Abstract
Background:
Essential thrombocythemia (ET) is a Philadelphia chromosome-negative myeloproliferative neoplasm that is characterized by thrombocytosis and an elevated risk of thrombosis and hemorrhage. We aimed to ascertain the demographics, treatment patterns, and risk of recurrent thrombosis among patients with ET in Taiwan.
Objectives:
To investigate the patient characteristics, proportion of treatment, age-specific treatment pattern, average daily dose, and risk of recurrence of thrombosis in patients with ET.
Design:
A population-based cross-sectional study was conducted between January 1, 2020 and December 31, 2021.
Methods:
This study utilized data from the National Health Insurance Research Database in Taiwan. Patients with ET were identified based on ICD-10-CM codes, and their demographic, clinical, and treatment data were analyzed. Descriptive and statistical analyses were employed to compare patient characteristics, treatment modalities, and recurrence of thrombotic events.
Results:
Among the 4503 patients with ET (prevalence: 19.3 cases per 100,000 population), the mean age was 58.1 years, with a female predominance (60.3%). Thrombosis occurred in 3.8% of patients during the study period, with a recurrence rate of 30.5% in patients with a history of thrombosis. Hydroxyurea (46.8%) and anagrelide (30.2%) were the most common treatments, with younger patients receiving higher therapeutic doses. Despite cytoreductive therapy, younger patients demonstrated higher recurrence rates of thrombosis. The cumulative incidences of secondary myelofibrosis and acute myeloid leukemia were 2.6% and 0.6%, respectively, over 2 years.
Conclusion:
This study highlights the burden of ET in Taiwan and reveals significant unmet needs in thrombosis prevention, particularly among younger patients. Real-world treatment patterns diverge from recommendations in guidelines, and this finding emphasizes the need for individualized therapeutic strategies to optimize patient outcomes. Further research, such as a longitudinal study, is warranted to investigate the risk factors of thrombosis, disease progression, and mortality in this cohort.
Plain language summary
Essential thrombocythemia (ET) is a rare blood disorder that causes the body to produce too many platelets, increasing the risk of blood clots (thrombosis) and bleeding. If left untreated, ET can also progress to more serious conditions, such as myelofibrosis (a scarring of the bone marrow) or acute leukemia. This study aimed to examine the characteristics, treatment patterns, and risks associated with ET in Taiwan. Using Taiwan’s National Health Insurance Research Database, which covers nearly the entire population, we analyzed data from 4,503 ET patients identified between 2020 and 2021. We examined their age, gender, treatment history, and risk of thrombosis. Key Findings: - The prevalence of ET in Taiwan was 19.3 cases per 100,000 people. - The average age of diagnosis was 58.1 years, and women (60.3%) were more affected than men. - Around 3.8% of ET patients had blood clots during the study period. Among those who had experienced thrombosis before, 30.5% had a second clot, despite receiving treatment. - The most commonly used medications were hydroxyurea (46.8%) and anagrelide (30.2%). - Younger patients were more likely to receive higher doses of hydroxyurea, yet they still had a high recurrence rate of thrombosis. - Over two years, 2.6% of patients developed myelofibrosis, and 0.6% progressed to acute leukemia. Conclusions: This study highlights the unmet needs in ET management, particularly in preventing blood clots among younger patients. Despite receiving recommended treatments, many patients continued to experience thrombosis, suggesting a need for more personalized treatment approaches. Additionally, treatment patterns observed in real-world practice do not always align with medical guidelines, emphasizing the importance of further research into disease progression, thrombosis risk factors, and long-term patient outcomes.
Keywords
Introduction
Essential thrombocythemia (ET) is a Philadelphia chromosome-negative myeloproliferative neoplasms (MPNs), along with polycythemia vera (PV) and primary myelofibrosis (MF). ET is frequently associated with one of three primary phenotype-defining mutations as follows: Janus kinase 2 (JAK2), calreticulin (CALR), or thrombopoietin receptor (MPL). 1 Clinically, ET is characterized by persistent thrombocytosis and, less frequently, leukocytosis. Common symptoms include fatigue, headache, and pruritus. 1 The major complications of ET are thrombotic events, followed by hemorrhagic events. 2 A small subset of patients with ET may progress to more severe conditions, such as secondary MF and acute myeloid leukemia (AML). 1
Based on the revised International Prognostic Score for ET (IPSET) model, patients with ET are classified into four risk groups: very low-risk, low-risk, intermediate-risk, and high-risk. 3 These classifications consider age, the differential presence of one of the phenotype-defining mutations, and history of thrombosis. For instance, very-low-risk patients are defined as those younger than 60 years, without CALR mutations, and without a history of thrombosis. In contrast, high-risk patients include those with a history of thrombosis, or those older than 60 years who harbor the JAK2V617F mutation. Treatment strategies differ by risk category, and range from observation to the administration of low-dose aspirin and/or medications, such as hydroxyurea (HU) or anagrelide (ANA). Interferons are seldomly used for the treatment of ET in Taiwan.
