Abstract
Background:
Secondary myelofibrosis (SMF) is characterized by the excessive deposition of fibrous tissue on top of the primary disease, often causing clinical manifestations to be overshadowed by the primary disease. Unfortunately, current staging systems do not incorporate myelofibrosis, leading to potential treatment delays for SMF.
Objectives:
To evaluate the prognosis of patients with multiple myeloma (MM) complicated with myelofibrosis
Design:
The study included the clinical data and treatment results of 208 newly diagnosed multiple myeloma (NDMM) patients who were treated in the Affiliated Hospital of Qingdao University from January 2014 to August 2020, and performed a retrospective analysis.
Methods:
All patients underwent bone marrow biopsy, and MF severity was classified into grades 0–3 according to the 2016 WHO criteria. Treatment efficacy was evaluated based on the International Myeloma Working Group (IMWG) standard and SPSS was used for analysis.
Results:
The MM patients without SMF exhibited better treatment response (
Conclusion:
Myelofibrosis emerges as an important prognostic indicator for predicting the survival outcomes of NDMM patients. In the era of new therapeutics, there is a pressing need to explore novel treatment strategies in order to improve the prognosis of patients with multiple myeloma complicated by myelofibrosis.
Introduction
Multiple myeloma (MM) is a complex disease characterized by the clonal expansion of plasma cells in the bone marrow, leading to monoclonal protein production and end-organ damage.1,2 Despite advancements in treatment modalities such as immunomodulatory agents, proteasome inhibitors, monoclonal antibodies, and stem cell transplantation, patient’s response and prognosis can vary greatly. 3 To improve outcomes, understanding prognostic factors and individualized treatment strategies are crucial.
Commonly used staging systems for MM include DS (Durie–Salmon System), ISS (International Staging System), and R-ISS (Revised-International Staging System), but they have limitations in predicting prognosis. For instance, DS staging focuses on tumor burden and may not accurately predict outcomes in specific MM subtypes like IgD and IgM. 4 Similarly, the staging of MM based on β2-microglobulin and serum albumin fails to provide a comprehensive evaluation of the biological characteristics of the disease. The incorporation of lactate dehydrogenase and genetic risk factors into the R-ISS still falls short in considering crucial factors such as age and comorbidities.
Myelofibrosis (MF) is a condition characterized by the abnormal deposition of reticulin fibers and collagen in the bone marrow and can arise from various malignant and nonmalignant diseases. It is currently believed that MF occurs due to the involvement of hematopoietic stem/progenitor cells in depositing these fibrous proteins, contributing to a compromised microenvironment rather than supporting normal hematopoiesis.5,6 It is characterized by varying degrees of blood cell reduction, extramedullary hematopoiesis, progressive splenomegaly, and weight loss, all of which are systemic symptoms impacting quality of life.7,8 MF encompasses two main types: primary myelofibrosis (PMF) and secondary myelofibrosis (SMF). PMF is a myeloproliferative disorder, whereas SMF involves the development of fibrous tissue in conjunction with an underlying primary disease. Various hematologic conditions can be associated with MF.9,10 SMF often remains undetected due to its overshadowed clinical manifestations by the primary disease, leading to potential treatment delays. In the era of novel drug therapies, limited research explores the relationship between clinical efficacy, prognosis, and immune-inflammatory markers in MM patients with MF.
In this study, we collected clinical data from 208 newly diagnosed MM patients treated at Qingdao University Affiliated Hospital between 2014 and 2020. We aimed to investigate the therapeutic efficacy, prognosis, and survival outcomes of MM patients with SMF. Additionally, we investigated the relationship between MM-SMF and immune-inflammatory markers, including neutrophil–lymphocyte ratio (NLR), monocyte–lymphocyte ratio (MLR), and platelet–lymphocyte ratio (PLR). Incorporating these factors, along with cytogenetics and immunophenotype, can yield more precise prognostic insights for individuals with MM.
