Abstract
Objective:
Here, we aimed to evaluate the prognostic values of the endothelial activation and stress index (EASIX) and its derivatives, modified EASIX (mEASIX) and simplified EASIX (sEASIX), in patients with multiple myeloma (MM).
Methods:
The data of 134 newly diagnosed MM patients between January 2020 and December 2024 were retrospectively analyzed. Patients were divided into groups based on EASIX and its derivatives, and the outcomes of survival rates in groups were compared. Optimal cut-off points were determined using the receiver-operating characteristic analysis, and mortality predictive values of the scores were investigated. The independent prognostic factors were evaluated through univariate and multivariate Cox regression analyses.
Results:
The optimal cut-off point for EASIX was detected as >1.03, and survival times were significantly shorter in patients with higher EASIX scores (
Conclusion:
EASIX, especially mEASIX, is a valuable prognostic tool for predicting survival in MM patients. EASIX can be easily integrated into clinical practice due to its simple computability and reliance on commonly used laboratory parameters. However, larger prospective studies are needed to determine how these scores can be integrated with traditional prognostic systems such as the International Staging System (ISS) and Revised-ISS.
Introduction
Characterized by clonal proliferation of malignant plasma cells, multiple myeloma (MM) is a heterogeneous hematological malignancy. 1 Clonal evolution and additional genetic events during the disease influence the progression from the asymptomatic to the symptomatic stage of the disease and lead to a refractory disease state. 2 In MM, increased production of monoclonal immunoglobulin and resulting complications such as bone marrow suppression, hypercalcemia, renal failure, and osteolytic lesions are frequently encountered. Despite significant advances in therapeutic approaches, MM is still far from being a completely curable challenge and the prognosis of MM varies widely among individuals. Therefore, reliable prognostic instruments are needed to more accurately assess the prognosis of MM patients and optimize the therapeutic processes.
Different biomarkers and risk scoring systems used in prognostic assessments contribute to the individualization of the treatment plans by allowing MM patients to be stratified into risk groups. The most common staging system used to estimate the prognosis of MM is the Revised International Staging System (R-ISS). 3 The R-ISS was created by incorporating chromosomal abnormalities (CA) and serum lactate dehydrogenase (LDH) into the original International Staging System (ISS). However, cytogenetic abnormalities in R-ISS do not include all genetic abnormalities in MM and can only be assessed in selected institutions capable of performing the analysis of genes. For such reasons, there is still an unmet need to develop a simple and useful risk stratification model for MM.
In recent years, the endothelial activation and stress index (EASIX) has attracted attention as a prognostic index in hematological malignancies. Initially developed to predict the prognosis of acute graft-versus-host disease following hematopoietic stem cell transplantation, EASIX has subsequently come to the fore with its potential prognostic value in MM and other hematological diseases. EASIX, which is simple and practical to formulate, is calculated through routine laboratory parameters, such as LDH, creatinine, and platelet (PLT) count.4–10 These parameters constituting EASIX are well-known prognostic factors for MM. Over time, such novel variations of EASIX as modified EASIX (mEASIX) and simplified EASIX (sEASIX) have been created.
In our center, located in Bursa, Türkiye, the standard treatment pathway for newly diagnosed multiple myeloma aligns with international guidelines and national practices. Transplant-eligible patients typically receive induction therapy with a bortezomib-based triplet regimen (e.g., VRD: bortezomib, lenalidomide, dexamethasone), followed by autologous stem cell transplantation (ASCT) and maintenance treatment. For transplant-ineligible patients, combinations of proteasome inhibitors, immunomodulatory agents, and corticosteroids are commonly used. This real-world cohort reflects patients treated according to this institutional algorithm.
In this study, we aim to compare the EASIX score and its derivatives in MM patients and to evaluate the effects of these scores on the prognosis. In particular, we aim to contribute to the prognostic evaluation process in MM patients by revealing the predictive values, clinical applicability, and superior characteristics of these scores over each other.
Material and method
Study design and selection of patients
A total of 238 newly diagnosed MM patients were initially screened. Among these, 104 patients were excluded due to missing data on key laboratory parameters required to calculate EASIX, mEASIX, or sEASIX scores, or due to incomplete follow-up information. Due to the retrospective nature of the study and the predefined time window, a formal sample size calculation or power analysis was not conducted. The study included all eligible patients meeting the inclusion criteria during the specified period (January 2020–December 2024). Demographic characteristics, laboratory values, and clinical variables of the patients were evaluated in detail. Patients having complete laboratory and clinical data that were required to calculate EASIX, mEASIX, and sEASIX scores, those with at least 6-month followed-up period after the diagnosis, and those having the determined mortality and survival status during the follow-up period were included in the study. All laboratory tests were performed on accredited hematology and biochemical analyzers used routinely in Hospital Central Laboratory. Even so, those with monoclonal gammopathy of undetermined significance, nonsecretory MM, amyloidosis, and plasma cell leukemia were excluded. Additionally, patients with incomplete clinical and laboratory data, insufficient follow-up period (<6 months), or those with another primary malignancy or severe systemic disease to affect the study data were also not included in the study.
