Abstract
This report describes the cases of two patients with mycosis fungoides and Sézary syndrome (MF/SS) who achieved clinical benefit with mogamulizumab combination therapies. Case 1 is a 56-year-old male with stage IIIB (T4NxM0B1) MF, which later progressed into SS, with ongoing skin symptoms (erythema, lichenified skin, and pruritis) and axillary and inguinal lymphadenomegaly. Skin-directed and systemic therapies failed to achieve a long-lasting response in this patient. Mogamulizumab (1 mg/kg once weekly for 4 weeks; once every 2 weeks thereafter) yielded temporary improvement in skin symptoms, but progression in the skin was confirmed after ~2 months. Subsequently, the combination of mogamulizumab with psoralen plus ultraviolet light A (PUVA) yielded a partial response; however, PUVA was discontinued due to phototoxicity and mogamulizumab was continued as monotherapy. At the latest evaluation, clinical improvement in the skin and reduced lymphadenomegaly were evident with ongoing mogamulizumab monotherapy; the patient is awaiting allogeneic hematopoietic stem cell transplantation. Case 2 is an 80-year-old male with stage IIIB (T4NxM0B1) granulomatous variant MF who presented with diffuse erythema with desquamation, ectropion, and inguinal lymphadenopathy. Treatment with oral prednisone and bexarotene failed to achieve adequate, long-lasting responses. Mogamulizumab (1 mg/kg once weekly for 4 weeks; once every 2 weeks thereafter) monotherapy yielded an initial improvement, characterized by less intense erythema, but the improvement was not sustained. Mogamulizumab was supplemented with oral prednisone and then PUVA; this combination resulted in improvement in the skin. PUVA was stopped due to unavailability, and methotrexate (10 mg once weekly) was initiated alongside continued mogamulizumab; this led to improvement in erythema. The patient continued mogamulizumab plus methotrexate with controlled clinical symptoms prior to their death, which was deemed to be unlikely to be related to treatment. Our experience suggests that, in principle, mogamulizumab can be used in combination with other therapies; however, further research is needed.
Keywords
Background
Mycosis fungoides (MF) and Sézary syndrome (SS) are subtypes of cutaneous T-cell lymphoma (CTCL). 1 Most patients present with early stage MF (stage IA–IIA), typically manifesting in the skin with patches and/or plaques with a generally indolent course and low risk of disease progression.1–3 However, advanced‑stage (stage IIB–IVB) disease can manifest with fungating tumors or erythroderma of the skin and disease involvement in the blood, lymph nodes, and viscera, leading to a median survival of 1.4–4.7 years.1–4 Achieving “curative” complete response is rare; the typical goals of treatment are to limit morbidity, improve quality of life, and restrict disease progression.1,5 Patients with early stage disease are generally managed with skin-directed therapies, whereas those with advanced-stage disease are generally managed with systemic treatment or the combined use of skin-directed and systemic treatments.5,6 Greater understanding of factors such as biomarkers, and stage- and compartment-specific heterogeneity may support the tailoring of treatments; indeed research in this area is ongoing, and many recent publications have explored these topics. 7
Mogamulizumab is a defucosylated humanized immunoglobulin G1 kappa monoclonal antibody targeting the C-C chemokine receptor 4 (CCR4). 8 It is approved as a second- or later-line therapy for adult patients with MF or SS who have received at least one prior systemic therapy.8,9 Mogamulizumab triggers antibody-dependent cellular cytotoxicity by binding to the CCR4 receptor expressed by malignant T-cell lymphomas.8–10 The results from the phase III, open-label, randomized, controlled MAVORIC study showed significant improvements in progression-free survival (PFS) in patients with stage IB–IVB relapsed or refractory MF/SS receiving mogamulizumab (1 mg/kg intravenously once weekly for the first 28-day cycle, then on days 1 and 15 of subsequent cycles) compared with those receiving vorinostat (400 mg/day; 7.7 months vs 3.1 months; hazard ratio: 0.53; 95% confidence interval, 0.41–0.69; p < 0.0001). 11 Based on these results, The National Comprehensive Cancer Network guidelines for primary cutaneous lymphomas recommend mogamulizumab as a systemic treatment regimen for patients with MF/SS (stage IB onward). 12 Although the MAVORIC trial assessed mogamulizumab as monotherapy, 11 combination treatment approaches using skin-directed therapy alongside systemic therapy may maximize clinical response in the skin. 5
