Abstract
Background:
We aim to analyze the efficacy and safety of Venetoclax (Ven) added to cladribine + cytarabine + granulocyte colony-stimulating factor (G-CSF) ± idarubicin or mitoxantrone (CLAG ± Ida/Mito) regimen as a salvage treatment of relapsed/refractory acute myeloid leukemia (RR-AML).
Methods:
A single-center, retrospective, cohort study was performed. Patients with RR-AML, being treated with CLAG ± Ida/Mito with versus without Ven, were retrospectively studied. The endpoints of this study were to evaluate the rate of composite complete remission (CRc), measurable residual disease (MRD), event-free survival (EFS), overall survival (OS), and relapse between CLAG and CLAG + Ven groups.
Results:
Sixty-nine patients were included, with a median age of 37 (range, 18–65) years. Thirty-one patients underwent one cycle of salvage treatment of CLAG ± Ida/Mito with Ven and 38 without. In the CLAG + Ven group, 24 (77.4%) patients acquired response, including 22 (71.0%) with composite complete remission (CRc) and 15 (48.4%) MRD-negative CRc, which was significantly higher than those (CRc 47.4%, p = 0.048; MRD-negative CRc 18.4%, p = 0.008) in the CLAG group. Subgroup analysis showed that patients without response after two courses of induction therapy, or patients with FLT3 mutations seemed to benefit more from CLAG ± Ida/Mito + Ven than CLAG ± Ida/Mito in acquiring CRc. With a median follow-up of 13 (95% CI 10.5–15.5) months, the CLAG + Ven group had a median OS of 22.9 (95% CI 19.6–26.2) months and EFS of 15.7 (95% CI 11.1–20.2) months. In contrast, the CLAG group had a median OS of 18.6 (95% CI 14.7–22.6) months and EFS of 10.7 (95% CI 6.6–14.8) months. Although not statistically significant, patients in the CLAG + Ven group showed a potential survival advantage compared to the CLAG group. AEs including all grade and grade 3/4 occurred at similar frequencies in the two groups.
Conclusions:
Ven added to CLAG ± Ida/Mito might improve the outcome of the patients with RR-AML, with well toleration, and a randomized controlled trial is needed to explored.
Plain language summary
Recent studies have shown the positive results of combining venetoclax (VEN) with intensive therapy for treating AML. The salvage regimen consisted of cladribine + cytarabine + granulocyte colony-stimulating factor (G-CSF) ± mitoxantrone or idarubicin (CLAG±Ida/Mito) was an optional alternative regimen for relapsed/refractory AML (R/R-AML) patients. However, whether the addition of the VEN to the CLAG±Ida/Mito regimen could improve the efficacy in R/R-AML is unknown. The authors assessed the effectiveness and safety of adding venetoclax to the CLAG±Ida/Mito regimen in RR-AML and demonstrated that the combined regimen is tolerable and induces higher response rates than CLAG did in relapsed/refractory(RR) patients.
Introduction
Patients with relapsed/refractory acute myeloid leukemia (RR-AML) have poor response to chemotherapy and need further study to improve their outcomes. According to the guidelines, clinical trials are the first choice for the treatment of RR-AML.1,2 As known, cladribine + cytarabine + granulocyte colony-stimulating factor (G-CSF) ± idarubicin or mitoxantrone (CLAG ± Ida/Mito) regimen is recommended for salvage treatment of patients with RR-AML being eligible for intensive chemotherapy,1,2 with a rate of complete remission (CR)/CR with incomplete count recovery (CRi) ranging 47.1%–58%.3–5 A recent prospective study showed that BCL-2 inhibitor Venetoclax (Ven) added to cladribine + cytarabine + idarubicin (CLA-Ida) as frontline treatment of AML acquired a composite CR (CRc) rate of 28/31 (90%) with well toleration. 6 In agreement with this, several studies reveal that Ven added to intensive chemotherapy might enhance the efficacy without increasing toxicity in the treatment of AML. A phase Ib/II study showed that Ven added to fludarabine + cytarabine + G-CSF + Ida (FLAG-Ida) obtained promising response not only in the frontline treatment of AML with a CRc rate of 90% but also in RR-AML with a CRc rate of 61%. 7 A real-world study further confirmed the efficacy and safety of FLAG-Ida-Ven in the treatment of RR-AML with a CRc rate of 14/30 (46.7%) and tolerable side effects. 8 Based on these, the use of Ven plus CLAG ± Ida/Mito in the salvage treatment of RR-AML could also enhance the response. Yet, the related studies are rare. Thus, we performed a single-center, retrospective, cohort study to analyze the efficacy and safety of CLAG ± Ida/Mito with versus without Ven as salvage therapy of RR-AML.