Despite clinical guidelines that provide a clear framework for ET management, the real-world data on ET patients in Taiwan remain scarce. Thus, there is a need for comprehensive data on key aspects of clinical care, such as the treatment patterns, the proportion of patients receiving various therapies, and the average daily doses of medications. A better understanding of how ET is managed in routine clinical practice is crucial to identify care gaps, support local decision-making, and inform future policy or clinical interventions.
This study aimed to address the gap in the literature by providing the first overview of ET in Taiwan to describe the demographic characteristics, clinical profiles, and treatment patterns of patients with ET.
Methods
Study design
This population-based, cross-sectional study was conducted between January 1, 2020, and December 31, 2021.
Rationale
This population-based, cross-sectional study wherein the study period is restricted to the years 2020–2021 is appropriate and feasible for evaluating treatment patterns, prescribing behavior, and real-world medication utilization at a specific timepoint and enables a snapshot analysis without the confounding effects of temporal changes in treatment practices over extended periods.
Data source
The current National Health Insurance program in Taiwan is a single-payer compulsory social insurance that provides healthcare access to 99% of the population. We accessed the National Health Insurance Research Database (NHIRD), which provides de-identified data on the entire population from 2016 to 2021. Extracted data included dates of birth and death, sex, diagnostic codes (e.g., International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM); International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM)), procedure codes, and drug codes. A list of variables of interest is available in Supplemental Table A. Lists of ICD-9-CM and ICD-10-CM codes that were used to define major thrombosis and hemorrhage are available in Supplemental Tables B and C.
Setting
The data used in this study were accessed through a secure, on-site data center authorized by the National Health Insurance Administration, located at Chang Gung University. Access was restricted to authorized personnel only, and the transfer of raw data outside the data center was strictly prohibited. All analyzed outputs were subject to a rigorous review process by the data center staff before being approved for release; this ensured a high level of data security and compliance with regulatory requirements.
Case identification process
Using the ICD-10-CM code D47.3, we identified patients with ET based on data from the NHIRD between January 1, 2016, and December 31, 2021. To ensure diagnostic accuracy, patients were included if they had at least two records of the ET diagnosis code spaced at least 90 days apart, which indicated continuity of care and minimized the risk of miscoding. To further refine the cohort, we excluded patients who, during the 90-day interval between the two ET-coded visits, had any ICD-10-CM codes that corresponded to other hematological diseases, including PV, MF, myelodysplastic syndrome (MDS), AML, or chronic myeloid leukemia. We then selected patients who had at least one ET-coded visit within the cross-sectional study period from January 1, 2020, to December 31, 2021. The date of the first ET-coded visit during this period was defined as the index date. Patients were followed retrospectively from their index date until the earliest occurrence of one of the following events: diagnosis of PV, MF, MDS, AML, or death, or until the end of the study period (December 31, 2021).
Variables of interest
The study investigated several information points pertaining to the demographic and treatment patterns related to patients with ET, including the number of patients by age group, history of thrombosis, and biological sex. We examined the age in the cross-sectional period and the history of bone marrow aspirate/biopsy. Comorbidities such as type II diabetes, hypertension, hyperlipidemia, hepatitis B/C, ischemic heart disease, chronic kidney disease, and splenomegaly were included, which were confirmed by at least two outpatient records or one inpatient record in a 365-day period before the index date between 2020 and 2021. Treatment patterns during 2020–2021 were also investigated, such as the average daily dose of HU and ANA.
Epidemiology definitions
Point prevalence was calculated as the total number of ET cases identified at the end of 2021 by the total population of Taiwan on July 1, 2021. Crude mortality rate per group was calculated as the number of deaths divided by number of ET patients, and both numbers were calculated for the period between 2020 and 2021.
Statistical analysis
Continuous variables between two groups were compared using the two-sample t test or Mann–Whitney U test. Continuous variables among four groups were compared using the Kruskal–Wallis test, and pairwise comparisons were followed by the Dwass–Steel–Critchlow–Fligner test. Categorical variables were compared between groups using the chi-square t test or the Fisher exact test. Multiple logistic regression was used to assess the simultaneous effect of multiple covariates—specifically the treatment effect—on the risk of thrombosis. Initially, age, sex, treatments, and all comorbidities listed in Table 1 were included in the model. Then, the most nonsignificant covariates were removed one by one, except for age, sex, diabetes, hypertension, and hyperlipidemia, which were considered confounders and retained for adjustment. Collinearity between variables is also checked to ensure the stability of the estimates. The model outputs adjusted odds ratios (OR) with corresponding p values, reflecting the strength and direction of these associations. Model fit was assessed using the p value from the Hosmer–Lemeshow test and the area under the curve. SAS version 9.4 (SAS Inc., Cary, NC, USA) was used for analysis, and p < 0.05 was set as the threshold of statistical significance.
