysis were included in the multivariate model. A two-sided p < 0.05 was considered significant. The results were displayed using GraphPad Prism software. For sample size determination, since EFS comparison between HD-MTX/Ifosfamide and HD-MTX monotherapy has never been reported. Therefore, we used the hazard ratio (HR) for EFS comparing HD-MTX/Cytarabine with HD-MTX as a reference. 34 The sample size was calculated based on the primary outcome (EFS), assuming an estimated HR of 0.54 for HD-MTX/Ifosfamide (ratio 1:1). With a significance level of 0.05 and a power of 80%, the target enrollment was estimated to be 83 patients.
Results
Patient characteristics and treatment outcomes of newly diagnosed PCNSL
Ninety-four patients were enrolled. The median age was 59 (range 16–82) years. The female-to-male ratio was 1.5:1. The majority of patients (52 patients, 55.3%) had an intermediate IELSG prognosis score. Fifty-two (55.3%) patients were in good performance status (Eastern Cooperative Oncology Group (ECOG) 0–1) at PCNSL diagnosis. The HD-MTX/Ifosfamide regimen and HD-MTX monotherapy were prescribed to 56 (59.6%) and 38 (40.4%) patients, respectively, based on primary physicians’ judgments. The HD-MTX/Ifosfamide cohort tended to be younger with better performance status, as shown in Table 1. Thirty-six (38.3%) patients who could afford to pay received rituximab combined with chemotherapy since the drug was non-reimbursable for PCNSL in Thailand.
Baseline characteristics and treatment response of newly diagnosed PCNSL patients.

CMT, chemotherapy; CR, complete response; ECOG, Eastern Cooperative Oncology Group; HD-MTX, high-dose methotrexate; IELSG, International Extranodal Lymphoma Study Group; PD, progressive disease; PR, partial response; SD, stable disease; WBRT, whole brain radiotherapy; XRT, radiotherapy.
The CR and PR rates of the entire cohort were 44.7% and 23.4%, respectively. The overall response rate (ORR) of patients who received HD-MTX/Ifosfamide and HD-MTX were 75% and 57.9%, respectively (Table 1). The toxicities led to chemotherapy discontinuation in seven (12.5%) HD-MTX/Ifosfamide-treated patients and nine (23.7%) HD-MTX–treated patients. The univariate analysis showed good performance status (ECOG 0–1) was the only factor related to the response (CR/PR) after chemotherapy, as shown in Table 2 (p = 0.013). Among 64 patients who achieved CR or PR after completing six cycles of chemotherapy, the WBRT (30.6–36 Gy) with boost lesion (up to 45 Gy) was administered to all 22 PR patients. Twenty patients who achieved CR after chemotherapy received WBRT (23.4–30.6 Gy) for consolidation treatment. The rest of the CR patients were observed without consolidation. Twenty-one PR patients subsequently obtained CR status after radiotherapy, but one patient experienced lymphoma progression. A total of 63 (67%) patients were in CR after completing first-line therapy.
Clinical factors related to overall response (CR and PR) after chemotherapy in PCNSL.