In terms of ethical approval, all procedures in the study were performed under the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Data accumulation
Such demographic characteristics of the patients as age, gender, accompanying diseases, types of MM, and clinical features were recorded. The laboratory parameters including the markers of MM, hemoglobin (Hb), white blood cell (WBC) count, lymphocyte, PLT, LDH, uric acid, creatinine, calcium, potassium, phosphorus, total protein, albumin, procalcitonin, and C-reactive protein (CRP) were evaluated. The data regarding treatment processes, chemotherapy protocols administered, ASCT status, immunomodulatory therapy, use of anticoagulant, responses to the treatment, and final outpatient follow-up were also analyzed. ISS, R-ISS, and EASIX were assessed at initial diagnosis. CA were evaluated based on conventional cytogenetic investigations or fluorescence in situ hybridization. While the high-risk CA was characterized by the presence of at least one of del(17p), t(4;14), or t(14;16), the standard-risk CA was characterized by the absence of previously mentioned abnormalities.
Calculation of EASIX, mEASIX, and sEASIX
The values of LDH, CRP, creatinine, and PLT count obtained at the time of diagnosis were used to calculate the EASIX, sEASIX, and mEASIX scores. The EASIX score was calculated according to the standard formula established by Luft et al.: (LDH (U/L) × creatinine (mg/dL))/PLT count (G/L). 7 The simplified version (sEASIX) was calculated as (LDH (U/L)/PLT count (G/L)), and the modified version (mEASIX) as (LDH (U/L) × CRP (mg/dL))/PLT count (G/L), as proposed by Pennisi et al. 11 These indices were then analyzed to assess their prognostic impact on mortality and survival outcomes in patients with MM.
Statistical analysis
The descriptive statistics are presented as the median and interquartile range (IQR = Q1 and Q3) for numerical variables and number (
Results
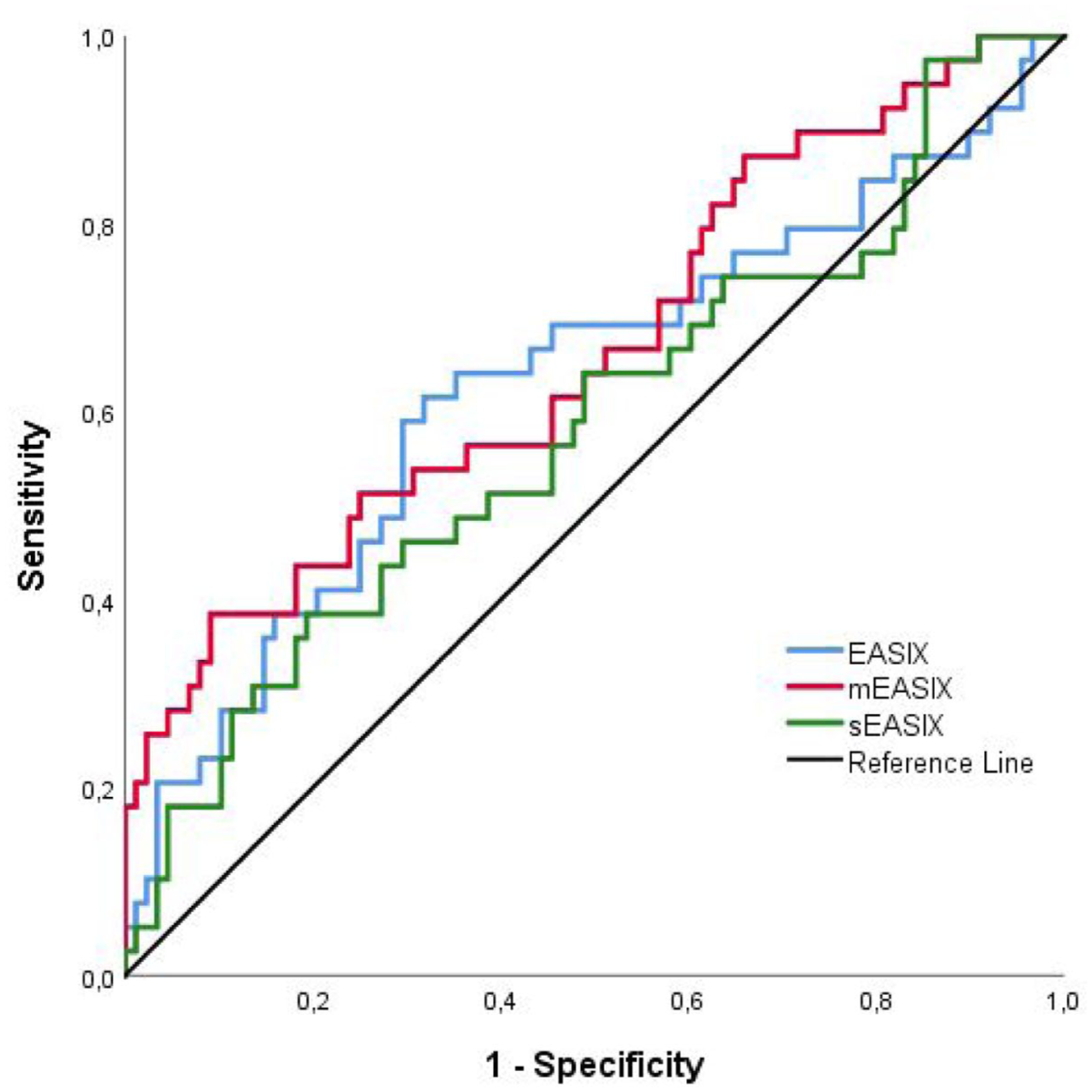
In our study, the mortality predictive power of EASIX and its derivatives (mEASIX and sEASIX) in MM patients was evaluated. The optimal cut-off point determined for EASIX was calculated as 1.03, and the patients with a cut-off of>1.03 were assessed to have a higher mortality risk (AUC = 0.631; 95% CI: 0.519–0.742). In the group of EASIX > 1.03, the mortality predictability was calculated as 61.5% for sensitivity and 68.2% for specificity. The optimal cut-off point for the mEASIX score was detected as 26.5, and the mortality predictability was stronger for patients above 26.5 (AUC = 0.663; 95% CI: 0.558–0.769). The sensitivity and specificity rates of the patients in the mEASIX group were found to be 38.5 and 90.9%, respectively. Even so, the cut-off point for the sEASIX score was calculated as 1.2, and the mortality predictability was lower for those above 1.2 (AUC = 0.586; 95% CI: 0.475–0.697). The sensitivity and specificity of the sEASIX group were determined as 38.0 and 80.7%, respectively (Table 1; Figure 1).
