Abstract
Background:
Thrombocytopenia is a common complication following hematopoietic stem cell transplantation, and there is currently no standardized therapeutic approach for its management.
Objectives:
This article aims to systematically review the existing clinical treatment regimens for this condition and assess their efficacy.
Design:
This is a systematic review and meta-analysis.
Data sources and methods:
We conducted a comprehensive literature search in several databases, including PubMed, Web of Science, Cochrane Library, and Embase, up until October 30, 2024. A total of 43 datasets involving 1154 patients were included for further analysis. Therapeutic effectiveness was defined as a patient’s platelet count being above 20 × 109/L and being off platelet transfusion for more than 7 consecutive days. Drug response rates and side effect values were statistically calculated.
Results:
Our study included a total of 42 articles. The results of our study show that Decitabine and Romiplostim show relatively high effectiveness in treating thrombocytopenia after hematopoietic stem cell transplantation (HSCT), with efficacy rates of 83% (95% CI: 57%–99%) and 87% (95% CI: 69%–98%), respectively. Eltrombopag and Avatrombopag also demonstrated good efficacy, with overall utilities of 68% and 73%. Subgroup analyses revealed that Eltrombopag was particularly effective in adolescent patients compared to adults. Most of the reported side effects were well tolerated, with an overall effect value of 14% (95% CI: 11%–18%, I2 = 41.08%, p = 0.04), and no patients discontinued treatment due to adverse effects. Furthermore, our network meta-analysis suggests that Decitabine-based therapy shows significant efficacy in managing thrombocytopenia after HSCT, although more high-quality clinical trials are needed to validate these conclusions.
Conclusion:
Our meta-analysis suggests a significant efficacy of both Romiplostim and Decitabine in the treatment for thrombocytopenia after HSCT, while Eltrombopag showed significantly better efficacy in the pediatric subgroup compared to elder patients, and the side effects of the drugs are within acceptable range.
Keywords
Background
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is an effective method for treating various hematological diseases. It involves the use of donor-derived hematopoietic stem cells to rebuild the recipient’s hematopoietic and immune function, effectively eliminating malignant cloned cells from the bone marrow niche. 1 However, complications can arise at various stages of the transplantation process, and thrombocytopenia is a common occurrence during immune reconstitution after HSCT, with an incidence ranging from 20% to 37%.2,3
Thrombocytopenia after HSCT can be defined by several criteria. Prolonged isolated thrombocytopenia (PIT) after HSCT was first described by First in 1985, defined as platelets below 20 × 109/L at 60 days after HSCT, and in 1998 Bieslki defined the time to 90 days.3,4 In 2001, Professor Bruno proposed secondary failure of platelet recovery (SFPR), defined as a drop in platelet count to <20 × 109/L for 7 days after initial implantation or the need for platelet transfusion. 5 With more research on HSCT, thrombocytopenia after HSCT has been delineated in more detail, which includes delayed platelet engraftment (DPE) and poor graft function (PGF). The former is defined as sustained platelet levels below 20 × 109/L after 35 days posttransplantation, and the latter as sustained platelet levels below 20 × 109/L on the 28th day after HSCT, accompanied by absolute neutrophil count below 0.5 × 109/L and hemoglobin below 70 g/L for at least 3 consecutive days.6,7
The mechanism of thrombocytopenia after HSCT is not fully understood. Young 8 found that peripheral blood transplantation resulted in superior hematopoietic recovery compared to bone marrow transplantation. Tang 9 reported on 93 cases of thrombocytopenia in a single center and identified viral infection, occurrence of graft-versus-host disease (GVHD), low number of CD34+ cells, and elderly patients as potential risk factors. Donor selection also played a role, as Gooptu 10 observed lower platelet engraftment rates in Haplo-HSCT patients receiving reduced intensity or myeloablative conditioning compared to HLA-identical donor transplantation. Joe 11 compared peripheral blood and bone marrow transplantation and found that peripheral blood transplantation exhibited better early platelet engraftment. The overall prognosis for thrombocytopenia after HSCT is poor. It was found that patients with delayed platelet recovery after HSCT exhibited higher transplant-related mortality during long-term follow-up. 12 Persistent thrombocytopenia may lead to long-term dependence on platelet transfusions, increasing the risk of infection and healthcare costs.