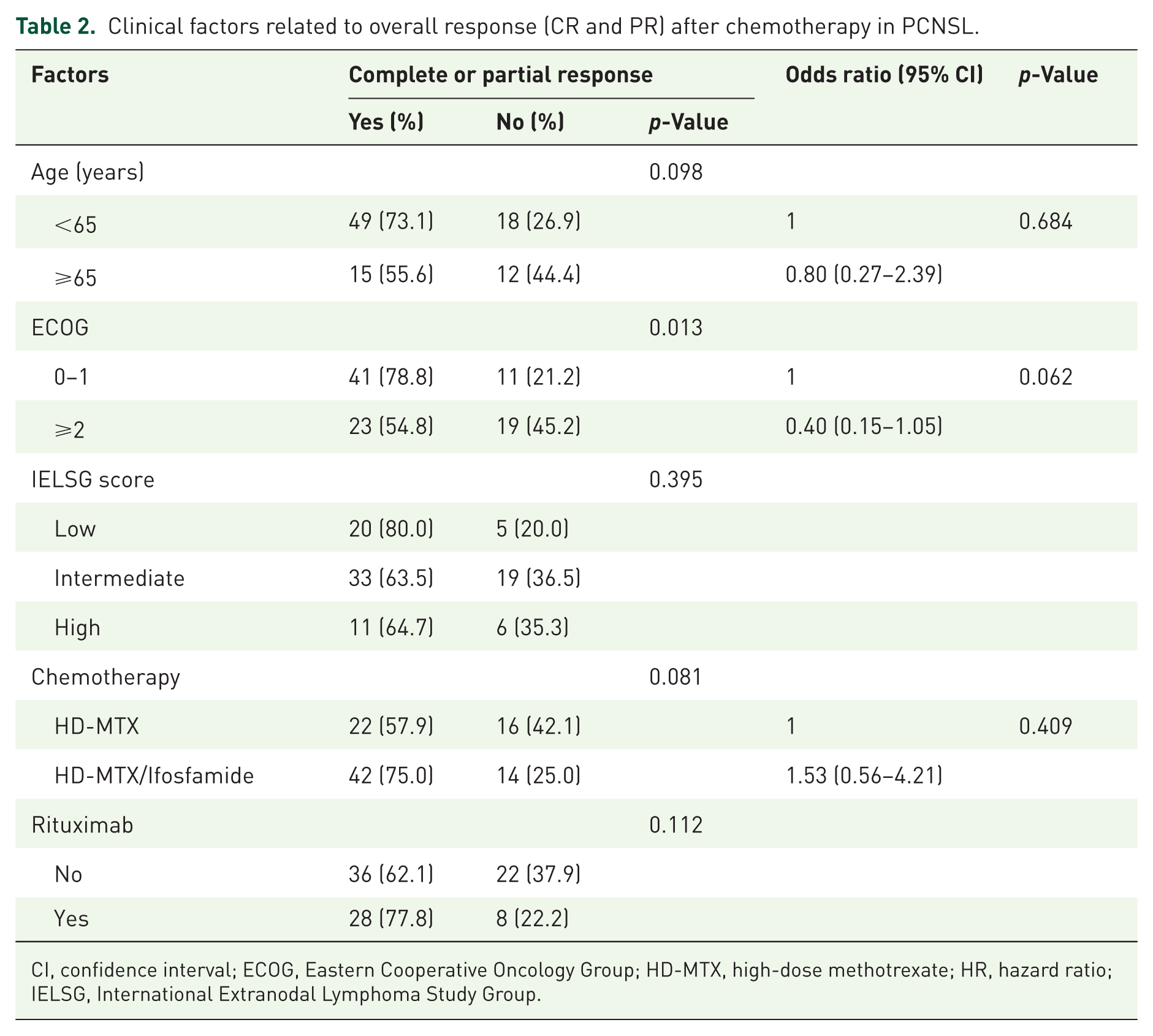
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HD-MTX, high-dose methotrexate; HR, hazard ratio; IELSG, International Extranodal Lymphoma Study Group.
Survival outcomes of PCNSL patients
For all 94 patients, the median follow-up time was 28 (range 1–166) months. The median EFS was 22 months, and the median OS was 64 months. The 2-year EFS and 2-year OS were 46.9% and 64.9%, respectively. The HD-MTX/Ifosfamide regimen was associated with superior EFS compared with the HD-MTX alone by univariate analysis (39 months vs 8 months, p = 0.021) (Figure 1(a)). An ECOG ⩾2 and intermediate/high IELSG scores were correlated with inferior OS (p = 0.007, p = 0.010, and p = 0.025, respectively). Factors associated with the EFS and OS are summarized in Table 3. The age difference between the HD-MTX and HD-MTX/Ifosfamide groups is a potential confounder for EFS. Using age and chemotherapy as covariates for EFS, the HR of HD-MTX/Ifosfamide was 1.76 (95% confidence interval (CI) 1.00–3.10, p = 0.051). Among 64 patients who achieved CR or PR after chemotherapy, 27 (42.2%) patients had relapsed lymphoma (N = 26) and PD (N = 1). Fourteen of the relapsed patients died from PCNSL progression.

(a) Event-free survival of newly diagnosed PCNSL patients compared between high-dose MTX/Ifosfamide and high-dose MTX regimens. (b) Progression-free survival of PCNSL patients who achieved complete and partial response after induction chemotherapy stratified by WBRT consolidation.
Univariate and multivariate analysis for EFS and OS of newly diagnosed PCNSL patients.

CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; EFS, event-free survival; HD-MTX, high-dose methotrexate; HR, hazard ratio; IELSG, International Extranodal Lymphoma Study Group; Ifos, Ifosfamide; mo, months; NR, not reached; OS, overall survival.
The median PFS was 47 months with a 2-year PFS of 63%. Baseline characteristics such as age, ECOG, IELSG score, chemotherapy regimen, rituximab, and achieving CR or PR after chemotherapy were not associated with PFS in the univariate analysis. Consolidation with radiotherapy was the only factor that significantly correlated with a superior PFS for chemo-responsive PCNSL patients in this cohort (Figure 1(b)). The 2-year PFS of patients who received and did not receive radiotherapy was 74.5% and 35.6%, respectively (p < 0.001).
Baseline characteristics of PCNSL and non-CNS lymphoma survivors in complete remission
Twenty PCNSL patients and 20 non-CNS lymphoma patients who were in CR of lymphoma were recruited. The median age of PCNSL and non-CNS lymphoma survivors at the time of cognitive function and HRQoL assessments was 60 (range 34–87) and 60 (range 42–78) years old, respectively. Fifty percent of patients in both groups were older than 60 years old. The age range of PCNSL and non-CNS lymphoma patients were similar (four patients of <50 years, six patients of 50–59 years, six patients of 60–69 years, and four patients of >70 years) The education level of a bachelor’s degree or more was found in 55% (N = 11) of PCNSL survivors and 45% (N = 9) of non-CNS lymphoma survivors (p = 0.343). All patients had an ECOG range 0–2. There was no difference in performance status between groups (ECOG 1–2: PCNSL 40% vs non-CNS 15%, p = 0.593). The median duration from CR to cognitive and HRQoL assessments was 37 (range 1–114) months among PCNSL survivors and 14 (range 1–49) months in the control group (p = 0.329). Eighty percent of PCNSL survivors received the HD-MTX/Ifosfamide regimen. Sixteen (80%) PCNSL survivors received WBRT, and five of them received a reduced dose of WBRT (23.4 Gy).
Cognitive impairment of PCNSL and non-CNS lymphoma survivors
Cognitive function screening using the MoCA test showed that 63% of PCNSL patients and 60% of non-CNS lymphoma patients had cognitive impairment. The mean MoCA score was not different between PCNSL and non-CNS lymphoma survivors (21.57 vs 23.7, p = 0.200). Attention and visuospatial attention were measured by digit span and symbol search tests, with no difference in mean scores between patient groups. Notably, 68% of PCNSL patients and 45% of non-CNS lymphoma patients had symbol search scores below the average (p = 0.147). Executive function evaluated with the WCST-64 test demonstrated that 73% of PCNSL patients and 45% of non-CNS lymphoma patients had scores in the range below the average value without any difference between groups (p = 0.071). The mean score of preservative responses and errors in PCNSL patients was in the range that reflected a mild executive function deficit. The mean score of preservative responses and errors in non-CNS lymphoma patients also indicated an executive function level just below the average value. The neuropsychological assessments between PCNSL and non-CNS lymphoma patients are summarized in Table 4. Baseline characteristics and cognitive function of PCNSL survivors who received different WBRT intensities are presented in Table 5.
Cognitive functions of PCNSL and non-CNS lymphoma patients in remission.

MoCA: normal ⩾25, mild cognitive impairment 18–24, dementia <18.
Digit forward: number of digits in the longest sequence correctly recalled.
Digit backward: number of digits in the longest sequence correctly recalled.
Digit sum: sum of digit forward and digit backward.
SS scaled score: extremely low = 1–3, borderline = 4–5, low average = 6–7, average = 8–11, high average = 12–13, superior = 14–15, very superior = 16–19.
WCST-64 standard score: severe <55, moderate to severe 55–61, moderate 62–69, mild to moderate 70–76, mild 77–84, below average 85–91, average 92–106, above average >107.
MoCA, Montreal Cognitive Assessment; SS, symbol search; WCST-64, Wisconsin Card Sorting Test-64.
Baseline characteristics and cognitive functions in PCNSL patients with different WBRT intensities.

MoCA: normal ⩾25, mild cognitive impairment 18–24, dementia <18.
Digit forward: number of digits in the longest sequence correctly recalled.
