Abstract
The treatment of immune thrombocytopenia (ITP) in adults has evolved rapidly over the past decade. The second-generation thrombopoietin receptor agonists (TPO-RAs), romiplostim, eltrombopag, and avatrombopag are approved for the treatment of chronic ITP in adults. However, their use in pregnancy is labeled as category C by the United States Food and Drug Administration (FDA) due to the lack of clinical data on human subjects. ITP is a common cause of thrombocytopenia in the first and second trimester of pregnancy, which not only affects the mother but can also lead to thrombocytopenia in the neonatal thrombocytopenia secondary to maternal immune thrombocytopenia (NMITP). Corticosteroids, intravenous immunoglobulins (IVIGs) are commonly used for treating acute ITP in pregnant patients. Drugs such as rituximab, anti-D, and azathioprine that are used to treat ITP in adults, are labeled category C and seldom used in pregnant patients. Cytotoxic chemotherapy (vincristine, cyclophosphamide), danazol, and mycophenolate are contraindicated in pregnant women. In such a scenario, TPO-RAs present an attractive option to treat ITP in pregnant patients. Current evidence on the use of TPO-RAs in pregnant women with ITP is limited. In this narrative review, we will examine the preclinical and the clinical literature regarding the use of TPO-RAs in the management of ITP in pregnancy and their effect on neonates with NMITP.
Keywords
Introduction
Thrombocytopenia, defined as a platelet count less than 150,000/µL, can occur in up to 7–11% of pregnancies due to multiple reasons (Table 1).1–5 Immune thrombocytopenia (ITP), defined as a platelet count less than 100,000/µL, is a common cause of thrombocytopenia in the first and second trimester of pregnancy and accounts for 1–4% of all causes of pregnancy-associated thrombocytopenia.2,3,6–8 Despite significant advances made in the treatment options for adult ITP, the management of pregnant patients presenting with acute ITP remains a challenge. Although all pregnant patients with ITP may not require treatment, when needed, corticosteroids and intravenous immunoglobulins (IVIGs) are the most widely used medications for treating such patients.3,6,9 Steroids take 3–7 days to achieve a response and nearly 2–3 weeks to achieve the maximum effect.
10