In the 20th century, platelet transfusion, glucocorticoids, and immunoglobulins are moderately effective in thrombocytopenia, but there are certain side effects associated with the treatment. 13 The abovementioned treatments are also currently considered conventional therapies. recombinant human thrombopoietin (rhTPO) was the first thrombopoietic drug applied to thrombocytopenia after HSCT. Studies have shown that rhTPO can significantly shorten the time of platelet implantation, reduce the need for blood transfusion, and demonstrate a good safety profile. 14 IL-11 has also been used to promote thrombopoiesis, but with less efficacy and safety than rhTPO. 15 In the last decade, TPO receptor agonists, such as Romiplostim and Eltrombopag, have gradually become an important treatment for thrombocytopenia after HSCT. These drugs activate the relevant signaling pathways by mimicking the function of TPO to promote megakaryocyte maturation and thrombopoiesis. 16 Additionally, preliminary studies have shown that oral N-acetylcysteine (NAC) prophylaxis can effectively prevent hematopoietic dysplasia by repairing damaged bone marrow endothelial cells. 17 Han Yue’s clinical study demonstrated that subcutaneous injection of decitabine for 3 days significantly improved platelet recovery after HSCT, making it a potential adjuvant drug for the treatment of thrombocytopenia. 18
Overall, the treatment strategies for posttransplant thrombocytopenia are diverse. However, there is limited research on the efficacy and side effects of Eltrombopag and Romiplostim in this context, with few studies included in the analysis of Thrombopoietin Receptor (TPOR) agonists. 19 Therefore, this article aims to comprehensively evaluate the current clinical drugs used for treating thrombocytopenia after HSCT and compare their treatment effectiveness and tolerability.
Material and methods
Literature and search strategy
A comprehensive literature search was conducted in preapproved journals using the PubMed, EMBASE, Web of Sciences, and Cochrane databases from the initiation to the current date of October 30, 2024. Boolean logic search rules were applied to connect the following subject headings and text words. The key terms for HSCT included “HSCT,” “Hematopoietic Stem Cell Transplantation,” “Bone Marrow Transplantation,” and “Stem Cell Transplantation.” Thrombocytopenia was represented by “Thrombocytopenia” or “Thrombopenia.” Synonyms for Eltrombopag encompassed “Eltrombopag,” “Promacta,” “SB497115,” or “Revolade.” For Romiplostim, the terms used were “Romiplostim,” “AMG531,” or “Nplate.” Decitabine was represented by “Decitabine,” “5-Aza-2′-deoxycytidine,” “2′-Deoxy-5-azacytidine,” or “5 Deoxyazacytidine.” Avatrombopag was denoted by “Avatrombopag,” “Doptelet,” “AKR501,” or “YM477.” rhTPO was indicated by “rhTPO” or “Recombinant Human Thrombopoietin.” Finally, Hetrombopag was represented by “Hetrombopag,” “Herombopag,” “Hydrazones,” or “Pyrazolones.” This meta-analysis was conducted in accordance with the recommendations of the Preferred Reporting Project for Systematic Review and Meta-Analysis (PRISMA). 20 Our meta-analysis is registered in PROSPERO. The PROSPERO registry was done after the completion of the manuscript. The registration number is CRD420251000342.
Eligibility criteria
Two researchers (W.H. and Y.C.) independently performed screening of the studies based on the following criteria:
The subjects of the study were patients who developed thrombocytopenia after HSCT, including DPE, PGF, SFPR, and PIT.
The medications used for treatment include thrombopoietin receptor agonists (TPO-RAs), Decitabine, NAC, or rhTPO. Treatment effectiveness was defined as the ability of patients to maintain a platelet count higher than 20 × 109/L without requiring platelet transfusions for at least 7 consecutive days.
Detailed data were available in the articles to assess the treatment response of the drugs and the probability of adverse events.
The follow-up time of the study needs to be longer than 2 weeks.
Systematic reviews, letters, comments, nonclinical studies were excluded from the screening process.
For duplicate studies and information on duplicate cases in the same center, the most recent data study was selected.
Only English-language articles were included in this meta-analysis.
In order to address interobserver variability, we adopted a standardized process, two-person independent extraction with cross-validation during data extraction, and when abnormal values appeared, we asked a third researcher to double-check until the values were consistent.
Data extraction
The basic information of 46 articles was extracted, including the author’s last name, publication time, sample size, types of thrombocytopenia, patients’ age, drug use, drug effect time, duration, response rate, and side effects. The quality of the included studies was comprehensively assessed using the Newcastle-Ottawa Scale (NOS) quantitative system. 21 Additionally, the case series studies were evaluated using the Agency for Healthcare Research and Quality (AHRQ) criteria.