Predicting mortality in patients according to the cut-off values of EASIX, mEASIX, and sEASIX.

AUC, area under the curve; CI, confidence interval; EASIX, endothelial activation and stress index; mEASIX, modified EASIX; NPV, negative predictive value; PPV, positive predictive value; SE, standard error; sEASIX, simplified EASIX.
Kaplan–Meier curves and overall survival averages of all cases.
When the patients were divided into two as the EASIX groups of the cut-off values of <1.03 and >1.03, the distributions in both groups were compared for each parameter; the distributions of the variables in both groups were examined under the EASIX cut-off points, and the characteristics of the evaluated groups are presented in Table 2. It was observed that the groups were similar in terms of the distributions of age, gender, BMI, and comorbidity (
Comparison of basic clinical features of cases in EASIX groups.

AF, atrial fibrillation; ASCT, autologous stem cell transplant; B-MG, beta 2-microglobulin; BMI, body mass index; BUN, blood urea nitrogen; CAD, coronary artery disease; CRP, C-reactive protein; CVE, cerebrovascular events; DEXA, dual-energy X-ray absorptiometry; DM, diabetes mellitus; EASIX, endothelial activation and stress index; ECOG, Eastern Cooperative Oncology Group; ESA, erythropoiesis-stimulating agent; FISH, fluorescence in situ hybridization; GFR, glomerular filtration rate; Hb, hemoglobin; HT, hypertension; IgA, immunoglobulin A; IgG, immunoglobulin G; IgM, immunoglobulin M; ISS, International Staging System; LDH, lactate dehydrogenase; LM, lenalidomide maintenance; MM, multiple myeloma; NSR, normal sinus rhythm; PLT, platelet; R-ISS, Revised-ISS; VGPR, very good partial response; WBC, white blood count.
The prognostic impacts of the clinical variables on the OS rates of the cases were evaluated (Table 3). The mean OS of all cases was calculated as (OS = 40.5 months; 95% CI: 35.4–45.61) using the Kaplan–Meier curves (Figure 2). Given the analyses of survival rates, the OS time of patients with high EASIX scores (OS = 30.19 months; 95% CI: 22.82–37.56) was found to be significantly shorter, compared to those in the low EASIX group (OS = 47.58 months; 95% CI: 41.53–53.63;
Comparison of overall survival according to the clinical characteristics of the cases using the log-rank test.