Characteristics of patients with essential thrombocythemia between 2020 and 2021 in Taiwan.

Risk definitions: Very low-risk: age ⩽60 years, no JAK2 mutation, and no history of thrombosis; Low-risk: age ⩽60 years, with JAK2 mutation, and no history of thrombosis; Intermediate-risk: age >60 years, no JAK2 mutation, and no history of thrombosis; High-risk: History of thrombosis at any age, or age >60 years with JAK2 mutation. p Values were derived from the chi-square test, Fisher’s exact test, unpaired t test, where applicable. Bold values indicate significant p values.
ET, essential thrombocythemia; Gp, Group; n, number; SD, standard deviation; w/, with; w/o, without.
The STROBE statement
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 4 A STROBE checklist is included as Supplemental Material D.
Results
Patient characteristics and female predominance
A total of 4503 ET patients were identified between January 1, 2020, and December 31, 2021 (Table 1). The prevalence rate was approximately 19.3 cases per 100,000 people (the denominator being 23,375,314, which was the population of Taiwan in 2021). The mean age ± standard deviation (SD) at the time of the cross-sectional study was 58.1 ± 17.7 years. There were 1790 (39.7%) male and 2713 (60.3%) female patients. The mean age of male patients was 59.5 ± 17.6 years, compared to 57.2 ± 17.7 years for female patients. A total of 2223 (49.4%) patients were older than 60 years, whereas 2280 (50.6%) patients were younger. A chi-square test of independence showed a significant association between sex and age group (p < 0.001).
Comorbidities and disease progression
Comorbidities, such as type II diabetes, hypertension, hyperlipidemia, ischemic heart disease, and chronic kidney disease, were more prevalent among patients who were older than 60 years. Among patients aged 60 years or younger, comorbidities were more common in those with a history of thrombosis. Splenomegaly was recorded in 2.1% of patients. The overall occurrence of hemorrhage between 2020 and 2021 was 29.7%, with 4.5% of these cases classified as major hemorrhage. The proportion of major hemorrhages was higher in patients with a history of thrombosis. Thrombosis occurred in 3.8% of patients during this period, with a higher proportion of thrombosis in patients with a previous history. A total of 116 patients (2.6%) progressed to secondary MF and 27 patients (0.6%) transformed into AML. A total of 246 deaths were recorded between 2020 and 2021, with both age and a history of thrombosis contributing to mortality risk.
Treatment patterns of patients with ET
Between 2020 and 2021, 582 (12.9%) patients received only anti-platelet (anti-PLT) treatment, namely aspirin and clopidogrel, 1373 (30.5%) received HU ± anti-PLT treatment, 627 (13.9%) received ANA ± anti-PLT treatment, and 734 (16.3%) received both HU and ANA ± anti-PLT treatment. A total of 1187 (26.4%) patients showed no record of active treatment, the majority of whom were ⩽60 years and had no history of thrombosis (Table 2). A high proportion of patients with a history of thrombosis received medication (80.9%). Similarly, 77.0% of patients over the age of 60 received medication. Age-specific treatment patterns (Figure 1) revealed an inverse relationship between age and the proportion of untreated patients. Younger patients tended not to receive active treatment, and the proportion of observation-only patients decreased as age increased. Up to 90% of patients under 20 years of age received no treatment, whereas only 20% of patients >79 years of age received no active treatment. More younger patients, particularly in the 30- to 39-year age group, received only aspirin or clopidogrel for thrombosis prevention, whereas a higher proportion of patients aged 40–49 years received only ANA. A direct relationship was found between age and the use of HU alone, with a less-pronounced association between age and the concurrent use of HU and ANA.
Stratification of patients with essential thrombocythemia by treatment pattern, age, and thrombosis history.

Bold values indicate significant p values.
ANA, anagrelide; HU, hydroxyurea; n, number; PLT, platelet; w/, with; w/o, without.