Statistical analysis
The statistical analyses were conducted using the Freeman–Tukey method to calculate odds ratio and 95% confidence intervals. All statistical analyses were performed using STATA15.1 Statistical Software (version 15.1, STATA Corporation, College Station, TX, USA). The Chi-square test was used to assess the significance of interstudy heterogeneity for qualitative statistical variables. If no significant heterogeneity was detected, a fixed-effects model was employed; otherwise, a random effects model was chosen. Subgroup analysis was conducted based on sample size, drug type, and patients’ age to explore the heterogeneity of results among the articles. 22 Meta-regression was employed to examine the influence of covariates, such as experimental or case characteristics, on the pooled effect in the meta-analysis, aiming to clarify the source of heterogeneity among studies. Publication bias was visually assessed using Begg funnel plots and statistically evaluated using the Egger regression test and Begg rank test, with a significance level of p < 0.05 defining statistical significance. 23 If publication bias was detected, fill and trim means were used to adjust for the bias, and the effect size estimates were reevaluated. Sensitivity analyses were conducted to assess the robustness of the study findings. However, for drugs with fewer than 10 studies, publication bias and sensitivity analyses were not performed. 24
Alternatively, we conducted a network meta-analysis to perform direct and indirect comparisons of multiple interventions. Network plots were created using the “networkplot” function to visually depict the interactions between different interventions. 25 The efficacy of drugs was ranked by comparing the area under the surface under the cumulative ranking curve (SUCRA). Charts were developed to quantitatively compare all interventions and determine if the differences in SUCRA for each pair reached statistical significance. To assess the presence of small-scale study bias that could contribute to publication bias in the network meta-analysis, a network funnel plot was generated and visually inspected based on the symmetry criterion. 26
Results
Study selection and characteristics
Figure 1 presents the literature selection process in a PRISMA flow chart. A total of 1436 articles were initially retrieved using the search strategy, and after removing duplicates through EndNote software, 1237 unique documents remained. Initial screening was performed by reviewing titles and abstracts, followed by a further assessment of the full text. Ultimately, 43 articles were included for analysis. One study by Wong 27 was excluded due to incomplete data integrity as it had only a two-week follow-up period. Therefore, a total of 42 articles were included in the subsequent analysis, including 1 case-control study, 4 cohort studies, and 37 cross-sectional studies. The total number of patients included in the analysis was 1154, with 7 different types of drugs investigated. There were 150 patients treated with Avatrombopag, 50 with decitabine, 604 with Eltrombopag, 91 with rhTPO, 97 with romiplostim, 30 with hetrombopag, and 132 with combination therapy. The outcome measures assessed the effectiveness of the drugs as well as their side effects. Basic characteristics of the included articles are summarized in Table 1.