Methods
Patients
In Nanfang Hospital, Southern Medical University, CLAG ± Ida/Mito is one of the common regimens for the salvage treatment of patients with RR-AML who are eligible for intensive chemotherapy. After May 2021, Kadia et al. reported that Ven + CLA-Ida showed promising outcomes and good toleration in the frontline treatment of AML, which prompted some physicians to explore to combine Ven with CLAG ± Ida/Mito in treating RR-AML. In this study, patients with RR-AML and being salvagedly treated with at least one cycle of CLAG ± Ida/Mito with/without Ven in our clinic from July 2021 to October 2024 were retrospectively enrolled. Patients were excluded from this study if they had acute promyelocytic leukemia or lacked treatment response assessment. The diagnosis and risk stratification of AML were based on the National Comprehensive Cancer Network 1 and European Leukemia Net (ELN) 2022. 9 Refractory AML was defined as acquiring no CRc after one cycle of standard induction therapy. Relapsed AML was defined as recurrence of blasts in the peripheral blood or bone marrow (BM) blasts ⩾5% or development of extramedullary disease after achieving CRc.9,10
Definition and response assessment
Generally, BM was evaluated at cycle 1 from day 28 to 42. Subsequent BM evaluations were commonly done before next cycle of chemotherapy, and then as clinically needed. Response was evaluated according to the criteria of the ELN 2022 guideline. 9 CR was defined as BM with less than 5% blasts and without extramedullary infiltration and peripheral blood blast and recovery of peripheral blood cells. CRi was defined as all the criteria for CR, except for persistence of neutropenia <1 × 109/L or thrombocytopenia <100 × 109/L. Morphologic leukemia-free state (MLFS) was defined as all the criteria for CR, except for nonrecovery of peripheral blood cells. Partial remission (PR) was defined as BM blasts of 5%–25% and a decrease of more than 50% as compared with pretreatment. CRc included CR and CRi. Nonremission (NR) was defined as a failure to obtain CRc, MLFS, or PR. Overall response included CRc, MLFS, and PR. Measurable residual disease (MRD) was assessed with multiparametric flow cytometry. The MRD levels of 0.1% were used as a threshold to distinguish MRD positive from MRD negative.10–12 OS was calculated from the salvage treatment to death or the last date of follow-up. Event-free survival (EFS) was calculated from the salvage treatment to the date of relapse or the death or the last date of follow-up. Nonresponding patients were considered as progressing on cycle 1 day 1 for EFS.
Patient data were censored at the date of their last visit or the date when they were last confirmed to be alive. Toxicity was graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (version 5.0).
Statistical analysis
Patient characteristics were summarized using median (range) and inter quartile range (IQR) for continuous variables, and frequencies (percentages) for categorical variables. A backward selection logistic regression procedure was performed to evaluate the factors for the achievement of CRc, patients with an unknown variable were included in the analysis using a dummy variable indicating missing data. Time-to-event endpoints were evaluated by the Kaplan–Meier method. Analyses were performed using SPSS 19.0 (SPSS Inc., Chicago, IL, USA) and R version 4.1.3 (R Development Core Team, Vienna, Austria), and statistical significance was defined as a p value of <0.05.
Results
Patients and disposition
From July 2021 to October 2024, 75 patients were enrolled, and 69 patients were included (Figure 1), with a median age of 37 (range, 18–65) years, male to female 34/35, refractory to relapsed disease 44/25, median 2 (range, 1–12) cycles of prior chemotherapy, 19 with prior hypomethylating agent (HMA) exposure and 3 underwent allogenetic hematopoietic stem cell transplantation (allo-HSCT). Thirty-one patients underwent one cycle of CLAG ± Ida/Mito with Ven (CLAG + Ven group), and 38 received one cycle of CLAG ± Ida/Mito without Ven (CLAG group) as salvage treatment. As shown in Table 1 and Figure 2, patient demographics, disease characteristics, and genetic patterns were similar between the two groups, except more patients underwent prior HMA (p = 0.016) and harbored TET2 mutation (p = 0.026) in the CLAG + Ven group.