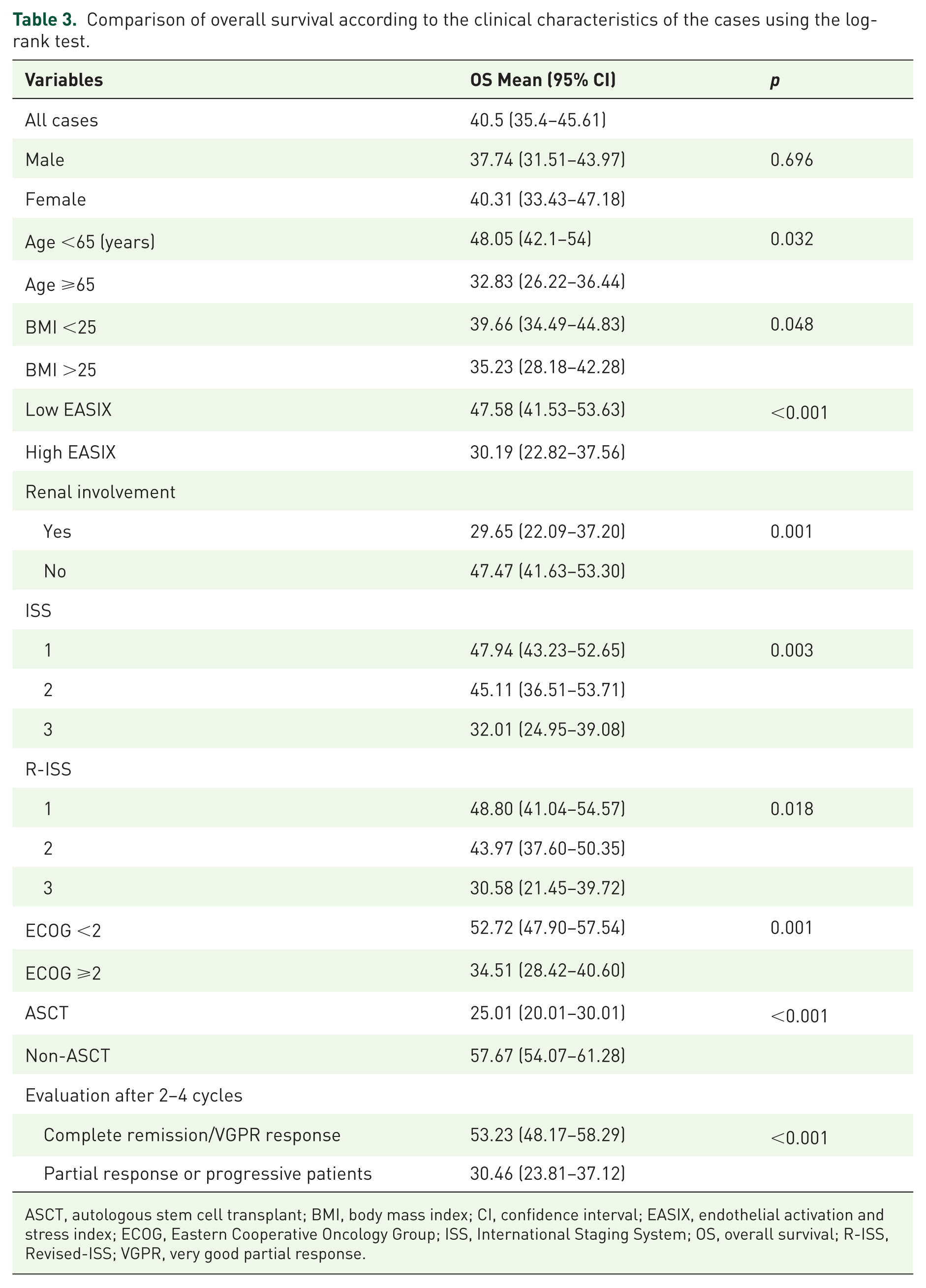
ASCT, autologous stem cell transplant; BMI, body mass index; CI, confidence interval; EASIX, endothelial activation and stress index; ECOG, Eastern Cooperative Oncology Group; ISS, International Staging System; OS, overall