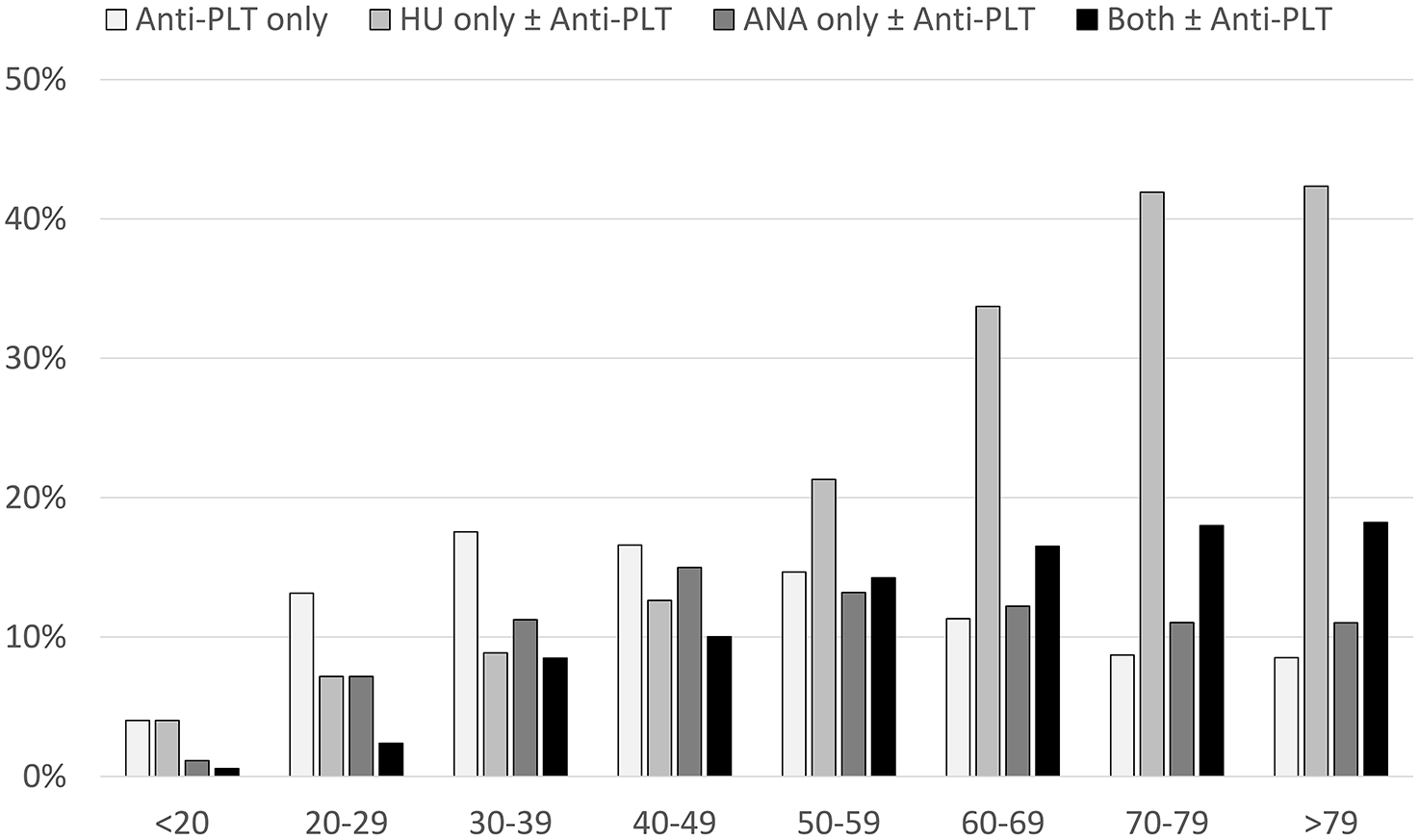
Age-specific treatment pattern in patients diagnosed with essential thrombocythemia between 2020 and 2021.
Dose patterns of HU and ANA
The average daily doses of HU and ANA were investigated for patients who had a complete 365-day follow-up after their first drug prescription between 2020 and 2021 (Table 3). Among patients on monotherapy, younger patients tended to receive higher doses of both HU and ANA. For patients without thrombosis history, those aged 60 years or younger received higher doses of HU (p = 0.008) and ANA (p = 0.001) compared to patients older than 60 years. Regarding combination therapy, there was no significant difference in HU doses between age groups; however, younger patients appeared to receive higher average daily doses of ANA, although this difference was not statistically significant.
Average daily dose of hydroxyurea and anagrelide as monotherapy or combined therapy, stratified by age and thrombosis history.

Dose of hydroxyurea presented in milligram. Dose of anagrelide presented in microgram. Bold values indicate significant p values.
ANA, anagrelide; Gp, group; HU, hydroxyurea; n, number; PLT, platelet; Q1, first quartile; Q3, third quartile; SD, standard deviation; v, versus.
Risk of recurrent thrombosis
The risk of recurrent thrombosis in patients with a history of thrombosis under different treatment regimens was investigated (Table 4). Among all patients with a history of thrombosis, 80.9% (284/351) received HU and/or ANA. Of patients ⩽60 years, 66.3% (67/101) received medication, compared to 86.8% (217/250) of patients older than 60 years. Despite receiving medication, 40.3% (27/67) of patients ⩽60 years experienced recurrent thrombosis, with an incidence rate of 37.2%, whereas 27.2% (59/217) of patients >60 years experienced recurrences, with an incidence rate of 21.2%.