Methods
This retrospective study aimed to investigate the clinical characteristics and outcomes of patients with MM-SMF. A total of 208 newly diagnosed MM patients treated at the Department of Hematology, Qingdao University Affiliated Hospital between January 2014 and August 2020 were included in the analysis, none of the patients had received treatment at the time of diagnosis. Patients with incomplete clinical data, previous diagnosis and treatment from other hospitals, existing infections, connective tissue diseases, or other malignant tumors were excluded.
All patients underwent bone marrow biopsy, and MF severity was classified into grades 0–3 according to the 2016 WHO criteria.11,12 In this study, patients with MF grades 1–3 were classified as the MM with SMF group, while those with MF grade 0 formed the MM without SMF group.
The induction therapy regimens used in all patients included VD (Bortezomib + Dexamethasone), RD (Lenalidomide + Dexamethasone), VRD (Bortezomib + Lenalidomide + Dexamethasone), VDT (Bortezomib + Thalidomide + Dexamethasone), PAD (Bortezomib + Doxorubicin + Dexamethasone), and VCD (Bortezomib + Cyclophosphamide + Dexamethasone).
Treatment efficacy was evaluated based on the International Myeloma Working Group (IMWG) standard, calculating the overall response rate (ORR) as the sum of complete response (CR), very good partial response (VGPR), and partial response (PR). Overall survival (OS) was defined as the time from treatment initiation to the last follow-up or death, while progression-free survival (PFS) represented the duration from treatment initiation to disease progression, relapse, or death. Patient follow-up was conducted by reviewing medical records and conducting telephone interviews until February 28, 2021.
Statistical analysis
Due to the noninterventional nature of this study, missing values were expected, and no statistical imputations were conducted. Statistical analysis was conducted using SPSS 22.0 software (IBM Corporation, Armonl, NY, USA). Count data were presented as the number of cases and percentages, and group comparisons were performed using the chi-square test or Fisher’s exact test. Receiver operating characteristic (ROC) curves were plotted and cut-off values were calculated. Survival analysis was performed using the Kaplan–Meier method, and the Log-rank test was employed for univariate analysis. Multivariate analysis was conducted using the Cox regression model. A
Results
Clinical and molecular characteristics
Among the 208 newly diagnosed multiple myeloma (NDMM) patients, there were 125 males and 83 females, with a median age of 62 years old (range: 37–83). The distribution of MF grades was as follows: MF-0 (23.6%), MF-1 (44.2%), MF-2 (30.3%), and MF-3 (1.9%) . Extramedullary infiltration was observed in 34 patients (16.3%). Eighteen cases underwent autologous hematopoietic stem cell transplantation.
Based on ROC curve analysis, the NLR demonstrated a critical threshold of 2.39, with an AUC (area under curve) of 0.626 (
Compared to the non-MM-SMF group, the MM-SMF group exhibited a significantly higher incidence of anemia (52.2% vs 34.7%,
Comparison of clinical chacteristics in MM-SMF (cases, %).