survival; R-ISS, Revised-ISS; VGPR, very good partial response.
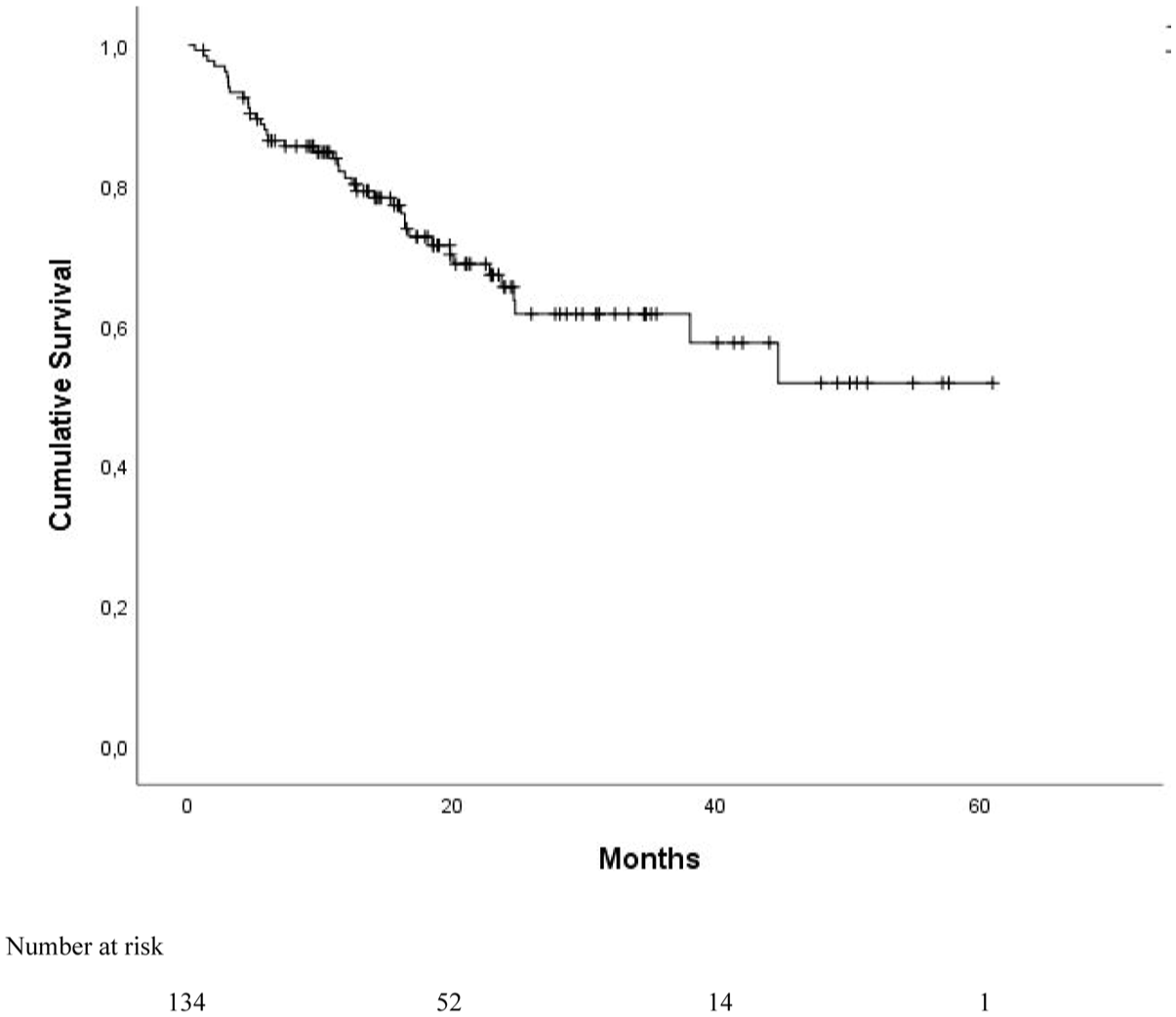
Kaplan–Meier curves for overall survival of all cases.
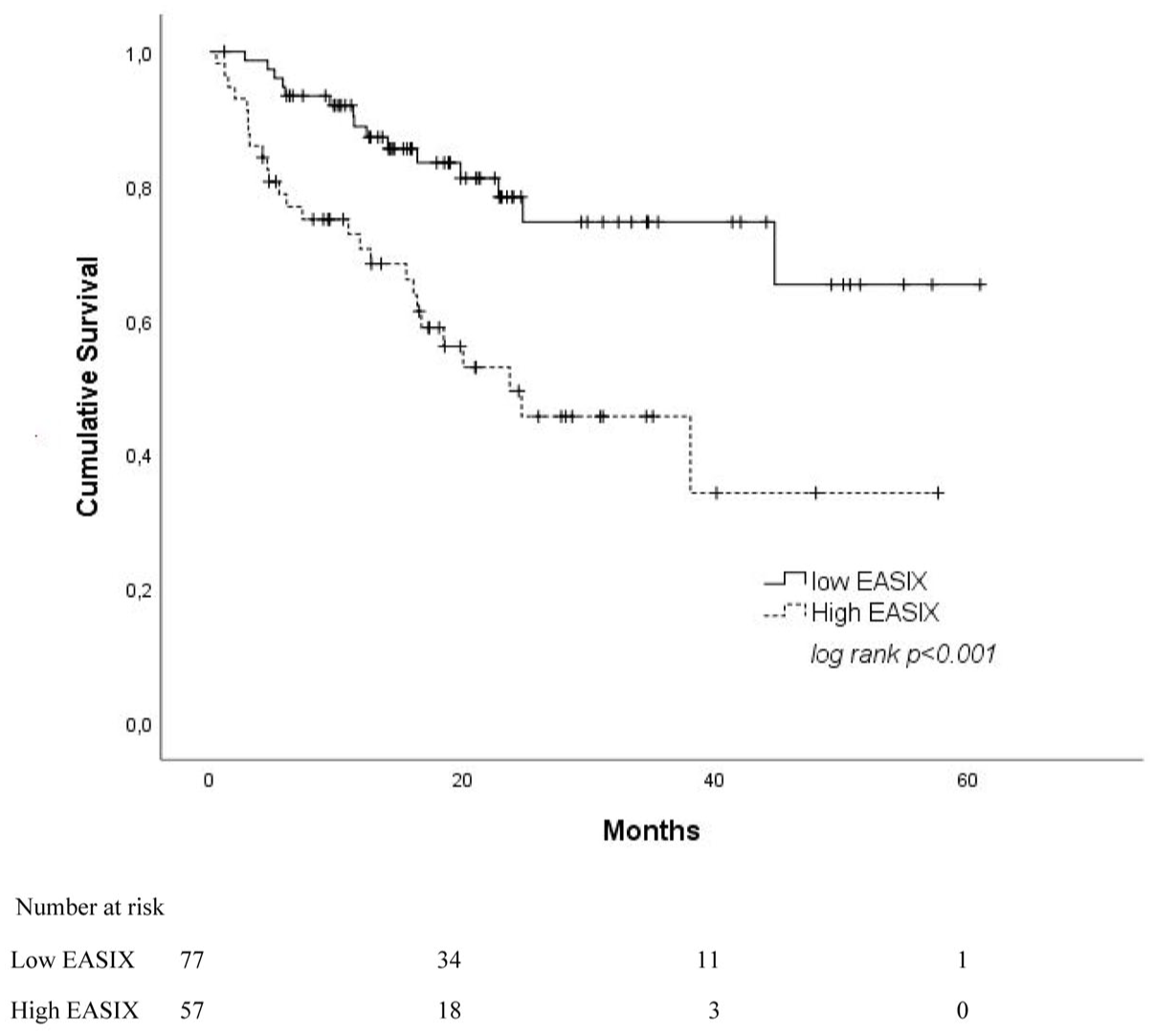
Kaplan–Meier curves for overall survival according to the EASIX groups.