Recurrence of thrombosis (person-years) between 2020 and 2021 in patients with a history of thrombosis, stratified by treatment pattern.

Bold values indicate significant p values.
ANA, anagrelide; HU, hydroxyurea; IR, incidence ratio; n, number; PLT, platelet.
Risk factors associated with recurrent thrombosis were assessed (Table 5). Multiple logistic regression models showed that, after adjusting for age, type II diabetes mellitus, hypertension, and hyperlipidemia, chronic hepatitis B infection was associated with a higher risk of recurrent thrombosis in patients >60 years (adjusted OR = 4.73; p = 0.007). The use of medications was associated with a lower risk of recurrence compared to patients who did not receive medication in patients aged 60 years or over (adjusted OR = 0.50; p = 0.013). The goodness-of-fit of was supported by the Hosmer–Lemeshow test (age <60 years: p = 0.825; age ⩾60 years: p = 0.220) and the area under the curve values (age <60 years: AUC = 0.669; age ⩾60 years: AUC = 0.683). The majority of these patients experienced recurrent arterial thrombosis, with subgroup analysis showing consistent results (data not shown). A subgroup analysis for recurrent venous thrombosis was not feasible owing to the low number of events.
Subgroup analysis of risk factors for thrombosis recurrence between 2020 and 2021 in patients with essential thrombocythemia and a history of thrombosis in Taiwan.
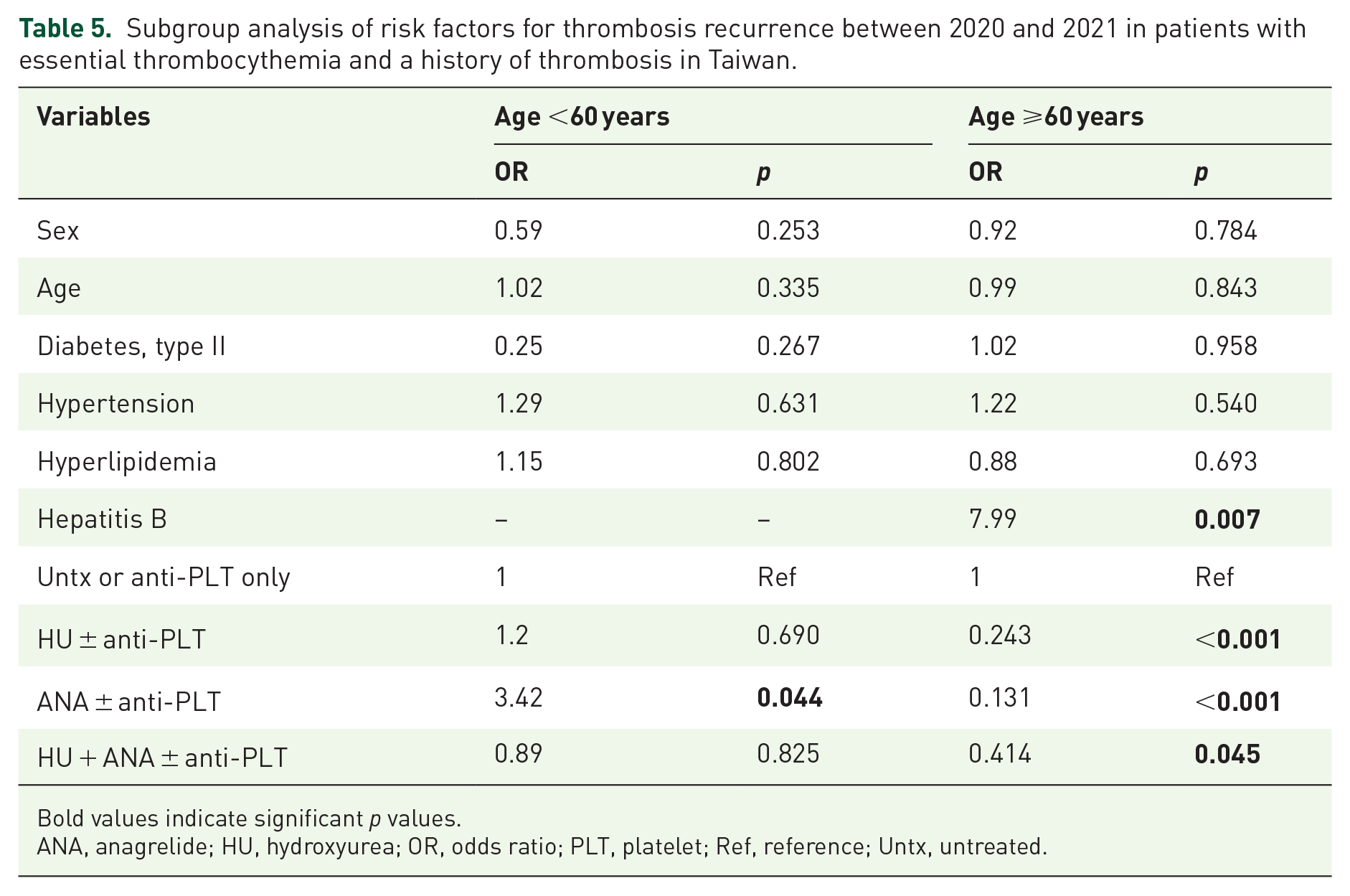
Bold values indicate significant p values.