Here, we report the cases of two patients with MF and SS refractory to skin-directed and initial systemic interventions. Patients 1 and 2 initially received mogamulizumab monotherapy for 3 and 10 months, respectively. The addition of psoralen + ultraviolet light A ((PUVA) alongside continued mogamulizumab for 4 and 3 months in patients 1 and 2, respectively) resulted in prolonged clinical response. These cases highlight the potential benefits of using mogamulizumab in a combination regimen for patients with refractory MF or SS. The reporting of these case studies conforms to the CARE guidelines. 13
Case 1
Clinical findings and diagnostic assessment
A 56-year-old White male presented with pruritic erythema in 2019. Due to ongoing symptoms despite initial treatment, he received a dermatology evaluation in December 2020 (see Supplemental Figure 1 for a timeline of the patient case, and Supplemental Table 1 for a summary of treatments and their outcomes for this case). The evaluation revealed axillary and inguinal lymphadenomegaly on physical examination and erythroderma covering the whole body, except the face. Skin was lichenified to touch, and the patient experienced intense daily itching. Skin biopsy in December 2020 revealed a morphologic and immunophenotypic picture compatible with cluster of differentiation (CD)4+ CTCL (Table 1).
Morphologic and immunophenotypic results of the skin biopsy (December 2020).

CD, cluster of differentiation; CXCL13, C-X-C motif chemokine ligand 13; EBER, Epstein Barr virus-encoded small RNA; PD1, programmed death 1.
Flow cytometry (FC) revealed atypical T-cell populations in peripheral blood, affecting 16% of total lymphocytes (CD3±/CD4+/CD26−/CD2±/CD7 bimodal/CD5+/CD8−; 300/mm3). Positron emission tomography with computed tomography showed axillary lymphadenopathy, with other lymph nodes weakly avid at the laterocervical, retronuchal, and bilateral supraclavicular levels. Additionally, there was metabolically active thickening of the subcutaneous soft tissues in the sacrococcygeal region. These results confirmed the stage IIIB (T4NxM0B1) MF diagnosis.
Therapeutic interventions
In January 2021, erythema covered 80% of the patient’s body surface area (BSA). A number of therapeutic interventions (extracorporeal phototherapy (ECP), methotrexate, clobetasol cream, and bexarotene) were initiated between January and October 2021; however, continued disease progression of the skin led to subsequent cessation of ECP, methotrexate, and bexarotene, while clobetasol cream was continued.
By November 2021, the patient experienced continued disease progression, with erythroderma covering >90% of the patient’s BSA and desquamation (Figure 1(a)). Intense itching and conjunctival involvement were reported. Lactate dehydrogenase (LDH) serum evaluation showed a slight elevation (231 U/L, normal value < 225 U/L). Mogamulizumab (80 mg (1 mg/kg IV weekly on days 1, 8, 15, and 22 of the first 28-day cycle, then infusions every 2 weeks thereafter on days 1 and 15)) was commenced. Mogamulizumab was initially successful, reducing itching and resulting in regression of skin disease (40% reduction in affected BSA from November 2021 within the first weeks of treatment) and conjunctivitis. FC performed during the fifth infusion of mogamulizumab revealed a CD4+/CD26− T-cell population affecting 68% of total lymphocytes in peripheral blood (1900/mm3), consistent with disease progression to secondary SS; however, due to the lack of baseline FC data before treatment, early changes in blood tumor burden during mogamulizumab therapy could not be confirmed.
By the end of December, erythema worsened, progressing to erythroderma in subsequent weeks with itching and desquamation on the back and thighs (Figure 1(b)), and serum LDH was slightly elevated (260 U/L). PUVA was added to the mogamulizumab treatment regimen in February 2022 (initially 0.5 J/cm2 twice a week, with increases of 0.5 J/cm2 every three sessions approximately; cumulative dose 62 J/cm2).

Disease progression in the skin: (a) diffuse erythroderma covering >90% of BSA and presence of desquamation in November 2021 and (b) worsening itching and desquamation in December 2021.