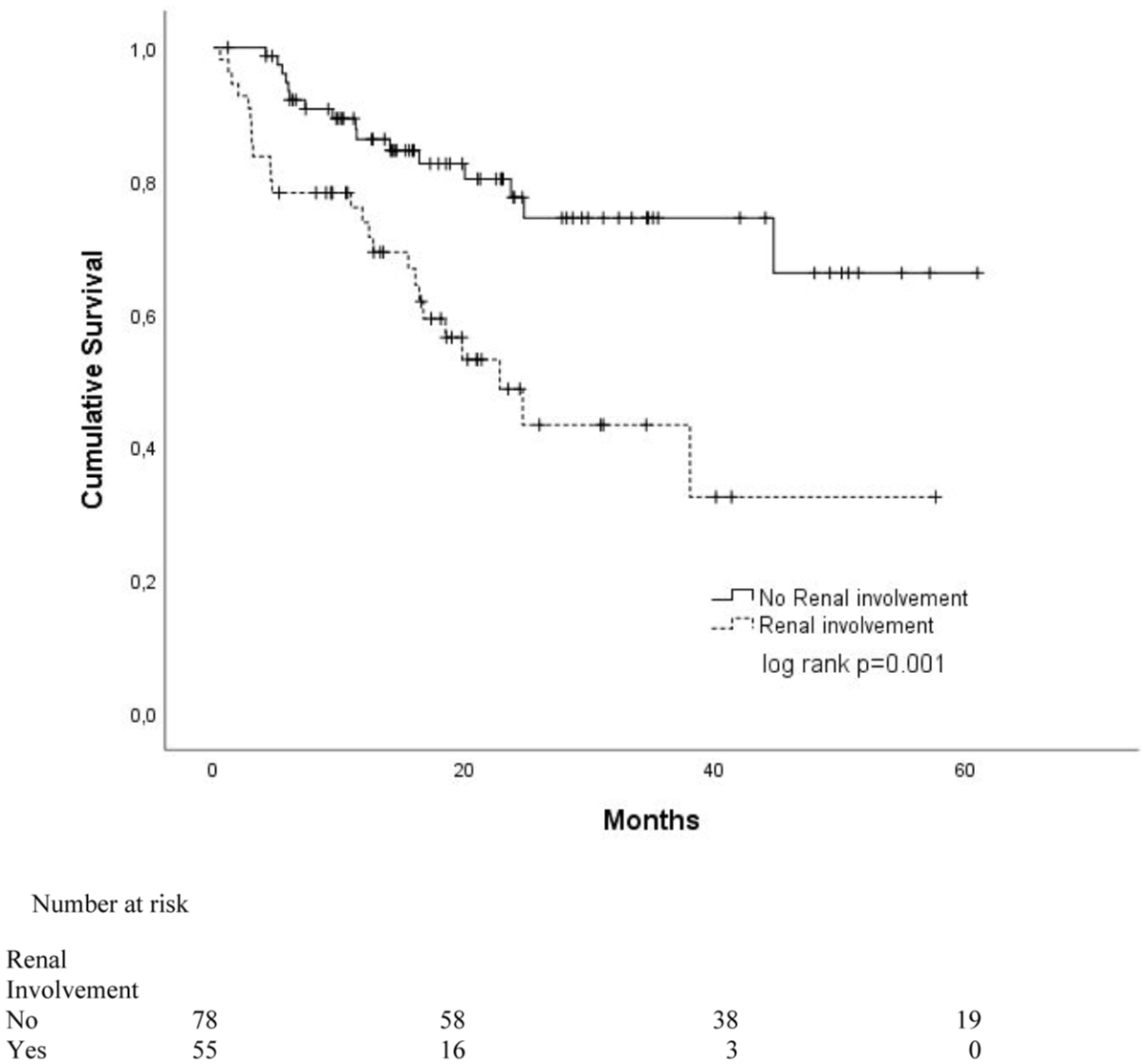
Kaplan–Meier curves for survivals according to the renal involvement rates of the patients.