In pregnant women, prednisone is preferred over dexamethasone due to a lower risk of transfer
Etiology of thrombocytopenia dependent upon the trimester of presentation.
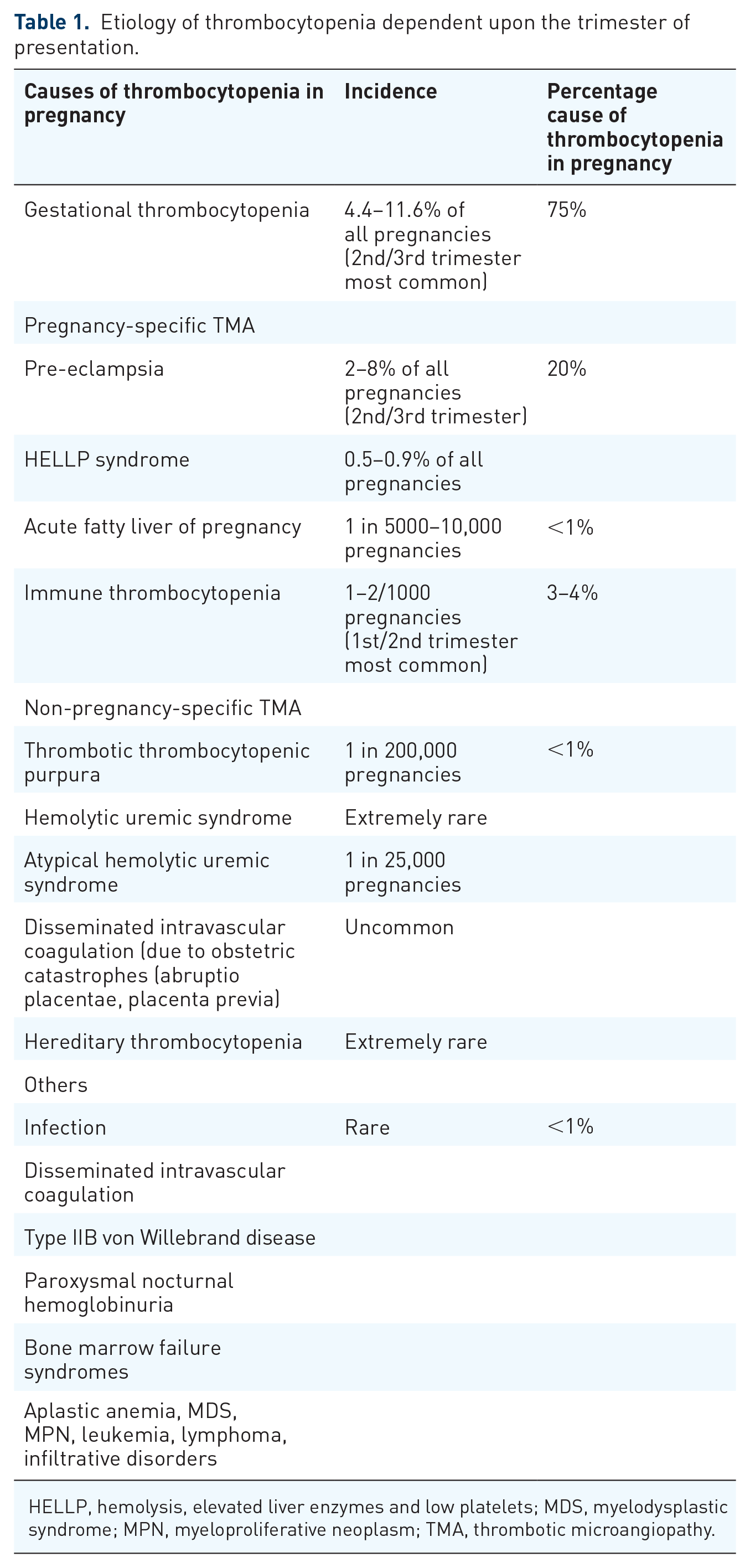
HELLP, hemolysis, elevated liver enzymes and low platelets; MDS, myelodysplastic syndrome; MPN, myeloproliferative neoplasm; TMA, thrombotic microangiopathy.
Thrombopoietin receptor agonists (TPO-RAs) act as thrombopoietin (TPO) mimetics to stimulate megakaryopoiesis in the bone marrow and yield an overall response rate of ~80%.16,17 TPO is a cytokine that regulates megakaryocyte and platelet production
TPO-RA is labeled as ‘category C’ by the US FDA, which means that animal studies have shown adverse effects on the fetus, but no adequate or well-controlled studies are present in humans. As a result, none of the trials exploring the use of TPO-RAs in adult patients with ITP include pregnant patients or lactating mothers. The recent guidelines from the American Society of Hematology and International Working Group also do not promote the use of TPO-RAs in pregnant patients.13,14 The current literature demonstrating the use of TPO-RAs in pregnancy is limited to off-label use in case reports and case series.33–42 Eltrombopag (Promacta), Avatrombopag (Doptelet), and Romiplostim (Nplate) are approved by the US FDA for adult patients with ITP.43,44 Studies of TPO-agonist antibodies [Minibodies (VB22B sc(Fv)2) and domain subclass-converted TPO agonist antibodies (MA01G4G344)] have not been reported as of yet. In this narrative review, we will review the preclinical and clinical literature associated with the use of TPO-RA in pregnancy and its impact on neonatal thrombocytopenia secondary to maternal immune thrombocytopenia (NMITP). A literature search was conducted using the PubMed electronic database from 1950 to 2018. The MeSH heading and/or text words ‘Romiplostim’, ‘Eltrombopag’, ‘Avatrombopag’, ‘Purpura’, ‘thrombocytopenia’, ‘idiopathic’, ‘thrombopoietin receptor agonists,’ ‘Pregnancy’, and ‘Pregnant’ were used. We also searched the Google Scholar database for any additional reports not listed in the PubMed database. In addition to this, the US FDA website and individual drug websites (Nplate, Promacta, Doptelet) were also searched for references. Finally, the bibliographies of all retrieved articles were examined for additional relevant citations.