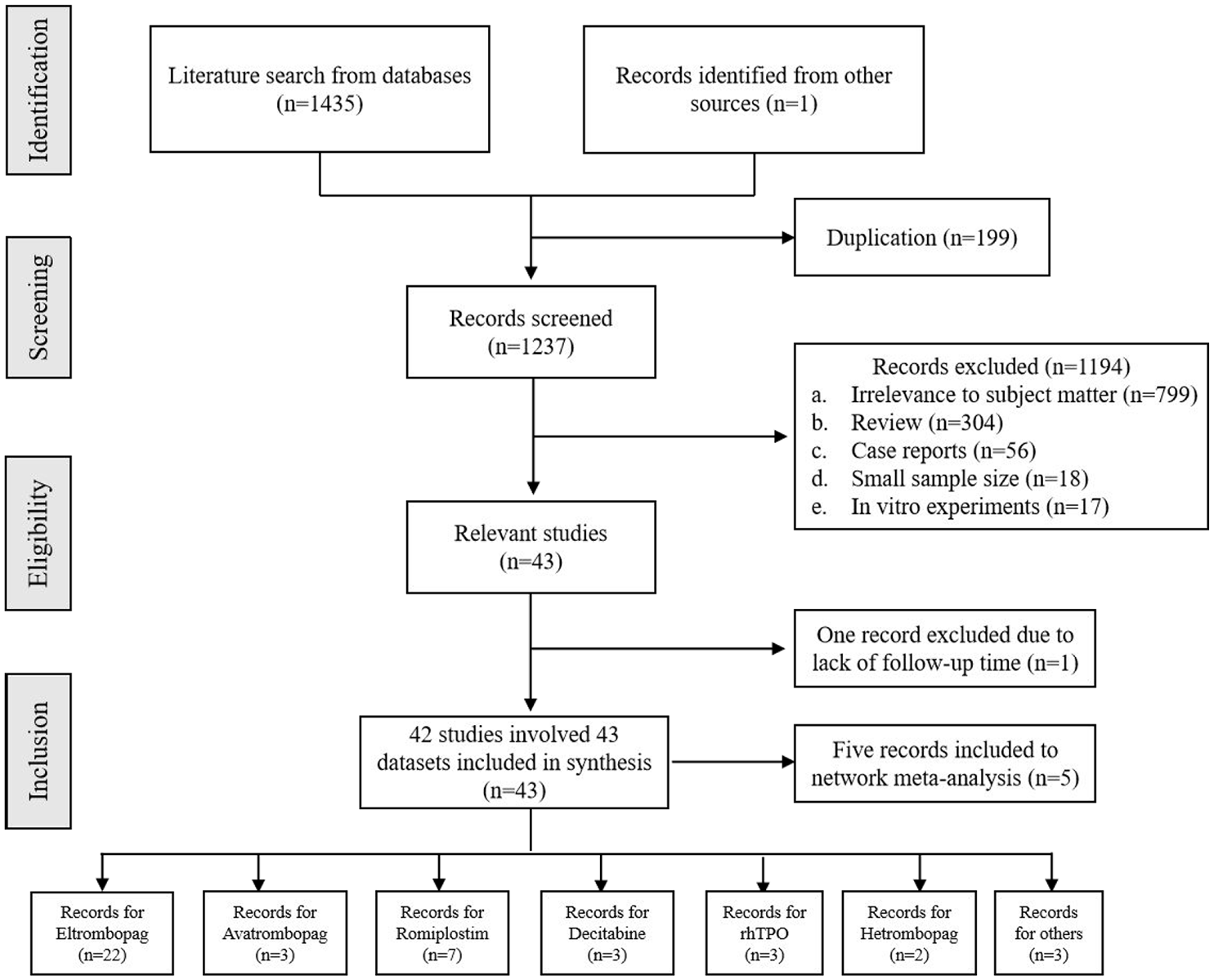
Systematic review flowchart for this meta-analysis.
The characteristics of included studies in this meta-analysis.

Adult = Patients included in the study were no less than 18 years old. Children = Patients included in the study were all under 18 years of age. Mixed = Patients included in the study included both minors and adults.
DPE, delayed platelet engraftment; MF, Myelofibrosis; MSCs, Mesenchymal Stem Cells; NA, not available; PGF, poor graft function; PIT, prolonged isolated thrombocytopenia; rhTPO, recombinant human thrombopoietin; SFPR, secondary failure of platelet recovery.
The overall NOS scores of the included studies ranged from 6 to 7 points, indicating moderate-to-good quality. For the case series studies, the AHRQ criteria were applied, and their scores exceeded 70% of the total score, signifying high-quality studies. Overall, all the included studies were of moderate or good quality (Table S1).
Overall and subgroup meta-analyses
Among the included studies, three articles reported the efficacy of rhTPO and decitabine in treating thrombocytopenia after HSCT. The overall response rate for rhTPO was 52% (95% CI: 18%–85%, I2 = 91.28%, p < 0.01, Figure S4), while for decitabine it was 83% (95% CI: 57%–99%, I2 = 69.74%, p = 0.04, Figure S3). The treatment effect of hetrombopag was evaluated in two studies, and the overall response rate was determined as 71% (95% CI: 52%–86%, I2 = 0.0%, Figure S2). Four studies evaluated Avatrombopag, with an overall effectiveness of 73% (95% CI: 65%–80%, I2 = 0%, p = 0.39, Figure S1). For Eltrombopag, 22 studies reported a response rate of 68% (95% CI: 61%–75%, I2 = 62.88%, p < 0.01, Figure 2(a)). Seven studies examined the use of romiplostim, showing an overall drug response rate 87% (95% CI: 69%–98%, I2 = 72.67%, p < 0.01, Figure 2(b)). A random effects model was applied due to the presence of heterogeneity in all these analyses. In the three studies that used combination therapy, the overall efficacy rate was 76% (95% CI: 62%–88%, I2 = 58.43%, p = 0.09, Figure S5).

Forest plots for response rate of patients with thrombocytopenia after HSCT patients to Eltrombopag (a), Romiplostim (b).
Given the large number of studies included in the Eltrombopag group, subgroup analysis was performed to explore the sources of heterogeneity. Subgroup analysis based on the average age of enrolled patients revealed that the treatment effect was better in the juvenile group compared to the elderly population, with an overall response rate of 82% (95% CI: 72%–91%). The effectiveness showed a decreasing trend with increasing age, with an effective rate of only 65% in elder patients. We investigated the factors contributing to heterogeneity in articles, including race, publication time, quality score and sample size; however, there was still some heterogeneity present (Table 2).
Stratified analysis of pooled response rate of patients with thrombocytopenia after HSCT using Eltrombopag.

HSCT, hematopoietic stem cell transplantation; NA, not available.