The findings of the univariate Cox regression model and the multivariate Cox regression model analyses of the clinical parameters considered to affect the OS times of the cases are presented in Table 4. Given the findings of the univariate Cox regression analysis, the patients >65 years of age, BMI >25, ECOG ⩾2, presence of atrial fibrillation, high EASIX, low albumin and glomerular filtration rate, high BUN, procalcitonin, B-MG, ISS and R-ISS scores, renal involvement, non-ASCT, non-LM, not receiving immunomodulatory therapy, erythropoiesis-stimulating agent, nonanticoagulant antigregant, venous thromboembolic event within 6 months of the diagnosis, and partial or stable response or progressive disease after 2–4 cycles were determined as the factors having a statistically significant effect on shorter survival rates in MM patients (
Univariate and multivariate Cox analysis for overall survival (

Bold values indicate
AF, atrial Fibrillation; ASCT, autologous stem cell transplant, B-MG, beta 2-microglobulin; BMI, body mass index; BUN, blood urea nitrogen; CI, confidence interval; CRP, C-reactive protein; CVC, central venous catheter; DEXA, dual-energy X-ray absorptiometry; EASIX, endothelial activation and stress index; ECOG, Eastern Cooperative Oncology Group; ESA, erythropoiesis-stimulating agent; FISH, fluorescence in situ hybridization; GFR, glomerular filtration rate; Hb, hemoglobin; HR, hazard ratio; IgA, immunoglobulin A; IgG, immunoglobulin G; IgM, immunoglobulin M; ISS, International Staging System; LDH, lactate dehydrogenase; LM, lenalidomide maintenance; MM, multiple myeloma; PLT, platelet; R-ISS, Revised-ISS; VTE, Venous thromboembolic event; WBC, white blood count.
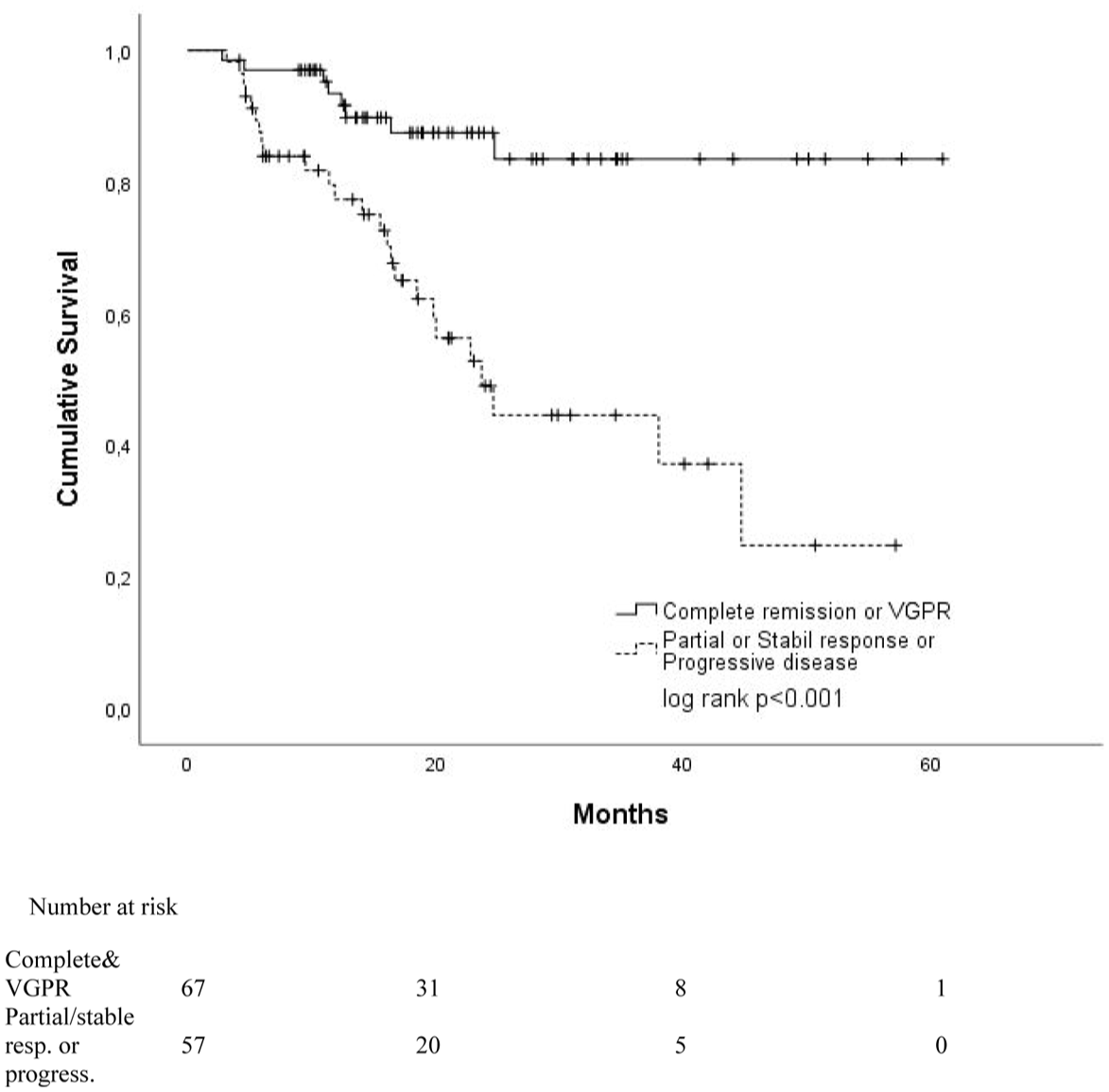
Kaplan–Meier curves for survivals according to responses after 2–4 cycles.