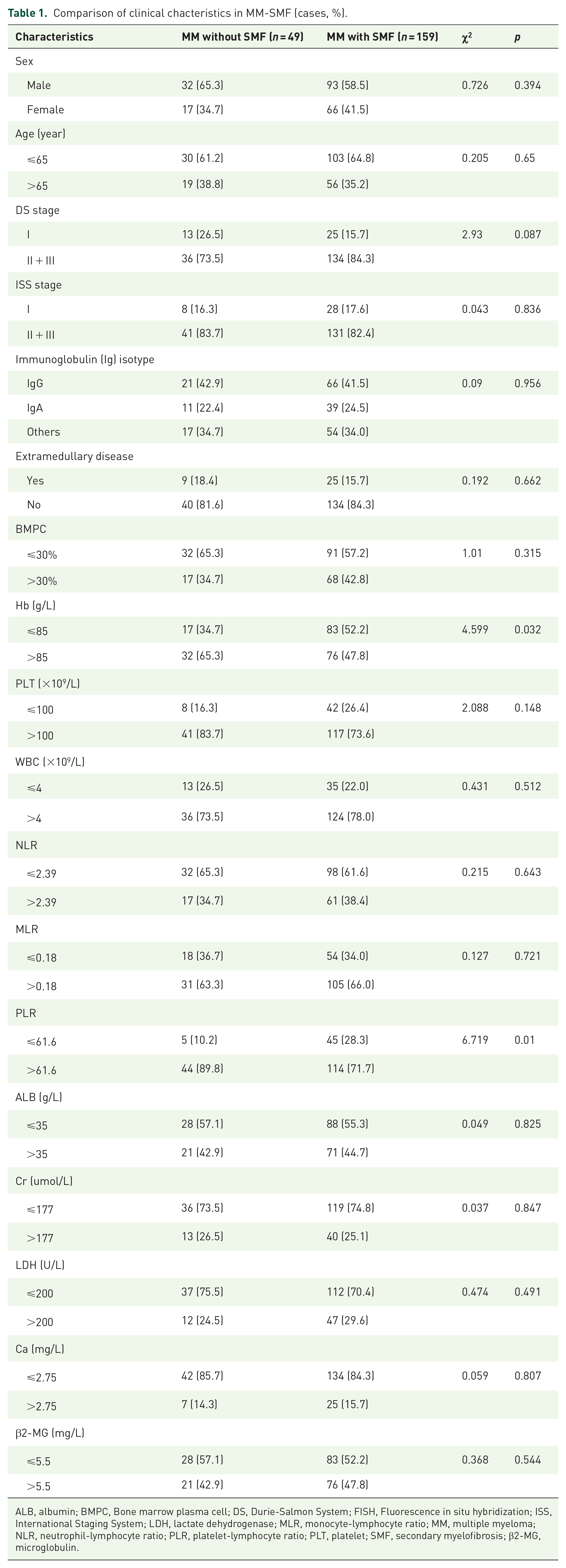
ALB, albumin; BMPC, Bone marrow plasma cell; DS, Durie-Salmon System; FISH, Fluorescence in situ hybridization; ISS, International Staging System; LDH, lactate dehydrogenase; MLR, monocyte-lymphocyte ratio; MM, multiple myeloma; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PLT, platelet; SMF, secondary myelofibrosis; β2-MG, microglobulin.
The detection rates of abnormal immunophenotypic expression in the MM-SMF group included CD56+ (51.4%) and CD117+ (21.2%). The detection rates for IgH rearrangement, P53 deletion, RB-1 deletion, and 1q amplification in the MM-SMF group were 56.8%, 9.1%, 38.6%, and 60.2%, respectively. Compared to the non-MM-SMF group, the proportion of CD56-negative cells was significantly higher in the MM-SMF group (
Comparison of immune typing and cytogenetics in MM-SMF (cases, %).

MM, multiple myeloma; SMF, secondary myelofibrosis.
Comparison of degree of myeloproliferation and efficacy
Comparing the degree of myeloproliferation between the non-MM-SMF group and the MM-SMF group, the non-MM-SMF group demonstrated significantly lower myeloproliferation than the MM-SMF group (32.7% vs 10.1%,
Comparison of the degree of myeloproliferation in MM-SMF (cases, %).

MM, multiple myeloma; SMF, secondary myelofibrosis.
In terms of therapeutic efficacy after induction chemotherapy, the analysis included 208 patients. The ORR in the non-MM-SMF group was 81.6% (40/49 cases), while the ORR in the MM-SMF group was 66% (105/159 cases). The non-MM-SMF group displayed superior therapeutic efficacy, and this difference was statistically significant (
Comparison of curative effects in MM-SMF (cases, %).

MM, multiple myeloma; SMF, secondary myelofibrosis.