TPO-RAs: preclinical data in pregnancy
Eltrombopag
Eltrombopag, developed under the name SB497115, is a non-peptide TPO mimetic belonging to the bioarylhydrazone class of compounds with an empiric formula of C25H22N4O4 and molecular weight of 445.2 D. 45 It has a metal chelation group in the center that binds to the TPO receptor causing phosphorylation and activation of the Janus kinase-2 (JAK-2), signal transducer and activator of transcription 5 (STAT5), mitogen-activated protein kinase (MAPK), and phosphoinositide-3 kinase (PI3K) pathway. 45 It is administered orally at a dose of 50–75 mg per day, achieves a peak concentration in 2–6 h, and has a half-life of 26–35 h in patients with ITP. It is excreted primarily in the feces, followed by urine. In clinical trials, eltrombopag showed a dose-dependent increase in platelet count by day 8–10 in healthy subjects. 45 Eltrombopag shows species specificity and is active only in humans and chimps. 46 Fetal toxicity in animals was seen only at doses whose equivalent doses would be toxic for human beings (Table 2). 47 However, no significant maternal–fetal toxicity was noted at doses that were equivalent to doses currently recommended in humans.47–49 Lactation studies in rats showed the continued presence of the drug in the F1 pup even after 22 h of commencing lactation, indicating that it was secreted in the milk. 47 A registry study in humans to observe the effects of eltrombopag in lactating mothers was launched but failed to recruit any patients in 5 years and was subsequently withdrawn (NCT01055600). 50 Eltrombopag did not affect male fertility in rats even at three times the human equivalent doses. 50 In addition to this, eltrombopag did not compete with TPO and hence had additive effects in combination with the native TPO. 46 Off-target toxicity in the form of cataracts was noted in young rodents when exposed to a dose that was four times the average human exposure. 51 Similar toxicity was also noted in human clinical trials; however, the patients in whom cataracts developed also had numerous other risk factors that predisposed them to develop cataracts (such as prior chronic treatments or use of steroids).48,51
Preclinical data from embryo-fetal developmental studies and lactation studies for eltrombopag, romiplostim, avatrombopag and fostamatinib.

ITP, immune thrombocytopenia; NOAEL, no observed adverse effect level; PND, postnatal day.
Romiplostim
Romiplostim, developed under the name of AMG531, is a 60-kDa recombinant protein called ‘peptibody’ with a molecular formula of C1317H2043N361O395S9.45,52 The molecule consists of a ‘peptide domain’ and an ‘Fc domain’. 45 The Fc portion of the molecule contains IgG1 heavy chains and kappa light chain constant regions bound by disulfide chains. 45 Two identical TPO-peptide sequences are linked to each arm of the Fcγ heavy chain by polyglycine linkers at the C-terminus. 45 In the initial stages of development, the 14-amino-acid long TPO peptide had a very short half-life and would have needed frequent administration to have meaningful clinical use. However, when linked with the IgG1-Fc fragment, the half-life of the drug extended dramatically (120–140 h). 45 It is administered as a subcutaneous injection at a dose of 1–10 µg/kg once every week. In patients with ITP, romiplostim achieves a peak concentration within 7–50 h (median 14 h).45,53 As with other TPO-RAs, romiplostim also showed maternal and fetal toxicity only at doses that were toxic to both humans and maternal test dams (Table 2). 53 However, more severe fetal adverse events such as stillbirth, fetal death, and significant malformations were noted at the toxic doses compared to eltrombopag. Romiplostim binds to the Fc receptor and is expected to be excreted in the milk of lactating mothers. 54 Within the western hemisphere, romiplostim (approved by the US FDA for ITP in 2008) has the longest safety record for the management of patients with ITP. In some case, it has been used for more than a decade. Although TPIAO (a TPO drug developed by 3SBio, a Chinese company), was launched in China in 2006 for chemotherapy-induced thrombocytopenia, it was not approved for ITP until 2011. Nearly 80% of patients on treatment with romiplostim either reduced or discontinued treatment and nearly a quarter permanently discontinued treatment.12,17 This finding correlated well with increased function of Tregs and Bregs; and also increased circulation of tumor growth factor-β, which suggested that TPO-RAs may play a key role in restoring immune homeostasis.12,55 Whether it is a direct effect of TPO-RAs or an immunogenic effect of increased platelet turnover is a matter of further debate.12,55–58
Avatrombopag
Avatrombopag, developed under the name of E5501 (formerly called YM477 and AKR 501), is a small molecule TPO-RA with a chemical formula of C29H34Cl2N6O3S2 and a molecular weight of 649.65 g/mol.59,60 Like eltrombopag, it is another non-peptide TPO mimetic with both
Drugs targeting Fc-FcγR interaction and spleen tyrosine kinase pathway
The Major Histocompatibility Complex (MHC)class I-like Fc receptor, neonatal Fc receptor (FcRn), is involved in a pH-dependent salvage pathway that recycles and subsequently prolongs the half-life of IgG. 62 This interaction helps to maintain humoral immunity in humans. However, in patients with autoimmune disorders, this interaction also maintains an abundant quantity of pathogenic IgG, which leads to humoral autoimmunity. 62 Drugs that block the interaction of FcRn and IgG are expected to enhance the degradation of the IgG and are being explored as a potential therapy for ITP. Many molecules, like ARGX-113 (efgartigimod), UCB7665 (rozanolixizumab), etc., are currently in clinical trials for adult patients with ITP. Fostamatinib (R788) is an oral spleen tyrosine kinase (syk) inhibitor that converts to its active metabolite R406 (by intestinal alkaline phosphatase) and blocks the downstream effect of Fc receptor activation in the mast cells and B cells. 63 It is approved as a second-line treatment for adult patients with ITP based on positive results from two randomized controlled trials. 64 Their use in pregnancy is contraindicated based on preclinical data in developmental studies, which show severe urogenital abnormalities in gravid rats and rabbits. 65 The US FDA recommends strict contraception before starting the medication, throughout treatment, and for more than one month after stopping the medication.