Publication bias analysis
We conducted a publication bias analysis for the studies included in the study on the efficacy of Eltrombopag. Publication bias was assessed based on the symmetry of the funnel plot, and quantitative judgments were made using Egger’s regression test and Begg’s rank test. Visually, the funnel plot appeared asymmetrical, and the Egger’s tests indicated the presence of publication bias with a significance level of p < 0.05 (Figure 3). We employed the trim-and-fill method for further analysis, and the results revealed no significant change in the effect size (Figure S6).

Funnel plots evaluating for publication bias for response rate in patients treated with Eltrombopag.
Sensitivity analysis
We conducted a leave-one-out sensitivity analysis to examine the influence of each individual study on the therapeutic efficacy of Eltrombopag. The results showed no statistically significant changes in our analysis (Figure 4), indicating the robustness of our findings.

Sensitivity analysis for the pooled estimates of Eltrombopag treatment.
Adverse events
Side effects were reported in 16 of the included studies, indicating that the drug was well tolerated by most patients. A meta-subgroup analysis was conducted, and the pooled rate of overall adverse effects was 15% (95% CI: 11%–20%, I2 = 41.04%, p = 0.04). The predominant adverse reactions associated with TPO-RAs included impaired hepatic function, nausea, and malaise. Relatively uncommon side effects included myelofibrosis and thrombocytosis combined with thrombosis. Additionally, in one study investigating patients after decitabine administration, the main adverse effect observed was neutropenia (Figure S7).
Network meta-analysis
A total of five eligible studies18,41,48,49,53 were included in the network meta-analysis, comparing five treatment regimens. Figure 5 provides a schematic comparison of the efficacy of different drugs in treating posttransplant thrombocytopenia, showing no significant difference in therapeutic efficacy between direct and indirect comparisons (p > 0.05). Decitabine ranked highest in terms of therapeutic efficacy among all the drugs evaluated, with a SUCRA value of 86.5%. In terms of the magnitude of the area under the curve, the rankings from highest to lowest were as follows: Decitabine-based study, rhTPO, Romiplostim, Eltrombopag, and conventional therapy (with a SUCRA value of 10.4%) (Figure 6). According to the data presented in Table 3, conventional treatment measures were significantly less effective compared to decitabine based study (OR = 0.09, 95% CI: 0.03–0.23), and rhTPO (OR = 0.16, 95% CI: 0.05–0.56) (Table 3).
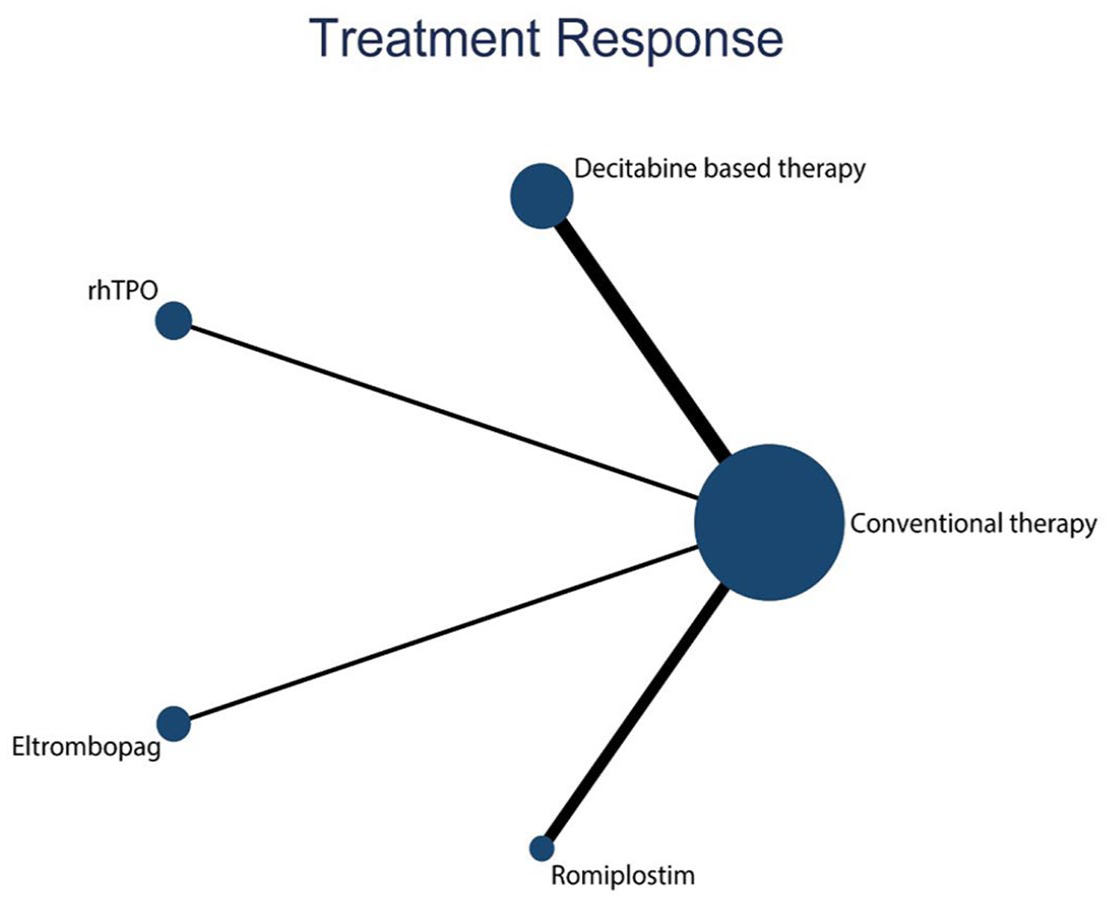
Network plot presenting the outcome evidence on the efficacy of different treatment types for posttransplant thrombocytopenia.