Prognostic analysis
Univariate analysis revealed that MM patients with IgA subtype, extramedullary lesions, MF stages 1–3, CD56−, P53+, 1q amplification, complex karyotypes, Hb ⩽ 85 g/L, PLT ⩽ 100 × 109/L, NLR > 2.39, MLR ⩽ 0.18, PLR ⩽ 61.6, and Ca > 2.75 mmol/L all exhibited significantly shorter PFS (
Multivariate analysis showed that NLR > 2.39 and MLR ⩽ 0.18 were independent adverse prognostic factors for PFS, while P53+, NLR > 2.39, and MLR ⩽ 0.18 were independent adverse prognostic factors for OS (Supplemental Table S2).
The effect of MF on the prognosis and survival
The survival analysis demonstrated that the median PFS for patients with MF-0, MF-1, MF-2, and MF-3 was 46, 25, 19, and 9 months, respectively (

Prognosis and survival curves in patients with MF. (a) Progression-free survival between grades of MF. (b) Overall survival between grades of MF. (c) Progression-free survival between patients with myeloma with or without myelofibrosis. (d) Overall survival between patients with myeloma with or without myelofibrosis.
To investigate the impact of immune-inflammatory markers on prognosis in MM-SMF patients, several indicators including NLR, MLR, PLR, C-reactive protein (CRP), CD56, and CD117 were analyzed. The results revealed that in the MM-SMF group, NLR > 2.39, MLR ⩽ 0.18, PLR ⩽ 61.6 all significantly shortened both PFS and OS (

Effect of inflammatory markers on patients with MM-SMF. (A) Progression-free survival of NLR among MM-SMF patients. (b) Overall survival of NLR among MM-SMF patients. (c) Progression-free survival of MLR among patients with MM-SMF. (d) Overall survival of MLR between MM-SMF patients. (e) Progression-free survival of PLR among patients with MM-SMF. (f) Overall survival of PLR among MM-SMF patients.
Treatment of MM with MF
To investigate the impact of bortezomib or lenalidomide-based therapy on the prognosis of MM-SMF patients, survival analysis was conducted. In MM-SMF patients, bortezomib-based treatment did not significantly affect prognosis (median PFS: 21 months vs 29 months,

Effects of different treatment modalities in patients with MM-SMF. (a) Progression-free survival with bortezomib in patients with MM-SMF. (b) Overall survival of bortezomib in patients with MM-SMF. (c) Progression-free survival of lenalidomide in patients with MM-SMF. (d) Overall survival of lenalidomide in patients with MM-SMF.
Discussion
The clonal proliferation of plasma cells in MM leads to impaired production of healthy blood cells, resulting in anemia. Additionally, the growth of abnormal plasma cells in the bone marrow can cause osteolytic lesions, frequent fractures, and elevated blood calcium levels due to bone breakdown. The accumulation of the M protein produced by malignant plasma cells can also impair kidney function. 13 Immune dysfunction in MM patients increases their susceptibility to infections. While the introduction of immunomodulators, proteasome inhibitors, and CD38 monoclonal antibodies has slowed disease progression and extended survival, most patients eventually succumb to the disease, and some may experience treatment-related complications.
Considering the high frequency of MM-SMF and the escalating global prevalence of MM, it is crucial to emphasize the significance of addressing MM-SMF in clinical practice.10,14 The clinical manifestations of SMF predominantly rely on the underlying primary disease. Typically, the degree of MF observed in SMF is lower compared to PMF, with the majority of cases classified as MF-2 grade, while only a small proportion progress to MF-3 grade.
15
Our study discovered that 77.6% of NDMM patients had MF, primarily at the MF-1 grade. Myeloproliferation was more active in the MM-SMF group compared to the non-MM-SMF group. Subramanian et al.
16
revealed that the median survival time for MM-SMF is only 11 months. Additionally, the research findings indicate that fibrous tissue hyperplasia has been observed in the bone marrow of MM patients, with varying degrees of proliferation, and some patients experienced a decrease in MF after treatment.
