Abstract
Core-binding factor acute myeloid leukemia (CBF-AML) is a subtype of AML characterized by specific genetic rearrangements, including t(8;21) and inv(16), which are associated with a relatively favorable prognosis with relapse rates around 30%. The mutational profiling of CBF-AML is highly heterogeneous in pediatrics, with mutations in the tyrosine kinase pathway (including KIT, FLT3, and N/KRAS), epigenetic regulators, cohesin, and additional cytogenetic abnormalities. The identification of high-risk mutations, such as those in KIT and FLT3, underscores the need for targeted therapies and highlights the importance of high-throughput sequencing technologies, providing critical insights into the prognosis and informing treatment strategies. Integrating targeted agents with existing treatment protocols has the potential to enhance treatment efficacy and significantly improve patient outcomes. However, CBF-AML presents significant heterogeneity in both pathophysiology and clinical characteristics, with cooperating cytogenetic mutations, leading to difficulties and uncertainties in the prognosis and treatment of CBF-AML in pediatrics. Given the relapse rates and the significant impact of specific mutations on prognosis, there is a critical need for improved risk stratification and personalized treatment approaches in pediatric CBF-AML. Ongoing research and clinical trials focusing on the molecular and genetic profiling of pediatric CBF-AML will be essential for developing more effective and targeted therapies, ultimately improving patient outcomes. This review summarizes the molecular genetics profiling in CBF-AML among pediatrics, targeting its effect and interactions on prognosis and treatment to provide an overview for further research based on mutations among CBF-AML in pediatrics.
Plain language summary
Core-binding factor acute myeloid leukemia (CBF-AML) has significant heterogeneity in both pathophysiology and clinical characteristics, with cooperating cytogenetic mutations, leading to difficulties and uncertainties in the prognosis and treatment of CBF-AML in pediatrics. Given the relapse rates and the significant impact of specific mutations on prognosis, there is a critical need for improved risk stratification and personalized treatment approaches in pediatric CBF-AML. Here, we demonstrate the mutational profiling of CBF-AML is highly heterogeneous in pediatrics, with mutations in the tyrosine kinase pathway (including KIT, FLT3, and N/KRAS), epigenetic regulators, cohesin, and additional cytogenetic abnormalities. Targeting the interactions on prognosis and treatment could provide an overview for further research based on mutations among CBF-AML in pediatrics.
Background
As childhood cancer is a notable cause of morbidity and mortality, leukemia stands as the predominant form of cancer found among children between the ages of 0 and 14 in both China and globally. 1 Acute myeloid leukemia (AML), which accounts for 20% of pediatric leukemia, is characterized by compromised differentiation and maturation of hematopoietic stem progenitor cells, malignant proliferation of abnormal blood cells, and impaired apoptosis. This is coupled with a high degree of heterogeneity in mutant genes, immunophenotypes, and epigenetics.2,3 Core-binding factor (CBF) is a heterodimeric transcription factor contains four subunits, including DNA binding and nonbinding subunits, which are fundamental in the initiation of hematopoiesis in the embryo. 4 In pediatric AML, CBF-AML constitutes approximately 30% of cases and is associated with various chromosomal aberrations disrupting the α and β subunits of CBF. The primary aberrations are t(8;21)(q22;q22), resulting in the RUNX1::RUNX1T1 fusion gene, and inv(16)(p13.1q22) or t(16;16)(p13.1;q22), leading to the CBFB::MYH11 fusion gene. RUNX1::RUNX1T1 fusion gene impedes cell differentiation and enhances leukemic self-renewal, as demonstrated in both cell line and transgenic expression models by inhibiting the differentiation of myeloid cells and promoting leukemogenesis through the suppression of RUNX1-mediated hematopoietic genes.5,6 CBFB::MYH11 encodes the CBFβ::SMMHC protein, which sequesters the transcription factor RUNX1 in the cytoplasm, thereby preventing it from regulating target genes essential for myeloid differentiation.7,8 Both t(8;21) and inv(16), although classified as favorable-risk subtypes, exhibit similar relapse rates of around 30%, comparable to other types of AML.9–11 Studies from the Therapeutically Applicable Research to Generate Effective Treatments (TARGET) database, the JPLSG AML-05 trial, and a tertiary hospital in China report relapse rates of 26.5%, 29.9%, and 30.8%, respectively.12–14 These findings highlight the persistent challenge of relapse in CBF-AML, even with its generally favorable prognosis, emphasizing the need for further research to refine treatment strategies and explore the molecular factors contributing to relapse to improve long-term outcomes.
Furthermore, the genomic profiling studies reveal that mutations involving tyrosine kinase pathway, epigenetic regulator, cohesin, and cytogenetics are related to the relapse rate and prognosis. A meta-analysis revealed that pediatric CBF-AML with a KIT mutation has a 69% increased risk of relapse.15–18 Among them, mutations in tyrosine kinase pathways like KIT and RAS are much more favored with controversial prognostic values.19–22 KIT mutation is common and associated with adverse prognostic effects in event-free survival (EFS) and recurrence-free survival (RFS) in patients with t(8;21), but not in those with inv(16). Conversely, RAS mutation is commonly seen in inv(16).23,24 Target therapy focusing on these mutation sites is gaining attention. Nevertheless, the prevalence of concurrent mutations poses challenges in the treatment process.