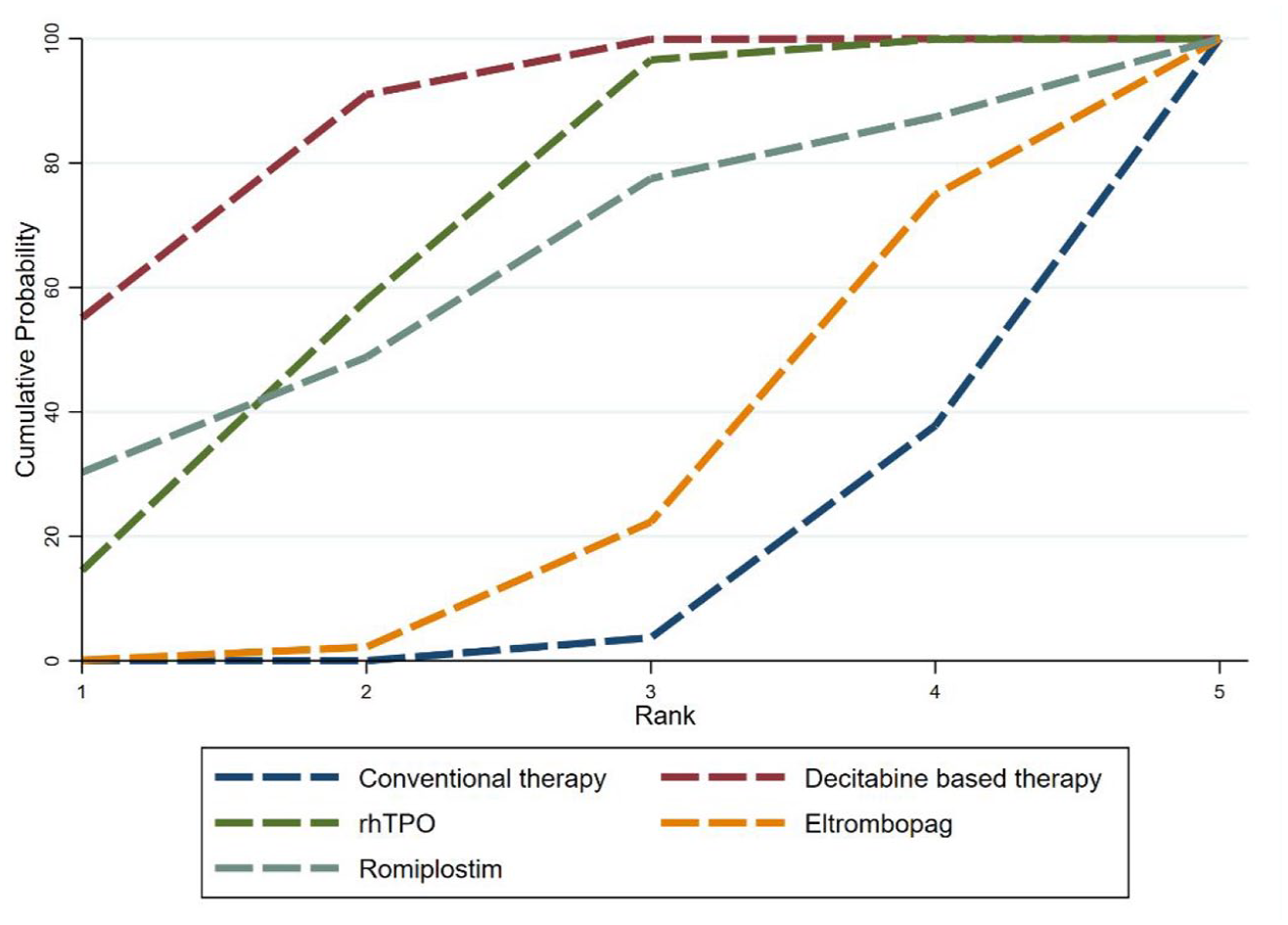
SUCRA ranking plot of drug response rates for five treatment options.