Patient enrollment and disposition.
Patient demographic and disease characteristics between CLAG + Ven and CLAG groups.

allo-HSCT, allogenetic hematopoietic stem cell transplantation; AML, acute myeloid leukemia; CLAG, cladribine + cytarabine + granulocyte colony-stimulating factor; ELN, European Leukemia Net; HMA, hypomethylating agent; Ven, Venetoclax.

Mutational landscape between CLAG + Ven and CLAG groups.
Treatment
The salvage regimen of CLAG ± Ida/Mito was performed according to the guidelines.1,2 In the entire group, 62 patients received CLAG, whereas 7 received CLAG plus Ida/Mito (4 in the CLAG + Ven group, 3 in the CLAG group, p = 0.493). The addition of Ven to CLAG ± Ida/Mito followed the usage of the previous study, 6 400 mg per day, from day 2 to day 8. The Ven dose was reduced by at least 50% in the patients receiving concomitant moderate or strong CYP3A4 inhibitors,10–12 In addition, 17 patients were combined with Fms-related receptor tyrosine kinase 3 (FLT3) inhibitors, including 8 with FLT3 mutation and 1 with AML1-ETO positive without FLT3 mutation in the CLAG + Ven group, and 8 with FLT3 mutation in the CLAG group, which was comparable between the two groups (Table 1).
Efficacy and the impacting factors
After the salvage treatment, 43 (62.3%) patients acquired response, including 23 (33.3%) CR, 17 (24.6%) CRi, 3 (4.3%) PR. As shown in Table 2 and Figure 3, in the CLAG + Ven group, 24 (77.4%) patients acquired response, including 22 (71.0%) CRc and 15 (48.4%) MRD-negative CRc, while 19 (50.0%) obtained response (p = 0.019), 18 (47.4%) CRc (p = 0.048), 7 (18.4%) MRD-negative CRc (p = 0.008), in the CLAG group. There was a significant difference in the rates of overall response, CRc and MRD-negative CRc, between the two groups. To balance the selection bias, we did a propensity score matching analysis with multiple logistic regression models between the two groups. Patients were selected by 1:1 matching without replacement using the nearest neighbor method, and a caliper width of 0.1 standardized differences for matching. Twenty-one patients of each group were score-matched, and their characteristics are shown in Supplemental Table 1. In the propensity score-matched population, the response rate in the CLAG + Ven group was also significantly better than that in the CLAG group (Supplemental Table 2), being consistent with the results before matching.
Treatment outcomes between CLAG + Ven and CLAG groups.

CI, confidence interval; CLAG, cladribine + cytarabine + granulocyte colony-stimulating factor; CR, complete remission; CRc, composite complete remission; CRi, CR with incomplete count recovery; MRD, measurable residual disease negative; NR, nonremission; ORR, overall response rate; PR, partial remission; Ven, Venetoclax.