17
The two-year survival rate of MM-SMF patients is significantly lower than that of non-MM-SMF patients (75% vs 95%). Another study found that MF was present in 48.2% of NDMM patients, with shorter median PFS and OS in patients with MF compared to those without MF (21.1 and 45.1 months vs 30.2 and 61.2 months).
18
In our study, we discovered that anemia incidence was significantly higher in the MM-SMF group, along with higher proportions of CD56−, complex karyotypes, and 1q amplification. The non-MM-SMF group exhibited better therapeutic outcomes. As the degree of MF increased, median PFS significantly decreased (
Inflammation-related responses play a critical role in the development of MF. 19 Tumor-associated macrophages support angiogenesis and mediate immunosuppression to protect tumor cells from apoptosis. Reduced lymphocyte counts lead to decreased immunity.20,21 MLR has been associated with various malignancies, including lymphoma, lung cancer, and nasopharyngeal carcinoma.22,23 Increased NLR indicates an enhanced inflammatory response and reduced body immunity, contributing to tumor occurrence and progression.24,25 Several studies have demonstrated the significance of NLR, MLR, and PLR in the prognosis of MM.26–28 Platelets promote tumor cell growth and metastasis by secreting growth factors and interacting with tumor cells.29,30 In the context of MF, higher leukocytes and lower platelet levels are considered poor prognostic factors.31,32 A study on NLR and PLR in MF showed that high NLR and low PLR were independent poor prognostic factors in MF patients. 33 The present study found that high NLR, low MLR, and low PLR significantly shortened median PFS and OS in MM-SMF patients. In studies investigating the levels of CRP in patients with PMF and polycythemia vera/primary thrombocythemia with fibrosis, it has been observed that fibrotic patients exhibit elevated levels of high-sensitivity CRP. 34 In this study, it was observed that MM-SMF patients with high NLR, low MLR, and low PLR exhibited significantly shortened median PFS and OS. While CRP levels did not show a significant difference among MM-SMF patients, it is worth noting that inflammation plays a crucial role in MF development. Inflammatory markers may reflect bone marrow proliferation itself rather than the degree of inflammation, offering a new direction for research on the pathogenesis and treatment of MF.
Bortezomib, a proteasome inhibitor, has been shown to reduce TGF-β1, bone sclerosis, and cytokines in a mouse model of MF, resulting in increased survival. 35 However, clinical efficacy of bortezomib in MF has been lacking, as demonstrated in phase I and II trials, where significant efficacy was not observed.36,37 Consistently, this study also found no significant improvement in the poor prognosis of MM-SMF patients with bortezomib-based therapy, underscoring the need for further research in this area.
Immunomodulatory agents have been shown to effectively inhibit the activity of NF-kB, a key transcription factor involved in promoting inflammation and apoptosis. In the context of MF, these agents play a crucial role in suppressing circulating levels of pro-inflammatory and apoptotic cytokines such as IL-2R, IL-6, IL-10, IL-8, transforming growth factor-β (TGF-β), and tumor necrosis factor-α (TNF-α). 38 Several clinical trials have reported encouraging outcomes with lenalidomide-based therapy, either as a monotherapy or in combination with short-term steroids, resulting in reductions in MF.39–41 Our study further supports the efficacy of lenalidomide-based treatment, revealing a significant extension in the median survival time of patients with MF. However, it is important to acknowledge the limited number of existing studies on lenalidomide’s role in MF; therefore, necessitating larger-scale multicenter investigations to elucidate its mechanism and potential benefits in MF.