Current evidence on the use of TPO-RAs in pregnancy
Since all trials of TPO-RAs in adult patients with ITP have excluded pregnant women and lactating mothers, the current literature is restricted to ‘off-label’ use in pregnant women. Till recently, a ‘Nplate Pregnancy Exposure Registry’ (NCT02090088) was open, which was terminated on 6 January 2015, and the results were never published in a journal. 66 Four patients, who had romiplostim exposure any time during the pregnancy, were registered during the trial period between March 2014 and January 2015, and all of the pregnancies resulted in live births. Adverse events such as preterm delivery, adrenal insufficiency, phimosis, decreased platelet count, and intraventricular hemorrhage were reported in the registry. As the results were never published formally, it is not possible to draw any conclusions regarding a causal effect between romiplostim and these adverse events. Eltrombopag and avatrombopag are pharmacologically active only in humans and chimpanzees. Hence the preclinical data from rats and rabbits cannot be interpreted conclusively for humans.46,49,60 A registry for eltrombopag use in pregnancy, named ‘Promacta Pregnancy Registry’ (NCT01064336 – February 2010–July 2016), listed only one patient and never reported any results.
Kong
Michel
Several case reports and small series have reported off-label use of TPO-RAs in pregnant women, which present more granular data regarding the use and adverse events related to TPO-RAs in pregnancy (Table 3). The majority of these patients had ITP (
Profile of patients and adverse effects associated with the use of thrombopoietin receptor agonists in pregnant patients.
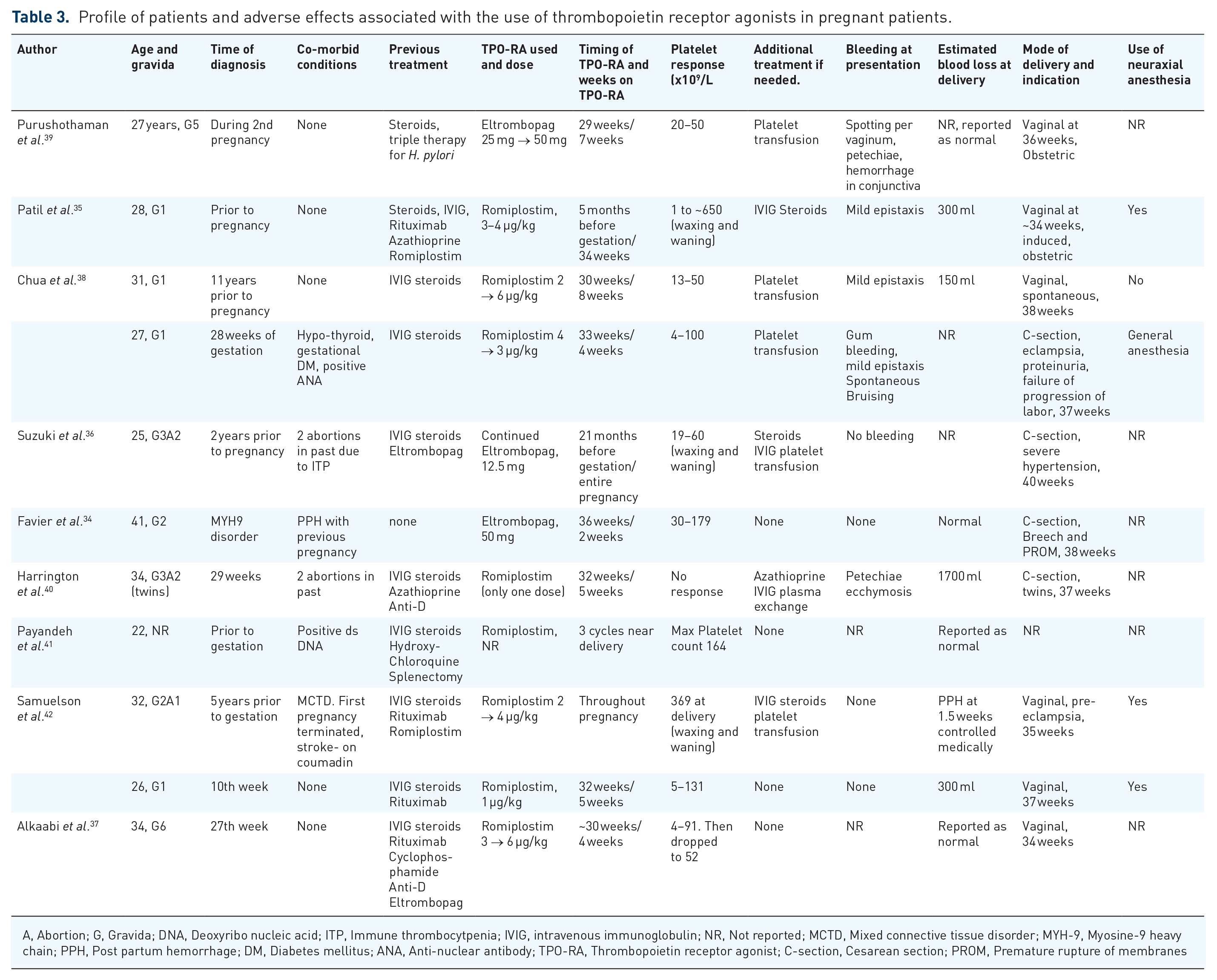
A, Abortion; G, Gravida; DNA, Deoxyribo nucleic acid; ITP, Immune thrombocytpenia; IVIG, intravenous immunoglobulin; NR, Not reported; MCTD, Mixed connective tissue disorder; MYH-9, Myosine-9 heavy chain; PPH, Post partum hemorrhage; DM, Diabetes mellitus; ANA, Anti-nuclear antibody; TPO-RA, Thrombopoietin receptor agonist; C-section, Cesarean section; PROM, Premature rupture of membranes
Neonatal thrombocytopenia associated with maternal ITP