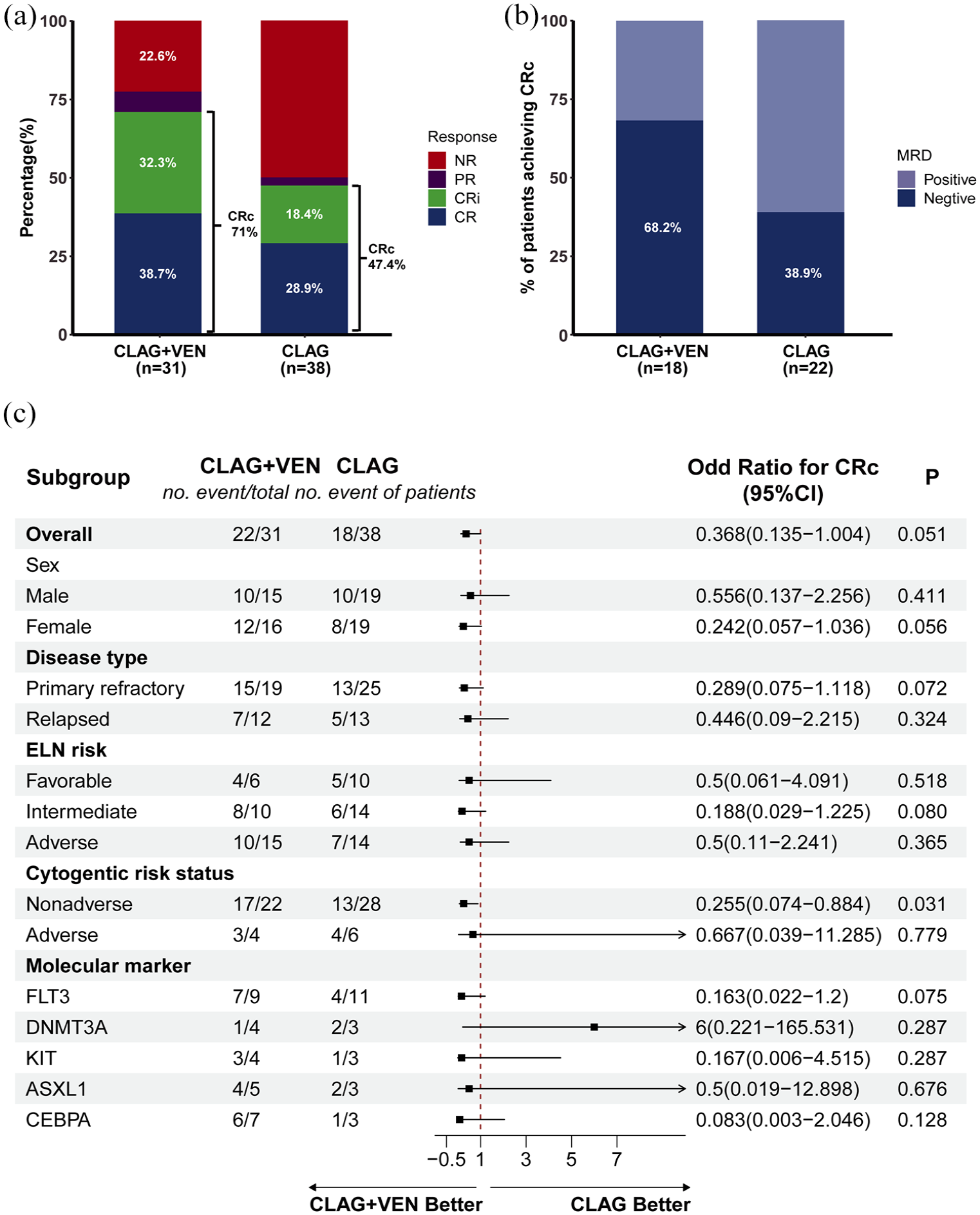
Treatment response and subgroup analysis of CLAG ± Ida/Mito + Ven versus CLAG ± Ida/Mito regimens. (a) The treatment response of CLAG+Ven versus CLAG, (b) The MRD-negative CRc rates of CLAG+Ven versus CLAG, (c) Subgroup analysis of the cohorts benefiting from the treatment of CLAG+Ven versus CLAG.
To assess the impacting factors of CRc, we did univariate analyses of the clinical characteristics, including gene mutations with an incidence of more than five cases. As shown in Supplemental Table 3, the CLAG + Ven regimen emerged as an independent factor favoring a positive response for CRc, while prior HMA exposure was a negative factor of acquiring CRc. Subgroup analysis showed that patients with prior HMA exposure had poorer CRc than those without even they were treated with CLAG + Ven regimen (Supplemental Table 4).
Treatment-emergent AEs in the CLAG + Ven versus CLAG group.

AEs, adverse events; CLAG, cladribine + cytarabine + granulocyte colony-stimulating factor; Ven, Venetoclax.