ANA, anagrelide; HU, hydroxyurea; OR, odds ratio; PLT, platelet; Ref, reference; Untx, untreated.
Discussion
The demographics of the current cohort were compared with those reported in the literature. The prevalence rate of ET in Taiwan (19.3 cases per 100,000 people) was higher than that in South Korea (7.1–9.0 cases per 100,000 people)5,6 and Norway (8.6 cases per 100,000 people), 7 but were lower than that reported in Europe on Orphanet (24 cases per 100,000 people), 8 as well as a study in the United States (24 cases per 100,000 people), 9 and a later study in the United States (38–57 cases per 100,000 people). 10
The age of ET patients in Taiwan during the study period was similar to that of other Asian populations but younger than that of Western populations. The mean age in Taiwan (58.1 years) closely aligned to that reported in South Korea (55.4 years). 6 Our findings are also consistent with the MERGE study, a multicenter observational registry that sampled 373 ET patients from Asia and the Middle East, reporting a median age of 55 years. 11 In contrast, cohorts from Western populations reported older median ages: 62 years in Austria (n = 620), 12 and 70 years in the United States (n = 1207). 13 An indirect comparison of age at diagnosis between Western and Asian populations supports our hypothesis that patients with ET in Asia are diagnosed at a younger age and tend to be younger overall. For instance, the median age at diagnosis was 62 years in Hungary (n = 280), 14 63 years in Spain (n = 1366), 15 65 years in Norway (n = 791), 7 68 years in Sweden (n = 1284), 16 and 67 years in Denmark (n = 96), 17 whereas it was comparable to Asia in Germany (54 years, n = 140). 18
Consistent with previous reports, female patients were more prevalent in this study (60.3% female). Female predominance in ET has also been documented in studies from the United States (60%–69%),10,19–21 Spain (63%), 15 Norway (60%), 7 Denmark (60%), 22 South Korea (58%–59%),6,23 New Zealand (56%), 24 and Japan (55%). 25 The MERGE study, which included data from various countries and regions in Asia and the Middle East, also reported a female predominance (57%). 11
Our data indicated that 2% of ET patients had recorded splenomegaly, which may be due to the lack of routine sonography and limited motivation to record such findings. 26 This is supported by the MERGE study, 11 where only 30% of MPN patients were assessed for splenomegaly, and 43.9% of those were found to have splenomegaly, equating to just 13% of the entire cohort, which was likely influenced by the inclusion of MF patients in the registry. Other studies have reported palpable splenomegaly in 13% of 1000 patients with ET from Italy, 27 and 12% of 1000 patients with ET from the United States. 28 A chart review study showed that approximately 15% of patients with ET had documented splenomegaly at diagnosis, 29 whereas another chart review study with an older cohort reported a rate of 22.8%. 30
The estimated annual incidence of thrombotic events in our study (1.9%) aligns with previous reports and includes both low-risk and high-risk patients. An Italian study involving 1063 patients reported an incidence of 2.3%, 31 whereas an international study with 891 patients reported a 1.9% incidence of fatal and nonfatal thrombotic events. 32 A meta-analysis reported an annual incidence of 2.0% for arterial thrombosis and 0.7% for venous thrombosis. 33 Although these overall incidence rates appear modest, it is important to note that high-risk patients face substantially greater risks. For example, one study observed a 14.4% incidence of thrombosis among patients with ET on cytoreduction and low-dose aspirin, and 24.8% among those on cytoreduction alone. 34
The recurrence rate of thrombosis in our study 30.5% (107/351) aligns closely with previous findings. A study of 120 ET patients with a history of thrombosis reported a recurrence rate of 28.3%, 35 whereas another study of 143 ET patients observed a recurrence rate of 30%. 36 Researchers identified leukocytosis and a JAK2V617F allele burden over 50% as risk factors for recurrent thrombosis in ET patients.36,37 These findings resemble those seen in PV, where leukocytosis and elevated JAK2 allele burden also serve as prominent risk factors for thrombosis.38,39
In our study, younger patients receiving less-intensive treatment (i.e., observation and anti-PLT therapy only) had a recurrence rate of 20.6%, compared to 54.2% among those receiving active treatment. This finding suggests that Taiwanese clinicians may selectively prescribe treatment based on additional factors, such as cardiovascular risk factors, leading to a lower rate of recurrence among those under observation or antiplatelet medication. However, there remain unmet needs in preventing thrombosis in patients who require active treatment, particularly in younger patients. Among patients aged over 60 years, 36.4% of those untreated or receiving anti-PLT therapy alone experienced recurrent thrombosis, indicating a need for more aggressive management. Furthermore, 30.3% of older patients receiving active treatment still had recurrent thrombosis, underscoring the necessity for improved preventive approaches across this group.