Follow-up and outcomes
By April 2022, there was clinical improvement in skin symptoms (erythema affecting 30% of the BSA) and partial improvement in lymphadenomegaly. The tests showed normalization of serum LDH (195 U/L). However, in May 2022, bullous lesions appeared on the patient’s upper limbs and trunk, indicative of a phototoxic psoralen reaction. PUVA treatment was immediately suspended, and the phototoxicity was resolved. PUVA was restarted 2 weeks later, at a reduced intensity (2.5 J/cm2 twice weekly; cumulative dose 74.5 J/cm2), but bullous dermatitis reappeared after five PUVA sessions. Therefore, PUVA treatment was discontinued in June 2022. In August 2022, lesions with a bubble-like appearance and superficial abrasions were noted on non-photo-exposed sites and near bony prominences (Figure 2).

Persistence of bullous lesions with bubble-like appearance following discontinuation of PUVA during combination mogamulizumab treatment: (a) patient’s back and (b) patient’s arm.
Skin biopsy of the lesion indicated a bullous pemphigoid (BP)-like reaction, likely induced by PUVA or mogamulizumab treatment (Table 2). Blood tests indicated the patient was negative for antibodies to desmoglein 1 (2.03 U/mL), desmoglein 3 (1.25 U/mL), and BP230 (1.04 U/mL), and positive for BP180 (78.58 U/mL). Stevens–Johnson syndrome/toxic epidermal necrolysis was suspected but excluded based on the morphologic presentation, as the lesions were not considered potentially life-threatening. The BP-like reaction was resolved by September 2022.
Morphologic and immunofluorescence results of the skin biopsy (August 2022).

CD, cluster of differentiation; CXCL13, C-X-C motif chemokine ligand 13; PD1, programmed death 1.
FC in November 2022 showed CD4+/CD26− T-cell populations comprising 42% of total lymphocytes (560/mm3), an increase from September 2022 (35%; 410/mm3), with serum LDH unaltered (182 U/L). Mogamulizumab monotherapy is currently ongoing (August 2024), maintaining the clinical stability of the skin disease (erythema affecting 20%–30% of BSA; Figure 3) and reducing lymphadenopathies (with a few pericentimetric laterocervical lymph nodes remaining).
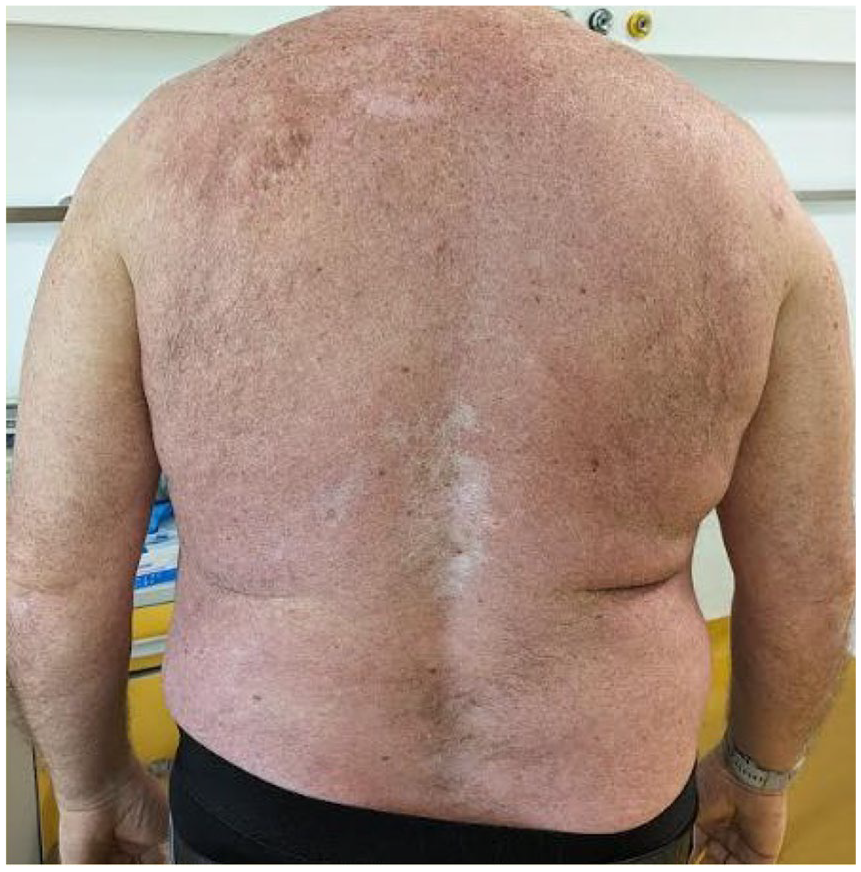
Presence of diffuse erythema on the trunk only, and residual effects of bullous lesions during ongoing mogamulizumab monotherapy (April 2023).