ITP in the mother can lead to neonatal thrombocytopenia and related complications. Up to 25–50% of neonates born to mothers with ITP develop thrombocytopenia.70–72 Out of these, 5–15% are born with platelet counts less than 50 × 109/L, and 1–5% are born with severe thrombocytopenia (platelet count less than 20 × 109/L).1,71–73 A maternal history of splenectomy (done for treating ITP) or a previous pregnancy with ITP that led to thrombocytopenia in the neonate are some recognized risk factors associated with NMITP.1,72–78 The timing of the diagnosis of ITP in the mother (whether diagnosed before or after pregnancy starts) as a risk factor for NMITP remains controversial.79,80 Passive transfer of maternal antibodies across the placenta is the most widely accepted mechanism behind NMITP. However, it must be noted that up to 40% of neonates will test negative for antiplatelet antibodies.72,81 Although breastfeeding is safe for mothers with ITP, prolonged thrombocytopenia (more than 3 months long) in the neonate has been observed in such cases. The reasons remain anecdotal and are attributed to the passive transfer of antibodies in breast milk. 82 A case–control study evaluating prolonged thrombocytopenia in neonates born to mothers with ITP demonstrated the presence of IgA antibodies targeting αIIbβ3 integrin. 83 In neonates born to mothers with ITP, a cord blood sample should be drawn to determine the platelet count.1,10 If the cord blood shows thrombocytopenia, then this should be confirmed with a venous blood sample.10,72 The platelet count usually reaches a nadir by postpartum day 1–5.1,2,10,72 IVIG is the drug of choice and is used when the platelet count drops below 30 × 09/L.1,2 Donor platelets are often transfused along with IVIG to raise the platelet counts quickly.1,10,72,80 Steroids are not frequently used in NMITP to prevent neonatal sepsis. However, if used then they are administered in low doses. 84 Intracranial hemorrhage (ICH) occurs in less than 1% of neonatres with NMITP.1,85,86 All neonates with NMITP exhibiting neurological manifestations indicative of ICH must be evaluated with a head ultrasound. 1 Concurrent NMITP and fetal-neonatal alloimmune thrombocytopenia (FNAIT) must be considered in the differential if a neonate develops ICH and in those born with a platelet count less than 10 × 109/L. 1 A full review of FNAIT is done elsewhere. 87
Current evidence for the use of TPO-RAs in neonates
Kong
Data pertaining to the effects seen in neonates whose mothers were treated with thrombopoietin receptor agonists during pregnancy.