Furthermore, 46.8% of patients received HU, and 30.2% received ANA for treatment. A nationwide database study in South Korea reported that HU was prescribed to 57.5%–63.5% of ET patients between 2007 and 2011, with a positive correlation between age and the proportion of HU prescriptions. 5 A German registry reported 49.3% of HU prescription and 23.5% of ANA. 18 Furthermore, a German study also reported 21.4% of patients who were on watch-and-wait approach, 27 which is comparable to the 26.4% observed in Taiwan.
Overall, our data suggest that HU was prescribed less frequently and ANA more frequently in Taiwan compared to Germany and the United States. Among patients receiving medication in our study, 77.1% were treated with HU, and 49.8% received ANA. A chart review study on first-line treatment in ET patients in the United States found that among those requiring cytoreduction, 93.3% (755/809) received HU, 3.7% received ANA monotherapy, and 1.4% received both HU and ANA. 26 A total of 10.1% (82/809) of patients discontinued HU treatment, with only 12 patients transitioning to ANA as second-line therapy. 26 The observational MOST study similarly found that 77% of low-risk patients and 82% of high-risk patients received HU, whereas 9% and 11%, respectively, received ANA. 13 Another chart review in Germany reported that 84.7% of ET patients received HU as first-line treatment, while 13.5% were treated with cytostatic agents, including ANA. 27
Regarding the daily dose of HU, our findings showed that the ET patients in Taiwan received a lower daily dose compared to those in the United States. A chart review of 755 ET patients reported that the most common starting doses were 1000 mg once daily and 500 mg twice daily. 26 In contrast, the median dose among 1758 ET patients in Taiwan was less than half of the dose.
Regarding the daily dose of ANA, our findings were comparable to those from Japan. A single-center study in Japan involving 35 ET patients found a median daily dose of 1.0 mg, 40 while a phase III single-arm study of 53 high-risk patients requiring second-line ANA reported a median daily dose of 1.9 mg. 41 In contrast, a multicenter study in South Korea with 70 patients reported a higher median daily dose of 3.0 mg, 42 whereas an earlier German study reported a median maintenance dose of 2.5 mg. 43
Our findings indicate that a prior history of thrombosis has a great impact on the recurrence of thrombotic events in ET patients in Taiwan. This aligns with a small study of 75 ET and 50 PV patients in China, which identified a history of thrombosis, the neutrophil-lymphocyte ratio, and TET2 mutations as stronger risk factors than age. 44 Similarly, a study of 150 ET patients in China found that age, a history of thrombosis, the neutrophil-lymphocyte ratio, and leukocyte count as predictors of future thrombotic events, with a history of thrombosis being the strongest risk factor. 45 In South Korea, a study of 108 ET patients identified a history of thrombosis as the sole major risk factor for thromboembolic events. 46 A collaborative multicenter study from Spain and Poland involving 909 ET patients further demonstrated that a history of thrombosis and cardiovascular risk factors were the most significant predictors of thrombosis. 47 Together, these studies support that a history of thrombosis is a strong predictor of future thrombosis in ET patients.
We compared real-world treatment patterns in Taiwan with the recommendations of the revised IPSET model. First, we found that most of very low-risk and low-risk patients (63.9%) were managed through observation alone or received prophylactic aspirin or clopidogrel. However, one-third of these patients still received HU and/or ANA, suggesting that treatment decisions may be influenced by symptoms or other clinical considerations. Second, we observed that some high-risk patients did not receive medications, a trend more common among younger patients (n = 34, 33.7%) than older patients (n = 33, 13.2%). Third, although the IPSET stratification effectively delineates the risks of recurrent thrombosis, our data indicate an ongoing unmet need in the prevention of recurrent thrombotic events.