Pairwise table showing comparative efficacy of five treatments (shown as odds radios and 95% confidence intervals).

The outcome is the OR in the treatment of the defined column compared to the OR in the treatment of the defined row. For efficacy, the treatment regimen in the defined column is better when the OR value is greater than 1. To obtain the OR value for the reverse comparison, the inverse should be taken. Bold values indicate statistically significant differences in efficacy between the two drugs.
rhTPO, recombinant human thrombopoietin.
Discussion
Thrombocytopenia after HSCT has a poor prognosis. Previous studies have indicated that these patients often experience increased mortality, disappointing quality of life, and significant medical expenses. 68 Bone marrow aspiration results have revealed dysfunction in megakaryocyte maturation in these patients. Furthermore, viral infections and the development of GVHD can disrupt the bone marrow microenvironment and affect platelet production through immune-mediated mechanisms. 69 As the pathogenesis of thrombocytopenia after HSCT remains unclear, current treatments mainly focus on comprehensive approaches that involve promoting megakaryocyte differentiation and platelet release, reducing platelet destruction, and regulating the immune system. 18
It is well known that megakaryocytes are critical for platelet formation, so impaired differentiation or maturation of megakaryocytes after HSCT can affect platelet production. Bielski reported that long-term isolated thrombocytopenia in HSCT patients is due to a decrease in stem cell-differentiated megakaryocytes, and that a low megakaryocyte count in the bone marrow prior to HSCT predicts the development of posttransplant thrombocytopenia. 4 Zhang found more hypoplastic megakaryocytes (⩽8N) in patients with thrombocytopenia after HSCT, suggesting the presence of impaired megakaryocyte maturation. 70 TPO-RA activate the JAK/STAT and PI3K/Akt pathways, which are essential for megakaryocyte maturation and thrombopoiesis, by binding to and activating the TPO receptor. 71
Our study primarily investigated the current drugs used for treating thrombocytopenia after HSCT, both domestically and internationally. This includes rhTPO and TPO-RAs, such as Romiplostim, Eltrombopag, Hetrombopag and Avatrombopag, which have been widely utilized. Increasing evidence has shown that decitabine plays an important role in improving platelet recovery after HSCT.
In our study, when grouped by drug type, romiplostim showed the highest efficacy among all drugs for the treatment of thrombocytopenia after HSCT, while rhTPO appeared to be relatively ineffective. This discrepancy may be attributed to the fact that most studies included in our analysis used hematopoietic agents during the bone marrow reconstitution process, among which rhTPO was commonly used. The advantages of romiplostim in enhancing platelet production have also been highlighted in previous studies in Immune Thrombocytopenia (ITP). A phase III clinical trial in Japan demonstrated that among 34 patients with chronic ITP who received either romiplostim or placebo, 95% of those in the romiplostim group achieved the target platelet response. 72 Romiplostim is a peptide with four domains that bind with high affinity to TPO-R and have no sequence homology to endogenous TPO. It binds in the same manner as endogenous TPO and is able to displace TPO from its receptor. Romiplostim activates numerous pathways identical to those of TPO, and thus in patients with ITP, sustained improvement in platelet counts can be achieved with continued treatment. 73 Romiplostim was significantly superior to Eltrombopag in terms of overall platelet response rate, whereas in terms of sustained platelet response rate, the difference was not significant, but Romiplostim still showed some advantages. 74 There are no studies directly comparing the therapeutic efficacy of romiplostim and oral TPO-RA in thrombocytopenia after HSCT. Our meta-analysis showed better efficacy of romiplostim, which may be due to the difference in the number of included studies. Previous meta-analyses of TPO-RAs in the treatment of posttransplant thrombocytopenia have reported similar overall response rates to our findings. 19 However, our study provides additional data and incorporates larger sample sizes to enhance the reliability of the results.