In addition, in Figure 3(c), subgroup analysis showed that female patients or patients with refractory AML or ELN-intermediate risk or nonadverse cytogenetics or FLT3 mutations tended to benefit more from CLAG ± Ida/Mito + Ven than CLAG ± Ida/Mito in acquiring CRc, though no statistical significance except in the cohort with nonadverse cytogenetics. We went further to analyze the treatment advantage of CLAG + Ven versus CLAG in the population with FLT3 mutation. Patients with FLT3 mutation distributed comparably as well as similar variant allele frequency and FLT3 inhibitors combination in the two groups (Supplemental Table 5). The benefit of CLAG + Ven versus CLAG not only in the salvage treatment of the patients with FLT3 mutation was observed, no matter as the whole cohort (CRc rate 7/9, 77.8% vs 4/11, 36.4%, p = 0.064), but also in those with FLT3 inhibitors combination (7/8, 87.5% vs 2/8, 25%, p = 0.012. Supplemental Table 6).
Furthermore, in comparison of CLAG ± Ida/Mito, CLAG ± Ida/Mito + Ven improved the CRc rate in the patients with at least two cycles of prior therapies, especially significantly in those acquiring NR after two courses of induction therapies (p = 0.040), but comparably in those with only one cycle of prior therapy (p = 1.000; Supplemental Figure 1(A)). We also analyzed the responses of the patients treated with CLAG or CLAG + Ven versus without Ida/Mito. As shown in Supplemental Figure 1(B), in the CLAG group, 17/35 (48.6%) patients without Ida/Mito acquired response, including 15 (45.7%) CRc and 5 (14.3%) MRD-negative CRc, while 2/3 (66.7%) cases with Ida/Mito obtained response (p = 0.547) as MRD-negative CRc (p = 0.025). In the CLAG + Ven group, 20/27 (74.1%) patients without Ida/Mito acquired response, including 18 (66.7%) CRc and 13 (48.1%) MRD-negative CRc; however, 4/4 (100%) cases with Ida/Mito obtained response as CRc (p = 0.170), including 2 (50.0%) MRD-negative CRc (p = 0.945), indicating that Ida/Mito added to CLAG or CLAG + Ven might tend to improve the response.
Survival and relapse
After the salvage treatment, 2 (6.5%) patients in the CLAG ± Ida/Mito + Ven group, and 6 (15.8%) in the CLAG ± Ida/Mito group received more than one cycle of other salvage therapy before proceeding to HSCT or other treatment modalities in the ongoing remission. After that, totally 34 (49.3%) patients were bridged to allo-HSCT, including 15 (45.5%) in the CLAG + Ven group, and 19 (50.0%) in the CLAG group (p = 0.894). With a median follow-up of 13 (95% CI 10.5–15.5) months, 12 out of 41 patients with CRc relapsed, with a cumulative incidence of relapse of 30.0%. A total of 18 (26.1%) patients died, including 16 that died of leukemia progression and 2 with transplant-related mortality. As shown in Table 2 and Supplemental Figure 2, 7/22 (31.8%) relapsed and 5 (16.1%) died in the CLAG + Ven group, with the median OS of 22.9 (95% CI 19.6–26.2) months and EFS of 15.7 (95% CI 11.1–20.2) months, while 5/18 (27.8%) relapsed and 13 (34.2%) died in the CLAG group, with the median OS of 18.6 (95% CI 14.7–22.6) months and EFS of 10.7 (95% CI 6.6–14.8) months. There was an apparent difference, though not significant, between the two groups. In the propensity score-matched population, the trend of survival benefit with the treatment of CLAG + Ven compared to CLAG was also observed (Supplemental Figure 3).
Among the whole cohort, patients obtaining CRc had significantly better survival (mOS, 24.9 (95% CI 22.4–27.4) months) than those without (mOS, 14.0 (95% CI 8.9–19.0) months, p < 0.001). Meanwhile, patients being bridged to allo-HSCT (mOS, 23.6 (95% CI 20.8–26.5) months) had significantly better OS than those who did not (mOS, 13.9 (95% CI 9.3–18.5) months, p = 0.007). Subgroup analysis also showed that achieving CRc (Supplemental Figure 2(C)) or bridging to allo-HSCT (Supplemental Figure 2(D)) might transfer into a survival benefit in both the CLAG + Ven or CLAG groups. The advantage was not statistically significant in the CLAG + Ven group (Supplemental Figure 2(D)) might be associated with a small case number and short term of follow-up.