Ruxolitinib, an oral JAK1/JAK2 inhibitor, is used to treat adults with intermediate or high-risk MF, including primary MF, postpolycythemia vera MF, and postessential thrombocythemia MF. Ruxolitinib has been shown to not only improve splenomegaly and the burdensome symptoms associated with MF but also increase OS.42–44 In recent years, the JAK family has been shown to play a role in the pathogenesis of MM. Cytokines in the bone marrow of MM patients have been shown to activate the JAK/STAT signaling pathway in tumor cells, promoting tumor growth, survival, and drug resistance. 45 JAK kinases play a crucial role in transmitting signals from cytokine and growth factor receptors to the nucleus. 46 Ruxolitinib combined with lenalidomide and dexamethasone has been shown to reduce the proliferation of MM cell lines U266 and RPMI8226, as well as primary tumor cells from MM patients. This inhibitory effect is greater when these drugs are used in combination compared to single-agent therapy. 47 A phase I trial using ruxolitinib combined with lenalidomide and methylprednisolone in relapsed/refractory multiple myeloma (RRMM) patients who had been treated with lenalidomide/steroids and proteasome inhibitors found that the JAK inhibitor ruxolitinib can overcome resistance to lenalidomide and steroids in RRMM patients. 48 In our future treatment processes, we can use ruxolitinib based on the patient’s condition, observe the efficacy and adverse reactions, and explore treatment regimens that are more beneficial for MM patients.
In summary, the complex nature of MM calls for a deeper understanding of prognostic factors and individualized treatment approaches. MF’s role in disrupting the bone marrow microenvironment adds another layer of complexity to the disease. MF is strongly associated with an unfavorable prognosis in NDMM patients and serves as a valuable prognostic indicator for predicting survival outcomes. The presence of MF correlates with PLR, while NLR, MLR, and PLR all serve as poor prognostic factors in NDMM patients with MF. Despite advancements in novel therapeutics, bortezomib-based therapy fails to significantly improve the dismal prognosis observed in MF patients. Conversely, lenalidomide-based therapy shows promise in extending median survival time for MF patients. As research into the JAK1/JAK2 inhibitor ruxolitinib deepens, patients with MF and MM both benefit, making it worthwhile to further investigate the treatment effects on SMF patients. Nonetheless, further exploration of treatment options is imperative to enhance the prognosis of MM patients with MF.
Supplemental Material
sj-png-1-tah-10.1177_20406207241292453 – Supplemental material for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Supplemental material, sj-png-1-tah-10.1177_20406207241292453 for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study by Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang and Xianqi Feng in Therapeutic Advances in Hematology
Supplemental Material
sj-png-2-tah-10.1177_20406207241292453 – Supplemental material for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Supplemental material, sj-png-2-tah-10.1177_20406207241292453 for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study by Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang and Xianqi Feng in Therapeutic Advances in Hematology
Supplemental Material
sj-xlsx-3-tah-10.1177_20406207241292453 – Supplemental material for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Supplemental material, sj-xlsx-3-tah-10.1177_20406207241292453 for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study by Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang and Xianqi Feng in Therapeutic Advances in Hematology
Supplemental Material
sj-xlsx-4-tah-10.1177_20406207241292453 – Supplemental material for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Supplemental material, sj-xlsx-4-tah-10.1177_20406207241292453 for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study by Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang and Xianqi Feng in Therapeutic Advances in Hematology
Supplemental Material
sj-xlsx-5-tah-10.1177_20406207241292453 – Supplemental material for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Supplemental material, sj-xlsx-5-tah-10.1177_20406207241292453 for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study by Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang and Xianqi Feng in Therapeutic Advances in Hematology
Supplemental Material
sj-xlsx-6-tah-10.1177_20406207241292453 – Supplemental material for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study
Supplemental material, sj-xlsx-6-tah-10.1177_20406207241292453 for Clinical features and treatment of newly diagnosed multiple myeloma with secondary myelofibrosis: a retrospective study by Han Xu, Yujie Xu, Mengying Wang, Chunxia Mao, Junxia Huang, Tianlan Li, Yan Gao, Shanshan Liu, Jingjing Zhou, Yi Zhang and Xianqi Feng in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