Digit backward: number of digits in the longest sequence correctly recalled.
Digit sum: sum of digit forward and digit backward.
SS scaled score: extremely low = 1–3, borderline = 4–5, low average = 6–7, average = 8–11, high average = 12–13, superior = 14–15, very superior = 16–19.
WCST-64 standard score: severe <55, moderate to severe 55–61, moderate 62–69, mild to moderate 70–76, mild 77–84, below average 85–91, average 92–106, above average >107.
CR, complete remission; MoCA, Montreal Cognitive Assessment; MTX, methotrexate; SS, symbol search; WCST-64, Wisconsin Card Sorting Test-64.
HRQoL between PCNSL and non-CNS lymphoma survivors
The HRQoL assessment of PCNSL and non-CNS lymphoma survivors is presented in Table 6. According to the EORTC QLQ-C30 questionnaire, PCNSL patients scored themselves significantly lower on global health status than non-CNS lymphoma patients (85.4 vs 92.5, p = 0.008). Assessment of specific functions of HRQoL by EORTC-QLQ30 found that PCNSL patients tended to have significant decreases in physical function compared to non-CNS lymphoma patients (92.7 vs 99.0, p = 0.053). The residual neurological deficits from prior CNS disease possibly impact the assessment by the HRQoL. However, other functional scales, such as role function, emotional function, cognitive function and social function, including the symptom scale, did not show any differences between PCNSL patients and non-CNS lymphoma patients. Brain-specific symptoms, assessed by EORTC QLQ-BN20, such as motor dysfunction and communication deficits, were more pronounced in PCNSL survivors without statistical significance.
HRQoL assessment of PCNSL and non-CNS lymphoma patients in remission.
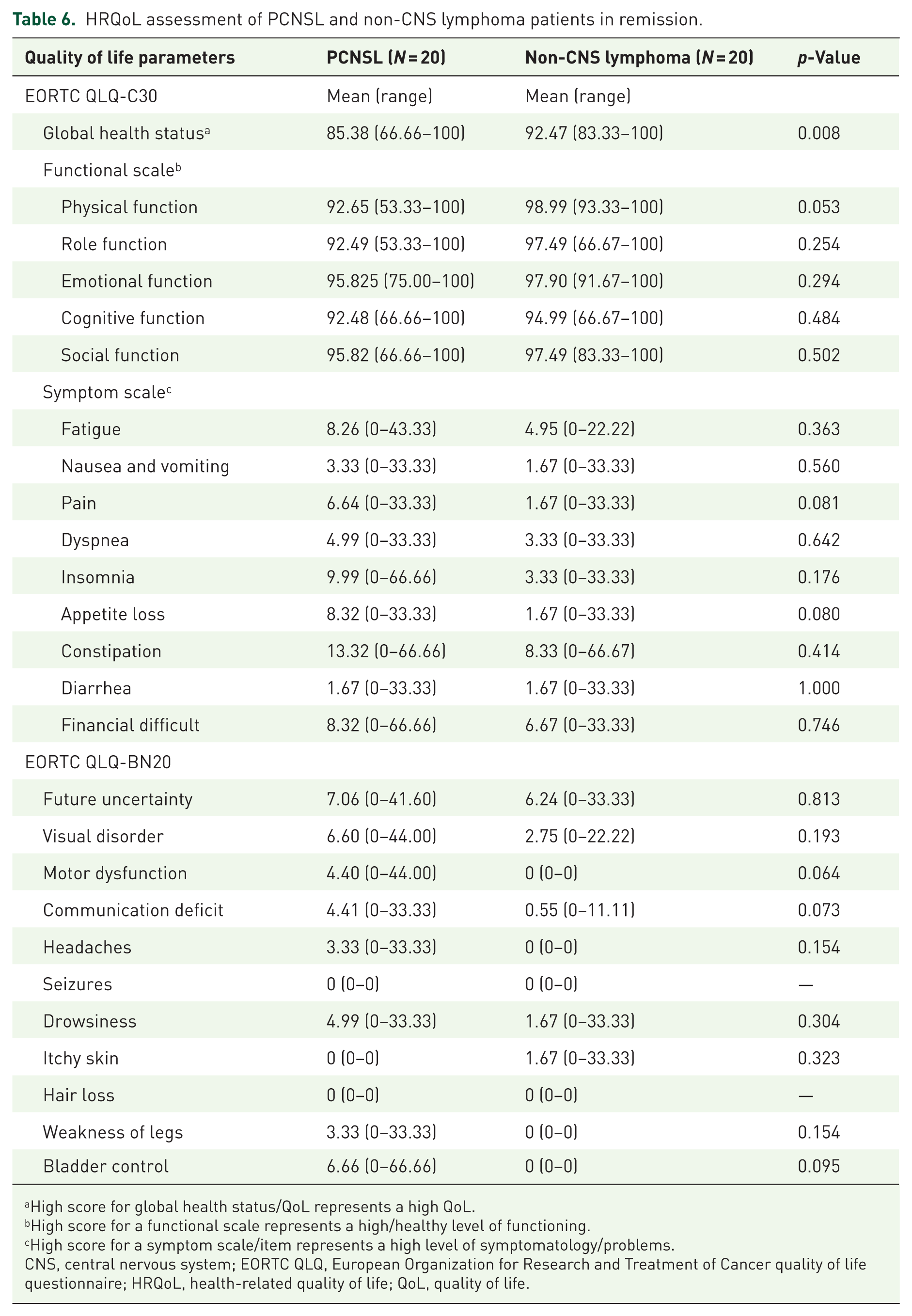
High score for global health status/QoL represents a high QoL.
High score for a functional scale represents a high/healthy level of functioning.
High score for a symptom scale/item represents a high level of symptomatology/problems.
CNS, central nervous system; EORTC QLQ, European Organization for Research and Treatment of Cancer quality of life questionnaire; HRQoL, health-related quality of life; QoL, quality of life.
Discussion
This study demonstrated that the HD-MTX/Ifosfamide ± rituximab protocol was more effective than the HD-MTX ± rituximab regimen by showing superior EFS and a potentially higher response rate in newly diagnosed PCNSL patients. However, the results may have been influenced by selection bias due to age and performance status reflecting real-life clinical practice. In the HD-MTX/Ifosfamide group, which mostly contained young and fit patients, the ORR was 75% with a median EFS and OS of 39 months and not reached, respectively. For the HD-MTX regimen, which mainly treated elderly and unfit patients, the ORR was 58% with a median EFS and OS of 8 and 40 months, respectively. These results displayed comparable efficacy to previous studies13,17–19,22 and suggest the positive impact of the ifosfamide