Furthermore, we found that Eltrombopag may be less effective in treating thrombocytopenia in elderly posttransplant patients, while previous literature has suggested that Eltrombopag is more effective in elderly patients with immune thrombocytopenia. The difference in outcomes may be related to the altered microenvironment of bone marrow hematopoietic reconstruction after HSCT. It is possible that TPO-RA not only affects platelet production but also partially influences the stimulation and proliferation of pluripotent stem cells, which may be relatively impaired in elderly patients. 75 However, further large-scale studies and clinical trials are needed to validate the age-related efficacy of other drugs.
Additionally, our study is the first comprehensive review that specifically examines the various types of agents used in the treatment of thrombocytopenia after HSCT, which adds data on decitabine and provides additional options for clinical management.
Based on the literature search, most of the side effects of the drugs were found to be within the tolerable range, with approximately one-third of the studies reporting related side effects. The common side effects of TPO-RAs included nausea, vomiting, diarrhea, dizziness, and so on. Liver function impairment and bilirubin increase were relatively serious side effects observed, and only one study reported a severe adverse reaction in the form of thrombus formation associated with platelet elevation. 53 The main adverse effect of decitabine was neutropenia, which had a high probability of occurrence.
Only five of the included studies had data on direct and indirect comparisons of drugs; hence, these five studies were eligible for network meta-analysis. Our results suggest that decitabine-based treatment had the highest SACRA value of 86.5%, which shows that this regimen has the best therapeutic effect. In vitro study found that decitabine can promote the maturation of megakaryocytes. Platelet counts increased by 30% in mice 12 h after decitabine injection, suggesting that decitabine promotes platelet release, and the mechanism may be related to the down-regulation of the methylation level of the P14 gene. 76 Currently in clinical therapy, decitabine is generally used in people with poor outcomes after conventional therapy and TPO-RA, and we have not seen any reports in the literature on the direct use of decitabine for the initial treatment of thrombocytopenia, which may also lead to publication bias by over-publication of positive results. The conclusion of the network meta-analysis indicated that TPO-RA was ineffective in the treatment of thrombocytopenia after HSCT. The main reason was that most of the clinical reports were cross-sectional studies, and thus could not be included in the multiarm comparison of the network analysis. It is important to note that further support for these conclusions is still required through large-scale, well-controlled studies.
However, it is important to acknowledge that there is some heterogeneity present in our study data. We have identified several factors that may contribute to this heterogeneity. First, the age subgroups included individuals with a wide range of ages based on average age criteria, which could potentially impact treatment outcomes. Second, the use of rhTPO in hematopoietic reconstruction during transplantation in many studies could be a confounding factor affecting the subsequent therapeutic effects. Additionally, the limited number of clinical studies and small sample sizes for some drugs contribute to the heterogeneity. Furthermore, our included studies were published in English, so there may be language bias in our article. Importantly, the use of individualized drug doses could also influence treatment outcomes. Lastly, given that the majority of the studies included in our analysis were cross-sectional in nature, there is a potential for observer variation within each study, this factor may also contribute to the heterogeneity observed in our results.
Regarding the subgroup analysis of Eltrombopag, we observed no source of heterogeneity among pediatric patients. Further refinement of the Galbraith plot by excluding studies outside the regression line of the confidence intervals resulted in reduced interstudy heterogeneity after reanalysis. In summary, for case series studies, there is a small amount of heterogeneity mainly attributed to the number of patients and individualized treatment regimens.
In conclusion, this meta-analysis evaluated the efficacy and adverse effects of pharmaceuticals for treating thrombocytopenia after HSCT. Based on our findings, Romiplostim demonstrated relatively high effectiveness, while Eltrombopag showed significantly better efficacy in the pediatric subgroup compared to elder patients. Future clinical trials are needed to further validate our findings, and assessment of efficacy and survival prognosis of the drug in different populations is necessary.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251353007 – Supplemental material for Efficacy of therapeutic strategies on thrombocytopenia after hematopoietic stem cell transplantation—an evidence-based meta-analysis
Supplemental material, sj-docx-1-tah-10.1177_20406207251353007 for Efficacy of therapeutic strategies on thrombocytopenia after hematopoietic stem cell transplantation—an evidence-based meta-analysis by Wenxi Hua, Yunyi Chen, Jiaqian Qi, Xiaofei Song, Meng Zhou and Yue Han in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207251353007 – Supplemental material for Efficacy of therapeutic strategies on thrombocytopenia after hematopoietic stem cell transplantation—an evidence-based meta-analysis
Supplemental material, sj-docx-2-tah-10.1177_20406207251353007 for Efficacy of therapeutic strategies on thrombocytopenia after hematopoietic stem cell transplantation—an evidence-based meta-analysis by Wenxi Hua, Yunyi Chen, Jiaqian Qi, Xiaofei Song, Meng Zhou and Yue Han in Therapeutic Advances in Hematology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