Our analysis identified an interesting association between active hepatitis B infection and recurrent thrombosis in ET patients, based on the results of logistic regression. While this observation has not been previously reported in the context of ET, it should be interpreted with caution given the absence of key genetic variables such as JAK2V617F mutation status in our dataset. Several biological mechanisms have been proposed in the literature to link hepatitis B infection with a prothrombotic state, including fibrinogen dysfunction, 48 elevated von Willebrand factor, factor VIII, and plasminogen activator inhibitor, as well as reduced levels of antithrombin, protein C, and plasminogen. 49 However, existing clinical evidence regarding the association between hepatitis B and thrombosis remains inconclusive. Some studies have reported associations with carotid atherosclerosis, 50 hemorrhagic stroke, 51 and intracerebral hemorrhage, 52 whereas others found no link with certain stroke subtypes,52,53 or even suggested a potential protective effect. 54 Further studies incorporating molecular markers are warranted to better understand the potential relationship between hepatitis B infection and thrombosis risk in MPN.
The limitations of this study stem from the nature of the NHIRD data. First, the NHIRD lacks laboratory and genetic testing data, which restrict our ability to stratify patients according to the revised IPSET model or examine the associations between disease progression and hematological parameters (e.g., hematocrit, platelet count, leukocyte count, neutrophil count) or JAK2V617F allele burden. Previous studies have indicated that platelet count, 55 leukocyte count, 56 and JAK2 allele burden contribute to thrombosis risk in ET patients.57,58 Moreover, the lack of laboratory and genetic testing data limited our ability to investigate further details, such as the remission rates among patients receiving different treatments, screen for potential misdiagnosis, in our analysis. Second, a limitation of our dataset is the lack of information about ET mutation type and peripheral blood data, as we were unable to differentiate patients by mutation type owing to these data limitations. Third, the use of ICD-9-CM prior to 2016, combined with the absence of a specific code for ET, limited our ability to determine the age at diagnosis for patients in this cohort.
Conclusion
This population-based cross-sectional study provides insight into the clinicodemographic characteristics and treatment patterns among ET patients in Taiwan. Although the prevalence of ET in Taiwan was higher than that in other Asian countries, it was lower than that in Western populations. ET patients in Taiwan are diagnosed at a younger age, and there is a predominance of female patients, which is consistent with global trends. A key finding is the significant contribution of a history of thrombosis on recurrent thrombotic events, which underscores the need for targeted preventive strategies in patients with a history of thrombosis. Cytoreductive therapy decreases the risk of recurrence; however, younger patients receiving active treatment had higher rates of recurrent thrombosis, which suggests an unmet need for optimized therapeutic approaches in this subgroup. The association of a history of hepatitis B and occurrence of ET thrombotic complications is another interesting finding. The insight from this study enhances the understanding of ET in Taiwan and thereby informs clinical decision-making for improved patient outcomes.
Supplemental Material
sj-docx-2-tah-10.1177_20406207251359651 – Supplemental material for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021
Supplemental material, sj-docx-2-tah-10.1177_20406207251359651 for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021 by Ming-Sun Yu, Lennex Hsueh-Lin Yu, Shih-Hao Huang, Tsung-Hsien Tsai, Alex Jia-Hong Lin, Josef T. Prchal, Min-Chi Chen and Kuan-Der Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-3-tah-10.1177_20406207251359651 – Supplemental material for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021
Supplemental material, sj-docx-3-tah-10.1177_20406207251359651 for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021 by Ming-Sun Yu, Lennex Hsueh-Lin Yu, Shih-Hao Huang, Tsung-Hsien Tsai, Alex Jia-Hong Lin, Josef T. Prchal, Min-Chi Chen and Kuan-Der Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-4-tah-10.1177_20406207251359651 – Supplemental material for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021
Supplemental material, sj-docx-4-tah-10.1177_20406207251359651 for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021 by Ming-Sun Yu, Lennex Hsueh-Lin Yu, Shih-Hao Huang, Tsung-Hsien Tsai, Alex Jia-Hong Lin, Josef T. Prchal, Min-Chi Chen and Kuan-Der Lee in Therapeutic Advances in Hematology
Supplemental Material
sj-xlsx-1-tah-10.1177_20406207251359651 – Supplemental material for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021
Supplemental material, sj-xlsx-1-tah-10.1177_20406207251359651 for Real-world characteristics and treatment patterns in essential thrombocythemia: a population-based cross-sectional study in Taiwan between 2020 and 2021 by Ming-Sun Yu, Lennex Hsueh-Lin Yu, Shih-Hao Huang, Tsung-Hsien Tsai, Alex Jia-Hong Lin, Josef T. Prchal, Min-Chi Chen and Kuan-Der Lee in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The Services Center for Health Information, Chang Gung University, facilitated the access to the National Health Insurance Research Database. This manuscript received editing services from SAGE Author Services.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