combination. Furthermore, the results emphasized that patients with good performance status (ECOG 0–1) at PCNSL diagnosis achieved a significantly higher response rate and OS. IELSG risk scores remain a key component in the survival prognostic model for newly diagnosed PCNSL. Among patients with chemosensitive disease (CR/PR), WBRT consolidation therapy exhibited significance as a factor correlated with improved PFS, supporting the role of WBRT consolidation for early relapse prevention. In Thailand, Thiotepa, rituximab, and temozolomide are unaffordable for most patients due to their high cost and non-reimbursement, which leads to inaccessibility for ASCT as a frontline consolidation therapy.19–21 Therefore, relapsed PCNSL patients usually proceed to palliative care due to the lack of an accessible and effective treatment regimen.
Neurocognitive toxicity from WBRT is a major concern, especially in elderly PCNSL patients. This study evaluated cognitive function between PCNSL and non-CNS lymphoma survivors who had similar age and education levels. Using the MoCA screening test, the scores did not differ between PCNSL and non-CNS lymphoma survivors. Normal MoCA scores were recorded in 37%–40% of patients. Applying specific standardized tests for attention/executive function assessment, the mean scores of digit span, symbol search, and WCST-64 standard scores were not different between the two groups. However, the majority of PCNSL survivors (68%) had scores below average for symbol search, but 55% of non-CNS lymphoma survivors had scores at or above average value. And 73% of PCNSL survivors had scores below average for the WCST-64 test, but 55% of non-CNS lymphoma survivors had scores at or above average value. Eighty percent of this PCNSL group received WBRT (23.4–36 Gy) to prevent relapse. Cognitive impairment from WBRT has been shown to deteriorate in studies with longer follow-up. 35 Our study demonstrated that PCNSL survivors who received WBRT 30.6–36 Gy had a lower score of attention domains (digit forward, digit sum, and symbol search) compared with PCNSL survivors who were in non-WBRT and WBRT 23.4 Gy cohorts. The median duration between CR to cognitive assessment in this study was 37 months in the PCNSL group. These cognitive declines in attention and executive function may reflect the sequelae of PCNSL and CNS-directed WBRT and/or chemotherapies. Previous long-term follow-up studies reported an increase in attentiveness/executive function and HRQoL impairment in patients who received WBRT (36 and 40 Gy) compared to those with ASCT.24,25 Correa et al. found no difference in cognitive performance decline and HRQoL between reduced-dose WBRT (<24 Gy) and ASCT patients after 3 years. 26 Our center provided WBRT doses of 23.4–30.6 Gy for CR patients and 30.6–36 Gy with boosted lesion to 45 Gy for PR patients. Larger studies to investigate the proper dose intensity of WBRT with the lowest cognitive toxicity and sustained disease control are needed.