Over the past three decades, official guidelines about myeloid neoplasms have been updated, leading to a swift progression in innovative strategies for treating CBF-AML, which has enhanced patient outcomes.25–27 However, few studies have discussed the prognosis of CBF-AML from different mutation sites, the efficacy of different treatment regimens, and the prognostic model in pediatric CBF-AML often yielding controversial conclusions. This review aims to consolidate the prognostic characteristics of pediatric CBF-AML based on different mutation sites, to provide information for upcoming novel treatment and prognostic model applications.
CBF-AML in pediatrics
Demographics
The exploration of gender differences in pediatric patients with CBF-AML reveals nuanced insights across various studies. Ben Sayed’s research indicates that 60% of the pediatric CBF-AML cohort were male, providing a demographic snapshot but not delving into gender-specific outcomes or prognostic differences. The median age of CBF-AML in children is about 9 years old with the range of 3–16 years old, highlighting the presence of this subtype in younger patients. 28
The percentage of CBF-AML in pediatric AML varies across different studies, reflecting a range of incidence rates due to geographical, ethnic, and methodological differences, with studies reporting 23%–46% among pediatric AML cases.29–32 In the research conducted by Charfi et al., it was found that 30% of pediatric patients with AML were diagnosed with CBF-AML from a monocenter study in Africa, consistent with another study by Neelum Mansoor, which reported a slightly higher frequency of 34% in India.32,33 Mao et al. found that 46% of pediatric AML patients had CBF-AML in the multiethnic region of Yunnan Province, China, highlighting a higher prevalence in specific geographical areas, potentially associated with race and ethnicity. 20
Differences in genetic mutations based on race and ethnicity have been noted in previous studies on CBF-AML, suggesting that genetic predispositions and environmental factors might influence the incidence and characteristics of CBF-AML in different populations. Specifically, Eastern Asian cohorts have a significantly higher ratio of CBF-AML, and the occurrence of inv(16) is more prevalent in Dutch, Belgian, and Swiss trials compared to Chinese and Japanese trials. 34 Another study conducted in Mexico has shown that the prevalence of CBF variants of AML was similar to that in Caucasian populations but with a significant presence of lymphoid markers expressed in the blast cells of individuals with t(8;21). 35 Furthermore, inv(16) is predominantly common in White races compared to t(8;21). 12
Ethnic disparities in pediatric leukemia relapse rates have also been observed, with younger Hispanic children displaying a notably elevated likelihood of recurrence in contrast to non-Hispanic Whites. 36 Transcriptome sequencing has also identified differences in gene expression and alternative splicing events between CBF-AML subtypes, suggesting subtle biological differences that could be influenced by ethnicity. 37 These variations accentuate the role of regional distribution, genetic diversity, and clinical heterogeneity in CBF-AML cases. 38
Clinical characteristics
CBF-AML, encompassing t(8;21) and inv(16), exhibits different clinical and molecular characteristics, influencing disease prognosis, treatment response, and risk stratification, with a summary shown in Table 1
Clinical characteristics of pediatric CBF-AML.

CBF-AML, core-binding factor acute myeloid leukemia; CNS, central nervous system; FAB, French–American–British; WBC, white blood cell.
In patients with inv(16), French–American–British (FAB) M4 is more frequent with a much higher initial white blood cell (WBC) count, while the M2 phenotype is more common in t(8;21), indicating inv(16) links to leukocytosis and extramedullary infiltrative manifestations.12,31,39 Qiu et al. also observed that chloroma was less frequently seen in individuals with inv(16) compared to those with t(8; 21). Chloromas, which are more frequently observed than in adults, can involve various anatomical sites, including the orbit, skin, lymph nodes, and central nervous system (CNS). 12
CNS involvement in CBF-AML has been documented in 3%–21% of cases, with a higher prevalence observed in specific subgroups like M4.31,40 Although CNS involvement in pediatric AML does not independently predict prognosis, it requires more aggressive CNS-directed therapy due to the increased risk of isolated CNS recurrence and associated psychological challenges, which are critical to children’s growth.38,41 Specific risk factors for CNS disease in CBF-AML include young age, elevated WBC counts, and certain morphologies such as M4.20,42
Diagnosis
The diagnosis of pediatric CBF-AML relies on a multimodal framework integrating morphology, immunophenotyping, cytogenetic analysis, and molecular profiling. Bone marrow aspiration and biopsy are essential diagnostic procedures by providing samples for morphological examination, cytochemical staining, and immunophenotyping. Morphological examination involves identifying myeloid blasts in the bone marrow or peripheral blood, with ⩾20% blasts required for a diagnosis of AML.25,27 Cytochemical stains, such as myeloperoxidase and nonspecific esterase, help differentiate myeloid from lymphoid lineage and subtype AML.