Toxicity
All grade and grade 3/4 adverse events (AEs) occurring in the CLAG + Ven and CLAG groups are displayed in Table 3. Among the whole population, grade 3/4 AEs occurring in ⩾10% of patients included anemia (100%), neutropenia (100%), thrombocytopenia (100%), febrile neutropenia (62.3%), pneumonia (20.3%), and nausea, vomiting (11.6%). AEs including all grade and grade 3/4 occurred at similar frequencies in the CLAG + Ven and CLAG groups. Meanwhile, the median time to ANC recovery (>1000/nL) was comparable between the two groups, presenting a median of 17 (7–58) days in the CLAG + Ven group, and 17 (9–32) days in the CLAG group (p = 0.566). Among the seven patients with Ida/Mito, the median time for ANC recovery was 24 (17–58) days, with all patients experiencing febrile neutropenia and presenting longer ANC recovery times compared to patients without Ida/Mito (17 (7–58) days, p = 0.062), as well as significantly higher incidence of febrile neutropenia (36/62 (58.1%) in the patients without Ida/Mito, p = 0.030).
Across all cohorts, 30- and 60-day mortality was 0% and 5/69 (7.2%). Deaths in the study within 2 months all occurred in NR patients (sepsis, n = 2; pneumonia, n = 2; cerebral hemorrhage, n = 1), including 1 (3.2%) in the CLAG + Ven group and 4 (10.5%) in the CLAG group.
Discussion
Ven added to CLA + IDA has been shown promising response and well toleration as frontline treatment in AML. 6 In the present study, we demonstrated that Ven added to CLAG ± Ida/Mito might also be a promising regimen in the salvage treatment of RR-AML, presenting that, as compared with CLAG ± Ida/Mito, CLAG ± Ida/Mito + Ven significantly improved the response, including CRc and MRD-negative CRc, and potentially transferred into survival benefit, and did not increase the toxicity.
As the recommendation of guidelines,1,2,9 the CLAG ± Ida/Mito regimen has been widely used in the treatment of RR-AML.3–5 In line with previous reports,3–5 this study showed that patients receiving CLAG ± Ida/Mito regimen acquired the ORR of 50% (95% CI 33.6%–66.3%), CRc rate of 47.4% (95% CI 31.3%–63.9%), and MRD- CRc of 38.9% (95% CI 18.2%–63.8%), respectively. Based on our data, patients with refractory AML acquiring no response after two cycles of induction chemotherapy or FLT3 mutation seemed to respond poor to CLAG ± Ida/Mito regimen, with the CRc rate of only 5/17 (29.4%) and 4/11 (36.4%), respectively. How to improve the efficacy of CLAG ± Ida/Mito in RR-AML needs further study.
As reported, BCL-2 was always aberrantly overexpressed in RR-AML and was associated with poor outcomes.13–15 Thus, BCL-2 has emerged as a promising therapeutic target for patients with RR-AML.1,9–11 Ven, as a selective small-molecule BCL-2 inhibitor, its combination regimens have been recommended for the frontline treatment of unfit AML and salvage therapy of RR-AML.1,2,9 Recently, several studies have tried to combine Ven with intensive therapy to frontlinely6,7,16 or salvagedly17–19 treat AML, including Ven added to CLA + IDA as frontline treatment of AML, 6 acquired a promising response and well safety. As reported, Ven plus FLAG-Ida (n = 33), 17 CPX351 (n = 18), 18 or high-dose cytarabine and Mito (n = 12), 19 to treat patients with RR-AML and acquired the CRc rate of 54.5%, 37%, 92%, respectively. In line with these results, in this study, we added Ven to CLAG ± Ida/Mito to treat RR-AML, and acquired the ORR of 24/31 (77.4%), CRc 71.0%, and MRD- CRc 47.4%. In comparison with the result of CLAG ± Ida/Mito therapy, Ven plus CLAG ± Ida/Mito significantly improved the response, even transferred into a trend of survival benefit, further supporting that Ven added to CLAG ± Ida/Mito might enhance the outcome of AML. As reported, Ida/Mito added to CLAG might strengthen the antileukemia effect in AML.20–22 In agreement with this, though with a small case number, our result indicated that Ida/Mito added to CLAG or CLAG + Ven might improve the response, especially MRD-negative CRc. Yet, our data also revealed that Ida/Mito added to CLAG-based regimen might significantly prolong the time of agranulocytosis and increase the incidence of febrile neutropenia, further supporting that CLAG-based regimens might increase the risk of early death in comparison with standard CLAG. 23 Since a certain number of patients could not tolerate the treatment of CLAG + Ida/Mito, based on our results, Ven might take the place of Ida/Mito to combine with CLAG, and acquire better response than CLAG, and comparable to CLAG + Ida/Mito, but have better toleration.