TPO-RA, Thrombopoietin receptor agonist; NMITP, Neonatal-maternal alloimmune thrombocytopenia; IVIG, Intravenous immunoglobulin; IVH, Intraventricular hemorrhage; NR, Not reported
Controversies in the effect of TPO-RA on the fetus and the newborn
The effect of TPO-RA on organogenesis in the human fetus is not clear from the current literature. TPO is known as a critical physiological regulator of hemangioblasts, which eventually differentiate into endothelial and hematopoietic cells.89,90 Excessive TPO exposure has long been speculated as a cause of limb defects in patients with hereditary thrombocythemia, an autosomal dominant disease that occurs either due to the mutation in the
The management of thrombocytopenia in the neonate aims to prevent catastrophic bleeding, particularly ICH. Platelet transfusion is optimal in the setting of acute bleeding secondary to NMITP. However, several studies have noted that ‘liberal’ transfusion of platelets prophylactically is associated with increased morbidity and mortality in neonates.97–100 In the recently concluded PlaNet-2 trial, a higher mortality and bleeding rate was observed in neonates who received platelet transfusion at a platelet count of 50 × 109/L compared to those who received it at a threshold of 25 × 109/L.
101
It is not clear whether the increased mortality is due to the higher platelet counts themselves or transfusion-related issues. Currently, there are no clinical data to support the use of TPO-RA in neonates with thrombocytopenia. In theory, the use of TPO-RA as an alternative to platelet transfusion in neonatal thrombocytopenia seems an attractive option.100,102 However, it usually takes 4–6 days of continuous administration of TPO-RA for the platelets to respond and at least 10–14 days to achieve the maximum response.45,103 Since nearly 80% of severe thrombocytopenia in the neonatal intensive care unit resolve within 14 days,104,105 the use of TPO-RA would be restricted to a minority of neonates who present with ‘persistent thrombocytopenia’ (thrombocytopenia for more than 14 days).100,102 Mahat
Strengths and limitations
The strength of this paper lies in the comprehensive review of literature pertaining to the use of TPO-RA in pregnant patients with ITP. The preclinical evidence presented here has never been explored in any manuscript and shows that although fetal toxicity was demonstrated in animal models, that is either not applicable to humans (as in the case of eltrombopag and avatrombopag) or does not lead to toxicity at human equivalent doses. However, the lack of prospective data and scarcity of information in the retrospective data is the major limitation of this review.
Conclusion
The treatment options for managing thrombocytopenia in pregnant patients with ITP are limited due to concerns for fetal toxicity. Steroids and IVIG are the most widely used drugs in the treatment of pregnant patients with ITP. Our review of the literature suggests that TPO-RAs can help raise the platelet count within 2–3 weeks in pregnant patients with ITP. In light of the existing literature, it seems that the use of TPO-RAs is safe in the second and third trimesters. However, their use during the first trimester when organogenesis is at its peak must be avoided till further evidence demonstrating fetal and maternal safety becomes available. The current literature does not suggest any untoward effect of using TPO-RAs in late pregnancy on the fetus or the neonate. However, there is no clinical evidence to suggest whether it helps in preventing thrombocytopenia in neonates with NMITP or not. There is a pressing need to conduct a prospective clinical trial or a registry study evaluating the use of TPO-RAs in pregnant patients with ITP.
Footnotes
Author contributions
NA: conception of the idea, data collection, data analysis, and interpretation, wrote the manuscript.
AM: Project leader, critical revision of the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
No financial support was received for this research, authorship, and/or publication of this article.
Availability of data and materials
The dataset supporting the conclusions of this article are included in the article.