Measurable residual disease (MRD) is another key determinant in guiding diagnosis, treatment, and predicting outcomes in CBF-AML, including flow cytometry and molecular MRD monitoring, offering insights into treatment response and relapse risk. Multiparameter flow cytometry (MFC) examines multiple surface and intracellular markers simultaneously, providing a highly specific immunophenotypic profile of residual leukemia cell in bone marrow and blood. MFC MRD monitoring based on cytogenetic stratification is useful for assessing susceptibility to chemotherapy and making risk stratification more accurate and instructive. In pediatric patients, the prognostic informativeness of MFC MRD monitoring varies widely among different genetic subgroups. For CBF-AML, which are prognostically more favorable, persisting MRD-positivity alone was not associated with an increase in the risk of relapse.30,43 However, the sensitivity of MRD in predicting relapse within pediatric CBF-AML remains low, as evidenced by 66% of relapses occurring in patients without detectable MRD in peripheral blood or bone marrow. 44 Furthermore, molecular MRD by quantitative polymerase chain based (qPCR) monitoring and droplet digital PCR detecting residue fusion gene in CBF AML have shown profound use, which was considered as a complementary tests in monitoring CBF-AML MRD.45,46 The European Leukemia Net (ELN) MRD Working Party suggests a failure to reach a 3-log to 4-log reduction of fusion gene by molecular MRD between the sample at diagnosis and at the end of treatment in adult patients with CBF-AML in adults would be a relevant marker for subsequent relapse, but further investigations are required to verify whether it could be applied to children as well. 47 Furthermore, results from the AML-BFM 2004 and 2012 studies of 91 pediatric patients with CBF-AML revealed persistent positive MRD by qPCR in the majority of cases, with relapse rates of approximately 18% in both MRD-positive and MRD-negative groups. 30 This is likely to be associated with the presence of adverse genetic mutations such as KIT, RAS, masking the underlying adverse genetic mutations, complicating the prognostic landscape. 48 Notably, findings from the Japan Children’s Cancer Group (JCCG) trial emphasized that KIT exon 17 mutations have a significant prognostic impact, independent of MRD status. These mutations necessitate treatment intensification regardless of MRD levels, underscoring their priority in risk stratification under MRD-guided therapy. 49 MFC and qPCR are complementary tests in monitoring the MRD of CBF-AML. The investigation of MFC and molecular MRD in pediatric CBF-AML remains limited. Further research should target with combining use of MFC and qPCR for MRD detection in pediatric CBF-AML, providing useful clinical information and therapeutic insights.
Treatment and its prospect
For pediatric CBF-AML, there is no specific official criteria or therapeutic approach. However, general therapeutic approach with conventional therapy and targeted therapy has been shown in Figure 1. 25

General therapeutic approach of pediatric CBF-AML.
Despite its complex clinical presentation, CBF-AML generally responds well to chemotherapy, with numerous research institutes documenting complete remission (CR) rates of up to 90% in both pediatrics and adults.12,31,50 Given the unique therapeutic challenges that children present, cytarabine has become a cornerstone of the pediatric treatment regimen. It has been proven that pediatric AML patients, particularly those with t(8;21), who received higher cumulative doses of cytarabine may experience improved outcomes, suggesting the importance of optimizing cytarabine dosage in treatment protocols. When used in targeted therapies, cytarabine is beneficial for pediatric patients with inv(16), consistent with the findings of a meta-analysis of five randomized controlled trials conducted in adults, suggesting the possibility of personalized treatment strategies based on specific genetic abnormalities. 12 In terms of clinical characters and treatment, the management of CBF-AML is progressively shifting toward a precision medicine strategy, where the identification of genetic and molecular characteristics guides the choice of individualized treatments and the stratification of individuals for therapeutic protocols.
Although CBF-AML typically has a favorable prognosis, this may change under certain circumstances. Concretely, in the presence of particular gene mutations, only half of CBF-AML patients are able to sustain long-term remission without relapse. 51 The progression of AML is recognized as a complex and multifaceted process that requires a series of sequential steps for full manifestation. While chromosomal translocations involving CBF are recognized as significant events in the pathogenesis of AML, they are not adequate on their own to trigger this disease.52,53 Furthermore, the comprehensive array of genetic mutations that may co-occur with CBF translocations in the context of AML remains undefined and warrants further investigation.