We further analyzed who might benefit more from Ven plus CLAG ± Ida/Mito. In comparison of CLAG ± Ida/Mito, Ven added to CLAG ± Ida/Mito significantly improved the response of the patients with primary refractory AML who could not acquire response after two cycles of induction therapy. Furthermore, female patients, and those with ELN-intermediate risk or nonadverse cytogenetics, as well as FLT3 mutations, appeared to derive better benefits from the treatment combining Ven with CLAG ± Ida/Mito. In contrast, patients with prior HMA or TP53 mutation seemed to respond poor to CLAG ± Ida/Mito with/without Ven. The true for poor response to CLAG ± Ven treatment in patients with prior HMA needs further studies. As reported, patients with TP53 mutation are expected to exhibit resistance to DNA-damaging chemotherapy, given that regimens containing cytarabine can lead to the clonal outgrowth of TP53-mutant cells.24,25 Badar T et al. highlighted that there was no benefit in adding Ven for patients harboring TP53 mutation. 26 In line with these reports, in this study, patients with TP53 mutation could only acquire the ORR of 2/6 (33.3%) with CLAG ± Ven treatment, compared to 41/63 (65.1%) in those with TP53 wild type (Supplemental Table 7).
Allo-HSCT is a vital treatment for curing RR-AML.27,28 In this study, 49.3% of patients were bridged to allo-HSCT and had significantly better OS than those who did not (p = 0.007). Subgroup analysis further supported patients might benefit from bridging to allo-HSCT regardless of salvage regimens, though the benefit was not significant in the CLAG + Ven group, which might be associated with small sample sizes and short time of follow-up. In addition, the OS of the patients with versus without prior HMA exposure was comparable, which might be attributed to almost half of the patients being bridged to allo-HSCT after salvage therapy. This finding suggests that allo-HSCT should be recommended as a subsequent treatment after salvage therapy, regardless of the salvage regimens.
In this retrospective cohort study, patients in the CLAG + Ven versus CLAG groups had similar side effects, with no early treatment-related mortality observed, indicating Ven added to CLAG ± Ida/Mito was tolerable, in agreement with that Ven added to intensive therapy does not increase toxicity.6–8,17–19 Grades 3 and 4 myelosuppression happened in all the included patients. The median recovery time of ANC was 17 (7–58) days and was comparable between the two groups. Febrile neutropenia, pneumonia, and nausea and vomiting were the most frequent grades 3 and 4 AEs, all of which were manageable. Taken collectively, this indicates that adding Ven to CLAG could be a potentially effective regimen with favorable tolerability.
Several limitations should be considered in our study. There was a single center, retrospective, cohort study, and patient selection bias might exist. Also, sample sizes were small, and the follow-up time of survival was relatively short. Third, the relative younger age in the population may not truly represent all RR-AML patients in real-world clinical practice.
Conclusion
The salvage regimen of Ven added to CLAG ± Ida/Mito might improve the outcome of the patients with RR-AML, especially in those with refractory disease acquiring no response after two cycles of induction or FLT3 mutation, without increasing toxicity. Multicenter, prospective, randomized-controlled trials are needed to explore this.
Supplemental Material
sj-docx-1-tah-10.1177_20406207251319603 – Supplemental material for Venetoclax added to CLAG regimen might improve the outcome of patients with relapsed/refractory acute myeloid leukemia
Supplemental material, sj-docx-1-tah-10.1177_20406207251319603 for Venetoclax added to CLAG regimen might improve the outcome of patients with relapsed/refractory acute myeloid leukemia by Yu Zhang, Zhao Yin, Zurong Yao, Dan Xu, Xuejie Jiang, Xiaqi Nie, Dandan Chen, Hongsheng Zhou, Pengcheng Shi, Hui Liu, Qifa Liu and Guopan Yu in Therapeutic Advances in Hematology
Footnotes
Appendix
Acknowledgements
We appreciate the data platform provided by Nanfang Hospital, Southern Medical University. We are also grateful to all the faculty members who participated in this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