Overall, mogamulizumab infusions were well tolerated, and the addition of PUVA improved clinical response. However, skin lesions resulting from combination therapy caused discomfort; the development of a BP-like reaction was possibly induced by the combination treatment. No other tolerability issues were observed. At the time of writing, the patient awaits allogeneic hematopoietic stem cell transplantation, pending identification of a suitable donor.
Case 2
Patient information and clinical findings
In April 2021, an 80-year-old White male presented to their dermatologist with diffuse erythema with desquamation, ectropion, and inguinal lymphadenopathy; these comorbidities had persisted for about 2 years and were progressively worsening (see Supplemental Figure 2 for a timeline of the patient case).
Diagnostic assessment
Lymphoproliferative disease was suspected; FC testing of peripheral blood revealed an atypical T-cell population (CD3±/CD4+/CD26−/CD2±/CD7 bimodal/CD5+/CD8−) comprising 19.7% of total lymphocytes (550/mm3), and skin biopsy from the right hip showed stage IIIB (T4NxM0B1) granulomatous variant MF. Abdominal ultrasound did not show splenomegaly or hepatomegaly, but some lymphadenopathy was observed in the groin.
Therapeutic interventions
Treatment with oral prednisone and bexarotene was initiated (see Supplemental Table 2 for a summary of treatments and their outcomes for this case); however, disease progression occurred, leading to discontinuation of the treatments in June 2021 and August 2021, respectively.
After excluding PUVA treatment due to logistic challenges, mogamulizumab (60 mg (1 mg/kg IV weekly on days 1, 8, 15, and 22 of the first 28-day cycle; followed by infusions every 2 weeks thereafter on days 1 and 15)) monotherapy was initiated in August 2021. Clinical improvement was achieved with mogamulizumab (erythema BSA, 50%–60%; atypical T cells, 2.8% of total lymphocytes (20/mm3)) in December 2021. Erythroderma and desquamation recurred in May 2022, and oral prednisone (25 mg/day) was added for 2 months before being stopped due to progression. Concurrently, there was an increase in serum LDH, which rose to 266 U/L, while previous values were within the normal range (<225 U/L). PUVA (2.5 J/cm2 weekly; cumulative dose 17.5 J/cm2) in combination with mogamulizumab was initiated in June 2022, resulting in improvement in the skin, resolution of itching and desquamation, and a decrease in serum LDH to 230 U/L in July 2022.
Follow-up and outcomes
In September 2022, PUVA was stopped due to a lack of local availability; the patient was unable to go to another center to continue PUVA. In October 2022, worsening erythroderma with desquamation over the entire BSA occurred. Similarly, there was an increase in serum LDH to 318 U/L. Oral methotrexate (10 mg once weekly) was initiated alongside mogamulizumab, with marked improvement in erythema (50%–60% of BSA) by November 2022 and a decrease in serum LDH (224 U/L) by December 2022. At the last visit in January 2023, symptoms were well controlled with mogamulizumab and methotrexate, and the patient’s erythema was stable without itching and desquamation. Mogamulizumab and methotrexate did not result in toxicity. The patient unexpectedly passed away in April 2023; this was judged by the authors as unlikely to be related to treatment.
Discussion
In this report, the cases of two patients with MF and SS refractory to skin-directed and initial systemic therapies who ultimately experienced clinical benefit with mogamulizumab combination treatment regimens are described.
Case 1 was a 56-year-old male with MF that progressed to SS during treatment, who did not experience adequate, long-lasting response to previous lines of treatment. Mogamulizumab was started as a monotherapy with some initial clinical improvement, but the addition of PUVA was beneficial in terms of increased disease control, suggesting combination therapy has a potential additional effect (although patients may experience additional toxicity). Case 2 was an 80-year-old male with granulomatous variant MF who did not experience adequate, long-lasting clinical response with oral prednisone or bexarotene. Mogamulizumab monotherapy yielded an initial clinical improvement before symptom recurrence; it was continued in combination with oral prednisone, followed by PUVA, and then methotrexate, each of which was associated with clinical improvement. At the time of the last evaluation prior to the patient’s sudden death, he continued to receive mogamulizumab plus methotrexate, his clinical symptoms were well controlled, and his skin condition was stable. Although the clinical course of our patients suggests a beneficial effect of combination therapy with PUVA and mogamulizumab, the additional effect of PUVA is unclear and not measurable.