Cooperating genetic mutations for CBF-AML in pediatrics
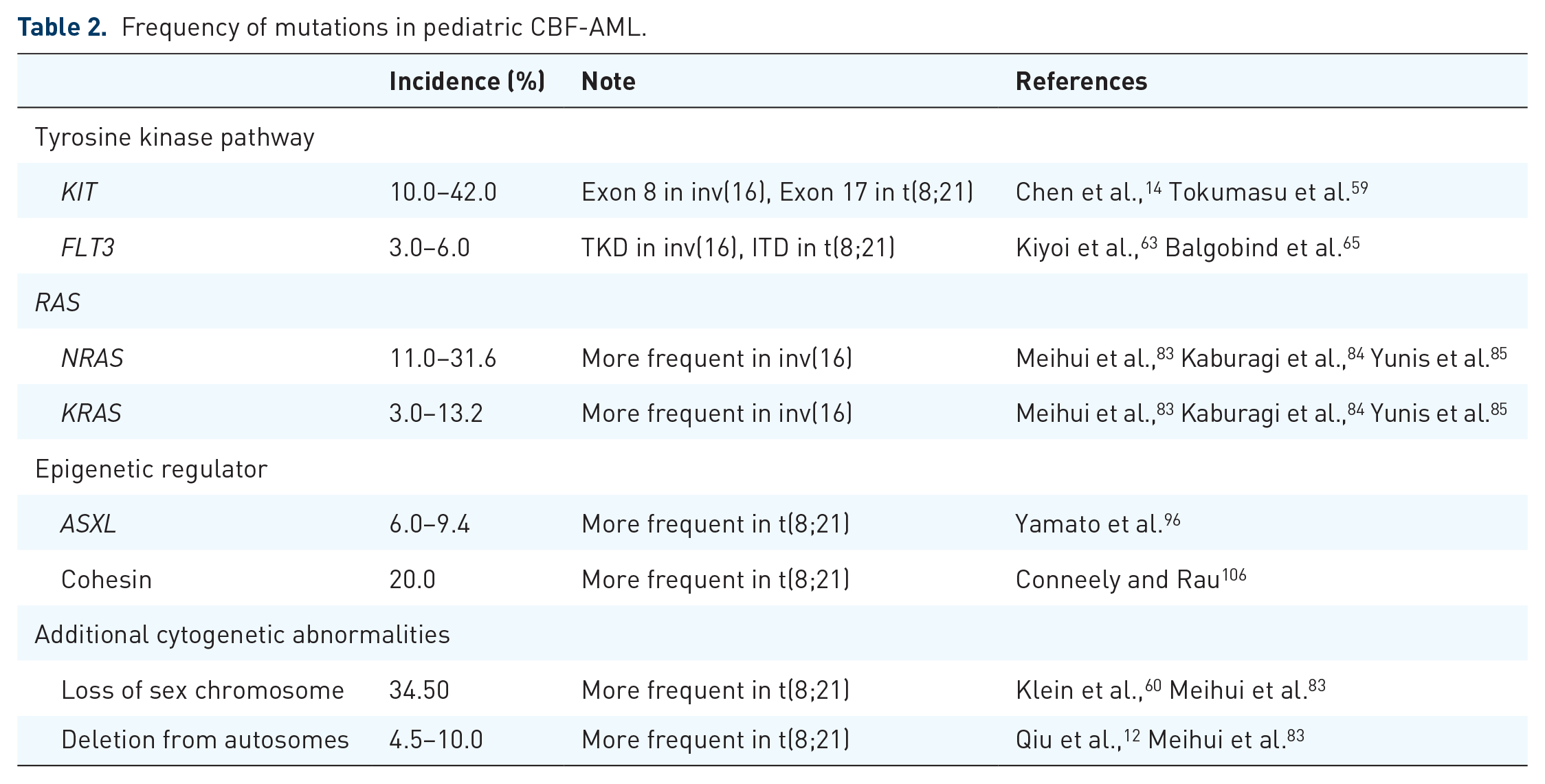
Patients with CBF-AML are generally associated with a favorable prognosis desipite the heterogeneity in genetic mutations, such as those in tyrosine kinase pathway (including KIT, FLT3, and N/KRAS), epigenetic regulators, and cohesion. The frequency of mutational profiling in pediatric CBF-AML is presented in Table 2, while the corresponding risk stratification based on the 2022 ELN recommendations is detailed in Table 3.
Frequency of mutations in pediatric CBF-AML.
Risk stratification of CBF-AML according to molecular status by 2022 ELN.

Not be used as an adverse prognostic marker if they co-occur with CBF-AML.
CBF-AML, core-binding factor acute myeloid leukemia; ELN, European Leukemia Net.