As a result, EASIX, especially mEASIX, stands out as an important instrument for the prognostic assessment of MM patients. A higher EASIX score was found to be associated with poor survival, and it may be recommended that EASIX be included in routine prognostic assessment processes in clinical practice.
Discussion
The presented study comparatively evaluated the effects of EASIX, mEASIX, and sEASIX scores on the prognosis in MM patients and examined the role of these scores in the prediction of mortality. The study findings revealed that higher EASIX scores were associated with a more severe course of the disease and shorter survival times. In particular, the patients with EASIX >1.03 cut-off point were found to have significantly shorter OS rates than those in the low EASIX group (
Such advantages of EASIX as the simple calculation method, reliance on laboratory parameters, and easy clinical practice make EASIX an attractive tool for risk assessment in hematological malignancies. Initially developed to predict the prognosis of graft-versus-host disease after the transplantation of allogeneic hematopoietic stem cells, EASIX has recently been shown to be an important predictive tool in malignancies, such as MM.8–10 Our study confirms that EASIX demonstrates a strong correlation with survival rates in MM patients, and therefore can be a useful tool in clinical decision-making processes. However, the prognostic power of EASIX in MM may be more limited, compared to other hematological malignancies. In the study by Luft et al., 6 while EASIX is reported to be successful in predicting the complications after the allogeneic transplantation the evidence for the use of EASIX in MM patients is also emphasized to be more limited. Although our study makes significant contributions to filling this gap, we consider that our findings need to be confirmed in larger patient groups.
mEASIX is a variant created by adding CRP to EASIX and targets to better reflect the inflammatory processes. 11 In this study, mEASIX was demonstrated to offer higher specificity, which is consistent with the findings of previous studies supporting the role of systemic inflammation in the prognosis of MM patients. 3 However, the relatively low sensitivity of mEASIX may limit its use, especially in early-stage patients.
However, the low performance of sEASIX in predicting mortality is noteworthy. The relatively lower AUC value of sEASIX suggests it may be less reliable for clinical decision-making compared to EASIX or mEASIX. In several previous studies, PLT count alone has been shown to have a prognostic value, although it is not strong enough when combined with other biomarkers in MM. 2 Therefore, the clinical use of sEASIX may be limited in clinical settings. Although the EASIX family scores showed statistical significance for predicting OS and PFS, their AUC values did not exceed 0.85. This may be attributed, in part, to the simplicity of the indices, which are based on readily available clinical biomarkers. However, these parameters are mechanistically relevant to MM pathophysiology. Elevated LDH levels reflect high tumor burden and increased cellular turnover. Serum creatinine is a marker of renal impairment, a frequent complication of MM due to light chain nephropathy and other disease-related mechanisms. CRP is an acute-phase reactant that signals systemic inflammation, often associated with poor prognosis in hematologic malignancies. Thrombocytopenia may indicate bone marrow infiltration or cytokine-induced suppression of megakaryopoiesis. When integrated, these variables offer a practical yet biologically grounded approach to risk stratification in MM. Therefore, despite their modest discriminatory capacity, the EASIX-based scores retain clinical relevance due to their strong physiological rationale and accessibility in routine practice.
Among the most commonly used staging systems to determine the prognosis in MM are ISS and R-ISS. 1 Our findings have revealed that higher EASIX scores are correlated with ISS and R-ISS, leading to a debate about whether EASIX is an independent prognostic tool. Such systems as R-ISS may provide a more comprehensive risk assessment due to including cytogenetic data. However, while genetic analyses may not be readily available in every hospital, the scores based on simple biochemical markers such as EASIX stand out as a more widely applicable alternative. 7 In our study, however, whether EASIX is an independent prognostic factor in MM patients could not be fully evaluated, compared to ISS and R-ISS. Therefore, we consider that it is important to investigate the additional contribution of EASIX in predicting prognosis when used together with ISS and R-ISS in future studies.
Limitations and study recommendations
Several limitations of this study should be acknowledged. First, its retrospective, single-center design may have introduced selection and information biases inherent to chart reviews. Second, although we included all eligible patients diagnosed between January 2020 and December 2024, no a priori power analysis was performed; the final cohort (
Conclusion
Our study reveals that EASIX and its derivatives, mEASIX and sEASIX, can be considered an important tool in the prognostic assessment of MM patients. In the study, higher EASIX scores were found to be associated with poor survival; therefore, it can be recommended that EASIX be included in the prognostic assessment processes in clinical practice. However, how EASIX can be integrated with such traditional prognostic systems as ISS and R-ISS requires further research. We consider that future prospective and larger-scale studies can determine better the place of such scores in clinical practice.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251387409 – Supplemental material for Clinical practice of the endothelial activation and stress index and its modified versions in multiple myeloma and the effects on prognosis
Supplemental material, sj-docx-1-tah-10.1177_20406207251387409 for Clinical practice of the endothelial activation and stress index and its modified versions in multiple myeloma and the effects on prognosis by Vildan Gursoy, Ozgur Omer Gul, Elif Kose and Sevil Sadri in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