In one section measuring global health status in the EORTC QLQ-C30 questionnaire, PCNSL survivors rated themselves significantly lower on performance scores than non-CNS lymphoma survivors. This impact on HRQoL among PCNSL survivors probably relates to the sequelae of neurological dysfunctions from disease and/or treatments, which are different from non-CNS lymphoma survivors. According to the EORTC QLQ-C30, the clinical significance between group differences is defined by a minimum important difference of 5–10 points or more on the 0–100 scale. The physical functional scale of PCNSL and non-CNS lymphoma survivors was 92.65 and 98.99, respectively. The functional and symptom scale results among PCNSL patients tended to report lower physical function scales than non-CNS lymphoma patients, which may have been caused by residual neurological deficits from the brain tumor. The mild decline in attention and executive function did not translate into a reduction in their self-reported cognitive domain and other functions of HRQoL.
The limitations of this study include the small number of patients and imbalanced baseline characteristics in the retrospective cohort, including the heterogeneity of some confounding factors in the prospective cohort, such as time to cognitive assessment, differing intensities of WBRT and a single timepoint of cognitive evaluation. Although there is no statistically significant difference in the duration from CR to cognitive and HRQoL assessment between groups. Due to a small number of patients, the p-value >0.05 should be interpreted with caution. The PCNSL patients were assessed at 37 months, which may represent more cognitive declination with a longer period. For control patients without CNS disease or treatment, the CR duration of 14 months should have less impact on cognitive function, but this shorter period impacts the HRQoL results. Furthermore, the WBRT dosage in this study may be different from other international trials due to the local practice. In countries with limited access to some medications, choosing between WBRT consolidation and observation in elderly PCNSL patients in remission after chemotherapy is a tough choice when balancing between relapse risk and cognitive impairment. The future perspective is to conduct a multicenter prospective study in PCNSL patients using R-MTX/Ifosfamide induction regimen and consolidation therapy with WBRT 23.4 Gy with serial monitoring of cognitive function, HR-QoL and survival outcome.
Conclusion
This study demonstrated the efficacy and tolerability of HD-MTX/Ifosfamide in young and fit newly diagnosed PCNSL patients. The findings highlight the benefit of incorporating ifosfamide into HD-MTX–based regimens for induction treatment. However, approximately two-thirds of CR patients who did not receive consolidation therapy experienced early relapses. Low-dose WBRT consolidation may reduce relapse risk, with only mild to moderate cognitive impairment compared with non-CNS lymphoma survivors. However, decreased measures of attention, executive function and HRQoL were observed in PCNSL survivors. Therefore, comprehensive discussions with patients and their families regarding treatment choices are critical, particularly when treatment options are limited.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251400429 – Supplemental material for Real-world survival, cognitive, and quality of life outcomes of methotrexate/ifosfamide plus whole brain radiotherapy for newly diagnosed primary central nervous system lymphoma
Supplemental material, sj-docx-1-tah-10.1177_20406207251400429 for Real-world survival, cognitive, and quality of life outcomes of methotrexate/ifosfamide plus whole brain radiotherapy for newly diagnosed primary central nervous system lymphoma by Warayoot Luangmaneerote, Natrada Rattanapong, Anthipa Chokesuwattanaskul, Tassapong Raiyawa, Thunyamon Chajuwan, Pitchayaporn Jongdee, Ponlapat Rojnuckarin and Chantiya Chanswangphuwana in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