Tyrosine kinase pathway
KIT mutation
The proto-oncogene, KIT, a type III receptor tyrosine kinase, plays a crucial role in pathogenesis in AML, mast cell leukemia, and gastrointestinal stromal tumors.54,55 KIT-activating mutations trigger homodimerization and activation of downstream pathways by binding with ligand stem cell factor, which is essential for the survival, proliferation, and differentiation of hematopoietic stem cells. 54 In general, KIT mutations are critical factors in the prognosis of pediatric CBF-AML involving Exon 17 and Exon 8, while their impact in adults appears less pronounced. 56 Mutations in Exon 17 are single nucleotide substitutions: most commonly in codon D816, and one each in codons N822 and L793, found in t(8;21), leading to constitutive activation of tyrosine kinase. KIT D816 mutation is responsible for encoding the activation loop within the tyrosine kinase domain, negatively impacting prognosis.50,51 Conversely, mutations occurring in Exon 8, which are common in inv(16), may not demonstrate a clear prognostic impact, with small insertions/deletions affecting residues, referred to as gain-of-function mutations. 14 In adults, the KIT mutation was found in 17%–46% of CBF-AML.57,58 A study by Chen et al. found that the incidence of KIT mutation in a Chinese pediatric cohort with CBF-AML is 30%, which is similar to the prevalence observed in both the United States and Asia, ranging from 10% to 42% with mutations clustered in Exon 8 and Exon 17.14,59 No prognostic significance was observed in pediatric patients with KIT mutations in the American and Taiwanese series. However, a Chinese study revealed that KIT mutation was linked with a negative survival outcome with t(8;21)in adults but not in children. 60 Ayatollahi et al. demonstrated in a meta-analysis of 22 published series that KIT mutations were associated with a worse prognosis, especially in t(8;21), which is consistent with another meta-analysis performed by Chen et al.51,52 Furthermore, additional pediatric studies found that KIT mutations adversely affected overall survival (OS), disease-free survival (DFS), and risk ratio in pediatric t(8;21) AML but not in inv(16).38,57,61
In some pediatric studies with prognostic significance, the cohort received a standardized chemotherapy regimen based on the Medical Research Council guidelines, facilitating the potential to detect different prognostic outcomes based on KIT mutations.14,61 On the contrary, high doses of cytarabine and anthracyclines were used in some studies, which may explain the failure to verify prognostic significance for KIT. 60
In the context of CD33-targeted antibody-drug conjugate gemtuzumab ozogamicin (GO) therapy, treatment outcomes in patients with Exon 17 mutation are comparable to that of CBF-AML patients without KIT mutation. GO therapy showed promising results in reducing relapse and enhancing OS, EFS, and DFS in patients with Exon 17 mutation compared to those who only received standard chemotherapy, though the clinical benefits were less evident in patients with Exon 8 mutation. 62 It is intriguing to note that the clinical advantages of GO in pediatrics strongly imply that the unsatisfying outcomes seen in CBF-AML patients with KIT mutations and CBF abnormalities can potentially be improved through the implementation of enhanced treatment strategies.
FMS-like tyrosine kinase 3
FMS-like tyrosine kinase 3 (FLT3), also classified as a type III receptor tyrosine kinase, is pivotal in hematopoietic cell functions, consisting of two major types: internal tandem duplication mutations in the juxta-membrane domain (FLT3-ITD) and point mutations or deletion in the tyrosine kinase domain (FLT3-TKD).63,64
The overall incidence of FLT3 mutation in CBF-AML was approximately 13% with an inconsonant prognostic value reported in a meta-analysis. FLT3-ITD mutations are more commonly found in t(8;21) AML, whereas FLT3-TKD mutations are frequently detected in inv(16) cases with the prevalence 3.0%–6.0% in pediatric CBF-AML.63,65 The discovery of novel mutations and the application of genomic profiling, including whole exome sequencing, have expanded the understanding of the molecular landscape of FLT3 mutated CBF-AML in adults, revealing unique mutational signatures that could influence diagnosis, prognosis, and treatment strategies.66–69
Nevertheless, a small number of cases with FLT3-ITD have been reported in pediatric CBF-AML, with Chen et al. reporting no instance of FLT3-ITD in their CBF-AML cohort, paying little attention to pediatric CBF-AML with FLT3 mutation.14,70
Possible roles of TKI in the treatment of CBF-AML
The treatment methods for pediatric CBF-AML have progressed significantly, especially with the use of tyrosine kinase inhibitors (TKIs). TKIs have shown efficacy in targeting aberrant signaling pathways in CBF-AML, potentially improving outcomes in pediatric patients with specific mutations like KIT and FLT3.71,72
TKIs such as imatinib, dasatinib, and radotinib have shown prospects in treating AML with KIT mutations, highlighting their clinical relevance in specific molecular subtypes of CBF-AML.73,74 Imatinib, as an agent targeted for KIT mutation, has proven effective with the disappearance of KIT mutations at relapse in a phase II study among adults. Combining imatinib with other treatments, such as conventional chemotherapy, has been considered for enhancing therapeutic outcomes by leveraging synergistic effects in CBF-AML.
Dasatinib demonstrated acceptable pharmacokinetics and safety profiles, with a major adverse event of low grade and a high rate of CR observed in pediatric CBF-AML. Yang et al. concluded that with the use of a regimen combining dasatinib and conventional chemotherapy, the first CR rate was 95% in pediatrics, which was similar to adults.75,76
FLT3 inhibitors have shown promise in treating CBF-AML, including both adult and pediatric cases, although direct evidence from the provided contexts regarding its use specifically in pediatric CBF-AML is limited. Gilteritinib, a second-generation FLT3 inhibitor, is well-documented with its efficacy in FLT3-mutated AML, with significant improvements in survival outcomes and response rates in relapsed/refractory (R/R) settings.77–79 However, the specific efficacy and safety of gilteritinib monotherapy in pediatric CBF-AML remain underexplored. The general tolerability and potential for use in children have been indicated, but clinical trials are necessary to determine the appropriate dosing and to understand the drug’s efficacy in this subgroup fully.