Published clinical data regarding the use of mogamulizumab in combination therapy regimens are limited. In MAVORIC, the use of concomitant steroids was permitted, as per the protocol. 11 In the only evaluation to consider the efficacy and safety of combination therapy from MAVORIC, a post hoc analysis showed greater improvements in PFS and overall response rate in patients receiving mogamulizumab with concomitant steroids versus the intention-to-treat population, with no unexpected differences in adverse event profiles. 14 A small number of case reports and studies suggest mogamulizumab may have some clinical benefit in combination with bexarotene,15,16 total skin electron beam therapy, 17 ECP,16,18 interferon, 16 or radiation therapy. 19 Further clinical experience is required to validate these observations. Trials to assess mogamulizumab in combination with ECP, total skin electron beam therapy, or brentuximab vedotin in CTCL are ongoing [ClinicalTrials.gov identifiers: NCT04930653, NCT05414500, and NCT04256018].
In Case 1, a BP-like reaction was noted following combination treatment with mogamulizumab and PUVA therapy, though causality was not assessed. The morphologic and immunofluorescence findings could be nonspecific and related to the dermal–epidermal damage caused by PUVA, 20 but such observations warrant additional investigation. No cases of BP reactions in patients receiving mogamulizumab in the MAVORIC trial were reported at the time of writing.
In the retrospective study by Masuda et al 2018., it was observed that four patients who received mogamulizumab and narrow-band UVB developed photosensitivity dermatitis. 21 The authors of this study observed that the photosensitive lesions had a CD8+ T-cell-dominant infiltrate, suggesting an autoimmune reaction similar to chronic actinic dermatitis, while Foxp3+ regulatory T cells were decreased in the photosensitivity lesions compared with lymphoma lesions. 21 The authors suggested that depletion of the CCR4-positive regulatory T cells by mogamulizumab potentially contributed to the activation of CD8+ cytotoxic T cells against epidermal keratinocytes; therefore, the photosensitivity observed may have been a unique immune-related adverse event. 21 In Case 1, a similar autoimmune activation may have occurred to that described in Masuda et al 2018.
In conclusion, data from the present case studies and those reported previously suggest that mogamulizumab could represent an option in combination with systemic and skin-directed therapies, with a potential synergistic effect. However, further studies are needed to evaluate the efficacy and toxicity profiles of these approaches.
Supplemental Material
sj-pdf-1-tah-10.1177_20406207251317165 – Supplemental material for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports
Supplemental material, sj-pdf-1-tah-10.1177_20406207251317165 for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports by Andrea Bernardelli and Carlo Visco in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-2-tah-10.1177_20406207251317165 – Supplemental material for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports
Supplemental material, sj-pdf-2-tah-10.1177_20406207251317165 for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports by Andrea Bernardelli and Carlo Visco in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-3-tah-10.1177_20406207251317165 – Supplemental material for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports
Supplemental material, sj-pdf-3-tah-10.1177_20406207251317165 for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports by Andrea Bernardelli and Carlo Visco in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-4-tah-10.1177_20406207251317165 – Supplemental material for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports
Supplemental material, sj-pdf-4-tah-10.1177_20406207251317165 for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports by Andrea Bernardelli and Carlo Visco in Therapeutic Advances in Hematology
Supplemental Material
sj-pdf-5-tah-10.1177_20406207251317165 – Supplemental material for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports
Supplemental material, sj-pdf-5-tah-10.1177_20406207251317165 for Management of mycosis fungoides and Sézary syndrome with mogamulizumab in combination with psoralen plus UVA: two case reports by Andrea Bernardelli and Carlo Visco in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors would like to thank the patients and their families for providing consent to use their medical information to make this article possible.
Declarations
Supplemental material
Supplemental material for this article is available online.
The timeline of interventions and outcomes from Case 1.
The timeline of interventions and outcomes from Case 2.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