RAS
Rat sarcoma gene (RAS gene), a group family proto-oncogenes, including neuroblastoma rat sarcoma viral oncogene homolog (NRAS) and Kirsten rat sarcoma viral oncogene homolog (KRAS). It encodes proteins responsible for the activation of RAF/MAPK signaling pathway, involving disturbances in the normal process of blood cell formation, interruption in cellular communication pathways, unchecked growth, and insufficient development resulting in the initiation and progression of leukemia.80,81
NRAS
The NRAS gene is crucial for regulating cell division, and its mutations lead to hyperactive RAS signaling, resulting in increased proliferation and decreased apoptosis of hematopoietic progenitors, which contribute to leukemogenesis. 82 According to research conducted by Yi et al. at the National Clinical Research Center for Blood Diseases in China, the incidence of NRAS was 31.6% in pediatric CBF-AML, which was similar to adults with CBF-AML but higher than the frequency of 13.4%–21.6% in pediatric AML.83–85 Studies have shown that NRAS mutations are commonly detected in CBF-AML, but their impact on prognosis, particularly in pediatric cases, varies, influenced by several factors, including co-occurring genetic alterations and specific NRAS mutations present. For instance, NRAS mutations were observed to be independent predictors of favorable outcomes in inv(16), suggesting a complex interplay between NRAS mutations and other genetic factors.84,86 On the contrary, NRAS mutations are associated with a poor prognosis regarding OS and DFS in adults. 58 However, some studies of NRAS mutations in CBF-AML revealed that these mutations, while prevalent, do not significantly affect the prognosis in terms of OS, suggesting that the presence of an NRAS mutation alone may not be a definitive prognostic marker and the need for a nuanced understanding of genetic mutations in pediatric CBF-AML. 87
The therapeutic landscape for pediatric CBF-AML with NRAS mutations presents a complex challenge since controversial impact on prognosis. Nowadays, bioinformatics analysis has further elucidated the deleterious effects of NRAS mutations on protein functionality, underscoring the potential of these mutations as targets for novel therapeutic interventions. 82 The emergence of drug resistance in CBF-AML, particularly against FLT3 inhibitors, has been linked to the acquisition of mutations in the RAS pathway, including NRAS, suggesting a need for alternative therapeutic strategies such as antisense oligonucleotides.88–90
KRAS
KRAS, another significant mutation in the RAS pathway, has been the subject of increasing attention, with an incidence of 13.2% among pediatrics CBF-AML in China, slightly higher than the cases of 3.7%–11.7% in pediatrics AML.83–85
KRAS mutations, as opposed to NRAS mutations, have been linked to poorer prognosis in AML in adults, characterized by shorter median OS and EFS, with the underlying mechanisms remaining unclear.91,92 One theory suggests that the presence of KRAS mutations might lead to resistance to initial treatment in AML, thus necessitating additional investigations to validate this hypothesis. 90
The prognosis and therapeutic prospects for pediatric CBF-AML with NRAS mutations are characterized by a multifaceted scenario, which is shaped by the intricate interactions of genetic changes and their treatment efficacy emphasizing the need for continued research and development of targeted therapies to improve prognosis and survival rates in affected children.
Epigenetic regulators
ASXL gene
The additional sex combs-like (ASXL) gene family consists of three distinct members—ASXL1, ASXL2, and ASXL3—that play crucial roles in the intricate process of epigenetic regulation. These three genes have been extensively studied for their significant impact on gene expression and chromatin remodeling. In various studies, mutations in ASXL1 and ASXL2 gene family have been consistently observed at elevated rates, particularly in myeloid neoplasms, including AML, shedding light on their potential involvement in the pathogenesis of AML.93,94 These discoveries have prompted significant interest in investigating the mechanisms and functions of ASXL proteins, their involvement in complex protein networks and the genetic alterations that contribute to the development of AML. The ASXL family plays a crucial role in the regulation of epigenetics, and when mutated, it typically results in the truncation of proteins at the C-terminal region before the plant homeodomain (PHD), ultimately promoting gene dysfunction and initiation of AML. This mutation-induced truncation of proteins at the C-terminal region disrupts the normal function of the gene, thereby causing a cascade of events that contribute to the pathogenesis of AML. 95 In a study with high-throughput sequencing technologies, a striking positive association of alterations and t(8;21) among pediatrics without independent impact on prognosis, consisting with another study showed that 6.0%–9.4% of patients with CBF-AML having ASXL1, ASXL2 mutation respectively, all of which is t(8;21) instead of inv(16).15,96
For CBF-AML patients with ASXL1 mutation especially those with higher WBC, research has shown that it is linked to poorer outcomes, including reduced OS and RFS, emphasizing the prognostic significance, consistent with findings across myeloid malignancies, where ASXL1 mutations are considered a poor prognostic factor.93,97–100 Moreover, the prognosis of pediatric AML with the ASXL1 mutation in a substantial Chinese multicenter cohort in the southern region of China was enhanced by HSCT (hematopoietic stem cell transplantation), aligning with findings in studies involving adult patients.97,101
The impact of mutations on epigenetic regulation like the ASXL family and its interaction with other genetic abnormalities underscore the need for further research to understand its role in disease progression and therapy response in pediatric CBF-AML.
BCOR and BCORL1
The genes BCOR and BCORL1 are situated on the X-chromosome and are responsible for encoding large nuclear proteins that exhibit widespread expression across various human tissues. Specifically, the BCOR protein functions as a corepressor of the BCL gene and can interact with different transcriptional factors, suggesting a pivotal role in the regulation of hematopoiesis. 102 In AML, mutations of BCOR and BCORL1 in leukemia disrupt the repressive function of PRC1.1 on target genes, leading to a cascade of events involving epigenetic reprogramming and the activation of abnormal cell signaling pathways. This interplay underscores the significance of BCOR and BCORL1 in the pathogenesis of AML, highlighting their potential as therapeutic targets for intervention strategies aimed at modulating aberrant gene expression and signaling cascades in leukemia. 103
Notably, mutations in BCOR and BCORL1 are infrequent occurrences within pediatric CBF-AML, posing a significant challenge in ascertaining their clinical significance and relevance. 103
Cohesin
The cohesin complex consists of four subunits, namely SMC1A, SMC3, RAD21, and STAG1/2. SMC1A, SMC3, and RAD21 create a central ring-shaped configuration, while STAG1/2 is an ancillary protein within the complex. This complex interplay of subunits forms a highly organized structure essential for the functional integrity of the cohesin complex underscoring its significance in various cellular processes, including DNA repair, chromosome segregation, and gene expression regulation. Mutations in the cohesin complex have a significant effect on the functionality of hematopoietic stem and progenitor cells, leading to alterations in the equilibrium between the processes of self-renewal and differentiation, a critical factor in the development of AML. 104
Several research studies, such as those conducted by Duployez and Faber, have observed cohesin mutations occurring alongside t(8;21) AML in approximately 18%–27% of cases. However, these studies found no instances of cohesin mutation in inv(16).15,105 In pediatrics, it has been observed that approximately 20% of patients with t(8:21) also present cohesin mutations; nevertheless, these mutations are notably lacking in cases of inv(16) AML. 106 Mutations in genes involved in epigenetic modifications or cohesin functions have been linked to an unfavorable prognosis in individuals with mutations in the tyrosine kinase pathway, indicating a potential synergistic interaction between these genetic events. 15 This association emphasizes the complicated interaction between different molecular mechanisms in influencing disease outcomes and suggests that a variety of collaborating mutations may impact the pathophysiology and clinical characteristics of CBF-AML, highlighting a potential distinct underlying mechanism for t(8;21) compared to inv(16). We hypothesize that the mutations under consideration, like the alterations mentioned above in epigenetic regulators, may disrupt the normal functioning of enhancers, leading to a dysregulation of transcriptional programs. This disruption is likely to impact the regulation of gene expression and potentially contribute to the development of various phenotypic traits and the progression of disease.
Additional cytogenetic abnormalities in pediatrics CBF-AML
The presence and influence of additional cytogenetic abnormalities in pediatrics with CBF-AML have been extensively investigated, and these findings can have a notable effect on the prognosis of the disease, accentuating its importance in the realm of treatment and the stratification of risk in pediatric CBF-AML.
Loss of sex chromosome
According to a retrospective analysis carried out by the Center for Blood Diseases in China, it was found that 34.5% of pediatric CBF-AML had a concomitant loss of sex chromosomes, a phenomenon that was observed more frequently in cases with the t(8;21), a finding that aligns with results in previous studies.15,39,60,83,107,108 Some research has indicated the correlation between the loss of a sex chromosome and improved outcomes in individuals across different age groups, showing higher rates of 5-year EFS and OS compared to those without the loss of a sex chromosome. This association has been documented in studies involving both pediatric and adult populations, suggesting a potential prognostic significance of sex chromosome loss in various medical conditions.12,60,109–111 However, Duployez et al. demonstrated that the absence of a sex chromosome does not significantly affect the outlook of patients with CBF-AML, which was established in a comprehensive study that included a diverse group of 73 children and 125 adults. 15 The results of this study suggest that the presence or absence of a sex chromosome does not play a major role in determining the prognosis for patients diagnosed with CBF-AML. This may have contributed to the resurgence of typical karyotypes following pathological remission underscoring its potential significance in disease surveillance and prognosis. Additional investigations are necessary to elucidate the precise repercussions of sex chromosome deficiency on the pathophysiology of CBF-AML, encompassing its involvement in leukemogenesis and stratification for individualized treatments.
Deletion from autosomes
Del(9q) is discovered in pediatric CBF-AML at a rate ranging from 4.5% to 10.0% based on data from the TARGET database and the Center for Blood Diseases in China, with all cases associated with the presence of t(8;21).12,83 Klein et al. demonstrated in their study that patients with CBF-AML who harbored del(9q) chromosomal aberrations exhibited a reduced CR rate compared to those without this deletion. In contrast, the investigation conducted by Qiu et al. did not identify any statistically significant disparities in survival outcomes and remission rates. This suggests that the prognostic implications of del(9q) in CBF-AML may vary across different studies and patient populations, warranting further research to elucidate its precise role in disease progression and therapeutic response.12,60
The cytogenetic abnormality del(9q) was no longer considered a significant factor for the diagnosis of AML with myelodysplasia-related changes conducted by the World Health Organization (WHO) since 2016, which has also been excluded in WHO 2022 and International Consensus Classification. This modification was introduced based on the frequent co-occurrence of del(9q) with mutations in CBF, which are pivotal in defining subgroups within AML.112–115 This approach enhances the understanding of CBF-AML genetics and highlights the importance of integrating multiple genetic alterations for accurate classification of hematologic malignancies.
Trisomy
In the realm of pediatric cases documented in the Nordic Society of Pediatric Hematology and Oncology (NOPHO) AML database, it is evident that there is an ongoing exploration related to the clinical significance and stratifications associated with cytogenetic variations, particularly those encompassing intricate translocations like t(8;21), as their infrequency poses challenges that warrant further scrutiny and investigation.116,117 Trisomy 4, a numerical abnormality observed in patients with t(8;21), is a rare occurrence that could indicate a more intricate disease progression and a less favorable prognosis. 118 Nevertheless, the infrequency of trisomy 4 in t(8;21) AML poses challenges in determining a conclusive prognosis and identifying the most effective treatment approaches for pediatric individuals affected by this condition.
Trisomy 8 mosaicism, classified as intermediate-risk AML according to the ELN classification, is characterized by intracranial abnormalities like agenesis of the corpus callosum and ventriculomegaly, leading to poor muscle tone and motor coordination. Distinctive features include deep palmar and plantar creases, dysmorphic facial features, and a single umbilical artery suggesting that allogenic HSCT should be considered as a consolidation therapy for affected patients.119,120 In CBF-AML, trisomy 8 is also observed in both t(8:21) and inv(16) and presents a range of prognostic implications, spanning from unfavorable to inconsequential on clinical results or even linked to extended survival, thus, the potential prognostic value of trisomy 8 necessitates thorough examination through extensive research studies.39,121–123 Trisomy of chromosome 21 is significantly more common in patients with inv(16) with specific clinical manifestations than t(8:21).39,124 Children with trisomy 21, also known as Down syndrome, are at a significantly increased risk of developing AML. 125 The common symptoms of trisomy 21 in pediatric AML can vary but often include various of hematologic and nonhematologic manifestations. Hematologic abnormalities are a hallmark of trisomy 21 AML, with patients frequently presenting with signs indicative of bone marrow failure, such as pallor, fatigue, and an increased susceptibility to infections due to neutropenia. Additionally, children with trisomy 21 may exhibit hepatosplenomegaly, a swelling of the liver and spleen caused by the infiltration of leukemic cells. 126 The risk of developing AML is significantly heightened in children with trisomy 21, with a greater than 100-fold increased risk compared to the general pediatric population. However, there was no noteworthy variation in the CBF-AML subgroup exclusively, indicating the necessity for additional research to elucidate the impact of trisomy 21 on the prognosis of pediatric CBF-AML.
Conclusion
The prognostic implications of CBF-AML mutations indicate that, despite the added complexity introduced by the presence of other mutations, the overall prognosis remains favorable. It is essential to detect, recognize, and target these mutational profiling to achieve personalized treatment.
Targeted therapies for CBF-AML are evolving, with promising strategies focusing on metabolic pathways, epigenetic regulation, and immunotherapy. For instance, midostaurin, a first-generation multitargeted TKI, has shown significant promise in treating AML with FLT3 and KIT mutations—genetic alterations often associated with poor prognosis. 127 The US Food and Drug Administration approved midostaurin for adult patients with newly diagnosed AML harboring the FLT3 mutation, in combination with standard induction and consolidation chemotherapy, marking a significant advancement in managing this aggressive leukemia.12,128 However, the role of midostaurin in pediatric CBF-AML remains unclear, as most research and approvals have focused on adult populations with the FLT3 mutation. As pharmaceutical research advances, newer TKIs like cabozantinib and lenvatinib mesylate are poised to dominate the market. Cabozantinib, a multitarget TKI, has shown substantial cytotoxicity in AML with specific genetic mutations, offering a versatile and potent therapeutic option by inhibiting multiple pathways, including KIT tyrosine kinase. 129 Similarly, lenvatinib mesylate has garnered attention for its efficacy in treating AML. The shift toward these newer agents is driven by their improved efficacy, manageable safety profiles, and ability to target multiple pathways, potentially offering better clinical outcomes and patient tolerability compared to older TKIs. 130 At the same time, small-molecule inhibitors targeting epigenetic regulators have been developed, aiming to reverse the aberrant chromatin modifications that drive leukemogenesis. 131 These compounds, such as inhibitors of histone methyltransferases and demethylases, have the potential to be integrated into combination therapies to enhance treatment efficacy and overcome resistance mechanisms.
Studies indicate that pediatric CBF-AML might represent a unique mutational profiling, necessitating a nuanced and personalized approach to treatment, wherein genetic mutations play a pivotal role in guiding clinical decision-making and optimizing patient outcomes, ultimately leading to notable differences in treatment outcomes. However, little study has targeted the mechanism of action, safety profile, and detailed usage in combination with therapy in pediatrics among CBF-AML. With unique pharmacokinetics among pediatrics, a combination of novel therapeutic strategies with a comprehensive risk assessment scale and patient-tailored treatment plans are needed for this challenging patient population.
Future research in pediatric CBF-AML will likely involve a combination of novel therapeutic strategies, improved risk assessment, and patient-tailored treatment plans to enhance outcomes for this challenging patient population.
